Abstract
Deep brain stimulation of the central auditory pathway is emerging as a promising treatment modality for tinnitus. Within this pathway, the dorsal cochlear nucleus (DCN) plays a key role in the pathophysiology of tinnitus and is believed to be a tinnitus generator. We hypothesized that high-frequency stimulation (HFS) of the DCN would influence tinnitus-related abnormal neuronal activity within the auditory pathway and hereby suppress tinnitus. To this end, we assessed the effect of HFS of the DCN in a noise-induced rat model of tinnitus. The presence of tinnitus was verified using the gap prepulse inhibition of the acoustic startle response paradigm. Hearing thresholds were determined before and after noise trauma by measuring the auditory brainstem responses. In addition, changes in neuronal activity induced by noise trauma and HFS were assessed using c-Fos immunohistochemistry in related structures. Results showed tinnitus development after noise trauma and hearing loss ipsilateral to the side exposed to noise trauma. During HFS of the DCN, tinnitus was suppressed. There was no change in c-Fos expression within the central auditory pathway after HFS. These findings suggest that DCN-HFS changes patterns of activity and results in information lesioning within the network and hereby blocking the relay of abnormal tinnitus-related neuronal activity.
Introduction
Subjective tinnitus is defined as an auditory perception that is not induced by an acoustic stimulus. This symptom is highly prevalent in the general population and can severely impair an individual’s quality of life. In recent years, numerous studies have provided further insight into tinnitus-related changes in auditory and nonauditory brain structures, which are usually triggered by hearing loss (Kaltenbach, 2011; Norena & Eggermont, 2003; Norena & Farley, 2013; Roberts et al., 2010).
The dorsal cochlear nucleus (DCN) is the first site of multisensory convergence in the auditory pathway. Previous studies have suggested an important role of the DCN in the pathophysiology of tinnitus (Kaltenbach, 2006; Kaltenbach & Godfrey, 2008; Wu, Martel, & Shore, 2016). It has been shown in animal models that a functioning DCN is necessary for tinnitus development (Brozoski, Wisner, Sybert, & Bauer, 2012; Kaltenbach, 2006). Therefore, the DCN has been referred as a tinnitus trigger and generator (Brozoski & Bauer, 2005). Following this theory, hearing loss leads to reduced input to the eighth cranial nerve, and as a compensatory mechanism the DCN shows hyperactivity, for example, increased synchrony and bursting (Wu et al., 2016). Multiple synaptic changes underlying hyperactivity have been described, such as degeneration of fibers, changes in excitatory neurotransmission, shifts in the balance of excitatory and inhibitory synapses, and adaptation of glycine receptor expression (Kaltenbach, 2007). Increased neuronal synchrony and bursting activity are also described in upstream areas of the auditory pathway, specifically in the inferior colliculus (IC) (Bauer, Turner, Caspary, Myers, & Brozoski, 2008; Luo, Pace, Zhang, & Zhang, 2014; Ropp, Tiedemann, Young, & May, 2014), medial geniculate body of the thalamus (MGB) (Kalappa, Brozoski, Turner, & Caspary, 2014), and primary auditory cortex (A1) (Luo, Pace, & Zhang, 2017; Norena, Moffat, Blanc, Pezard, & Cazals, 2010). These findings suggest that a neural code for tinnitus emerges in the DCN but affects the full network.
Despite decades of efforts to develop an effective therapy, it is still highly challenging to treat tinnitus. Multiple preclinical and clinical studies suggested deep brain stimulation (DBS) as a promising treatment option in severe, refractory tinnitus (Smit et al., 2016; van Zwieten et al., 2018; van Zwieten, Smit, Jahanshahi, Temel, & Stokroos, 2016). Furthermore, electrical stimulation of the DCN with an auditory brainstem implant (ABI), which is used to elicit auditory sensations, can alter tinnitus loudness. (Soussi & Otto, 1994). DBS is a less invasive modality to electrically stimulate a subcortical structure, yet implantation of the DCN with a DBS electrode has not been performed in a clinical setting.
Although the exact mechanism behind the effects of DBS remains unclear, there is evidence that especially high-frequency stimulation (HFS) disrupts abnormal neuronal activity (McConnell, So, Hilliard, Lopomo, & Grill, 2012; McIntyre & Hahn, 2010). It has been proposed that a high-rate conditioner stimulus induces stochastic characteristics of resting-level spontaneous activity, resulting in tinnitus suppression (Rubinstein, Tyler, Johnson, & Brown, 2003; Tyler et al., 2008). In Parkinson’s disease, hypersynchrony and bursting activity can be suppressed with HFS of the subthalamic nucleus (McConnell et al., 2012; Whitmer et al., 2012). Therefore, we hypothesized that HFS of the DCN would suppress tinnitus with a similar mechanism.
