Abstract
Objective. To describe and quantify hearing impairment among children referred to the audiology clinic in Princess Marina Hospital, a public referral hospital in Botswana. Methods. In a retrospective case series, we reviewed medical records of children aged 10 years and younger whose hearing was assessed between January 2006 and December 2015 at the audiology clinic of Princess Marina Hospital in Gaborone, Botswana. Results. Of 622 children, 50% were male, and median age was 6.7 years (interquartile range = 5.0-8.3). Hearing impairment was diagnosed in 32% of clinic attendees, comprising sensorineural (23%), conductive (25%), and mixed (11%) hearing loss, while 41% of children with diagnosed hearing impairment did not have a classification type. Hearing impairment was mild in 22.9%, moderate in 22.4%, severe in 19.4%, profound in 16.9%, and of undocumented severity in 18.4%. Children younger than 5 years were 2.7 times (95% confidence interval = 1.29-5.49; P = .008) more likely to be diagnosed with sensorineural hearing impairment compared with those older than 5 years. By contrast, children older than 5 years were 9.6 times (95% confidence interval = 2.22-41.0; P = .002) more likely to be diagnosed with conductive hearing loss compared with those under 5 years. Conclusion. Hearing impairment was common among children referred to this audiology clinic in Botswana. Of those with hearing impairment, more than a third had moderate or severe deficits, suggesting that referrals for hearing assessments are not occurring early enough. Hearing awareness programs individually tailored to parents, educators, and health care workers are needed. Neonatal and school hearing screening programs would also be beneficial.
Introduction
Childhood-onset hearing impairment is very common, with global estimates that 5 of every 1000 infants are born with or develop disabling hearing impairment in early childhood. 1 Hearing impairment is estimated to be the most common disabling condition worldwide. 2 Global estimates by the World Health Organization (WHO) indicate that close to 360 million people have disabling hearing impairment, of which 32 million are children. 1 Two thirds of people with hearing impairment worldwide live in developing countries, 3 where 2000 infants with hearing impairment are born daily. 4
Disabling hearing impairment in children is defined by a hearing loss greater than 30 decibels (dB) in the better hearing ear. 5 The severity of hearing impairment is classified as mild (26-40 dB hearing level [dB HL]), moderate (41-60 dB HL), severe (61-80 dB HL), and profound (81 dB HL or greater).5,6
Hearing impairment in children has both developmental and economic impacts. It causes significant delays in language development and academic achievement.3,7 These delays are apparent even with mild to moderate hearing impairment.2,8-11 Without early intervention, the poor academic performance of children with hearing impairment persists. In these children, their hearing impairment may only be identified much later in their education. 12 Hearing impairment in children also has an economic impact. 13 It is associated with lower employment levels during adulthood. 14 In 2000 in the United States, severe to profound hearing impairment was estimated to cost society $297 000 over the lifetime of an affected individual, mostly due to reduced work productivity. 15 Furthermore, in 2015 in South Africa, the total average medical costs for a child for the first 5 and 10 years post–cochlear implantation were $27 000 and $40 000, respectively. 16 Conversely, outcomes are excellent in infants in whom hearing impairment is identified early and an intervention is initiated by 9 months of age. These infants develop language at a rate that has the potential to match that of their peers who have no hearing impairment.3,4,17
There is, therefore, an urgent need to identify infants and children with hearing impairment as early as possible so that appropriate interventions can be instituted. The Joint Committee of Infant Hearing, the American Academy of Pediatrics, and the American Academy of Audiology have published guidelines that recommend early detection of and intervention for infants with hearing impairment.18,19 The goal of “Early Hearing Detection and Intervention” is to maximize linguistic competence and literacy development for children who have hearing impairment so that they do not fall behind their hearing unimpaired peers in communication, cognition, reading, and social-emotional development. 18
The magnitude of hearing impairment among children in Botswana is not known. Given that at least 2 newborns are born deaf for every 1000 live births in developing countries, 3 and using Botswana’s 2012 birth cohort of 47 700 live births, 20 at least 96 newborns would be expected to be born deaf each year in Botswana. There are only 2 government-run audiology clinics in Botswana. Recently, audiology services are also offered at one private hospital. The Botswana government funds special education programs for children with severe hearing loss. 21 Botswana’s public health service also provides hearing aids to selected children with significant hearing loss. Universal infant hearing screening is not currently a component of newborn health care in Botswana.
