Abstract
Transcatheter aortic valve implantation (TAVI) can be safely performed in old patients. Increasing longevity raises often the question whether TAVI can be still useful for patients beyond a certain age limit. Data on long-term outcomes of elderly patients after TAVI are sparse. We sought to assess the impact of very advanced age on long-term outcomes after transfemoral (TF)-TAVI. Data of 103 patients undergoing TF-TAVI with the balloon-expandable bioprosthesis between May/2014 and May/2019 were analyzed. We divided the cohort into two age groups: ≥85 years (group1: n = 37; 87.5 ± 2.6 years; STS-Score 3.9 ± 1.4) versus < 85 years (group2: n = 66; 80 ± 3.1 years; STS-Score 3.4 ± 1.8). We conducted up to 6 years clinical follow-up. Overall mortality at 30 days was 3.8% without significant differences between the two age groups. Incidence of major vascular injury (8.6 vs. 6.3%, p = .695) and stroke (2.8 vs. 3%, p = 1) was not significantly different between group 1 and 2, respectively. More than mild paravalvular leakage was found in 1 patient (group 1). The mean long-term survival probability was 51.3 months [95% CI: 42.234–60.430] in group 1 versus 49.5 months [95% CI: 42.155–56.972] in group2 (p = .921). Long-term outcomes of very old patients after TF-TAVI show a similar treatment benefit compared to the younger patients.
Introduction
Long-term data on outcomes after transfemoral transcatheter aortic valve implantation (TF-TAVI) are sparse due to serious comorbid- and age-related conditions limiting life expectancy. The available data on the effect of very advanced age on long-term clinical outcomes are even more limited. Therefore, physicians are often questioning the treatment benefit of TF-TAVI in very old patients.
An additional reason for the scarce data on long-term postprocedural outcomes of very old patients could be the fast-paced development in the area of TAVI. For instance, in the last decade, shifting from higher to lower risk patients was rapid and additionally accompanied by growing interventional experience and industrial developments. Therefore, the global focus on long-term follow-up data is mainly concerning “younger” TF-TAVI patients by use of the last generation transcatheter heart valves (THV). Moreover, drawing conclusions from current data regarding the possible long-term benefit of TF-TAVI in very old patients remains difficult not only due to inconsistencies such as use of various THVs and access routes but also due to the lack of data in general (Pepe et al., 2020; van der Kley et al., 2016; Yokoyama et al., 2019). Thus, we sought to analyze long-term data after TF-TAVI using the balloon-expandable THV and assess the impact of very advanced age on long-term outcomes.
Methods
This single-center retrospective study was conducted to evaluate age-related early and long-term clinical outcomes after TF-TAVI using the Edwards SAPIEN 3 (ES3) (Edwards Lifesciences Inc., Irvine, CA, USA) bioprosthesis. Data from 103 consecutive patients with symptomatic aortic valve stenosis undergoing TF-TAVI in our center between May 2014 and May 2019 were analyzed. The decision for TAVI was made by an interdisciplinary heart team. All patients underwent coronary angiography and thoraco-abdominal computed tomography angiography before the TF-TAVI for assessment of coronary arteries, aortic valve, aorta, and femoral arteries. THV-size selection and TF-TAVI procedures were performed according to standard techniques (Kappetein et al., 2012). We divided the cohort into two age groups: patients ≥85 years (group 1) and patients <85 years (group 2). Clinical endpoints were defined according to the Valve Academic Research Consortium (VARC II) definitions (Kappetein et al., 2012). Paravalvular leakage (PVL) was classified as none, trace–mild and moderate and severe. We collected the data from hospital records, through telephone interviews with patients and referring physicians. The cause of death was obtained from the last physician involved in the patients’ treatment. All patients gave informed consent for data collection.
of Early and Long-Term Follow-Up Timing
The early follow-up timing was defined as the first 30 postoperative days. The long-term follow-up timing was defined as a time interval from the second postoperative month up to 6 years. The mean follow-up was 30 months.
