Abstract
The purpose of this study was to describe the development of the Gastrointestinal and Gastroesophageal Reflux (GIGER) Scale for Infants and Toddlers, and determine its factor structure and psychometric properties. Items were developed to comprehensively assess gastrointestinal (GI) and gastroesophageal reflux (GER) symptoms observable by a parent. Exploratory factor analysis on 391 responses from parents of children under 2 years old resulted in a 36-item scale with 3 subscales. Internal consistency reliability was acceptable (α = .78-.94). The GIGER total score and all 3 subscales were correlated with the Infant Gastroesophageal Reflux Questionnaire-Revised (I-GERQ-R) (P < .05) and Infant Gastrointestinal Symptoms Questionnaire (IGSQ) (P < .05). GIGER total score was higher in infants with a diagnosis of GER (P < .05) or constipation (P < .05) compared to those without. The GIGER is a parent-report measure of GI and GER symptoms in children under 2 years old with adequate psychometric properties.
Introduction
Parent concerns about symptoms along the gastrointestinal (GI) tract, including GI distress and/or gastroesophageal reflux (GER), are common in young children.1,2 These symptoms are often non-specific and it can be difficult for clinicians to differentiate symptoms that fall within the range of normal from symptoms that may warrant further evaluation, intervention, and/or referral. For clinical practice, valid and reliable assessment tools can complement clinical assessment to guide decision-making and personalized care, or provide reassurance if symptoms fall within the range of normal. For research, parent-reported assessment tools can be used to compare symptoms between populations and can be used longitudinally to measure change over time or in response to interventions. Currently-available parent-report measures of GI distress and GER for children under 2 years old are not written in parent-friendly language and do not comprehensively assess symptoms along the entire GI tract. The purpose of this study was to describe the development of a new parent-report measure, the Gastrointestinal and Gastroesophageal Reflux (GIGER) Scale for Infants and Toddlers, determine the factor structure of this new measure, and evaluate its psychometric properties, including internal consistency reliability, convergent validity, and known-groups validity.
Methods
Ethical Approval and Informed Consent
This study (Protocol Numbers: 20.200.01e,20.258.01,20.267.01,21.073.01e) was approved by the Institutional Review Board at Boston College. Written informed consent was obtained from participants prior to participation.
Item Development
A literature review was conducted in PubMed and Cumulative Index to Nursing and Allied Health Literature (CINAHL) using the following search terms: (((assessment) or (tool) or (questionnaire) or (survey)) and (((gastrointestinal) or (gastroesophageal) or (intestinal)) and (symptoms)) and ((infant) or (neonate) or (child) or (toddler))). The following limits were placed on the search: English language, full-text, humans, ages 0 to 23 months, and research articles. Two years old was chosen as the limit for this scale to obtain parent-reported information about children who are unable to speak or to articulate discomfort symptoms. While this is a large age range and there is considerable development of the GI tract across this time, a single measure with items relevant across this age range would allow for consistent measurement over time. Age-specific reference values can provide guidance on expected development over time.
Article titles/abstracts were screened for inclusion and exclusion criteria. To be included, the article had to be a research study reporting on the development of or use of a parent-report measure of gastrointestinal or gastroesophageal reflux symptoms in children under 2 years of age. Articles were excluded if they were a review article, child age was >2 years old, no assessment tool was used specifically to measure GI or GER symptoms, or the article reported on a diagnostic assessment (eg, laboratory test, pH probe) or clinician assessment.
After initial screening of irrelevant articles, full-text articles were reviewed for inclusion/exclusion criteria. Assessment tools used in each article that met inclusion/exclusion criteria were identified and a separate search was conducted for each of these to identify psychometric properties of the tools. Items from each tool were qualitatively coded for concept(s) measured by the item. We compiled a list of concepts measured across all of the tools, then mapped these concepts to items on the Neonatal Eating Assessment Tool (NeoEAT)3-5 or Pediatric Eating Assessment Tool (PediEAT); 6 these items are written in parent-friendly language and had previously undergone extensive content validity testing with parents to ensure that they were understood as intended.7,8 New items were added using similar parent-friendly language, as needed, to comprehensively measure all concepts identified.
Setting
Data were collected via online surveys on both the Qualtrics (Provo, UT) and Research Electronic Data Capture (REDCap) platforms hosted at Boston College.9,10 REDCap requires the inclusion of the following statement: REDCap is a secure, web-based software platform designed to support data capture for research studies, providing (1) an intuitive interface for validated data capture; (2) audit trails for tracking data manipulation and export procedures; (3) automatic export procedures for seamless data downloads to common statistical packages; and (4) procedures for data integration and interoperability with external sources.
