Abstract
Amino acid-based formulas (AAF) contain free amino acids as their protein source and are indicated for certain gastrointestinal and medical conditions. When switching formulas, monitoring tolerance helps assure adequate nutritional management. A cross-sectional retrospective study was conducted using U.S. insurance claims data from 402 children switched from one AAF (Formula1) to a different AAF (Formula2) between June 2021 and April 2023 during the nationwide formula shortage. Database was analyzed for age, sex, comorbidities, clinical symptoms, and healthcare encounters. Mean age was 5.3 ± 4.7 years (60% male). Significantly fewer children experienced abdominal distention, diarrhea, flatulence, nausea/vomiting, and allergy symptoms 1-, 3-, 6-months post-switch to Formula2 (P < .05). Significant reductions for total mean adjusted costs, emergency department, inpatient, outpatient, urgent care, and telemedicine visits were observed 1-, 3-, and 6-months post-switch (P < .001). Children in post-acute care settings who transitioned to an alternate AAF (Formula2) experienced significantly improved clinical outcomes and reduced healthcare costs.
Keywords
Introduction
Amino acid-based formulas (AAF) are hypoallergenic formulas, made from a protein composition of free amino acids. AAF play a significant role as a sole source of nutrition in the dietary management of children with cow’s milk protein allergy (CMPA), multiple food allergies, a history of anaphylaxis, eosinophilic gastrointestinal diseases, food protein induced enterocolitis syndrome 1 (FPIES), intestinal failure, 2 short bowel syndrome 3 (SBS), and other gastrointestinal disorders.
Many AAF in the U.S. have varying percents of fat as medium chain triglycerides (MCTs). AAF with MCT are recommended for children with fat malabsorption or maldigestion, or SBS where there may be decreased digestive and/or absorptive capacity. 3 MCTs do not require traditional fat metabolism and are more easily absorbed directly into the enterocyte and are transported through the portal vein to the liver.3,4
Breastfeeding is the optimal source of nutrition for all infants. In infants with CMPA who are breastfed, maternal dietary exclusion of cow’s milk protein is recommended to improve symptoms in the infant. Having CMPA does not necessitate cessation of breastfeeding. However, some infants, with or without CMPA are not exclusively breastfed for a variety of reasons. In the case of the non-breastfed infant, most treatment guidelines recommend AAF as first-line nutritional management in non-breastfed infants with severe CMPA symptoms, such as anaphylaxis, and severe faltering growth.2,5 -8 AAF is also recommended as a first therapeutic option for multiple food allergies, FPIES, and to control inflammation in patients with eosinophilic gastroenteritis and eosinophilic esophagitis (EoE) who do not respond to food-elimination diets. 2
Families who must purchase AAF for their children can experience a financial burden due to the cost of AAF which is greater than the cost of a standard formula. Within the category, however, pricing is similar for AAFs. Conditions that require an amino acid-based formula can be costly to manage, resulting in significant financial burden on families and the healthcare system. Researchers estimate that 32 million Americans have food allergies, including 5.6 million children under the age of 18, and approximately 9500 hospital discharges per year with a diagnosis related to food allergy has been shown.9,10 Direct medical costs were $4.3 (95% CI, $2.8-$6.3) billion annually, including clinician visits, emergency department visits, and hospitalizations. 9 Likewise, annual direct costs of eosinophilic GI disorders, such as EoE, in the United States ranged from $503 million to $1.36 billion in a systematic review that evaluated health-related quality of life and costs associated with EoE. 11 In addition, in children who have SBS, care is resource-intensive, often beginning at the time of birth or shortly thereafter and requiring prolonged hospital admission and multiple surgical procedures within the first year of life along with specialized nutritional support from AAF. Total cost of care per child with SBS in the U.S. over a 5-year period was estimated in a retrospective review to be nearly $1.6 million. 12 Therefore the cost of care for children who require an amino acid-based formula can be significant.