In this study, the effect of bilateral HFS of the DCN on tinnitus was assessed in a noise-induced rat model of tinnitus. Auditory brainstem responses (ABRs) were recorded before and after noise exposure. Gap prepulse inhibition of the acoustic startle (GPIAS) response paradigm was used to assess tinnitus. To elaborate on the effects of HFS as well as noise trauma on neuronal activity, the expression of an immediate early gene (c-Fos) was measured in the IC, MGB, and A1.
Materials and Methods
Subjects
Ten male Sprague Dawley rats were used in the study, weighing approximately 250–300 g at time of surgery. Rats were individually housed in standard Makrolon™ cages, with feed and water ad libitum. Conditions in the room were constant, with a temperature of 20℃ to 22℃ and a humidity of 60% to 70%. The light–dark cycle was reversed, and experiments were conducted within the dark period. The study protocol was approved by the Animal Experiments and Ethics Committee of Maastricht University.
Study Design
A repeated measures design was used. All subjects were tested at baseline and after noise trauma. All rats underwent surgery at the beginning of the experiment (Figure 1).
Timeline of the experimental procedures. GPIAS = gap-prepulse inhibition of the acoustic startle reflex paradigm for tinnitus assessment; ABR = auditory brainstem response recordings; HFS = high-frequency stimulation. (a) Representative photomicrograph of coronal brain section stained for Nissl, showing histological verification of the electrode location (symbol *) in the dorsal cochlear nucleus (white dotted line). (b) Anatomic placement of all electrode tips (symbol •), shown schematically in one hemisphere.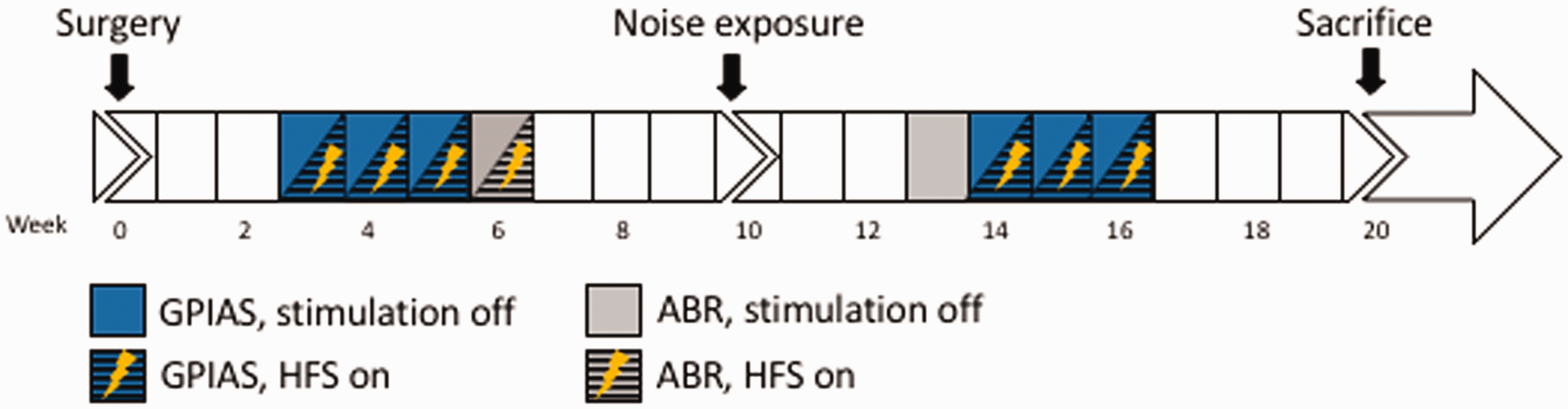

Surgical Procedure
Anesthesia was induced by an intraperitoneal injection of Xylazine (10 mg/kg) and Ketamine (90 mg/kg) and maintained with Ketamine (60 mg/kg/h). A rodent stereotact (Stoelting, Wood Dale, IL, USA, model 51653) was used with blunt ear bars to prevent damage to the middle ears. Stimulation electrodes (coaxial gold-plated with a platinum-iridium inner wire, shaft diameter of 250 µm, and tip diameter of 50 µm [Technomed, Beek, the Netherlands]) were bilaterally implanted in the DCN (coordinates from Bregma: anterior-posterior −11.1; medial-lateral 3.9; dorsal-ventral 7.8; Paxinos, 2006; Tan et al., 2010). In addition, two recording electrodes (Teflon-coated stainless-steel wire electrodes with exposed tip) were permanently secured to the scalp of the rats, one on the vertex and one at the mastoid bone, to record ABR. The electrode construct was secured with five stainless steel screws and dental cement (Paladur, Heraeus Kulzer GmbH, Hanau, Germany).
