Abstract
We report the case of a 2-month-old infant with incomplete Kawasaki disease that presented as an apparent urinary tract infection. The patient’s fever persisted despite antibiotic treatment. Intravenous immunoglobulin and aspirin therapy cured both the incomplete Kawasaki disease and bacterial pyuria. Renal sonography, voiding cystourethrography, and renal parenchyma radionuclide scanning did not detect any abnormalities. Temporary dilation of the coronary artery was noted. In a urine specimen obtained through transurethral catheterization, the growth of 105 colony-forming units/mL of extended-spectrum β-lactamase–producing Escherichia coli was detected. Polymerase chain reaction analysis revealed that the enzyme genotype was CTX-M-8, which is a rare type in Japan. In conclusion, attention should be paid to a misleading initial presentation of fever and pyuria, which might be interpreted as urinary tract infection in patients with Kawasaki disease. Furthermore, pediatricians should consider incomplete Kawasaki disease when patients present with fever and pyuria, which are consistent with urinary tract infection, but do not respond to antibiotic treatment.
Case Report
It is sometimes difficult to differentiate Kawasaki disease (KD) from urinary tract infection (UTI) in the early period of KD. In this article, we describe the case of a patient with KD initially presenting with fever and pyuria.
The patient was a previously healthy 2-month-old male Japanese infant who was the third child of healthy non-consanguineous parents. He was delivered at 39 weeks 5 days of gestation. His birth weight was 2989 g and body length was 51.0 cm. He suffered from a fever of 39°C and was taken to a local physician, who prescribed ampicillin because blood test revealed mild inflammatory reaction. However, fever continued until the next day, and the inflammatory reaction was exacerbated compared with the previous day. Hence, he was admitted to our hospital. On admission, his height was 60 cm (+0.95 SD); body weight, 5.1 kg (−0.59 SD); temperature, 38.6°C; heart rate, 190 beats/min; respiratory rate, 46 breaths/min; and blood pressure, 106/65 mm Hg. Physical examination findings were unremarkable except for a maculopapular rash on the chest, abdomen, and back.
Hematological values were as follows: a white blood cell count of 13 100/µL with 65.9% neutrophils, 25.3% lymphocytes, and 2.5% monocytes; hemoglobin of 9.7 g/dL; and platelet count of 42.4 × 104/µL. Blood chemistry and serologic findings showed levels of total protein of 6.1 g/dL; albumin, 4.2 g/dL; urea nitrogen, 6.0 mg/dL; creatinine, 0.21 mg/dL; aspartate aminotransferase, 45 U/L; alanine aminotransferase, 42 U/L; sodium, 134.9 mEq/L; potassium, 5.0 mEq/L; chloride, 105 mEq/L; and C-reactive protein, 4.44 mg/dL. Urine analysis revealed a 3+ test for leukocyte esterase and 1+ test for nitrite. Chest and abdominal X-ray findings were normal.
Hospital Course
On admission, the initial presentation of fever and pyuria was presumed to be UTI. Hence, he was initially treated with 100 mg/kg/day of cefotaxime. On day 2 of admission (3 days after the onset of fever), 2 g/kg of intravenous immunoglobulin (IVIG) and oral aspirin (30 mg/kg/day) were administered because the patient exhibited conjunctival injection and red lips, in addition to persistent fever and a rash on his trunk; these findings were suggestive of incomplete KD. The Kobayashi risk score for predicting IVIG resistance was 3, indicating a low risk. 1 The treatment for KD was effective, and antibiotic therapy was discontinued. However, 105 colony-forming units (cfu)/mL of extended-spectrum β-lactamase (ESBL)–producing Escherichia coli was isolated from a urine specimen obtained through transurethral catheterization on admission, and polymerase chain reaction (PCR) using specific primers for blaCTX-M detected an enzyme (CTX-M-8) that is rarely observed in Japan. Briefly, PCR was performed: 10 ng of template DNA, 50 pmol of forward and reverse primers, and 400 nmol of NTP were mixed in buffer (10 mM Tris-HCl, 50 mM KCl, 2.5 mM MgCl2; pH 8.3) to make a final volume of 25 µL; the mixture was then incubated at 95°C for 5 minutes, followed by 30 consecutive cycles of 95°C for 30 seconds, 60°C for 30 seconds, 72°C for 1 minute, and 72°C for 7 minutes for final extension. PCR primers to detect CTX-M gene were the following: forward: 5′-TTT GCG ATG TGC AGT ACC AGT AA-3′ and reverse: 5′-CTC CGC TGC CGG TTT TAT C-3′. To determine the sequences, PCR products (520 bp) were purified by 1.0% agarose gel electrophoresis. A portion of the purified DNA was used in DNA sequencing using the forward or reverse PCR primers. 2 Antimicrobial susceptibilities and ESBL production were determined according to the Clinical and Laboratory Standards Institute guidelines. 3 Susceptibility of the isolated pathogen to antimicrobial agents is shown in Table 1. Interestingly, it was susceptible for non-carbapenems such as cefmetazole, flomoxef, and fosfomycin. Negative bacteriologic cultures of the throat and blood were noted. On day 3 of admission, urine analysis revealed negative for leukocyte esterase and nitrite. Ten days past admission, voiding cystourethrography did not detect vesicoureteral reflux. Urine analysis revealed a 2+ test for leukocyte esterase, and negative for nitrite. A total of 105 cfu/mL of ESBL-producing E coli was asymptomatically isolated from a urine specimen obtained through transurethral catheterization. Renal parenchyma radionuclide scanning with technetium-99-m-DMSA (99Tc DMSA) showed no cold spots. At 2 weeks and 3 months after discharge, urine analyses revealed negative for leukocyte esterase and nitrite. Dilation of the coronary arteries was identified with echocardiography. Z scores were calculated according to the lambda-mu-sigma method. 4 The diameters of the left main tract and right coronary artery were 2.0 mm (Z score, +2.01) and 2.01 mm (Z score, +2.84), respectively, but there was no clot. The diameters of the left anterior descending tract and left circumflex tract were 1.38 mm (Z score, +0.78) and 1.15 mm (Z score, +0.33), respectively. At 1 month after the onset of KD, the left main tract and right coronary artery remained slightly dilated, but this finding resolved spontaneously after 3 months and was interpreted as a transient dilation.
