Abstract
Background. Extra-uterine transition difficulties including BP abnormalities have been documented following perinatal asphyxia. Aim. The study aimed to determine the BP changes in the early neonatal transitioning period. Method. This comparative, cross-sectional study was conducted at the Ekiti State University Teaching Hospital, Ado-Ekiti, Nigeria. Result. A total of 246 term babies were analyzed. Their mean (standard deviation) systolic, diastolic and mean arterial blood pressures (SBP, DBP, and MBP) at birth were 68.2 (9.0), 39.9 (7.0), and 49.4 (6.9) mmHg and 67.4 (7.9), 37.1 (6.1), and 47.2 (5.8) mmHg, respectively. Asphyxiated neonates had significantly higher initial DBP (P < .001) and MAP (P = .008), lower SBP (P < .001) and MAP (P = .024) at 6 hours and lower SBP at 12 hours after birth, statistically. Conclusion. This study showed that while the BP of healthy neonates steadily increases in the first 48 hours after birth, it fluctuated markedly within the first 12 hours of birth among asphyxiated babies.
Introduction
At birth, the mechanical expansion of the lungs and removal of the placenta results in a reduction in pulmonary vascular resistance, increased systemic vascular resistance, reversal of ductus arteriosus shunts, and subsequent closure.1,2 The increased volume of pulmonary blood flow returning to the left atrium increases left atrial volume and pressure sufficiently to close the foramen ovale functionally, although the foramen may remain probe patent. The removal of the placenta also leads to the closure of ductus venosus, 1 all these changes lead to a gradual rise in systemic BP and a fall in pulmonary BP. Abnormalities in any of the above processes may delay the transition from fetal to neonatal circulation and also affect the BP of a neonate. In neonates with perinatal asphyxia, the cardiovascular physiologic response following perinatal asphyxia is to protect the vital organs such as the brain, the heart, and adrenals. 3 This is achieved by increased catecholamine secretion, peripheral vasoconstriction, and vasodilation of the vessels of the heart, brain, and adrenals.3,4 The BP is either maintained fairly constant or mildly raised at the early phase. 4 With prolonged perinatal asphyxia and persistent secondary apnea, there is usually attendant bradycardia, acidosis, and ischemia, these result in ischemic injury to the heart muscles, ventricular dysfunctions, reduced stroke volume, and cardiac output, and eventual hypotension if there is no prompt and adequate intervention. 5
What constitutes normal BP in neonates is unknown as there is no universally acceptable cut-off. Attempts at describing the normal BP pattern in neonates have been made in different regions of the world,6-9 one of such attempts is a nomogram that describes the normal pattern of BP expected in neonates of various gestational ages designed by Dionne et al 6 One of the shortfalls in the development of this nomogram includes the fact that studies involving measurement of BP in sick neonates were included.
In Nigeria, there is a paucity of data on BP patterns in neonates. Sadoh and Ibhanesebhor 7 in 2009 took 473 readings of BP among Nigerian neonates and reported the mean (standard deviation) on day 1 to be 66.8 (7.7), 38.5 (6.3), and 47.9 (6.3) mmHg for the systolic, the diastolic, and the mean arterial BP readings respectively.
Nwokoye et al 10 in a study done in Enugu in 2015 reported a relatively lower mean (standard deviation) of the systolic, diastolic, and mean arterial BP of 63.3 (5.5) mmHg, 36.8 (5.3) mmHg, and 46.4 (5.2) mmHg. The authors’ adduced reason for the lower BP values compared to Sadoh and Ibhanesebor’s findings was because large-for-gestational-age and small-for-gestational-age babies were excluded from their study. These two Nigerian studies did not set out to determine the trend or BP changes as they checked BP just once per day. More frequent BP checks in the early hours after birth will most likely give a better understanding of the trend and changes in BP as newborns transit to extra-uterine life. This study provides data on BP changes in the early hours after birth among asphyxiated babies as no such previous data was found among asphyxiated neonates in Nigeria.
Methods and Materials
Design
This study design was a hospital-based, comparative, cross-sectional design.
Study Location
This study was conducted at the Special Care Baby Unit (SCBU) and post-natal ward of Ekiti State University Teaching Hospital (EKSUTH), a tertiary hospital in Ado-Ekiti, the capital city of Ekiti State, South-west Nigeria.