Deep Brain Stimulation
Rats were tested in two conditions: stimulation off (attached to the stimulation cable) and during HFS (100 Hz, 60 -µs pulse width, and 100-µA amplitude). Electrical stimulation was bipolar with monophasic pulses, applied with a stimulator (DS8000, WPI, Berlin, Germany) connected to a constant-current isolator (DLS100, WPI, Berlin, Germany). The chosen stimulation parameters were based on our previous experiments (Smit et al., 2016; Tan et al., 2010; van Zwieten et al., 2018). For GPIAS and ABR measurements, electrical stimulation was applied continuously during the full acquisition time, for GPIAS this included 10 min of habituation.
Tinnitus Induction
Subjects were anesthetized (induction with intraperitoneal injection of Xylazine (10 mg/kg) and Ketamine (90 mg/kg) and maintained with Ketamine (60 mg/kg/h) and unilaterally exposed to a 16 kHz octave-band noise at 115 dB for 90 min. The sound paradigm was designed by a custom-made Matlab script, processed with an external soundcard with a sampling rate of 192 kHz (Creative E-MU 0204) and amplified (Ultrasonic power amplifier and Ultrasonic Dynamic Speaker Vifa [Avisoft Bioacoustics, Berlin, Germany]). A loudspeaker was placed at a standard distance of 3 mm to the right ear, while the left ear was protected with a plug of modeling clay. Complete removal of the plug after the experiment could be easily achieved and confirmed with visual inspection. After noise exposure, subjects were not tested for two weeks.
Gap Prepulse Inhibition of the Acoustic Startle
To assess the presence of tinnitus, GPIAS testing was performed as previously described (Smit et al., 2016; van Zwieten et al., 2018). There were four conditions (a) at baseline, stimulation off, (b) at baseline, with HFS, (c) after noise-exposure, stimulation off, and (d) after noise exposure, with HFS. The rats were placed in a cylinder made of vertical aluminum bars and polyethylene floor (diameter 17 cm, height 40 cm), inside an acoustic chamber. The stimulation electrode with swivel was attached to the electrode construct. Startle force was measured with a piezo transducer (FSG15N1A, Honeywell, Canada) underneath the cylinder floor. In the ceiling of the testing chamber, a speaker was mounted (Ultrasonic Dynamic Speaker Vifa; Avisoft Bioacoustics, Berlin, Germany). Sounds were amplified (Ultrasonic power amplifier; Avisoft Bioacoustics) and calibrated (Bruel & Kjaer 2231 decibel meter with a 4191 microphone).
Background signals consisted of broadband noise (BBN) or narrow-band noise of 10, 12, 16, or 20 kHz at 75 dB. The startle stimulus was a 20 ms long 115 dB peak equivalent sound pressure level (peSPL) BBN burst. In gap trials, there was a silent gap of 50 ms, prior (100 ms) to the startle stimulus. For each of the different background signals, 10 gap trials and 10 startle-only trials were presented with a random variable stimulus interval of 20 ± 5 s. The gap:no-gap ratio was calculated by dividing the amplitude of each gap startle by the corresponding mean of no-gap startles. For each condition, two complete sessions were performed on separate days, and the mean of these gap:no-gap ratios was used for further analysis. Responses that contained too disturbing (moving) artifacts were excluded from analysis. Prior to every session, subjects were acclimatized for 5 min in the startle chamber, followed by 10 startle-trials in order to habituate the startle response. Furthermore, one complete session was performed at the start of the experiment for habituation to the testing procedure.
Auditory Brainstem Responses
Hearing thresholds were assessed before noise trauma and two weeks after noise trauma using ABR measurements. Furthermore, hearing thresholds during HFS were assessed before noise trauma. The exact procedure is described elsewhere (Smit, Jahanshahi, Janssen, Stokroos, & Temel, 2017). Briefly, subjects were anesthetized (see protocol earlier) and placed in a sound-attenuating Faraday cage. Cables were connected to the sockets of the recording electrodes in the construct on the animal’s head and a ground electrode was placed on the left front paw. One thousand 5 ms tone bursts of 10, 12, 16, 20, 24, and 32 kHz and a cos2 rise and fall filter were created with Matlab and presented unilaterally with a frequency of 50 Hz at decreasing intensities from 110 to 0 dB peSPL with steps of 10 dB. The contralateral ear was plugged with clay. Auditory stimuli were calibrated (Bruel & Kjaer 2231 decibel meter with a 4191 microphone) and digitally triggered. ABRs were recorded in LabChart Pro 7 (ADInstruments, Castle Hill, Australia), and raw data were imported into Matlab. The evoked responses were amplified 100,000 times, band-pass filtered (300–3000 Hz), and averaged. Data containing DBS artifacts were automatically removed based on a peak-detection analysis using a manual depicted maximal baseline value. The auditory threshold was defined as the lowest decibel level (peSPL) of the stimuli that produced a distinctive ABR, in which at least two peaks (positive or negative) had to be clearly visible (Figure 4(b)).