Many infants and children in the U.S. rely on specialty formulas, like AAF, for all or some of their nutrition. Formula changes happen for many reasons, including price, medical indications, palatability, tolerability, brand preferences, or even availability. The nationwide powdered formula shortage in 2022 significantly affected formula availability and resulted in necessary formula switches for infants and children using specialty AAF. The shortage of formula in the United States can be attributed to a combination of factors, including industry-wide supply chain issues following the COVID-19 pandemic and a significant recall of Abbott’s powdered formulas. During the pandemic the formula industry for infants and children faced supply challenges like many other industries including ingredient and packaging shortages. Then in early 2022, Abbott Nutrition, one of the largest manufacturers of formula in the U.S., issued a recall of several powdered formula products due to contamination concerns. This recall removed a significant portion of the formula from the market and intensified the existing shortages, as Abbott’s products represented a substantial share of the market and ultimately resulted in a nationwide formula shortage.
During the unprecedented formula shortage, the American Academy of Pediatrics (AAP) released recommendations to help parents and caregivers feed their children, which included switching formula brands if the formula was the same type. 13 Reports of parents who employed switching formulas during the shortage range from 13.6% 14 up to 87%. 15 Healthcare providers reported that caregivers gave a new formula to 99% of their patients. 16 Switching formulas may have a potential impact on tolerance and other clinical outcomes and may lead to increased healthcare resource utilization (HCRU) and economic impact. Data are limited regarding documented clinical outcomes after changing formulas within the same class (eg, AAF). The objective of this study was to evaluate impact of switching AAF on clinical outcomes including gastrointestinal (GI) intolerance, allergy symptoms, and health economic measures during the 2022 U.S. formula shortage. In addition, demographic and clinical characteristics, and medications for patients receiving these formulas will be described.
Methods
A retrospective study was conducted using nationally representative U.S. claims data obtained from the Decision Resources Group Real World Evidence Data Repository (Clarivate), which covers 98% of U.S. health plans, including medical and pharmacy claims. Medical claims and prescription claims were used to analyze longitudinal patient-level data. Inclusion criteria were patients from birth to 18 years, in a post-acute care setting, not receiving palliative or end-of-life care, with a history of formula usage with a specific AAF followed by at least 7 days of formula usage after switching to another AAF.
Patient characteristics (sex, age, race/ethnicity), comorbidities, and clinical outcomes (GI intolerance, allergy symptoms) were assessed in children ≤18 years with a history of receiving EleCare® Infant or EleCare® Jr formulas (Formula 1, Abbott Nutrition, US) and having switched specifically to Alfamino® Infant or Alfamino® Junior formulas (Formula 2, Nestlé HealthCare Nutrition, US; Table 1) in post-acute care settings between June 2021 and April 2023. The US formulations of these products did not contain fiber or human milk oligosaccharides. The primary difference between the formulas is the fat composition. Alfamino® formulas have a higher MCT content than the comparator Elecare® formulas, as well as other AAF brands. By comparing the switch from the recalled Elecare® specifically to Alfamino® formulas, it was hypothesized that if clinical differences observed after the switch from the recalled Elecare® formula, they would be most evident with Alfamino® formulas due to the unique nutrient profile with high MCT. There was no comparator group in this study as the focus of this study was observations of individuals switching to Alfamino® formulas after the recall of Elecare®. Because other AAF available at the time of the recall were similar in macronutrient amounts and sources to Elecare®, clinical differences due to nutrient profile would not be expected when switching; therefore switches from other AAF were not studied. This retrospective study intended to collect real-world evidence on what patients experienced during the unprecedented formula shortage and whether the switch to another AAF containing more MCT made a clinical difference.
Nutrition Information for Amino Acid-Based Formulas Used.

Source: Abbott Nutrition product guide as of December 2024, Nestlé Medical Hub as of December 2024.
Abbreviations: ARA, arachidonic acid; DHA, docosahexaenoic acid; MCT, medium chain triglycerides.
The index date was defined as the date when patients switched to Formula 2. The pre-switch period was defined as 6 months prior to the index date. Post-switch periods were 1-, 3-, and 6-months after the index date.