Study Population
The subjects of this study were term babies with perinatal asphyxia admitted into the SCBU, EKSUTH and the controls were term apparently healthy neonates delivered in EKSUTH. Perinatal asphyxia was defined as an APGAR score of 6 or less at the fifth minute of life. For babies with perinatal asphyxia delivered outside EKSUTH, documented APGAR score at birth from referral center and/or failure to cry at birth requiring extensive resuscitation and abnormal neurologic findings on clinical examination were used.11,12
Sampling
Term neonates with perinatal asphyxia admitted to the SCBU who are within 2 hours of birth and term apparently healthy neonates in post-natal ward. Neonates with the following were excluded; presence of a murmur, obvious structural congenital anomaly, for example, encephalocele; birth injury such as fracture to the humerus and a significant disparity in blood pressure measurement from both arms.13,14 All consecutive term asphyxiated neonates admitted into the SCBU of EKSUTH who met the study criteria were recruited, the apparently healthy neonates were matched with the asphyxiated ones such that the weight difference was not more than 0.5 kg. 15
Data Collection
Asphyxiated babies were promptly resuscitated after birth (in-patients) or at presentation (for out-patients). Asphyxiated babies were recruited into the study after successful resuscitation.
Before the recruitment of the participants, the researcher obtained informed consent from the mother or primary caregiver, where the mother was not available. A screening examination focused on the cardiovascular system including blood pressure measurement in both arms, checking for murmurs, and bounding dorsalis pedis pulsation was performed for each potential participant. Where the baby was eventually recruited, the blood pressure readings taken during this screening examination were documented as the first measurement.
For recruited participants, relevant information obtained from the mothers/primary caregivers was filled into the appropriate section of the study proforma, a detailed examination including weighing, length measurements, temperature check, and examination of the respiratory and cardiovascular systems were performed, and findings recorded appropriately.
The blood pressure was measured using an oscillometric device; the Welch Allyn™ Propaq Encore 206EL ©, serial number DA108534. Blood pressure measurement was done with the babies laid quietly and calmly in the supine position. The appropriate cuff was chosen (with the length of the bladder covering 80%-100% of the arm circumference, and the width to arm circumference ratio of 0.45-0.55). 16 After the cuff had been wrapped around the mid-portion of the arm (half-way between acromion and olecranon process), babies were allowed to rest for 15 minutes following which 3 blood pressure measurements were taken within an hour at least 2 minutes apart. 16 The mean of the last 2 blood pressure measurements was used in the analysis because the first measurements are likely to be significantly higher than the rest. 16
Babies’ blood pressure was first measured within an hour of birth for babies delivered at EKSUTH and within 1 hour of admission for babies referred to EKSUTH, then at the 6th, 12th, 24th, 36th, and the 48th hour of life. If a baby was noticed to be unduly agitated as evidenced by crying or jitteriness during a blood pressure measurement, the reading was suspended and repeated after the baby had been calm for at least 5 minutes. The data collection lasted a 9 months period from March to November 2020.
Ethical Considerations
Formal approval of the Ethics and Research Committee of Ekiti State University Teaching Hospital with protocol number EKSUTH/ A67/2018/09/012 was obtained before the commencement of the study. Written informed consent was obtained from the parents or the primary caregiver (where parents were not available) of eligible neonates before recruitment into the study.
This study was at no cost to the participants. The researcher bore the cost of the transportation for babies who required follow up visits for blood pressure measurement.
Data analysis
The analysis was done using the software Statistical Package for the Social Sciences (SPSS) version 25.0. Means with their standard deviations (SD) were reported for continuous data that were normally distributed. The means of the blood pressure of the 2 groups were compared using independent sample t- test. The mean difference was compared for 24th hour BP and 48th hour BP in each group using dependent sample t-test. Results were presented in tables and figures. Analysis of variance was used to compare means of blood pressure across different classifications of birth weights
Statistical significance was established when the probability value (P) was less than .05 and a 95% confidence interval (CI) excluded unity.
Results
The study was conducted over 9 months; 246 participants met the requirements for analysis. The study participants were term neonates delivered between estimated gestational ages of 37 and 42 weeks with a mean estimated gestational age of 38.5 (1.2) weeks. There were more females 124 (50.4%) across both groups, more males 68 (55.3%) among the subjects and more females 69 (56%) among the controls.