Sample
To obtain a large, diverse sample of children, participants were recruited from multiple sources, including Qualtrics panels, online communities of parents of young children, and a pediatric dental office in the northeastern United States that sees infants and toddlers for revision of ankyloglossia. To be included in this data analysis, the parent or primary caregiver (hereafter referred to as parent) had to be at least 18 years old, self-identify as being proficient in English, have access to the internet to complete the survey, and have a child under 2 years of age. Each parent was asked to only report on 1 child. Parents completed a single online survey that included questions about the parent, the child’s family and medical history, and the GIGER. We asked specifically if the child had received a formal diagnosis of GER or constipation by a medical provider. A subsample was also invited to complete the Infant Gastrointestinal Symptoms Questionnaire (IGSQ) 11 and Infant Gastroesophageal Reflux Questionnaire-Revised (I-GERQ-R). 12
Sample Size
The recommended sample size for factor analysis is at least 5 participants per item. 13 With 39 items on the initial GIGER, the required sample size was at least 195 participants.
Measures for Convergent Validity
Infant gastrointestinal symptoms questionnaire (IGSQ)
The IGSQ is a 13-item parent-report measure of digestion and elimination behaviors. 11 Each item is scored on a 5-point scale, with higher scores indicating more symptoms. A total score is calculated as a sum of all responses, with a possible range of scores from 13 to 65. The IGSQ has evidence of acceptable internal consistency reliability, test-retest reliability, and known-groups validity. 11
Infant gastroesophageal reflux questionnaire-revised (I-GERQ-R)
The I-GERQ-R is a 12-item parent-report measure of gastroesophageal reflux symptoms in the week prior. 12 A total score is calculated as a sum of all responses. Higher scores indicate greater symptom burden, and scores range from 0 to 42. The I-GERQ-R has evidence of acceptable internal consistency reliability, test-retest reliability, and known-groups validity. 12
Data Analysis
IBM SPSS Statistics 25 was used for all data analyses. Cases with >10% missing data for the GIGER were excluded from the overall analysis. Cases with >10% missing data on the IGSQ or I-GERQ-R were excluded pair-wise from the convergent validity analyses. A missing data analysis was conducted prior to all other statistical analyses. For all statistical tests, an alpha of .05 was used to define statistical significance.
Item analysis
Inter-item correlations were calculated using Pearson’s product-moment correlation. First, the correlation matrix was evaluated to identify item-item correlations ≥.8, indicating these items were measuring the same concept. If 2 items correlated at ≥.8, 1 item was removed. Next, items that failed to correlate with any other item at ≥.3 were removed because this indicated the item was measuring something unrelated to the other items.
Exploratory factor analysis
We conducted an exploratory factor analysis using principal components analysis with varimax rotation. We followed accepted guidelines for factor analyses of health-related instruments. 13 First, the Kaiser-Meyer-Olkin (KMO) statistic and Bartlett’s test of sphericity were evaluated to determine whether the sample size was adequate to proceed. Then, we examined the scree plot for the bend in the curve to determine a range of factor solutions that would provide a parsimonious, but comprehensive model. Exploratory factor analyses were conducted for each of the possible solutions by forcing the number of factors. We evaluated each possible factor solution by considering the variance explained by each additional factor, the number of items on each factor, and conceptual clarity of the factors.
After a factor solution was selected, item loadings were evaluated. If an item failed to load on any factor at ≥.3, it was considered for removal. Items that loaded on more than 1 factor at ≥.3 were considered for movement to another factor based on conceptual fit. Factor names were assigned after all items were assigned to their final factor and were chosen to represent the theme of the items within the factor; items that loaded highly on the factor were weighted more heavily in the naming decisions. After factor names were assigned, the factors were then referred to as subscales.
Internal consistency reliability
Prior to evaluating internal consistency, item-total correlations were calculated for each item. If the item-total correlation was <.3, the item was considered for removal. 13 We used Cronbach’s alpha to evaluate the internal consistency reliability of the full scale and each subscale. A Cronbach’s alpha ≥.7 was considered acceptable. 14 Each item was evaluated to determine whether the Cronbach’s alpha would increase significantly if the item were to be removed. After all final decisions were made regarding item removal, Cronbach’s alpha were again calculated for each subscale and the full scale.