Antibiotic Susceptibility of Isolated ESBL-Producing Escherichia coli Strain.
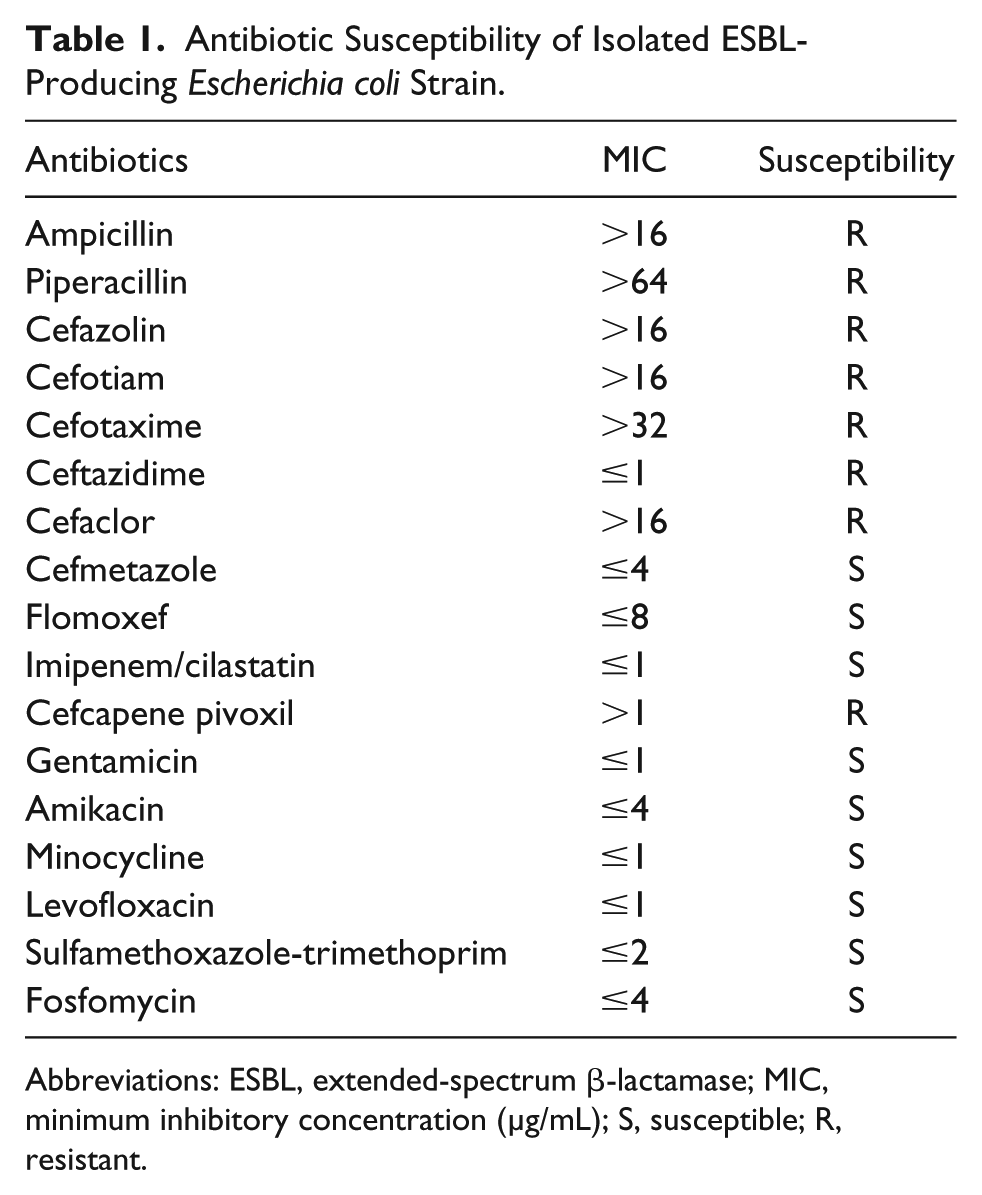
Abbreviations: ESBL, extended-spectrum β-lactamase; MIC, minimum inhibitory concentration (µg/mL); S, susceptible; R, resistant.