The mean (SD) birth weight of the total study participants was 3.05 (0.66) kg. The male participants’ average birth weight of 3.10 (0.70) kg was slightly higher than females’ 3.00 (0.64) kg [P = .660]. The majority of the study participants (175; 71.1%) had normal weight, 49 (19.9%) were low birth weight (LBW); the others were macrocosmic. Also, (167; 67.9%) of the study participants had appropriate weights for gestational age, 40 (16.3%) were small for gestational age and 39 (15.9%) were large for gestational age (LGA).
The length of the participants ranged from 40.0 to 56.0 cm. The mean (SD) length of the babies was 47.9 (2.8) cm. The mean (SD) length of the males was 48.0 (3.1) cm while that of the females was 47.8 (2.5) cm (P > .05). There was no significant difference in the length between the controls and the subjects (P > .05).
Also, there was no significant disparity in the BP across the gender, weight groups and weight appropriateness for age at birth (Table 1). The participants’ temperature, respiratory rate and heart rate were classified as shown in Table 2, the mean (SD) of temperature is significantly lower among the controls 36.2 (0.6) versus 36.4 (0.3) among the subjects P = .002 and respiratory rate was significantly higher among the controls 68.9 (18.2) versus 55.3 (10.9) among the subjects P = .000. There subjects had higher mean (SD) heart rate 149.3 (16.1) compared to the controls 145.8 (12.1) P = .055.
Blood Pressure Disparity Across Gender and Weight Classifications.

Abbreviations: t*= independent test; LBW = low birth weight; AGA = appropriate for gestational age; t = analysis of variance; SGA = small for gestational age; LGA = large for gestational age.
Comparison of Clinical Parameters of the Study Participants.

Abbreviations: * = independent sample t-test value; N = total number; n = number of cases; + = Pearson chi square value; cpm = cycles per minute; bpm = beats per minute.
Across both groups, the means (SD) of the SBP, DBP, and MAP at birth were 67.8 (8.4), 38.5 (6.7), and 48.3 (6.4) mmHg respectively. It was 68.2 (9.0), 39.9 (7.0), and 49.4 (6.9) mmHg respectively at the admission of the subjects and 67.4 (7.9), 37.1 (6.1), and 47.2 (5.8) mmHg respectively at birth for the controls.
Among the controls, the mean (SD) of the SBP, DBP, and MAP increased with postnatal age from birth to the 48th hour. However, among the subjects, the mean (SD) of the SBP, DBP, and MAP showed an initial dip at the sixth hour, after which it gradually rose till the 48th hour. (See Tables 3-5 for details of the changes in BP means with postnatal age and Figures 1-3 for the patterns).
Comparison of the Means of Systolic BP of the Study Participants.
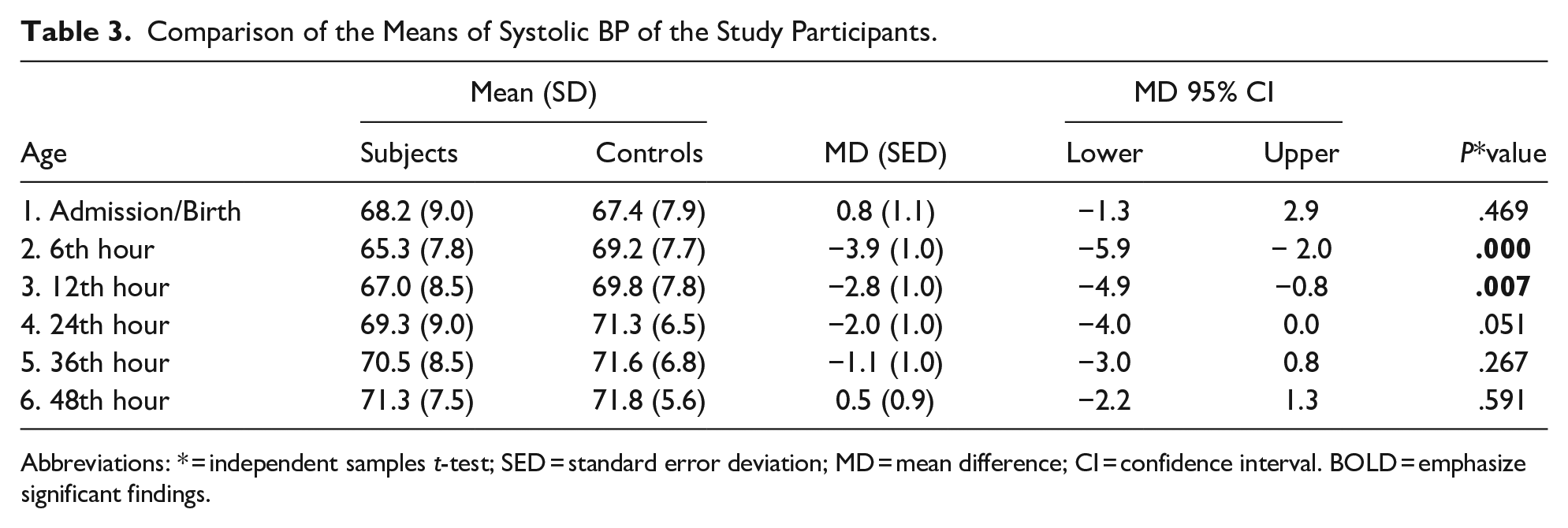
Abbreviations: * = independent samples t-test; SED = standard error deviation; MD = mean difference; CI = confidence interval. BOLD = emphasize significant findings.