Convergent validity
To evaluate convergent validity, bivariate correlations were calculated using Pearson’s product moment (r, 2-tailed) between the GIGER subscales and total score and the IGSQ total score and I-GERQ-R total score.
Known-groups validity
To evaluate known-groups validity, independent samples t-test was used to determine whether the GIGER subscales and total score were different between (1) infants with a medical diagnosis of GER and infants without a diagnosis of GER and (2) infants with a medical diagnosis of constipation and infants without a diagnosis of constipation. For the diagnoses of GER and constipation, parents were asked to report “yes” or “no” to questions about whether a healthcare provider had diagnosed their child with gastroesophageal reflux or constipation.
Results
Item Development
After removal of duplicates, the literature search identified 572 unique articles. Of these, 38 articles were reviewed in full-text. An additional 5 articles were identified through review of reference lists and included in the full-text review. There were 23 articles that met inclusion/exclusion criteria. From the 23 articles, the following parent-report measures of GI and/or GER symptoms were identified: GERD Symptom Questionnaires for young children (GSQ-YC) and infants (GSQ-I); 15 Infant Gastroesophageal Reflux Questionnaire (I-GERQ) and its modified versions, including the Infant Gastroesophageal Reflux Questionnaire-Revised (I-GERQ-R);12,16-21 Infant Gastrointestinal Symptoms Questionnaire (IGSQ); 11 and the Questionnaire on Pediatric Gastrointestinal Symptoms (QPGS) Form B. 22 The Total GERD Symptom and Severity Scale was stated to be used by Haddad and colleagues 23 in children ages 1 to 11, but the tool they referenced was the GERD Activity Index, 24 which had been developed for use in adults and had not been validated for use in children under 2 years old, so it was excluded. Psychometric properties and constructs measured by these tools are provided on Table 1. None of these measures met the requirements of both comprehensively assessing GI and GER symptoms across the first 2 years of life and having adequate psychometric properties.
Currently-Available Parent-Report Measures: Constructs Measured, and Psychometric Properties.

Abbreviation: GERD, gastroesophageal reflux disease.
From these measures, we identified 34 concepts related to GI and GER symptoms in children less than 2 years old. There were 25 concepts that mapped to items on the NeoEAT and 3 additional concepts that mapped to items on the PediEAT. Eleven new items were added to comprehensively measure all identified concepts, resulting in 39 items on the initial version of the GIGER. Response options for each item were on a 6-point Likert scale indicating frequency of the symptom from “Never” to “Always.” Items were scored such that higher scores indicated more problematic symptoms. Positively worded items were reverse-scored.
Sample