GI intolerance was assessed by reporting abdominal distention, abdominal pain, constipation, diarrhea, flatulence, gagging and retching, and nausea and vomiting. Allergy symptoms included reporting of anaphylaxis (reaction to milk or other foods), atopic dermatitis, eczema, hives, other allergic reactions, or rash based on ICD-10 codes. Although both atopic dermatitis and eczema have the ICD-10 L20 “parent” code, eczema was distinguished from atopic dermatitis by specific “child” codes (eczema: flexural eczema, infantile eczema, intrinsic eczema; atopic dermatitis: atopic dermatitis, Besnier’s prurigo, other atopic dermatitis, atopic neurodermatitis, other atopic dermatitis, unspecified atopic dermatitis).
Medications of interest were categorized as either GI or antiallergy medications. GI medications included antacids, antidiarrheal agents, antiemetic agents, H2 blockers, laxatives, pancrelipase, proton pump inhibitors, and others. Antiallergy medications included antihistamines, leukotriene inhibitors, steroids, and sympathomimetic agents. In addition, HCRU were reported as the mean number of healthcare facilities visits and each place of service (inpatient visits, outpatient visits, emergency department [ED], urgent care, and other places of services). Unadjusted and adjusted HCRU costs were reported for each place of service and as total costs of all places of service.
The pediatric comorbidity index (PCI) is a single numerical index tool that can be used in real-world data studies to summarize the underlying health status of children and to address confounding bias for risk adjustment in nonrandomized studies. 17 The pediatric comorbidity index provides a summary measure of disease burden and can be used for risk adjustment in epidemiologic studies of pediatric patients. It was designed as a baseline variable for comparison between groups rather than a measure of change after intervention. It was also used for risk adjustment in multivariate regression. Twenty-four comorbidities were included in the PCI, which predicts the 1-year risk of hospitalization. The score ranges from 0 to 29, with each point increase associated with 1.39 increased odds of hospitalization.17,18 Individuals with multiple comorbidities are represented in all applicable categories.
Patient demographics, clinical characteristics, comorbidities, GI intolerance, allergy symptoms, HCRU rates, and unadjusted costs were analyzed using descriptive statistics (median, mean, and standard deviation). Cumulative post-switch costs reflect the dollar amount recorded at certain time points, where costs at post-switch 6 months includes the costs from previous periods (1 and 3 months). Univariate analysis and paired t-tests were performed using Python, while descriptive statistics were analyzed in Microsoft© Excel. Appropriate statistical tests (paired t-test and chi-squared test) at the alpha ≤ .05 level of significance were used for between-group comparisons. Adjusted costs were calculated using a multivariate generalized linear model (GLM) adjusted for age, sex, and PCI in pre- and post-switch periods. Multivariate analysis was performed using R programing language.
Ethical Approval and Informed Consent
This records-based research study used existing commercially available secondary data that was de-identified and retrospectively analyzed. It was not considered human subjects research and was deemed exempt from Institutional Review Board oversight and informed consent under US Code of Federal Regulations 45 Part 46. The study followed Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Results
A total of 402 children aged 0 to 18 years from all U.S. regions met inclusion criteria and were found to have switched from Formula 1 to Formula 2 between June 2021 and April 2023 (mean [standard deviation [SD]] age 5.3 [4.7] years). Patient characteristics are summarized in Table 2. Most subjects were male (60%), 1 to 3 years old (n = 174, 43%), and from the South (n = 174, 43%). The majority of children (n = 249, 62%) had a commercial payer as opposed to Medicaid. Children may have had more than 1 comorbidity, with the most common comorbidities pre-switch identified as GI conditions (51%, n = 205), congenital conditions (49%, n = 198), and developmental delays (27%, n = 107). Based on medical history, 70% (n = 283) of subjects had diseases of the digestive system. 12% (n = 29) of all subjects reported CMPA. Nearly half of all children had a higher PCI score reflecting the medical complexity of the population. In 355 patients (88%) with ≥1 comorbidity, the mean (SD) PCI score was 4.8 ± 3.4.
Patient Characteristics (N = 402).