Comparison of the Means of Diastolic BP of the Study Participants.

Abbreviations: * = independent samples t-test; SED = standard error deviation; MD = mean difference; CI = confidence interval. BOLD = emphasize significant findings.
Comparison of the Means of the Mean Arterial BP of the Study Participants.

Abbreviations: * = independent samples t-test; SED = standard error deviation; MD = mean difference. BOLD = emphasize significant findings.
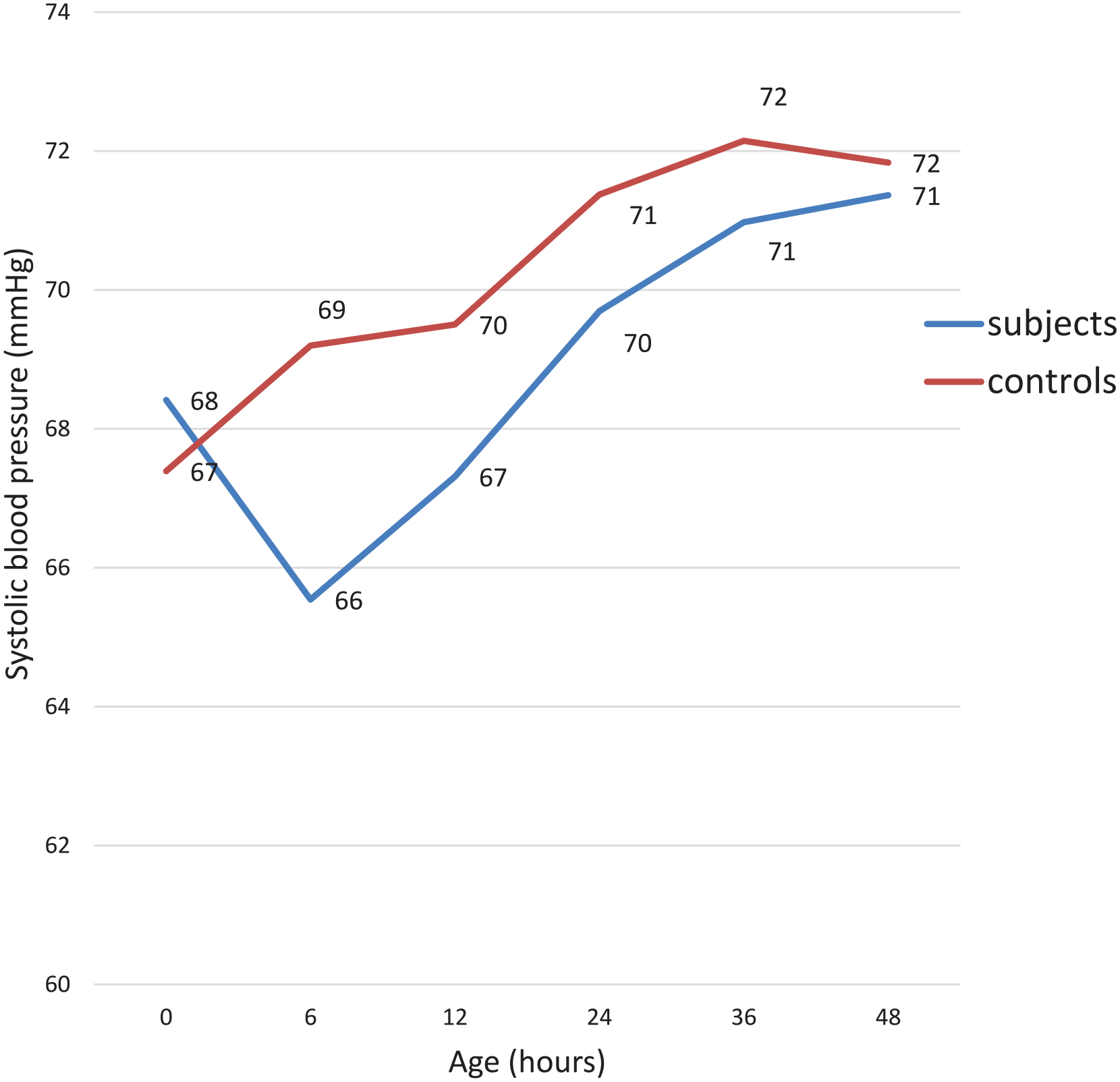
Variation of systolic BP of study participants with age.