There were 391 participants with <10% missing data on the GIGER items. Missing data analyses revealed no missing data. Sample characteristics are provided on Tables 2 and 3. The majority of participants were from the United States (98%), with representation from nearly every state; there were 2 participants from Australia, 2 from the United Kingdom of Great Britain and Northern Ireland, and 1 each from Canada, Portugal, Singapore, and Sweden.
Child Age and Sex Distribution (N = 391).

There were 2 parents who preferred not to share their child’s exact date of birth, but the child was less than 7 months of age.
Characteristics of Participants and their Child (N = 391).
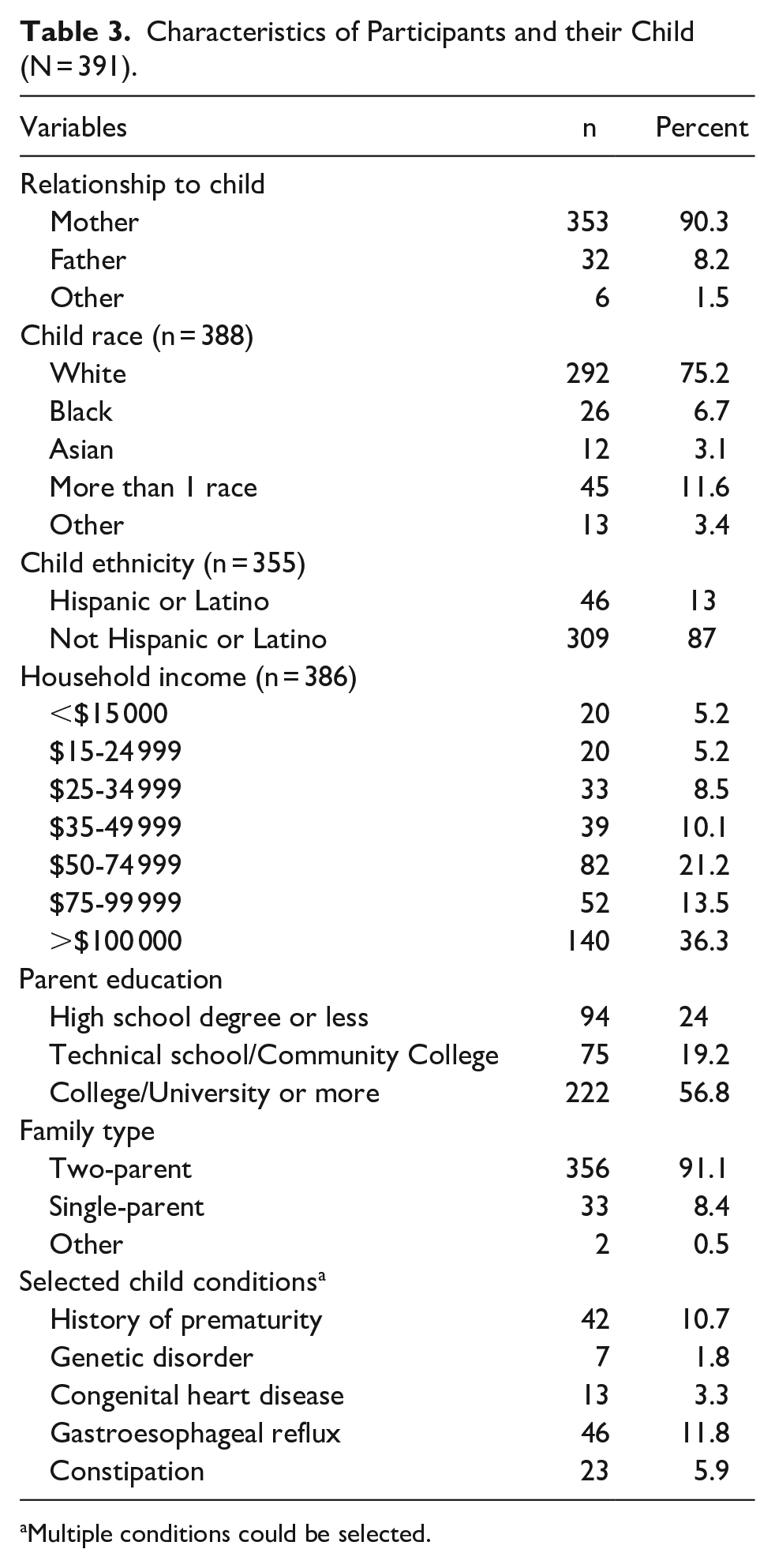
Multiple conditions could be selected.
Item Analysis
The items “My child throws up during feeding” and “My child throws up in between feedings” were found to correlate at .8. The item “My child throws up during feeding” was chosen for removal. The item “My child stools/poops at least once per day (24 hours)” was not correlated with any other item at ≥.3, so it was removed.
Factor Analysis
The initial factor analysis was run with 37 items. The sample was determined to be sufficient to proceed: Kaiser-Meyer-Olkin (KMO) = 0.93 and Bartlett’s test of sphericity was significant (P < .001). Upon examination of the scree plot, it was determined that either a 3 or 4 factor solution would be appropriate. The 3 factor solution explained 49.2% of the total variance, while the fourth factor would add 4.2% to the variance explained for a total of 53.3% variance explained. Both a 4 factor model and 3 factor model were forced. In the 4 factor model, the fourth factor only contained 2 items, which did not hold together conceptually. The 3 factor model was determined to be the most parsimonious and conceptually-sound solution. Item placement within each subscale and factor loadings are available on Table 4.
Factor Loadings for Principal Component Analysis with Varimax Rotation of the GIGER Scale for Infants and Toddlers.

Internal Consistency Reliability
Evaluation of item-total correlations revealed that the item “My child has hard stools/poop” was not at all correlated (r = 0); this item was removed, resulting in 36 items. Subscale 1 was named “Common GI and GER symptoms” and had 15 items (Cronbach’s α = .94) that described common symptoms that would become more concerning with increasing frequency. Subscale 2 was named “Compelling GI and GER symptoms” and had 13 items (Cronbach’s α = .86) that described uncommon, highly concerning symptoms if occurring with any frequency. Subscale 3 was named “Difficulties with Self-regulation” and had 8 items (Cronbach’s alpha = .78) that describe an infant’s ability to self-regulate, including eating, sleeping, and being consoled. All items on the “Difficulties with Self-regulation” subscale were positively worded and reverse-scored, such that higher scores indicate more difficulties with self-regulation. The Cronbach’s alpha for the full 36-item scale was .94. There were no items that would result in significant increase in Cronbach’s alpha if removed, so the final scale included 36 items.