Primary and Secondary Endpoints
The primary endpoints of the study were the 30-day mortality and long-term survival, the 30-day stroke and long-term freedom from stroke and the 30-day new permanent pacemaker implantation (PPI) and long-term freedom from PPI. The secondary endpoints were procedural major vascular injury rates and the paravalvular leakage (PVL) rates at discharge.
Statistics
Distributions of quantitative variables are described as means (±SD) and compared with the use of the Mann–Whitney U test. Qualitative variables are summarized by count and percentage and compared with the use of the chi-square test or Fisher’s exact test. The Kaplan–Meier method was used to calculate the mean survival time and the group comparisons were made with the log-rank test. The functional evaluation was conducted using the McNemar test. Data were managed with the SPSS statistical package, (IBM SPSS Statistics for Windows, Version 23.0.0.2 Armonk, NY: IBM Corp.). A two-sided p value of less than 0.05 was considered to indicate statistical significance. No adjustment for multiple testing was performed. All the analyses were considered to be exploratory.
Results
Patient Characteristics
Baseline Characteristics.
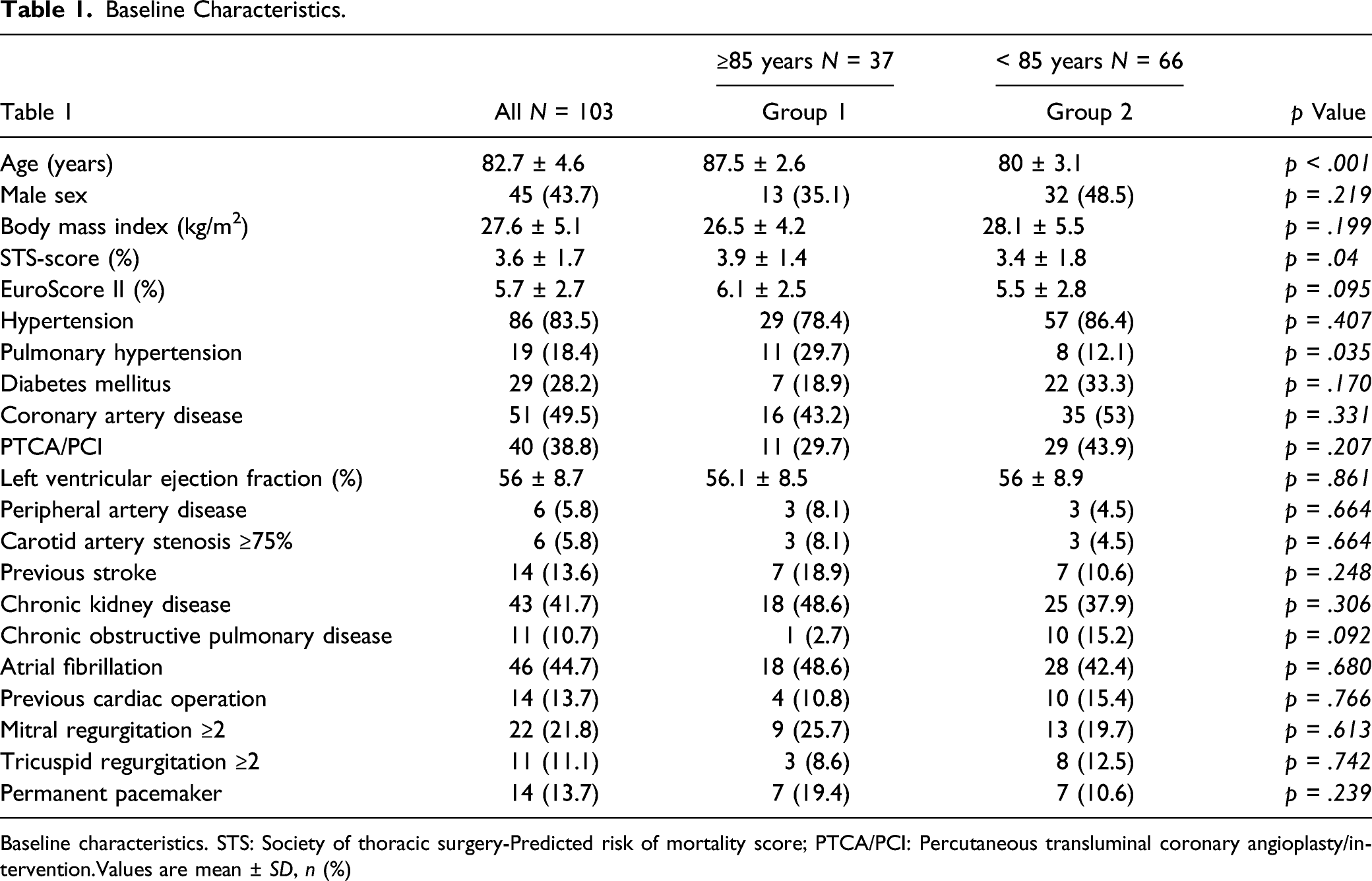
Baseline characteristics. STS: Society of thoracic surgery-Predicted risk of mortality score; PTCA/PCI: Percutaneous transluminal coronary angioplasty/intervention.Values are mean ± SD, n (%)
Procedural Characteristics
Procedural characteristics.
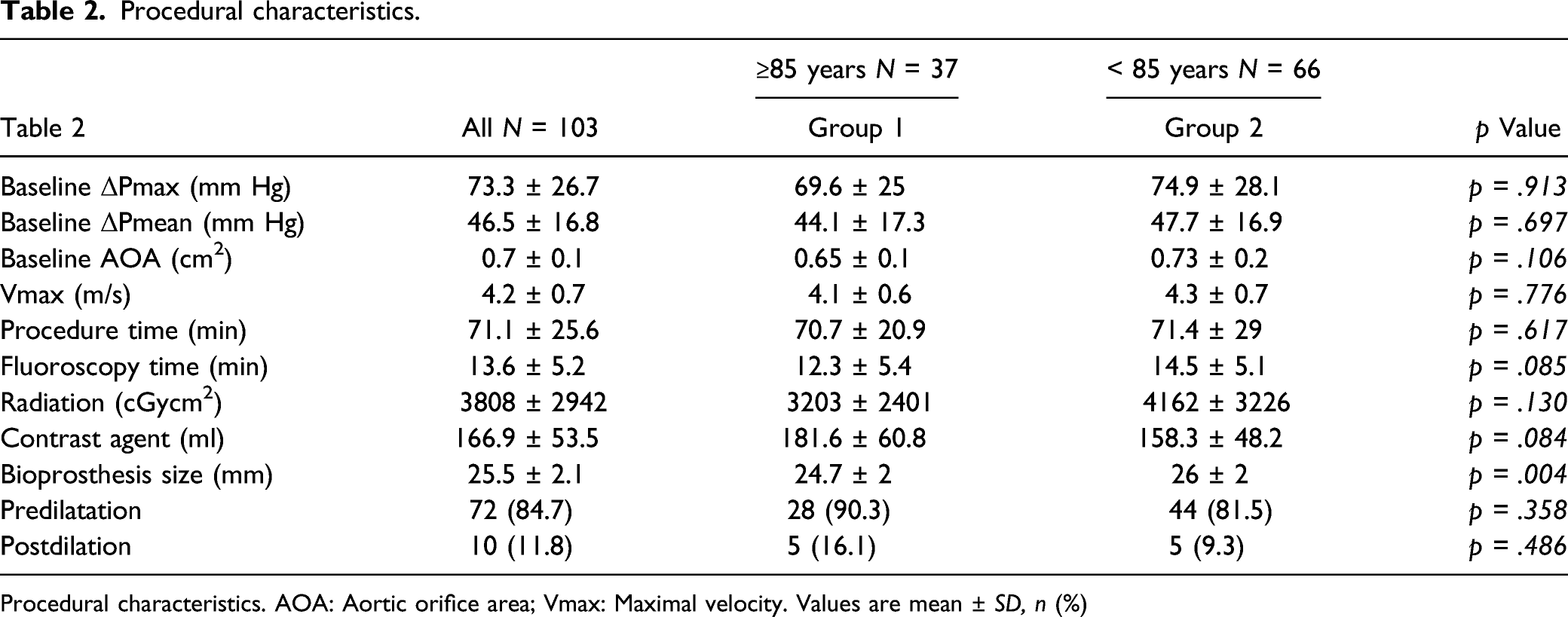
Procedural characteristics. AOA: Aortic orifice area; Vmax: Maximal velocity. Values are mean ± SD, n (%)
Procedural Complications
Procedural complications.