Hearing outcomes of children in Botswana have not been well studied. Using data from children referred to an audiology clinic at Botswana’s largest hospital, we address this gap, summarizing referral reasons, age at first referral, and the nature and magnitude of hearing impairment, if identified.
Methods
Study Site, Participants, and Design
Princess Marina Hospital (PMH), located in Gaborone, Botswana’s capital city, is the nation’s largest hospital and serves as 1 of 2 public service audiology clinics in Botswana. Using a retrospective descriptive design, we reviewed records of children aged 10 years and younger, with audiology assessments between January 2006 and December 2015 at PMH’s audiology clinic.
Procedure
Data were extracted from archived paper patient medical records. Variables of interest included age of the child at the time of referral, sex, reason for referral, type of hearing test done, whether hearing impairment was present, and type and severity of hearing impairment using the WHO classification system.3,5
Data Analysis
We generated descriptive statistics and compared proportions of children with or without hearing impairment, including severity, using the χ2 test. P < .05 was considered statistically significant. We used SAS 9.4 software (SAS Institute, Cary, NC; 2012) for analyses.
Ethical Approval
Approval was obtained from the institutional review boards of the University of Botswana, Princess Marina Hospital, and the Botswana Ministry of Health.
Results
Demographics and Study Population
We screened 666 patient records, excluding 44 (7%) that had an inconclusive hearing test, resulting in 622 (93%) children with a conclusive hearing test for analysis (Figure 1). Fifty percent of the children were males, and the median age was 6.7 years (interquartile range [IQR] = 5.0-8.3; Table 1). A quarter of the children were less than 5 years old when first seen at the audiology clinic.
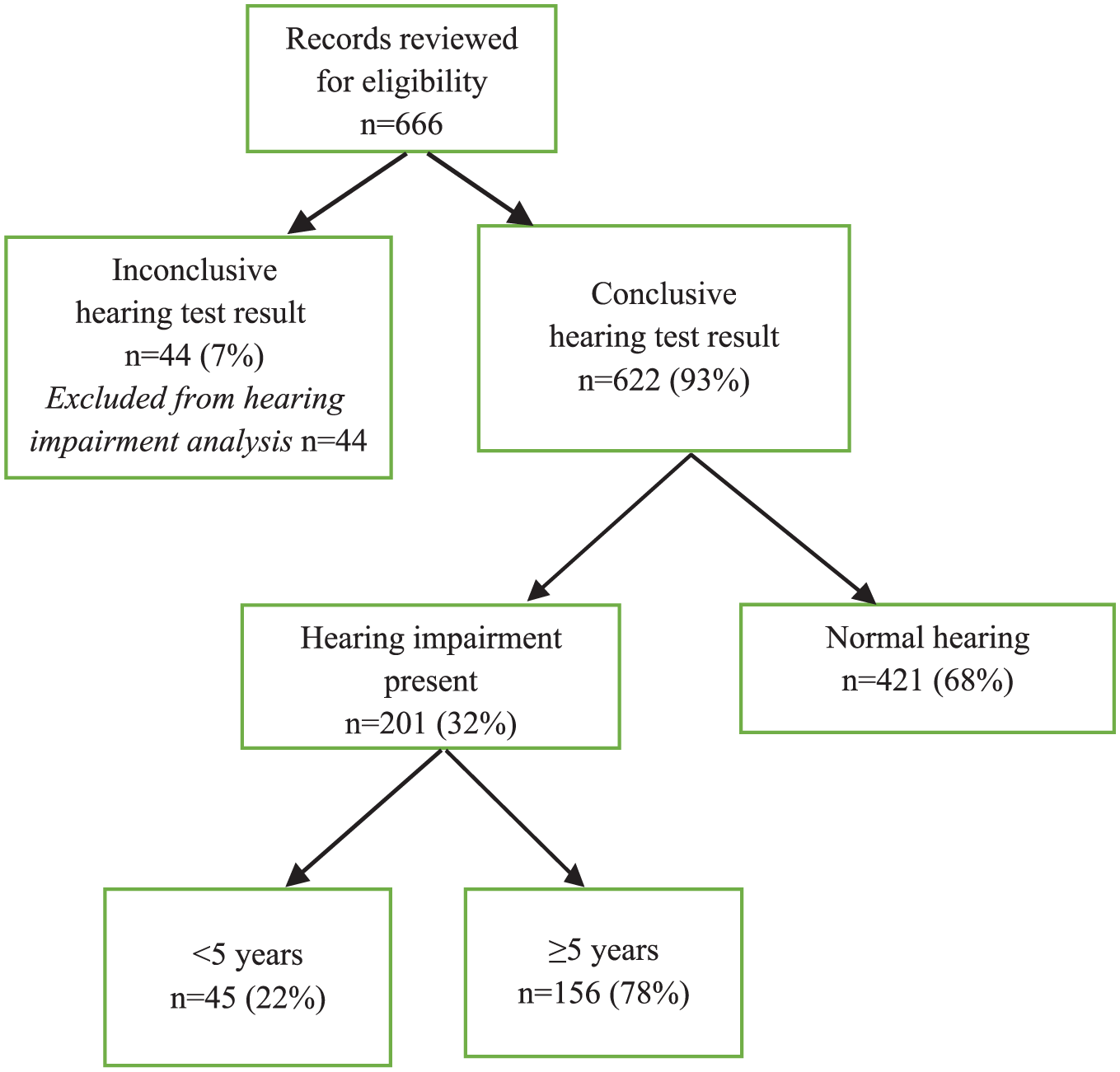
Study flow diagram.
Age, Sex, and Reasons for Referral of Children in the Study (N = 622).