Convergent Validity
The IGSQ total score was found to be significantly correlated with the GIGER total score (n = 19; r = .76, P < .001), subscale 1 Common GI and GER symptoms (r = .52, P = .02), subscale 2 Compelling GI and GER symptoms (r = .73, P < .001), and subscale 3 Difficulties with Self-regulation (r = .47, P = .04). The I-GERQ-R total score was also found to be significantly correlated with the GIGER total score (n = 44; r = .76, P < .001), subscale 1 Common GI and GER symptoms (r = .78, P < .001), subscale 2 Compelling GI and GER symptoms (r = .43, P < .01), and subscale 3 Difficulties with Self-regulation (r = .48, P < .01).
Known-Groups Validity
Infants with a medical diagnosis of GER (n = 46) had higher scores on the GIGER total score (M = 53.8 ± 24.3) than infants without a diagnosis of GER (n = 345; M = 36.2 ± 23.7; P < .001). Infants with a medical diagnosis of GER also had higher scores on subscale 1 Common GI and GER symptoms (M = 28.2 ± 15.6 vs M = 17.4 ± 13.6; P < .001) and subscale 3 Difficulties with Self-regulation (M = 16.8 ± 7.7 vs M = 11.8 ± 6.8; P < .001). The difference between infants with and without GER was not statistically significant for subscale 2 Compelling GI and GER symptoms (M = 8.8 ± 5.3 vs M = 7 ± 7.5, P = .1).
Infants with a medical diagnosis of constipation (n = 23) had higher scores on the GIGER total score (M = 39.3 ± 21.8) than infants without a diagnosis of constipation (n = 245; M = 29.7 ± 20.9; P = .04). Infants with constipation also had higher scores on subscale 3 Difficulties with Self-regulation (M = 12.7 ± 6.7 vs M = 9.5 ± 6.1; P = .02). The difference between infants with and without constipation was not statistically significant for subscale 1 Common GI and GER symptoms (M = 17.8 ± 12.2 vs M = 13.3 ± 10.6; P = .05) or subscale 2 Compelling GI and GER symptoms (M = 8.7 ± 5.9 vs M = 6.9 ± 7.8; P = .28).
Discussion
While there are several existing parent-report GI-related measures for young children, there is currently no measure written in parent-friendly language, that comprehensively assesses symptoms along the entire GI tract, and has adequate evidence of psychometric properties. Evidence of adequate psychometric properties ensures that an assessment measures what it intends to measure and does so in a consistent manner. We developed the GIGER Scale for infants and toddlers to complement clinician assessment of GI symptoms in children under 2 years old who unable to communicate well. We also developed the GIGER to provide researchers with a parent-reported outcome measure for research related to GI symptoms in infants and toddlers that has adequate psychometric properties. We developed items by identifying the range of symptoms described in the literature related to GI and GER symptoms and tested it with a sample of 391 parents of children under 2 years old. A factor analysis identified a 3 factor structure, with acceptable internal consistency reliability, convergent validity with the IGSQ and I-GERQ-R, and known-groups validity. Future testing of the GIGER should include evaluation of test-retest reliability, inter-rater reliability between parents, and should seek further testing in more diverse samples of children. Reference values for specific ages that represent the differences in development across the first 2 years of life, with a large, diverse sample, will be critical to guide determination of symptoms that fall outside of the range of typical for a child’s age for research and clinical practice.
Conclusion
The GIGER scale for infants and toddlers is a 36-item parent-report measure of GI and GER symptoms in children under 2 years old with adequate psychometric properties. The GIGER scale is written in parent-friendly language and can be used for both clinical care and research to improve the care of infants and toddlers. The GIGER may be helpful in identifying young children with symptoms that warrant further evaluation and management as well as measuring response to treatment.
Footnotes
Acknowledgements
The authors would like to acknowledge the families who participated as well as the healthcare providers at the study sites who facilitated this research.
Author Contributions
BFP: contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
CR: contributed to acquisition, analysis, and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
RRH: contributed to acquisition, analysis, and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Britt Pados owns Pediatric Feeding Analytics, LLC, a small private feeding consultation practice. Christine Repsha and Rebecca Hill declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Boston College Vice Provost for Research through an Ignite Grant and Faculty Research Funds, and by the Massachusetts General Hospital Institute for Health Professions School of Nursing Seed Grant.