Gap-prepulse inhibition of the acoustic startle reflex paradigm for tinnitus assessment before and after exposure to the 16 kHz tone, during stimulation off and HFS. Notice the increased gap:no-gap ratio after noise trauma at 16 kHz background noise. HFS of the DCN caused a decrease of the gap:no-gap ratio. Data are presented as Tukey boxplots. *p < .05.
Tissue Collection
Subjects were divided into sham (n = 5) and HFS groups (n = 6). For postmortem analysis, one noise-exposed animal who completed the whole protocol but received sham stimulation, was added. Two hours prior to transcardial perfusion, the HFS group received HFS for 60 min, while the sham rats were only connected to the stimulation cable without stimulation. This was followed by 60 min of rest in their normal cages. Afterwards, rats were anesthetized with an overdose pentobarbital and perfusion-fixation was performed with Tyrode solution (0.1 M) followed by fixative containing 4% paraformaldehyde, 15% picric acid, and 0.05% glutaraldehyde in 0.1 M phosphate buffer (pH 7.6). Brains were collected and postfixed overnight in paraformaldehyde at 4℃ and subsequently in 1% NaN3 at 4℃ for long-term storage. Brains were cut serially on a vibratome (Leica®, Wetzlar, Germany) into 30 -µm-thick coronal sections while embedded in 10% gelatin (Sigma-Aldrich, Zwijndrecht, the Netherlands).
Histological and Immunohistochemical Staining
For electrode verification, sections containing the electrode trajectories were processed for a cresyl violet (Nissl) staining. For c-Fos immunohistochemistry, sections were incubated for two nights with polyclonal rabbit anti-c-Fos primary antibody (1:500; Santa Cruz Biotechnology Inc, Santa Cruz, USA) followed by biotinylated donkey anti-rabbit secondary antibody (1:400; Jackson Immunoresearch Laboratories Inc., Westgrove, USA) and avidin-biotin peroxidase complex (1:800; Elite ABC-kit, Vectastain®, Burlingame, CA, USA). The staining was visualized by 3,3′-diaminobenzidine combined with NiCl2 intensification.
Quantitative Cell Counting
The immunohistochemically stained sections were used to evaluate total number of c-Fos positive cells within the MGB, IC, and A1. Stereological quantification was carried out with a stereological computer microscopy system (Stereo Investigator, Microbrightfield Bioscience, Williston, VT, USA). In all sections, MGB (6 sections per rat), IC (5 sections per rat), and A1 (10 sections per rat) were delineated, and total number of c-Fos positive cells was estimated with the optical fractionator probe (Schmitz & Hof, 2000, 2005; West, Slomianka, & Gundersen, 1991). If questionable, boundaries of brain areas were verified in corresponding Nissl-stained sections. Established stereological counting methods have been described previously (Temel et al., 2006).
Statistical Analysis
Because of our small sample size, nonparametric tests were used for statistical analysis. For analysis of GPIAS test and ABRs, Wilcoxon signed rank tests were performed. For c-Fos, left and right hemispheres were compared using Wilcoxon signed rank test. The effect of stimulation on the amount of c-Fos positive cells was assessed using Mann–Whitney U test. If applicable, Holm-Bonferroni corrected p values are presented. p values < .05 were considered significant. All calculations were performed with SPSS (version 22.0 for Mac, SPSS, Chicago, IL, USA).
Results
Electrode Localization
In all animals, electrode tips were localized in Nissl stained sections. Exact localization of electrode tips is presented in Figure 2. No histological damage was observed at microscopic level.
Gap Prepulse Inhibition of the Acoustic Startle
GPIAS testing results are presented in Figure 3. One animal lost the electrode construct and was therefore excluded from GPIAS analysis. Only at 16 kHz background noise, gap:no-gap ratios were significantly increased after noise exposure (Z = −2.668, p = .023). This finding is similar to results in our previous experiments (Smit et al., 2016; van Zwieten et al., 2018). After noise exposure, the gap:no-gap ratio decreased during HFS in the 16 kHz background sound (Z = −2.310, p = .042). At baseline, there was no effect of HFS (p > .05 for all frequencies).