Abbreviation: IQR, interquartile range.
Other reasons included developmental delay or autism spectrum disorder, syndromic appearance, epilepsy, ear pain, poor school performance, visual impairment, cleft palate, ear trauma, and cerebral palsy.
Reason for Referral and Finding of Hearing Impairment
Of the 139 children with a documented reason for referral (Table 1), the 4 most common reasons were speech delay (8%), reduced hearing (4%), recurrent ear infections or ear discharge (3%), and combined speech delay with reduced hearing (2%). There were no associations between reason for referral and either age or sex (data not shown).
Tests for Hearing and the Finding of Hearing Impairment
Children underwent a variety of tests during their hearing assessments. Of 622 children, 447 (72%) had otoscopy and 232 (37%) had tympanometry. The types of hearing tests performed were audiometry, otoacoustic emissions, acoustic reflexes, and brainstem auditory evoked response. Some children had more than one test performed (Table 2).
Type of Hearing Test Performed and the Results (N = 622).

Abbreviation: BAER, brainstem auditory evoked response.
Some children had more than one test preformed, so we have not reported the percentages.
Of the 622 children with known hearing outcome, 201 (32%) had hearing impairment, while 421 (68%) had normal hearing (Table 2). Eight children had a history of bacterial meningitis, and 6 (75%) of these had hearing impairment. Six children had a history of head trauma, and 3 (50%) had hearing impairment. We were unable to compare laterality of hearing impairment because of insufficient documentation in medical records.
Diagnosis and Degree of Permanent Child Hearing Impairment
Among the 201 children with hearing impairment, 23% had sensorineural hearing loss, 25% conductive hearing loss, 11% mixed hearing loss, and 41% unclassified hearing loss. Among children with hearing impairment, 22.9% had mild impairment, 22.4% moderate, 19.4% severe, 16.9% profound, and 18.4% no documentation of severity of impairment (Figure 2).