Significantly fewer children experienced at least 1 GI intolerance symptom at 1-, 3-, and 6-months post-switch to Formula 2, compared with pre-switch (P < .001; Figure 1). For any GI intolerance symptoms, 37% (n = 148) of subjects reported having at least 1 intolerance symptom pre-switch compared to 15% (n = 61) at 1-month post-switch (P < .001), 20% (n = 80) at 3 months post-switch (P < .01), and 23% (n = 94) at 6 months post-switch (P < .001). Significant reductions in the percentage of children reporting abdominal distention, diarrhea, flatulence, and nausea and vomiting were observed at all time-periods post-switch on Formula 2 compared with pre-switch (P < .05). Constipation was significantly reduced at 1- and 3-months post-switch compared to pre-switch (P < .05).

Percentage of children reporting Gl intolerance symptoms, pre- and post-switch.
At 1-, 3-, and 6-months post-switch, significantly fewer patients experienced at least 1 allergy symptom compared with pre-switch 20% (n = 82), 12% (n = 47) at 1-month, 13% (n = 54) at 3 months, 15% (n = 59) at 6 months post-switch, P < .05; Table 3). There were no differences in incidence of anaphylaxis, atopic dermatitis, or eczema. A significant reduction in the percentage of children reporting hives (P = .025) and other allergic reactions (P = .019) was observed at 1-month post-switch. There was no difference in the use of gastrointestinal or anti-allergy medications pre-switch while on Formula 1 compared to any time point post-switch to Formula 2.
Significant difference pre- versus post-switch shown in bold.
Chi-square test (pre- vs post-switch), alpha = .05 level of significance.
Significantly fewer patients required emergency department (ED), inpatient, urgent care (UC), telemedicine, or other services at 1-month post-switch compared with pre-switch (P < .05). A significant reduction was maintained for ED, inpatient, and telemedicine visits at 3 months post-switch (P < .001; Figure 2a). Mean total visits (P < .001), outpatient (P < .001), inpatient (P = .004), ED (P < .001) and UC (P = .014) visits per patient were significantly lower at 1-month post-switch. Significant reductions in mean total number of visits and outpatient visits were also observed up to 3 months post-switch (P < .001).