Variation of diastolic BP of study participants with age.
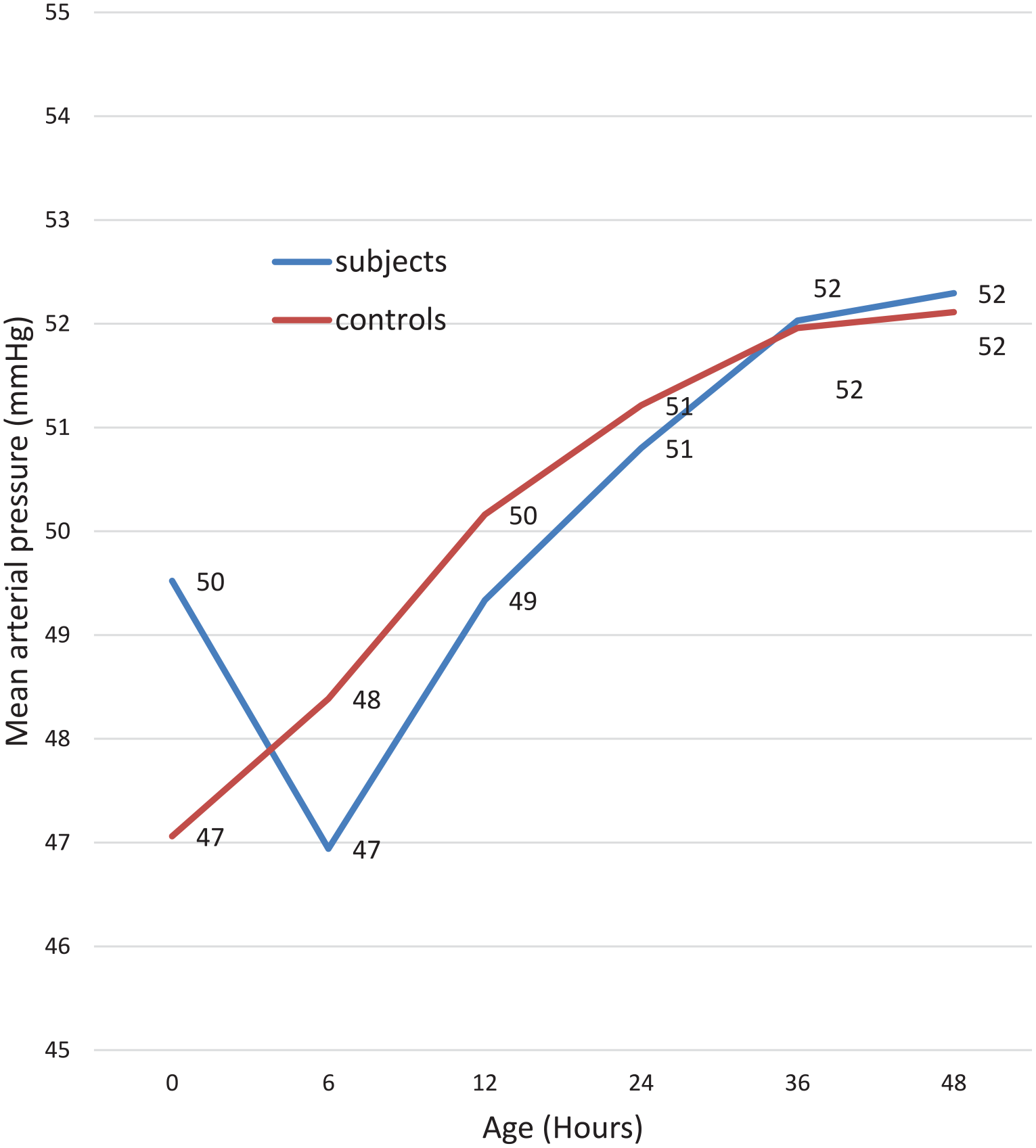
Variation of mean arterial BP of study participants with age.