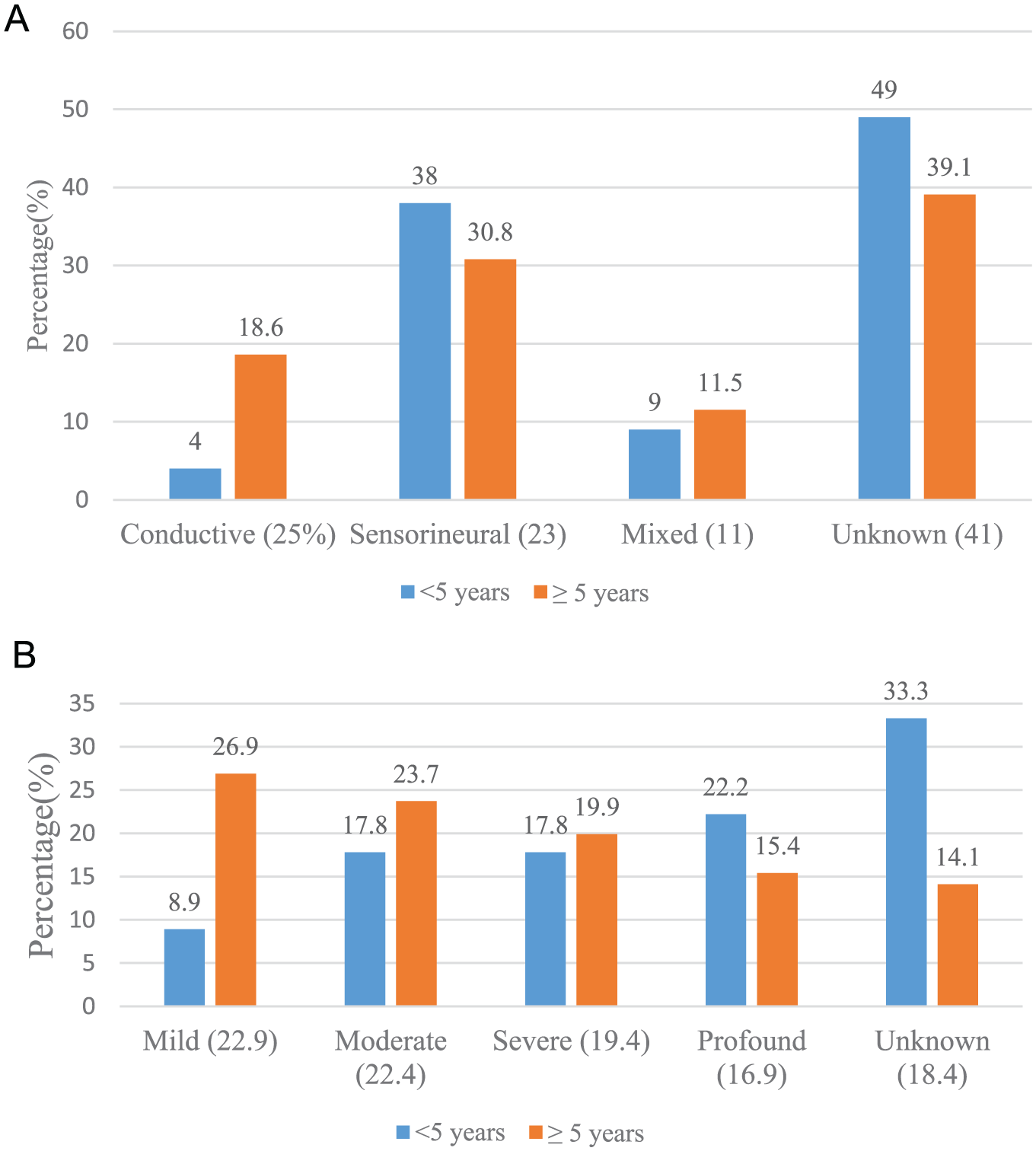
(A) Type and (B) severity of hearing impairment (n = 201).