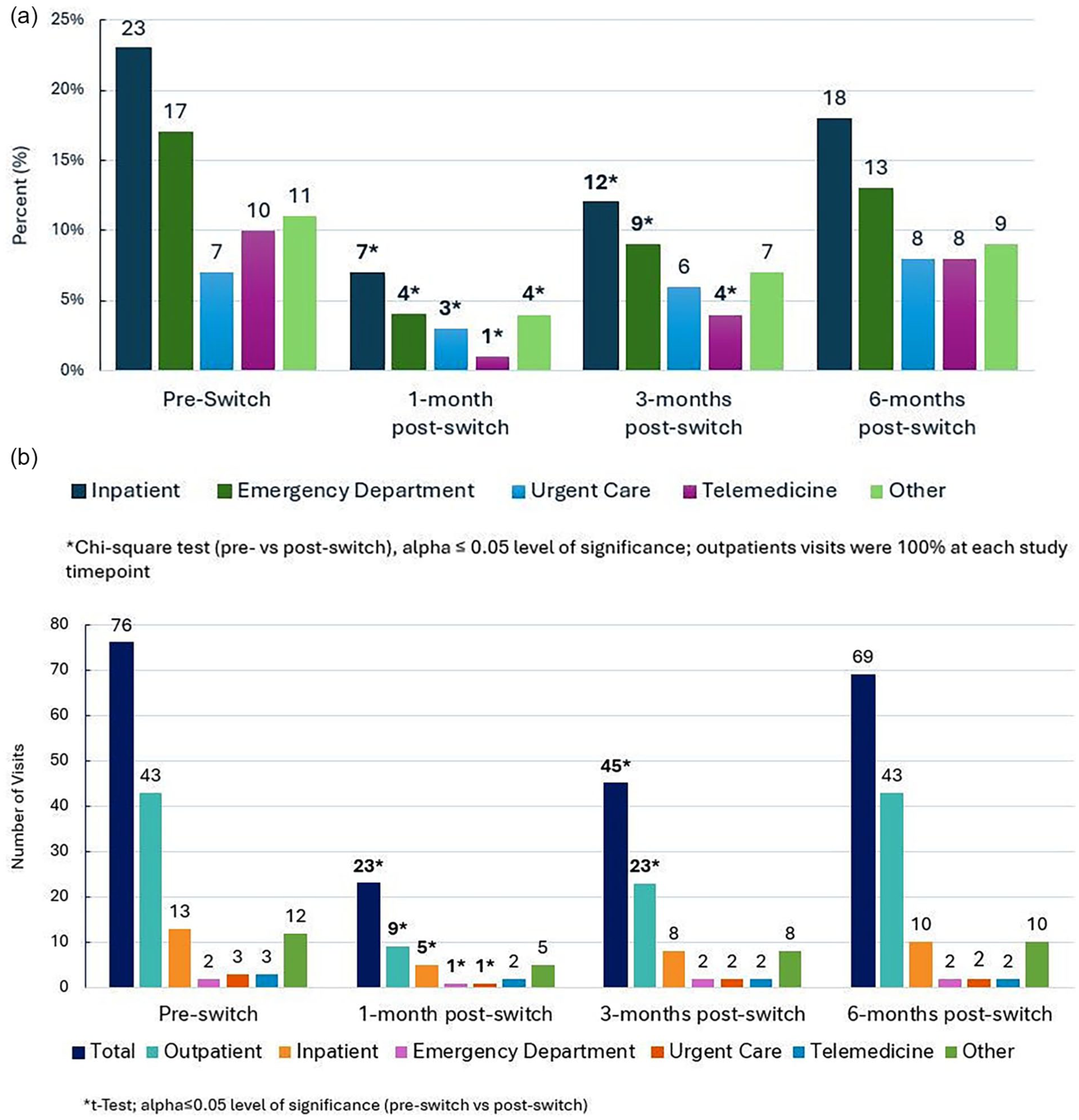
(a) Percentage of children requiring care by place of service. (b) Mean number of visits by place of service.
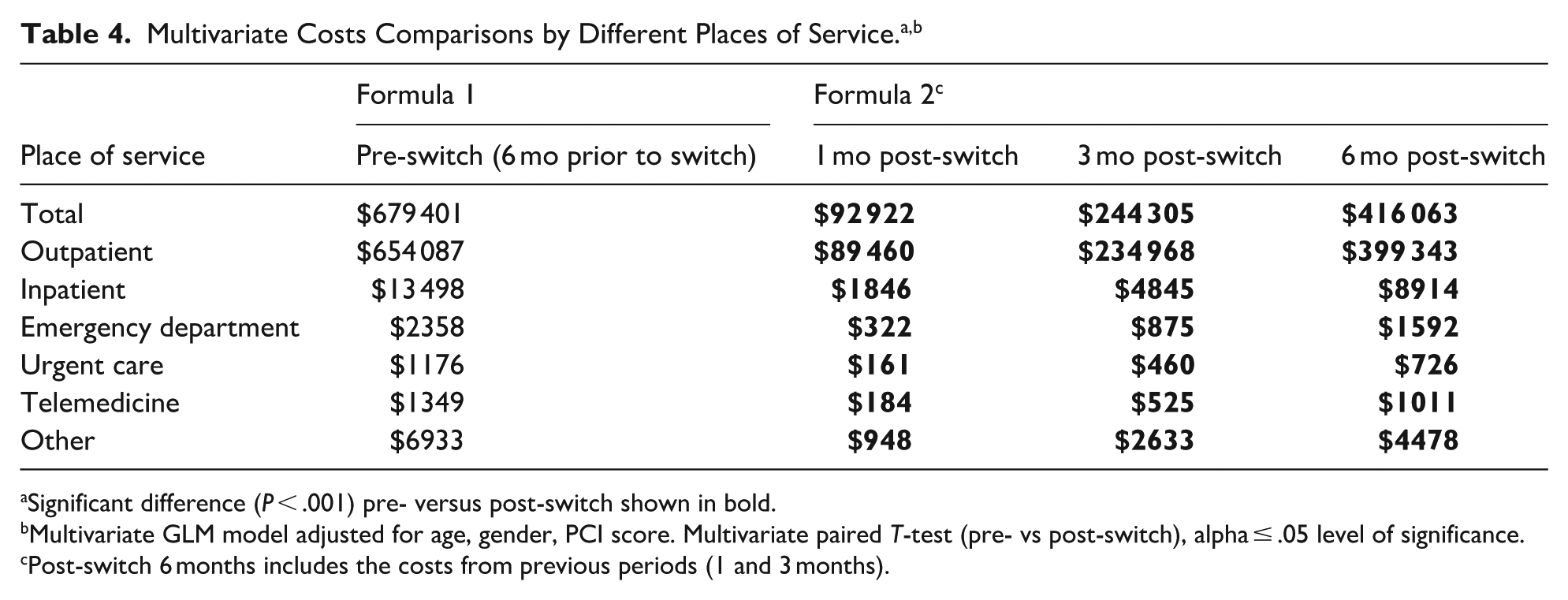
Significant reductions (P < .001) in costs were observed for emergency department, inpatient, outpatient, urgent care, telemedicine, and other visits at all time periods after the switch to Formula 2 (Table 4). Total mean adjusted costs significantly (P < .001) decreased from $679 401 to $92 923, $244 305, and $416 063 at 1-, 3-, and 6-months post-switch, respectively.
Significant difference (P < .001) pre- versus post-switch shown in bold.
Multivariate GLM model adjusted for age, gender, PCI score. Multivariate paired T-test (pre- vs post-switch), alpha ≤ .05 level of significance.
Post-switch 6 months includes the costs from previous periods (1 and 3 months).
Discussion
This retrospective study showed that switching AAF due to a nationwide infant formula recall in a population of infants and children in a post-acute care setting was well-tolerated and associated with improvements in clinical and health economic outcomes. The patients included in this study had various diagnoses covering conditions for which AAF are indicated such as digestive diseases (eg, intestinal malabsorption, short gut syndrome), and allergy (eg, CMPA, anaphylaxis). Regardless of whether the symptoms were allergy or GI-related, the switch from Formula 1 to Formula 2 did not result in a worsening of symptoms, and in fact, was associated with significantly reduced reporting of certain GI intolerance and allergy symptoms. In this study of medically complex children, significant reductions in GI and allergy symptoms were associated with significant reductions of total mean visits up to 3 months post-switch. Improvements in clinical and HCRU outcomes were associated with a significant reduction in costs up to 6 months post-index, for both adjusted (based on age, sex, PCI) and unadjusted cost values. These data reinforce success of the transition in this case, particularly in combination with the significant reduction in gastrointestinal and allergy symptoms. Data are limited documenting clinical and HCRU outcomes in children switching from 1 AAF to another. However, one study showed that when infants switched AAFs with different carbohydrate sources, a minimal improvement in tolerance was observed. 19