Final Diagnosis
Incomplete Kawasaki disease associated with urinary tract infection.
Discussion
Our patient had bacteriuria, which was consistent with UTI, and KD was subsequently detected. The organism isolated from his urine was ESBL-producing E coli, and it was resistant to cefotaxime in vitro and in vivo. Antibiotic therapy with cefotaxime had no effect on the fever, but IVIG was effective. These findings indicate that our patient concurrently developed KD and UTI due to ESBL-producing E coli.
Sterile pyuria is a common feature in KD patients, and it originates from the urethra, the kidney as a result of a subclinical renal injury, and/or the bladder due to cystitis. 5 By contrast, pyuria is not always sterile in KD. Jan et al reported that 10.7% of KD patients have bacterial pyuria. 6 In addition to our case, there are only 4 cases in the literature of children with UTI who subsequently developed KD (Table 2).7-9 In our patient, UTI was suspected due to the presence of pyuria, and finally, ESBL-producing E coli was isolated from his urine. Regarding KD with UTI and urinary tract abnormalities, Shiono et al reported on a KD patient with left voiding cystourethrography and UTI due to E coli. 10 Consistent with our patient, Horikawa et al and Husain et al have also reported on KD patients with UTI due to E coli and without urinary tract abnormalities.7,9 Wu et al reported on 2 patients with UTI and incomplete KD. 8 In 1 patient without urinary tract abnormalities, Proteus mirablis was detected in the urine, whereas in another patient with bilateral hydronephrosis on renal sonography, the urine culture, voiding cystourethrography, and DMSA renal scan findings were negative. These results indicate that KD patients can have concurrent UTI regardless of urinary tract abnormalities. It is difficult to clarify whether the fever was due to KD or upper UTI in our patient. Apparent improvement in this patient was not seen after antibiotic treatment because cefotaxime was ineffective due to resistance of the pathogen to this antibiotic. If the fever was induced by KD, it is likely that bacterial pyuria was not due to upper UTI but lower UTI. Interestingly, treatment with IVIG and aspirin finally resolved both KD and the pyuria. Thus, it is likely that UTI might be a trigger and/or one of the causes of KD, as previously reported. 11 Our patient exhibited transient dilation of the coronary arteries; however, the presence of UTI does not alter the clinical phenotype of KD or its effect on coronary arteries.5,11
Urine Culture Revealed a Urinary Tract Infection in Children Who Subsequently Developed Kawasaki Disease.

Abbreviations: KD, Kawasaki disease; CFU, colony-forming unit; ESBL, extended-spectrum β-lactamase; UTI, urinary tract infection; IVIG, intravenous immunoglobulin; VUR, vesicoureteral reflux.
The incidence of pediatric UTI caused by ESBL-producing pathogens is increasing, and CTX-M-producing E coli is also an important cause of pediatric UTI.2,12 In Japan, a recent nationwide surveillance of hospitalized patients revealed that the CTX-M-9 group (64.6%) was predominant in E coli. 13 In the community setting, CTX-M-9 was likewise reported to be dominant; CTX-M-1 and CTX-M-2 groups (27.3%) were also detected. 14 In our previous community setting study, CTX-M-14 (53.3%) was the most predominantly encountered ESBL genotype in Tokyo’s Jonan Ward, which is located at southern side of 23 wards in Tokyo. 2 In our case, the rare genotype of CTX-M-8 was detected. To our knowledge, it was not detected previously in Japan. It was first described in Brazil and was recently detected in Germany. 15 This implies that CTX-M-8 transmission may have occurred within the community through conjugative plasmid transfer even in Japan. The CTX-M genotype might differ locally, and the prevalence of the CTX-M genotype might be changing in Japan. Thus, constant surveillance for the emergence of ESBL-producing bacteria is required in both hospitalized patients and outpatients.
Conclusion
We should pay attention to the misleading initial presentation of fever and pyuria, which can be presumed to be UTI, in patients with KD. Pediatricians should consider incomplete KD when patients present with fever and pyuria, which are consistent with UTI, but do not respond to antibiotic treatment.
Footnotes
Authors’ Note
A preliminary report was presented at the 631st Japan Pediatric Society Tokyo Chapter, 2016.
Author Contributions
KK: Contributed to analysis; drafted the manuscript; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity.
YA: Contributed to conception and design; contributed to analysis; drafted the manuscript; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity.
TK: Contributed to analysis; gave final approval.
KU: Contributed to analysis; gave final approval.
TM: Contributed to analysis; gave final approval.
KF: Contributed to conception and design; contributed to analysis; drafted the manuscript; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity.
MT: Contributed to conception and design; gave final approval.
KI: Gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A part of this work was supported by Grants-in-Aid for Scientific Research (C) No. 17K09021.
Ethical Approval
This study was approved by the Ethics Committee of Showa University School of Medicine (No. 2141), and the study was performed in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments.