Among the asphyxiated neonates and compared to the controls, the DBP and MAP were statistically significantly higher at birth (P < .001 and P = .008 respectively), the SBP and MAP were lower at the sixth hour (P < .001 and .024 respectively), and the SBP was lower at 12th hour. The details of the mean differences and P-values at 95% CI are provided in Tables 3 to 5. Among the controls, the rate of BP rise was higher on the first day while for the subjects; the rate of BP rise was higher on the second day (See Table 6 for details).
Comparison of the Rate of Rise of BP of the Study Participants on the First and Second Day.
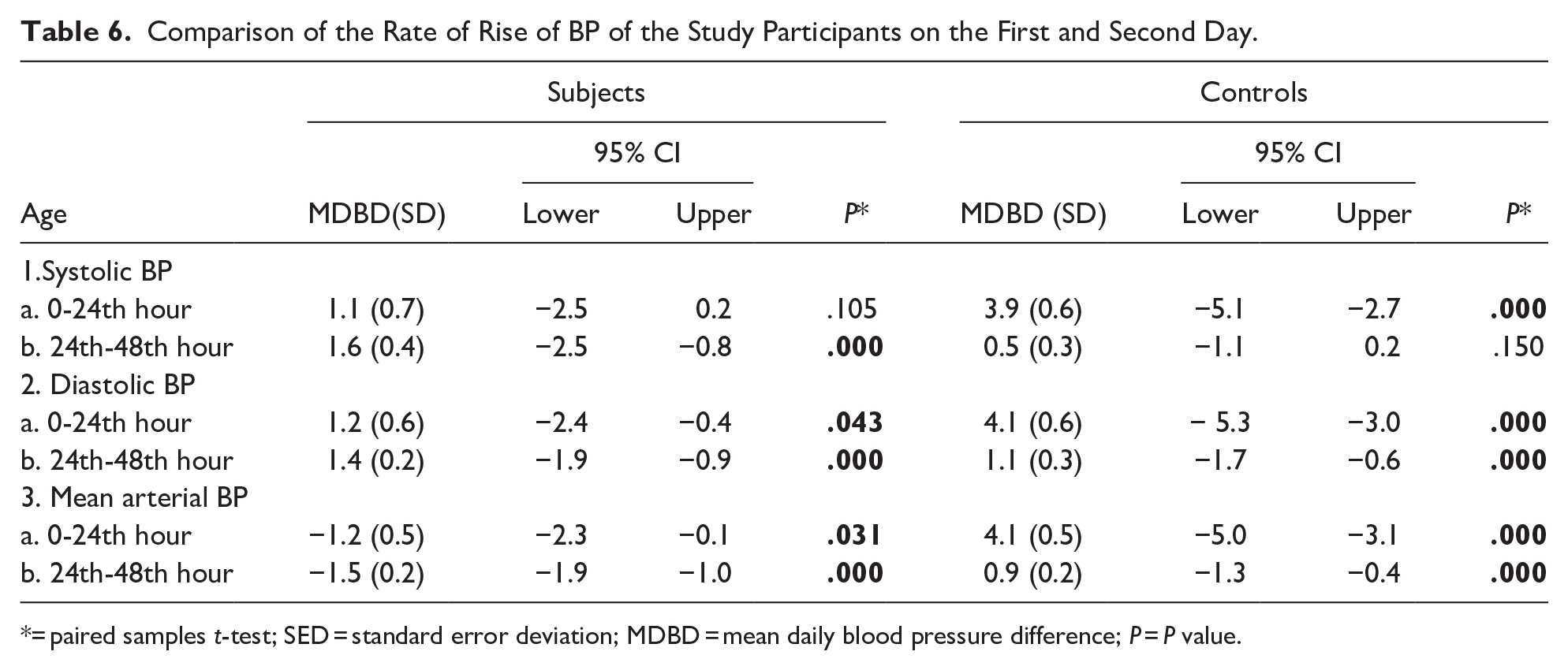
= paired samples t-test; SED = standard error deviation; MDBD = mean daily blood pressure difference; P = P value.