While there are multiple reasons that children may need to switch from one AAF to another, such as intolerance and palatability, the national formula shortage of 2022 undoubtedly had great implications, including an increased intent to breastfeed among pregnant women during this period. 20 However, among those parents who were actively using formula during the shortage, self-reported anxiety and depression were significantly increased. 21 From the healthcare provider perspective, it has been reported that there was an increase in malnutrition-related diagnoses like failure to thrive and deceleration in z-score and associated symptoms, likely resulting from feeding practices that are not generally recommended. 16
Data collected from a cross-sectional retrospective survey during the formula shortage, showed that 87% of children switched formulas (brand or type), and 21% of those surveyed reported their infants required a specialty formula due to a metabolic or medical condition, such as allergy. Overall, 61% of respondents reported their infants had one or more problems in response to switching, and that those requiring specialty formulas experienced more problems than those who did not, with vomiting driving the significant difference. 15 Unlike the present study, the survey did not examine switching only within a category of formula and instead considered both formula brand and type of switch. The authors found an increased use of intact cow’s milk formula and the number of infants experiencing problems in response to a formula switch was higher in children requiring specialty formulas. AAF are considered specialty formulas often reserved for children with the most severe forms of nutrition intolerance, and the step from an AAF to a more general formula is likely to be poorly tolerated. It is possible that children requiring specialty formulas may have switched to a standard intact cow’s milk formula, soy formula or other plant-based formulas for which their underlying conditions may not have been indicated, resulting in an increase in reported problems which was not seen in our study where switches were limited to only 2 AAF product brands.