Procedural complications. SAVR: Surgical aortic valve replacement; TAVI: Transcatheter aortic valve implantation
Thirty-Day Clinical Outcomes
The 30-day all-cause mortality of the entire cohort was 3.8%, with no death in the group 1. The all-stroke rate was 2.9% without significant differences between the study groups.
Thirty-day outcomes.

Thirty-day outcomes. AKIN: Acute kidney injury network; LBBB: Left bundle branch block. PPI: New permanent pacemaker implantation
Long-Term Clinical Outcomes
The mean survival probability was similar in both age groups. Long-term survival probability was 51.3 months [95% CI: 42.234–60.430] in group 1 versus 49.5 months [95% CI: 42.155–56.972] in group 2, p = .921 (Figure 1a). a. Long-term survival of the two age groups. The mean survival probability was similar in both age groups (p = .921). b. Long-term freedom from stroke. c. Long-term freedom from postprocedural permanent pacemaker implantation. Long-term freedom from postprocedural permanent pacemaker implantation (PPI) was similar between both age groups (p = 0.958).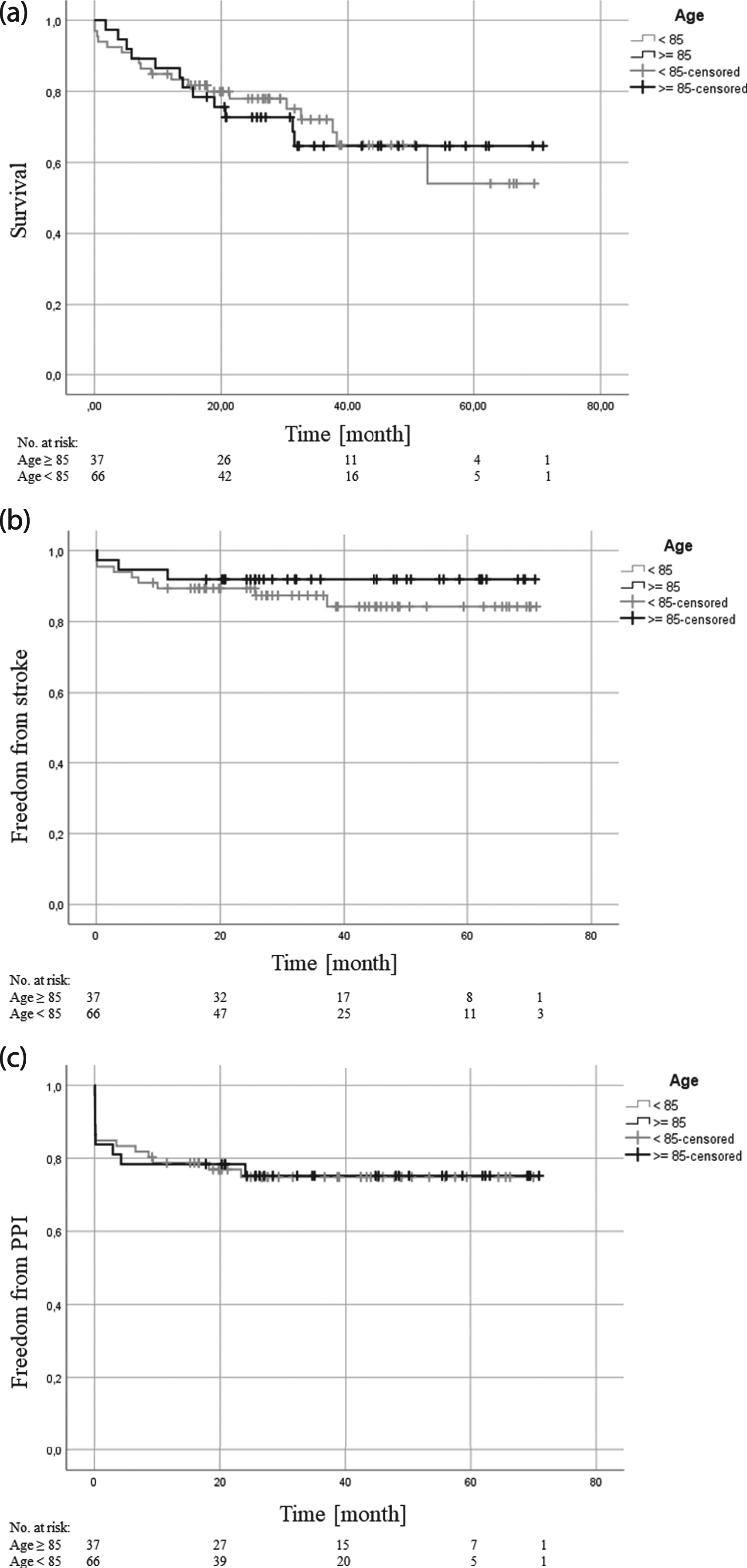