Discussion
This study observed significant differences in the early changes in blood pressure (BP) of asphyxiated neonates compared to the healthy controls.
Generally, the BP of the neonates was higher on the second day compared to the first day, this observation is similar to the findings in other studies in Nigeria and other countries. It is perhaps due to the rising systemic BP expected as the shunts closed and the placenta removed. The mean values of BP on day one and day two in this study are also similar to the mean BP readings reported by other studies7,10,17 The findings of these studies form a useful tool for interpretation of BP readings among hospitalized babies in neonatal intensive care units and special care baby units in Nigeria.
Among the asphyxiated neonates, the mean BP at admission was higher but later dipped significantly by the sixth hour after birth, followed by a slow rise till the 48th hour after birth. The higher BP at admission and the subsequent dip among the asphyxiated neonates can be partly explained by the early cardiovascular response to perinatal asphyxia that includes increased cardiac output, and peripheral vasoconstriction following catecholamine release all leading to increased BP. 3 This is an adaptive mechanism to maintain adequate cerebral blood flow. 3 If the asphyxia persists longer (more than 6 hours) without significant intervention, the cardiac output will eventually decrease, due to the prolonged hypoxia and ischemia of the cardiac muscles; the BP will also reduce with time and the injury to the brain and other tissues will worsen.4,5
The majority of the asphyxiated neonates were admitted within an hour of delivery and early intervention was instituted, however, there was no facility for the inducement of therapeutic hypothermia which has been touted to reduce the effects of hypoxic-ischemic injury in affected neonates. Following the dip in the BP noticed at the sixth hour, there was a steady rise of the BP in the second half of the first day and through the second day. The rate of rise of the BP was however higher on the second day after birth compared to the first day.
This is contrary to what was observed among the normal controls in whom the rate of rise of BP on the first day was more than 2 times higher than what happened on the second day. The finding of a progressive increase in BP from birth to the 48th hour with a higher rate of rise on the first day among the controls was consistent with reports of previous studies among healthy neonates in the USA by Zubrow et al, 18 in Australia by Kent et al 8 and Nigeria by Sadoh and Ibhanesebhor. 7
The disparity in the rate of rise of BP between the subjects and controls in this study may be explained by the difficult transition of the asphyxiated neonates from fetal to neonatal circulation and generalized depressed functions of body tissues especially the nervous and cardiovascular systems following perinatal asphyxia.
The asphyxiated neonates were observed to have a significantly lower temperature at birth compared to the healthy controls. Similar observations have been made by other authors.19-21 This finding may have contributed to the higher mean BP readings recorded at birth among the asphyxiated neonates as low temperature causes vasoconstriction, increased vascular resistance, and raise BP.22,23 It could also be explained as an adaptive mechanism to downregulate oxygen consumption and metabolic activities aimed at ameliorating the pathophysiological processes that follow cerebral insults; mild hypothermia attenuates blood-brain barrier damage, reduces the release of excitatory neurotransmitters, lessens free radical production, and causes increased IL-10 production.24,25
Conclusion
This study has documented the changes expected in BP of normal neonates in the early neonatal days; it, therefore, serves as a useful tool to determine the normal BP of neonates during this period. It also showed the remarkable fluctuation in the BP of asphyxiated neonates within the first 24 hours after birth, emphasizing the need for continuous monitoring during this period. This will help to detect any clinically significant change that may occur and institute appropriate intervention.
Limitation
This study did not do continuous/hourly BP measurements of the neonates in both groups, which may have provided a more robust outline of BP patterns in this age group as they adjust from intra- to extra-uterine life, especially within the first 6 hours of birth.
Footnotes
Author Contributions
All authors contributed to the preparation and design of this manuscript. ABT, JAO, EOO Conceptualised, drafted, and editted the manuscript. OAT analysed data and editted the manuscript . OAB and OAO drafted manuscript and collected data . All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