Auditory Brainstem Response
Consistent with previous experiments (Smit et al., 2016; van Zwieten et al., 2018), ABR thresholds (Figure 4) were significantly higher after noise trauma compared to baseline in the traumatized (ipsilateral) side along all frequencies (Z = −2.522, p = .011, Z = −2.687, p = .007, Z = −2.716, p = .007, Z = −2.539, p = .011, Z = −2.716, p = .007, Z = −2.555, p = .011, for 10, 12, 16, 20, 24, 32 kHz, respectively). In the contralateral side, hearing thresholds were not increased after noise exposure (p > .05 for all frequencies). Hearing thresholds during HFS could not be determined due to disturbing unspecified artifacts in 13 out of 42 recordings. These recordings were therefore excluded from analyses. HFS did not change hearing thresholds (p > .05 for all frequencies) in seven subjects that were recorded during HFS.
(a) Auditory thresholds measured with auditory brainstem responses at baseline without HFS (round, solid), during HFS (diamond, dashed-dotted) and after noise exposure in the traumatized ear (square, dashed) and contralateral ear (triangle, dotted). Hearing thresholds at baseline during HFS auditory thresholds are presented as means ± SEM. *p < .05. (b) Example of a recorded auditory brainstem response with an auditory threshold of 50 dB peSPL.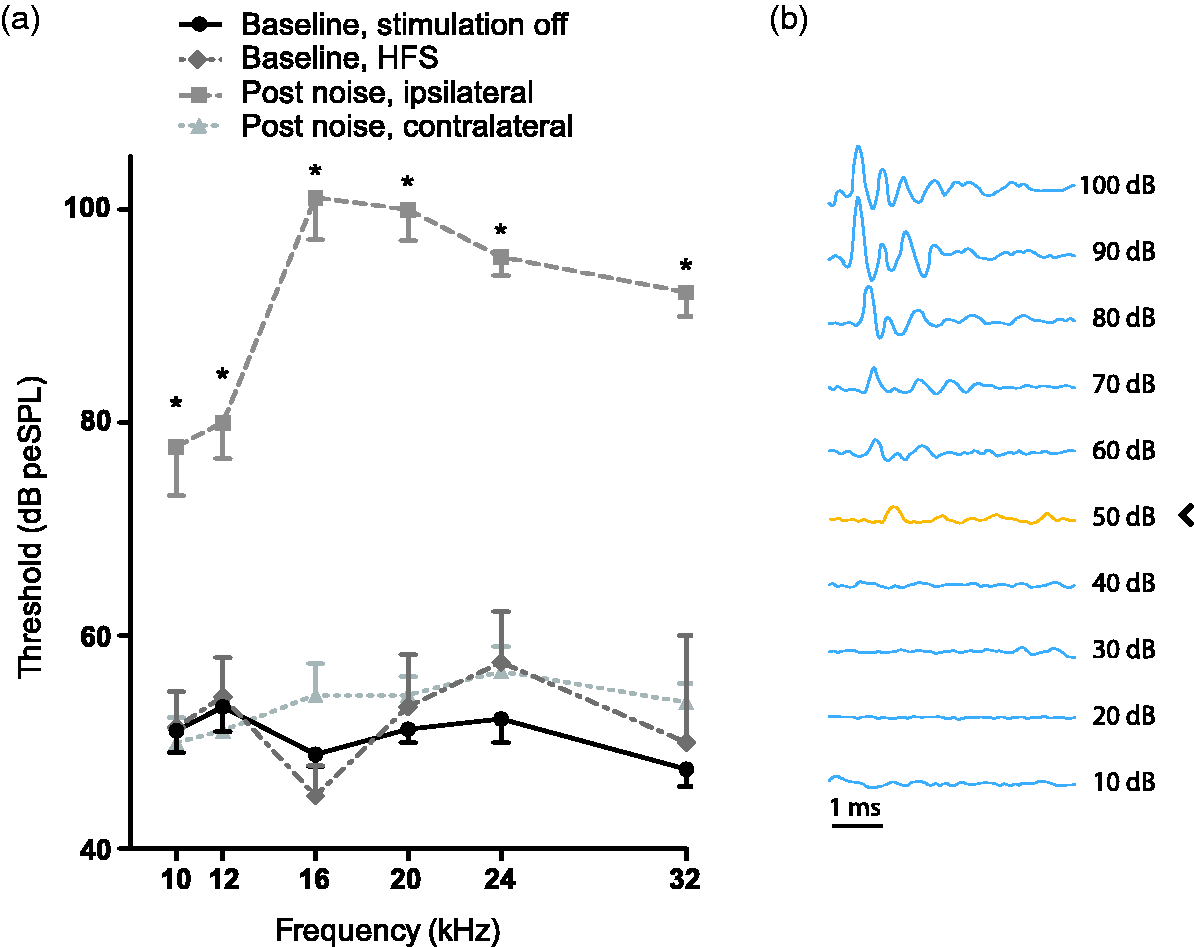
C-Fos
No significant effects of HFS on the total number of c-Fos positive cells in the MGB, IC, and A1 (p > .05 for all areas) were found (Figure 5). Noise trauma was applied in all animals in the right ear. There were significantly less c-Fos positive cells in the left compared to right IC in both sham (Z = −2.023, p = .043) and stimulated (Z = −2.201, p = .028) groups. In MGB, c-Fos expression was lower in the right side in the sham group only (Z = −2.023, p = .043). In A1, there was no difference between left and right in sham (Z = −0.674, p = .500) and stimulated (Z = −1.782, p = .075) groups.