Freedom from long-term stroke was similar between both age groups. The mean freedom from stroke was 65.6 months [95% CI: 59.761–71.368] in group 1 versus 62 months [95% CI: 56.419–67.530] in group 2, p = .395 (Figure 1b).
The mean freedom from PPI in the long-term analysis was 54.3 months [95% CI: 44.851–63.771] in group 1 versus 53.6 months [95% CI: 46.711–60.654] in group 2 (p = .958) (Figure 1c).
At long-term we registered one degenerated valve (group 2), exhibiting high grade aortic stenosis and mild regurgitation requiring Re-TF-TAVI (6 years after initial implantation). Two patients suffered on prosthesis endocarditis on the 6th and 10th postoperative month (group 1). Both patients deceased on endocarditis.
Causes of Death
Overall, cardiac death was the most common cause with a rate of 10.7% (Figure 2). Causes of death (MOF: Multiple organ failure).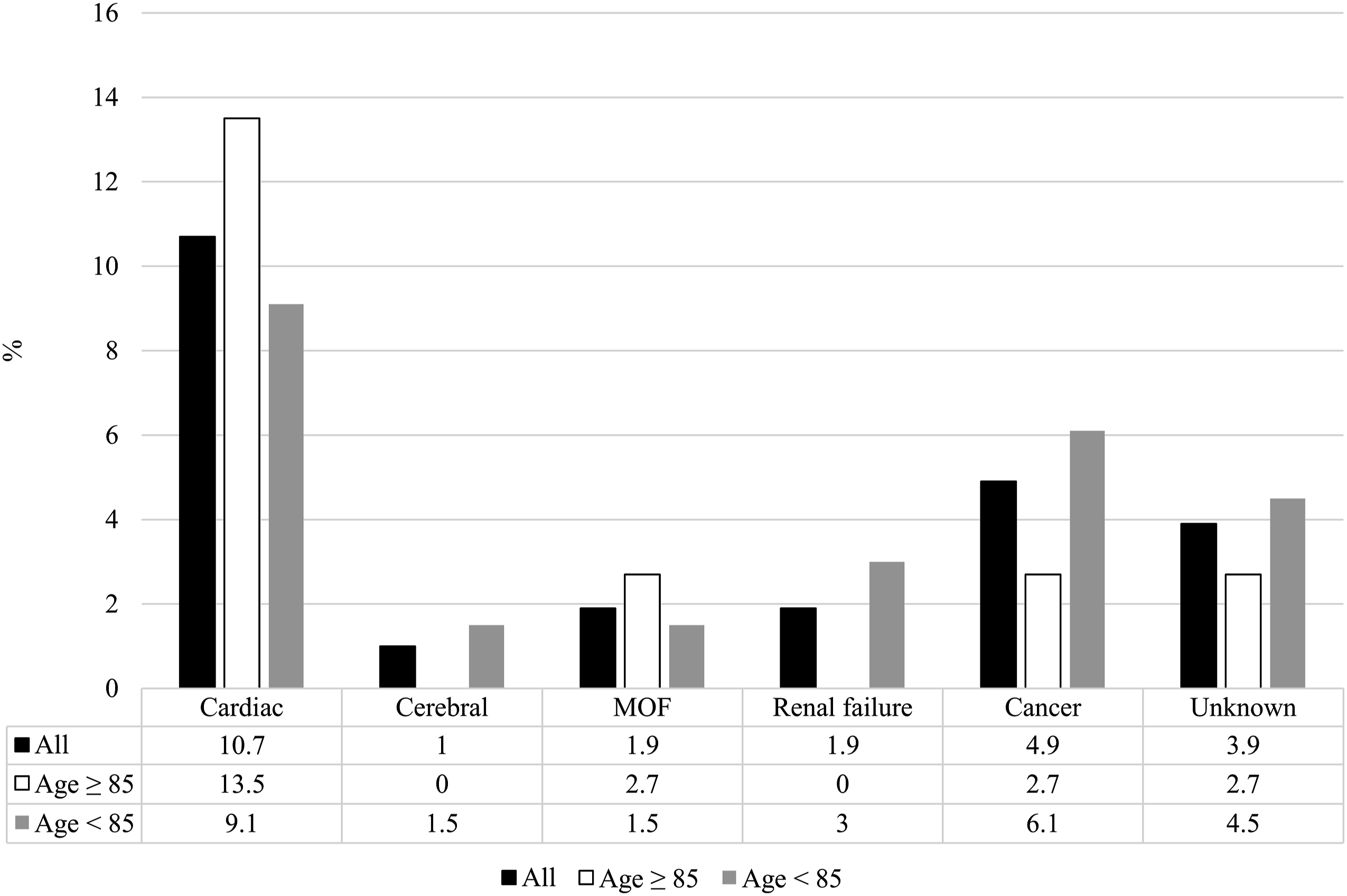
Hemodynamics
At discharge the peak/mean THV-gradients were similar in both groups: 19.6 ± 7.6/11 ± 4.3 mmHg in group 1 versus 19.9 ± 7.3/11 ± 3.7 mmHg in group 2, respectively (p = .785). At discharge the maximal flow velocity was similar between both groups: 2.1 ± 0.5 m/s in group 1 versus 2.2 ± 0.3 m/s in group 2 (p = .632). Incidence of PVL was similar between the two groups at discharge. Severe PVL did not occur. In group 1 no PVL was observed in 45.9% of the patients, trace–mild PVL was found in 37.8% and moderate in 2.7% of the patients. In group 2 no PVL was observed in 50% of the patients, trace–mild PVL was found in 31.8% and moderate in 0% of the patients.
Functional Evaluation
According to the New York Heart Association (NYHA) 92.3% of the patients at the baseline were classified in NYHA class III/IV. Long-term follow-up after TF-TAVI showed a significantly improved functional capacity of the patients after the procedure (81.7% of the patients were classified in NYHA class I/II), as assessed by the McNemar test; p < .001.