Among the 45 children under 5 years of age with documented hearing impairment, 4% had conductive hearing loss, 38% sensorineural, 9% a combination of the two, and 49% with unclassified hearing loss. Hearing impairment type was different among the 156 children who were 5 years and older, with 30.8% having conductive hearing loss, 18.6% with sensorineural loss, 11.5% with a combination of the two, and 39.1% with unclassified hearing loss.
Children under the age of 5 were 2.7 times (95% confidence interval = 1.3-5.5; P = .008) more likely to be diagnosed with sensorineural hearing loss compared with children aged 5 years or older. By contrast, children aged 5 years or older were 9.6 times (95% confidence interval = 2.2-41.0; P = .002) more likely to be diagnosed with conductive hearing loss compared with children younger than 5 years. The occurrence of mixed hearing loss did not vary significantly by age, with 4% (6/161) of children younger than 5 years having mixed hearing loss compared with 4% (16/461) of children 5 years and older (P = .81).
Discussion
In our study, speech delay was the most common reason for audiology referral. The median age of first referral for hearing screening was 6.7 years, with hearing impairment prevalence of 32%. A higher proportion of under-5 children had sensorineural hearing loss.
While the reason for referral was often not listed in our study, among children whose medical record included a referral reason, speech delay was the most common reason. Other published work, which was population-based, has shown that the most common cause of hearing impairment in children in Swaziland and Botswana is chronic otitis media. 22 Additionally, meningitis and rubella together are responsible for more than 19% of childhood hearing loss worldwide, 23 and close to 50% of African preschool children may develop hearing impairment due to cerumen impaction, according to one systemic review. 24 All these causes of hearing impairment are most likely to occur during the first 10 years of life, a critical period of speech development.
The median age of 6.7 years at which children were first referred for hearing screening in our study is significantly later than in many other reports. Retrospective data analysis in an audiology clinic in the Free State Province of South Africa showed median ages of 3.4 years and 3.7 years at first visit and at diagnosis, respectively. 25 To further highlight the need for early timely referral, le Roux et al characterized a cohort of children from Gauteng and Free State Provinces in South Africa as being diagnosed “late” with a mean age at diagnosis of profound hearing impairment of 1.3 years. They attributed delays in initial hearing aid fitting occurring at 1.57 years, delays in early intervention services at 1.62 years, and delays in cochlear implant placement at 3.6 years to the initial delay in diagnosis. 16 Late referral of children to the PMH audiology clinic may have contributed to delayed development, as early diagnosis and intervention of hearing impairment has been shown to be very effective in mitigating the negative impact of hearing impairment on speech, language, educational achievement, and vocational outcomes.3,4,12-14,16,17,26-29
The prevalence of hearing impairment in our audiology clinic–based study was 32%. Varying prevalence figures have been reported for hearing impairment in the medical literature and depend on the cohort sampled. The prevalence of hearing impairment during screening assessments in schools was 32.8% among school children in rural India 30 and 37% among school children in urban Tanzania. 31 By contrast, hearing impairment prevalence was only 2.2% among urban school children in South Africa 32 and 3.1% among school children in Uganda. 33 The reported prevalence of hearing impairment among children in the general population of sub-Saharan Africa varies from 2.2% to 13.9%.32,34 Our prevalence cannot be compared with population estimates, as we were only sampling those referred to an audiology clinic. Despite this, it is concerning that a large number of children referred to the audiology clinic were diagnosed with hearing impairment, suggesting that only those children with a high likelihood of hearing impairment may be getting to the PMH audiology clinic. However, the delay in recognition of the need for referral is even more concerning. Although the high prevalence of hearing impairment from our work does not reflect the prevalence of hearing impairment among children in Botswana overall, as our study population was referred for hearing assessment, these findings suggest the need for educational materials specifically tailored to parents and health care providers that highlight symptoms of hearing impairment and promote the value of early referral to audiology clinics.
The finding of a significantly higher proportion of under-5 children having sensorineural hearing loss is in keeping with South African data where 95% of under-5 children with profound hearing impairment had sensorineural hearing loss. 16 This finding might be related to an interplay of environmental age-related risk factors involved in the etiology of hearing impairment in children.