Effect of HFS on c-Fos neural activity. (a) Total number of c-Fos positive cells in the IC, MGB, and A1 in both hemispheres of sham (stimulation off) and HFS groups. Data are presented as mean ± SEM. (b) Representative micrographs of c-Fos immunohistochemical staining of 30 -µm-thick sections of a nonstimulated animal, showing the IC (I–II), MGB (III–IV), and A1 (V–VI) in both hemispheres. IC = inferior colliculus; MGB = medial geniculate body of the thalamus; A1 = primary auditory cortex; HFS = high-frequency stimulation.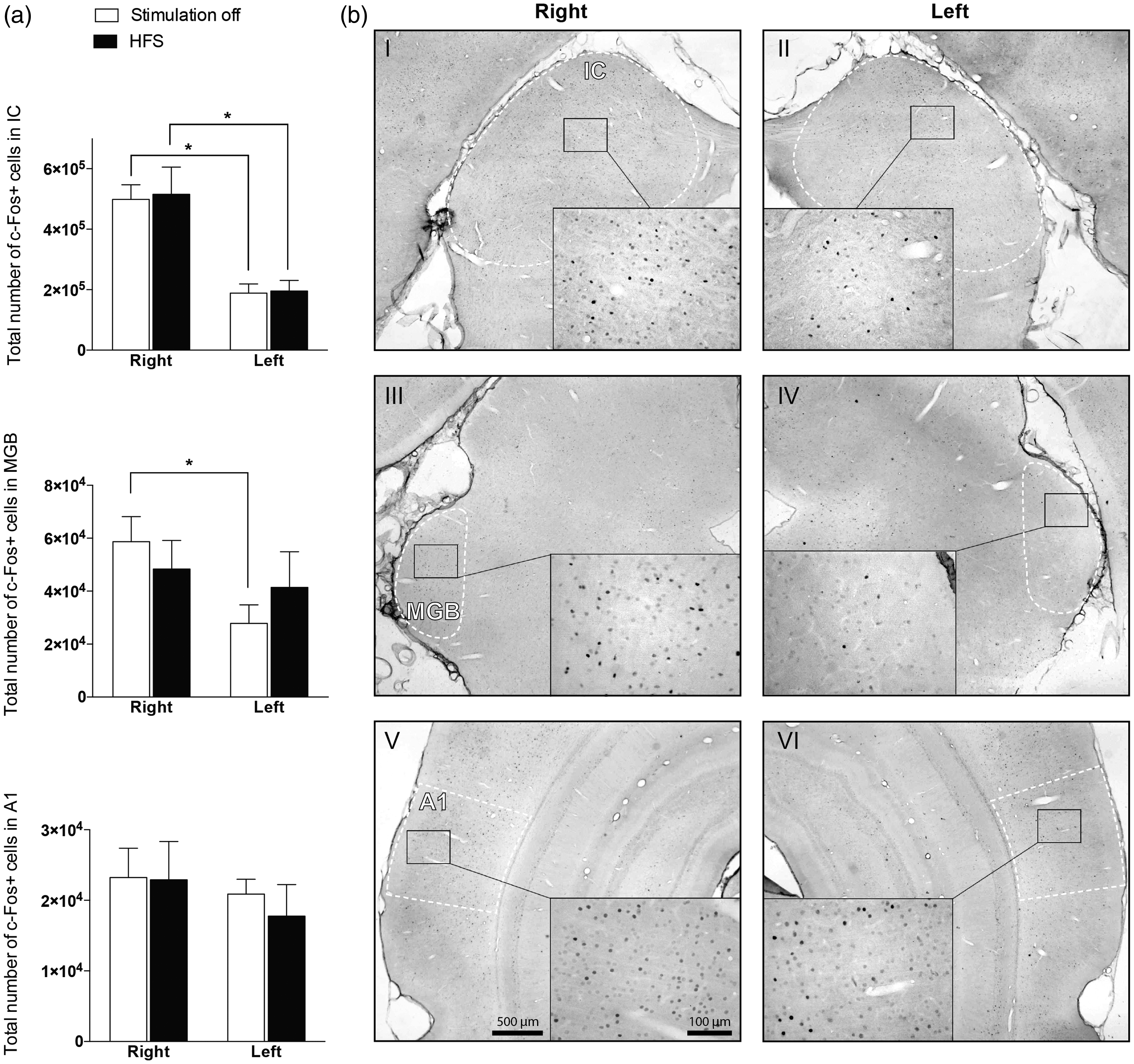
Discussion
In this study, we demonstrated that bilateral HFS of the DCN suppressed tinnitus-like behavior in a noise-exposed animal model of tinnitus. HFS did not increase ABR hearing thresholds. There was no effect of HFS on neural activity within the IC, MGB, and A1, measured by c-Fos immunoreactivity. Furthermore, unilateral noise exposure was associated with a contralateral decrease in c-Fos expression in the IC and MGB, but not in A1.