Discussion
To the best of our knowledge, the current study represents the longest clinical follow-up for very old patients treated with the new generation balloon-expandable THV. The present study is the first to demonstrate that long-term outcomes of very old patients undergoing TF-TAVI (long-term survival, long-term freedom from stroke and from rhythm disturbances requiring PPI) are similarly good compared to the younger population.
Data on long-term outcomes after TAVI are remarkably rare, especially in the very old patient-groups. The majority of the studies with elderly patients focus on clinical follow-up data up to 1 year. As a result of more serious comorbid conditions and age-related natural causes limiting life expectancy, long-term outcomes of very old patient undergoing TF-TAVI are sparse. Therefore, the operator is frequently faced with the question, whether very old patients have a similar treatment benefit compared to the younger patients. Taking the most frequent short and long-term complications (e.g., relevant PVL and vascular complications) into consideration increases the complexity of this question. Studies trying to address this issue had to modify the definition of long-term follow-up accordingly due to the advanced age of the patients (van der Kley et al., 2016; Yokoyama et al., 2019). Thus, a distinct long-term follow-up does not exist in case of elderly TAVI-populations. van der Kley et al. (2016), similar to Yokoyama et al. (2019)., reported a 3-year follow-up period for older patients who underwent TAVI, and defined this timeframe in their study as long-term.
The learning curve, improved interventional skills, continuous development of sophisticated THVs and THV-delivery systems, improved patient selection, and accurate annular-sizing have moved boundaries of the possible to achieve the desired post-interventional results. Therefore, in our opinion, this analysis shows that patients who are most profiting from this evolution are those who are more fragile and very old. Very old patients are commonly more prone to short- and long-term complications. Pepe et al. (2020) observed significantly higher 30-day mortality in patients older than 85 years compared to their younger counterparts. Similarly, Arsalan et al. (2016) and Yokoyama et al. (2019) reported a higher 30-day mortality in nonagenarians than in the younger ones. In our study mortality at 30-days was 0% in the ≥85 years cohort. Additionally, it has been shown that very old patients are more prone to procedural complications. A higher incidence of stroke (van der Kley et al., 2016; Vendrik et al., 2018; Yokoyama et al., 2019) and vascular complications (Arsalan et al., 2016; Yamamoto et al. 2012; Yokoyama et al., 2019) have been reported in the older patients. In the current analysis we registered similar early stroke-rates between the two age groups and an excellent probability of long-term freedom from stroke in both groups. Incidence of major vascular complications was similar between the two age groups.
Various data show a trend towards higher mortality in very old patients after TF-TAVI (Arsalan et al., 2016; Deharo et al., 2020; Pepe et al. 2020). In our study, patients aged ≥85 years had similar long-term survival rates compared to patients aged <85 years. Two main reasons could explain these differences. First, in the above-mentioned studies the surgical risk score was higher than in our analysis and various THVs and delivery systems were used (including older generation THVs). Notably, Pibarot et al. (2020) showed recently a significant lower rate of structural valve deterioration of the ES3 than in SAPIEN XT after 5 years.
Heart team approach is key for selection of the appropriate access route. Mostly due to severe peripheral artery disease the transapical route for TAVI still plays a role in treating patients (17,2–23,7%) (Auffret et al., 2017; Leon et al., 2016). Due to numerous technological advances, improved interventional skills, and reported better outcomes, the transfemoral approach gains continuously ground against the other access routes (Siontis et al., 2016). However, even in the most recent studies the use of the ES3 and ES3 Ultra reported up to 12.3% major vascular complication (Rheude et al., 2020). Recent studies including very old patients at high operative risk reported a peripheral artery disease (PAD) rate ranging from 16% to 25.7% (Arsalan et al., 2016; Pepe et al. 2020; Yokoyama et al., 2019). Procedural vascular injuries can be associated with higher mortality (Généreux et al., 2012). This was not the case in our study. Nevertheless, baseline data showed a PAD - incidence of only 5.8%. Therefore, accurate pre-procedural screening and patient selection based on “heart team” consensus is of great importance.