It is well-known that children can outgrow certain food allergies, including CMPA. 22 Time was accounted for partially by gathering data at exact intervals (1, 3, and 6 months), and in this way, we examined if time was a major factor in improvement of symptoms. While it is true that symptoms may improve over time for some of the underlying conditions studied, these improvements usually occur over a greater period of 6 months. 2 Cow’s milk protein allergy or intolerance resolves in 90% of children by the age of 6 years. Half of infants will have developed tolerance at age 1 year, and more than 75% will have resolution by 3 years of age. 2 Most infants that are started on AAF or breastfed by a mother on a cow’s milk free diet will need to remain on that diet for at least 6 months. At that point, the child may be challenged with cow’s milk, and if no reactions, milk can be introduced into the child’s diet. The most common diagnosis of our study population was digestive system diseases, including malabsorption. Cases of transient malabsorption would not necessitate use of an AAF, so it could be assumed that most cases of our study population were chronic digestive disorders which typically would not resolve within a month-time period. With the wide age range of the subjects, it is unlikely that improvements seen were due simply to recovery with time. Statistically, future studies with survival analysis including Cox proportional hazard model could be considered to account for time.
Tolerance and improvement of symptoms with AAF usually occur in 4 weeks or less, and improvement of symptoms/tolerance due to growth or weight gain usually require at least 6 months to occur. Most studies and clinical trials show tolerance to AAF and disappearance of allergic symptoms within 4 weeks or less of switching to an AAF.19,23 -25 These factors seem to indicate that patients did not have an adequate response to their original formula that was recalled, and that these patients benefited from the switch due to the persistence of residual symptoms and observed improvements in symptoms post-switch. The tolerance pattern of a hypoallergenic formula is thought to be determined by its residual allergenicity or maldigestion of its non-protein components.
The composition of both formulas used in this study are similar (Table 1), with protein from free amino acids, carbohydrate sources including corn syrup solids, similar osmolality, and a fat blend that contains MCT. That said, Formula 2 has a different carbohydrate blend and slightly different levels of micronutrients, such as potassium, which may have contributed to improvements in diarrhea, flatulence, and abdominal distention; additionally, the MCT content as a percent of total fat in Formula 2 (43% and 65%; infant and junior formulas respectively) is higher than that of Formula 1 (33% and 33%; infant and junior respectively) and it is possible that this may have improved GI symptoms, with the increased amount of MCT supporting absorption of fats in this population. MCT were first introduced in 1950 for treatment of lipid absorption disorders because of their shorter chain length compared to long chain triglycerides (LCT).4,26 Based on the structure of MCT compared to LCT, MCT are digested and absorbed differently. Because of their shorter chain length, MCT do not require bile salts nor pancreatic enzymes for their digestion and absorption; instead they are directly absorbed via the portal vein, and are therefore “easy to absorb.” This has been demonstrated in in vitro, 27 animal studies, 28 and has been the topic of several reviews.4,26,29
Specific to specialty formulas, studies have been conducted after the formula shortage looking at key factors parents were seeking when switching formulas. Parents who switched their child’s AAF were most interested in tolerability, assurance, safety, cost-effectiveness, worth, and brand trustworthiness. 30 This coincides well with pediatric healthcare providers practicing during the formula shortage who cited safety, efficacy, and tolerability among the top factors they considered as “extremely important” when switching to another AAF. 31 Formula 2 used in the post-switch period of this study was shown to be safe, well-tolerated, and demonstrated efficacy with improved symptoms and reduction in financial burden.