The severity of hearing impairment was evenly distributed in our study. This differs from many studies reporting that the majority of children have mild hearing impairment. Taha et al in a study performed in both rural and urban schools in Egypt reported an overall prevalence of 48.7% for mild hearing loss. 35 However, Mann et al in Chandigarh, India, reported separate prevalence figures for rural and urban schools. In urban areas, they reported prevalence figures of 1.94% and 4.36% for mild and moderate hearing impairment, respectively. In rural areas, they reported prevalence figures of 6.25% and 26.56% for mild and moderate hearing impairment, respectively. 30 Children who have more severe hearing impairment tend to be diagnosed earlier, and so, they tend to receive attention earlier than those with less severe degrees of hearing loss. 26 Even mild hearing impairment has been associated with developmental consequences. Poor language and school performance, including a failure rate of 37% for at least one grade among students with minimal hearing impairment versus 3% for normal hearing students has been reported. 36 Children with minimal hearing impairment have also been found to have a reduced capacity to multitask. 37 Their timely identification of hearing impairment, regardless of the severity, is needed to optimize developmental outcomes. The WHO guidelines call for priority for hearing aids and services for children with a mean hearing impairment in the range 31 to 80 dB HL in the better ear in the frequency range 500 Hz to 4 kHz as these are the children who are expected to derive the most benefit from hearing interventions. 38 It is essential in Botswana that all children with hearing loss are identified early, including those with mild to moderate impairment. This will require uniform screening.
This study was limited by missing data, a phenomenon intrinsic to retrospective study designs. Although the accumulated data were readily accessible from the audiology clinic, undesirably, data were missing on several occasions. This reduced our sample size and hence the power of the study. 39 Due to missing biographical data in patients’ records, we were unable to analyze known risk factors for hearing impairment including neonatal intensive care unit stay for more than 5 days,16,40,41 serious illness in the neonatal period, 42 neonatal hyperbilirubinemia,16,43,44 congenital infections, 45 and bacterial meningitis in childhood.46-48 Missing data may also explain why speech delay was the main reason for referral to our auditory clinic. Furthermore, hearing impairment was unclassified in 49% of the cases, and the severity of hearing impairment was recorded in only 18% of the children. This limitation speaks to the importance of improving data capture in the PMH audiology clinic. We plan to repeat this study but with a prospective study design.
Given that Botswana is a middle-income country and that the government is committed to preventative care including early screening and cost-effective interventions, earlier detection of hearing impairment would represent programming in keeping with national health care and early child development values. Programs to consider include a universal neonatal auditory screening program and school-based hearing screening. In South Africa, the school health policy prescribes that every child should be screened for hearing before starting the foundation phase of learning. 49 There are certainly resource implications in rolling out such a policy. However, there are also novel, low-cost solutions including a validated smartphone-based hearing screen that has been used in primary health care clinics in South Africa. 50 In Egypt, Taha et al demonstrated that health care professionals including nurses with limited audiology background can be trained to correctly conduct hearing tests, 35 the results of which, if abnormal, can then be verified by diagnostic audiology tests involving specialists in the audiology clinic.
Conclusion
Hearing impairment was found to be highly prevalent among children referred to an audiology clinic in Botswana. Late referral, as reflected in the severity of hearing impairment, was common and represents a missed window of opportunity to intervene before development sequelae occur. Hearing awareness programs are needed for parents, educators, and public and health care workers. Introduction of a universal neonatal hearing screening program and a school health policy that mandates hearing screening before enrolment into the foundation phase would represent a public health investment with evidence-based benefit.
Footnotes
Acknowledgements
We would like to thank the staff of the audiology clinic in Princess Marina Hospital for being very helpful to the authors during data collection.
Authors’ Note
Any underlying research materials can be accessed through email to the corresponding author (
Author Contributions
FMB: Contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
KMP: Contributed to design; contributed to analysis and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
ABM: Contributed to acquisition and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MM: Contributed to acquisition and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
KDW: Contributed to interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
TD: Contributed to interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
APS: Contributed to conception and design; contributed to interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