A typical complication after TAVI is PPI. After the release of the ES3, the initial data reported an incidence of PPI between 13 and 25.5% (Murray et al., 2015; Webb et al., 2014). Experience over time and research on the underlying mechanisms leading to PPI have contributed to a certain further decrease in the PPI rate using the ES3. Recent studies reported PPI rates between 6.6% (Mack et al., 2019) and 16% (Husser et al., 2016). Our PPI rate was 19.1% and consistent with the reported initial data. Nevertheless, higher incidence than in the more recent studies could be explained by factors such as learning curve of less experienced operators (in house proctoring). In addition, we did not exclude patients who are known to be at higher risk for PPI (e.g., patients with complete right bundle branch block or high calcification burden at the height of the non-coronary cusp, which have been shown to be predictors for PPI) (Husser et al., 2016; Mack et al., 2019; Murray et al., 2015; Webb et al., 2014). Another relevant reason might be the indication spectrum for PPI after TAVI, which can differ from center to center. Nevertheless, data on long-term rhythm disturbances after TAVI requiring PPI are sparse. Our analysis showed no necessity for PPI at long-term even in the very old patient-group, which constitutes a novelty.
One of the major post-procedural complications after TAVI represents acute kidney injury (AKI). Reasons for post-procedural AKI are complex. Independent predictors for AKI occurrence such as previous peripheral artery disease, history of chronic kidney disease, and transapical access have been determined (Wang et al., 2017). Furthermore, hypertension, atrial fibrillation, congestive heart failure, diabetes mellitus, and stroke have been found also to be risk factors for TAVI-associated AKI (Wang et al., 2017). Other and potentially influenceable factors associated with AKI occurrence have recently been identified, such as inflammatory response, hypoperfusion, and oxidative stress (Navaratnarajah et al., 2020), and the fact that the applicated periprocedural contrast media exhibits nephrotoxic features in this population is beyond doubt (Becker et al., 2006; Jochheim et al. 2014; Yamamoto et al., 2013).
In a recent published relevant study of the Society of Thoracic Surgeons/American College of Cardiology National Cardiovascular Data Registry–Transcatheter Valve Therapy Registry including 107 814 study patients was reported a rate of 10.7% of post-procedural AKI (Julien et al., 2021). Among patients who developed AKI in this study (Julien et al., 2021), 9.5% experienced stage 1 AKI, 0.1% stage 2 AKI, and 1.1% stage 3 AKI. Here, only the AKI stage 3 appeared to be of clinical relevance increasing dramatically the mortality after TAVI (Julien et al., 2021).
In our study we registered almost identical AKI rates (AKI stage 1: 11.4%, AKI stage 2: 1.3% and AKI stage 3: 1.3%) without differences between age groups. The fact that the AKI stage 3 rate in our study was only 1.3%, suggests a very satisfactory AKI related outcome in our study.
Conclusion
Long-term outcomes of very old patients after TF-TAVI show a similar treatment benefit compared to the younger patients. Not age-related patient selection and continuous procedural optimization is necessary in order to further decrease TAVI-associated complication rates.
Limitations
This is a retrospective and non-randomized single-center study using a limited number of patients.
Footnotes
Acknowledgments
The authors thank Hildegard Christ PhD for the statistical support.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Polykarpos C Patsalis is proctor for Edwards Lifesciences. All other authors declare no potential conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
IRB Approval
All patients provided informed consent for data collection, and approval for the study was obtained from the Ethics Committee of the Medical Faculty of the RUHR University, Bochum, Germany; Date: 19.10.2018; Reg. No. 18-6339.
Ethical Approval
The Ethics Committee approval has been obtained (Reg. No. 18-6339).
Data Availability Statement
Due to the nature of this research, participants of this study did not agree for their data to be shared publicly, so supporting data is not available. This is also mandated from the local Ethic Committee and documented upon the Ethic vote for this study.