Effect of HFS on Neuronal Activation
Neuronal expression of the immediate early gene c-Fos is a valuable marker of brain activity with cellular resolution. Previously, it has been shown that electrical stimulation of the cochlea with 50 Hz as well as acoustic stimulation causes an increase in Fos expression in the central auditory system, especially in neonatally deafened rats (Jakob, Doring, & Illing, 2015). These results suggest that cochlear and acoustic stimulation restore input and could herewith generate sound perception. In this study, DCN-HFS did not induce any changes in c-Fos expression in the IC, MGB, and A1. Thus, the effect of DCN-HFS cannot be explained by a simple restoration of the lack of input or an inhibitory or excitatory effect on neuronal activity in the network. More likely, DCN-HFS disrupts pathological patterns of activity or temporal coherence, which results in restoration of information flow and blockage of the relay of abnormal signals. However, it should be noted that c-Fos alone might not be a reliable readout, since changes in cell activity could take place during HFS while overall c-Fos activation remains constant.
Electrical Stimulation of the Central Auditory Pathway for Tinnitus Suppression
Here, we showed a beneficial effect of DCN-HFS on tinnitus-like behavior. The effect of the prepulse gap on the startle response was restored during HFS at the believed tinnitus-pitch, which was the 16 kHz background sound.
In a clinical study in neurofibromatosis type 2 patients, direct electrical stimulation of the DCN with ABI suppressed tinnitus in six out of seven patients who used the device on a daily basis (Soussi & Otto, 1994). The electrical stimuli of ABI are analogues of acoustic waveforms, band-pass filtered between 250 and 4000 Hz, with the purpose to restore hearing. Despite these encouraging results, it remains questionable if and how the results of this study can be extrapolated to potential clinical use of electrical DCN stimulation for tinnitus suppression. First, only one patient experienced complete suppression of tinnitus. Second, the patient group in this study was generally not representative for patients with severe refractory tinnitus. These particular patients did not have a functioning auditory nerve, and the primary goal of the ABI was to restore auditory sensations, not to suppress tinnitus.
A beneficial effect of DBS in the DCN on tinnitus-like-behavior has been confirmed in a noise-exposed rat model for tinnitus (Luo et al., 2012). However, in this preclinical study, a low-frequency stimulation paradigm of 10 Hz was used, and mechanisms underlying this suppression remain unclear. Previously, we have reported that HFS of the IC and MGB suppresses tinnitus in rats as well (Smit et al., 2016; van Zwieten et al., 2018). Interestingly, we did not find beneficial effects of low-frequency stimulation of the MGB in tinnitus. To our knowledge, the positive effect of HFS of the DCN on tinnitus has not been reported before.
The positive effect of HFS in three different targets within the classical auditory pathway on tinnitus-like behavior is noteworthy. Interestingly, in Parkinson’s disease, a positive effect can also be obtained when different targets within the cortico-basal ganglia thalamic loop are stimulated (Chiken & Nambu, 2014; Vitek, 2002). It can be postulated that tinnitus is also a network disorder, and disrupting the pathological information flow within the auditory network at any structure may suppress tinnitus (van Zwieten et al., 2016). The exact mechanisms underlying the effects of high-frequency brain stimulation remain unclear. Current research has revealed that the effect of HFS-DBS is beyond simple excitation or inhibition of the local neural elements at the stimulation target. These mechanisms are rather multifactorial and include immediate neuromodulatory effects, synaptic plasticity, and long-term neuronal reorganization (Ashkan, Rogers, Bergman, & Ughratdar, 2017).