Strengths of this retrospective include the use of a database covering >98% of U.S. insurance which allowed the inclusion of 402 pediatric patients from all U.S. regions. To minimize the potential of a selection bias, multivariate analysis was performed controlling for age, gender, and PCI score.
As in other real world evidence studies, several biases were identified, and efforts were made to control for confounders. Patients selected for this study were truly representative across the US, and the database utilized is representative of 98% of US healthcare plans, eliminating selection bias, providing generalizable, and reliable results. The multivariate regression model accounted for age, sex, and PCI score, which are common confounders in epidemiological studies. We also acknowledge certain biases that were limited by the nature of the study and limitations of the database. Patient anthropometrics were not available, so growth-related outcomes were not analyzed. A survival analysis including Cox proportional hazard model could not be performed as patient follow-up for longer duration of time was limited.
Limitations of the study include those related to data source and clinical variables not available to perform additional analysis. Not all clinical data was available from the U.S. claims database that may affect the observed tolerance outcomes (eg, specific diagnoses, timing and method of severity of the GI symptoms, and availability of nutritional team support). Data was not available to determine the exact reason for the switch, only that the period occurred during the nationwide formula shortage. In terms of diagnoses, no standard definition of CMPA or malabsorption was utilized. Note that retrospective, claim-based analyses rely on accurate diagnostic and medical coding and any error in the data could affect the results. The data were analyzed for the entire study population, not separated for age groups, as the population under the age of 1 year and presumably using the Infant formulations was only 12% of the study population. Having the data broken down by age group within the 1 to 18 year-old subjects with meaningful sample sizes would be beneficial for clinicians to understand whether specific age groups experienced a larger benefit due to the switch. Since authors were employees or received direct or indirect payment from the study sponsor for this work, potential biases were minimized by presenting both significant and non-significant results. Finally, retrospective analyses can only highlight the connection between decreased GI symptoms and HCRU utilization in patients on AAFs but cannot draw causative conclusions.
Conclusion
This retrospective study conducted during the time of the nationwide formula shortage found that children in post-acute care settings successfully transitioned from one AAF (Formula 1) to another (Formula 2) with significant reduction in reported allergy symptoms and GI intolerance symptoms, including abdominal distention, constipation, diarrhea, flatulence, and nausea & vomiting up to 6 months post-formula switch. Additionally, significant reductions in medical visits and costs of care were observed in children who switched to Formula 2.
Footnotes
Acknowledgements
The authors would like to thank Dr. Krysmaru Araujo Torres for her thorough and critical review of the manuscript and our medical writer, Laura Czerkies at LC Consulting, for her valuable support in preparing the manuscript.
Ethical Considerations
This records-based research study used existing commercially available secondary data that was de-identified and retrospectively analyzed. It was not considered human subjects research and was deemed exempt from Institutional Review Board oversight and informed consent under US Code of Federal Regulations 45 Part 46. The study followed Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Author Contributions
P. Cekola, A. Desai, P. Kumar, and A. Klosterbuer equally contributed to the conception and design of the research; P. Cekola, J. Boccella, A. Desai, P. Kumar, S. Mondal, Y. Vikaykadam, C. Samal and A. Klosterbuer contributed to the acquisition and analysis of the data; P. Cekola, J. Boccella, A. Desai, P. Kumar, S. Mondal, Y. Vikaykadam and A. Klosterbuer contributed to the interpretation of the data; and P. Cekola, J. Boccella, A. Desai, A. Klosterbuer and KH Thakkar drafted the manuscript. All authors critically revised the manuscript, agree to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Nestlé Health Science.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KHT is employed by Memorial Herman Medical Group, Sugar Land, TX, and provided consulting services to Nestlé Health Science on this project. PK, SM, and YV are employees of Clarivate Data Analytics, Bangalore, Karnataka, India, which was contracted by Nestlé Health Science for this project. PC, JB, AD and AK are employees of Nestlé Health Science.
Data Availability Statement
All relevant study data has been included in the article. Data is not publicly available as data is part of a larger data set (the claims database) that continues to have updates. Individual requests for data will be considered directly from the corresponding author.