Up to now, it remains unclear which subcortical target can be stimulated with the least side effects and risks, and on the other hand best tinnitus reduction. Based on the results in this study, DCN-HFS might have potential to suppress tinnitus loudness and could therefore be a therapeutic option to treat tinnitus. Considering a bottom-up approach, the DCN is the first central region that can be stimulated. Since this area is less modulated in comparison to more upstream targets, it hypothetically has a higher potential to suppress pathological activity. However, it has been suggested that hyperexcitability in central neurons increases with time, which eventually changes spontaneous firing to intrinsically generated pulses in upstream nuclei (Mulders & Robertson, 2011). Contemplating this progressive centralization and the undetermined rostral effects of DCN-HFS, it is uncertain if DCN stimulation also has a positive effect on long standing tinnitus.
The surgical approach to the DCN is not straightforward. The DCN has been stimulated in humans before with an ABI to improve hearing function. This surgical procedure is invasive and complex, and to date has only been performed in patients without a functioning auditory nerve. From a neurosurgical point of view, the MGB of the thalamus is best accessible via stereotaxy and might therefore be the target of first choice. DCN modulation should not be ruled out as a treatment option, as fast progress is made in the development of noninvasive neuromodulation techniques. Examples are focused ultrasound (Lipsman et al., 2013) and magnetothermal stimulation (Chen, Romero, Christiansen, Mohr, & Anikeeva, 2015).
The Effect of Unilateral Noise Trauma on Neuronal Activity and Hearing
In all subjects, hearing thresholds were increased unilaterally after acoustic over exposure in the right ear. A reduction in c-Fos expression was found in the left compared to right IC (both groups) and MGB (sham group), but not in A1. A lack of input due to hearing loss can cause a lower baseline c-Fos (Harrison & Negandhi, 2012; Pernia et al., 2017). C-Fos is a general marker for neuronal activity, and a decrease in c-Fos expression can both mean a reduction in inhibitory or excitatory neurotransmission.
Interestingly, a left-right difference was not found in A1. This finding is consistent with the results of a study in which they only found a short-term effect of hearing loss on c-Fos expression, but no long-term effect (Pernia et al., 2017). There was a relative increased c-Fos expression in the traumatized side, which might reflect increased spontaneous activity in A1. It is thought that tinnitus-related activation arises within the thalamocortical loop of the auditory system, ultimately leading to increased spontaneous activity in auditory cortices, more than in downstream structures of the auditory pathway (Wallhausser-Franke et al., 2003). A1 is a more upstream structure, in which the effects of noise trauma are more modulated, due to connectivity with other brain regions. This makes subcortical structures more suitable for neuromodulation. A limitation of this study is that there was no control group, so immunohistochemical findings could not be compared to a healthy state.
The potential confounding effect of hearing loss cannot be ruled out in this study. However, it should be noted that gap:no-gap ratios were only found in the 16 kHz background frequency while unilateral hearing thresholds were increased in all frequency bands. These findings suggest that increased gap:no-gap ratios reflect actual tinnitus instead of a hearing loss-induced temporal processing deficit.
Hearing and DCN-HFS
A significant concern when stimulating the central auditory pathway is the effect on hearing. In our study, ABR showed that there was no deterioration of hearing during HFS. In a previous study, bilateral DCN lesions did not increase hearing thresholds as assessed with ABR (Brozoski et al., 2012). It seems that a functional DCN is not obligatory for normal hearing thresholds. Furthermore, HFS of other auditory brain structures such as the IC seems not to impair hearing in rats (Smit et al., 2017).
It is known from clinical studies that electrical stimulation of the DCN (Schwartz, Otto, Shannon, Hitselberger, & Brackmann, 2008) and IC (Lim, Lenarz, & Lenarz, 2009) is able to induce auditory perceptions. Electrical stimulation of the DCN with low frequency pulses (10 pps) induces neural activation in auditory cortex (Zhang & Zhang, 2010). It is unknown if stimulation with continuous HFS will cause auditory perceptions as well.
Conclusion
Neuromodulation is a promising treatment modality to alleviate tinnitus. Here, we showed that DCN-HFS reduces tinnitus-like behavior in an animal model without increasing ABR hearing thresholds. If seen as a network disorder, the DCN might be the first station that can be targeted with HFS to disrupt the pathological tinnitus signals. Although DCN-HFS is effective, many questions remain unanswered. We therefore encourage neuroscientists, medical specialists, and audiologists to further explore neuromodulation for the treatment of tinnitus both preclinically and clinically.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
