Abstract
Affective processing appears to be altered in tinnitus, and the condition is to a large extent characterized by the emotional reaction to the phantom sound. Psychophysiological models of tinnitus and supporting brain imaging studies have suggested a role for the limbic system in the emergence and maintenance of tinnitus. It is not clear whether the tinnitus-related changes in these systems are specific for tinnitus only, or whether they affect emotional processing more generally. In this study, we aimed to quantify possible deviations in affective processing in tinnitus patients by behavioral and physiological measures. Tinnitus patients rated the valence and arousal of sounds from the International Affective Digitized Sounds database. Sounds were chosen based on the normative valence ratings, that is, negative, neutral, or positive. The individual autonomic response was measured simultaneously with pupillometry. We found that the subjective ratings of the sounds by tinnitus patients differed significantly from the normative ratings. The difference was most pronounced for positive sounds, where sounds were rated lower on both valence and arousal scales. Negative and neutral sounds were rated differently only for arousal. Pupil measurements paralleled the behavioral results, showing a dampened response to positive sounds. Taken together, our findings suggest that affective processing is altered in tinnitus patients. The results are in line with earlier studies in depressed patients, which have provided evidence in favor of the so-called positive attenuation hypothesis of depression. Thus, the current results highlight the close link between tinnitus and depression.
Introduction
Tinnitus is a hearing disorder generally defined as the perception of a phantom sound perceived by the affected person in the absence of measurable external stimuli and may affect anyone at any time during the lifetime (Jastreboff, 1990). Despite intensive research efforts, the pathogenesis of tinnitus is still poorly understood. Different forms of tinnitus have been described and it is now known that tinnitus involves both peripheral and central auditory pathways. Tinnitus is further often accompanied by comorbid depression, anxiety and stress (Dobie, 2003). The complex pathology and lack of a measurable physiological marker of the phantom sound make tinnitus a debilitating condition that is difficult to stage and challenging to follow up regarding treatment responses.
The whole phenomenon of tinnitus not only involves the phantom sound but also the affective reaction to it. Previous research has suggested that the limbic system may attribute emotional significance to sounds, thus determining the individual’s emotional response to stimuli (K. S. Kraus & Canlon, 2012; Kumar, von Kriegstein, Friston, & Griffiths, 2012). Structural and functional alterations in the limbic system have also been associated with tinnitus (Carpenter-Thompson, Akrofi, Schmidt, Dolcos, & Husain, 2014; Golm, Schmidt-Samoa, Dechent, & Kröner-Herwig, 2013; Maudoux et al., 2012; Mühlau et al., 2006; Vanneste et al., 2010). This interaction with affective processing has also been incorporated into models of tinnitus. In Jastreboff’s (1990) neurophysiological model of tinnitus, the affective component enhances the tinnitus percept, thus creating a vicious circle eventually leading to gradual worsening of symptoms. The model also suggests that the interplay between pathological auditory activity and associated emotional reactions is mediated by the limbic system. Rauschecker, Leaver, and Mühlau (2010) also emphasized the role of the limbic system in tinnitus. Despite some differences between these models, they formalize and highlight the central role of the emotional aspects of auditory processing that take place in tinnitus. One unresolved question in emotional processing associated with tinnitus is whether the affective mechanisms that maintain tinnitus are specific to tinnitus, or whether these mechanisms influence all sounds in general, making some individuals more susceptible to developing disturbing tinnitus than others. Investigating the reactions to emotion-carrying sounds may provide additional information about how the auditory system processes sounds in tinnitus patients.
Here, we investigated autonomic activation in response to affective sounds in tinnitus patients by measuring changes in pupil diameter and also studied the effects of transcutaneous vagal nerve stimulation (tVNS) on that sound-evoked activity. The pupil diameter is known to closely reflect the balance of the autonomous nerve system because its diameter is determined by the relative activity of two iris muscles, a sphincter, and a dilator controlled by the opposing autonomic nerves. Whereas pupil constriction is mainly driven by parasympathetic activity, pupil dilation is predominantly triggered by the sympathetic pathway (Eckstein, Guerra-Carrillo, Miller Singley, & Bunge, 2017). Accordingly, several studies have demonstrated that emotional arousal during affective picture viewing or auditory emotional stimulation is associated with enlarged pupil diameter (Bradley, Miccoli, Escrig, & Lang, 2008; Partala & Surakka, 2003). In consequence, processes that are invoked to modulate emotional responses should alter pupillary responses as well, and it has shown that pupil dilation can be influenced by cognitive factors (Van Der Meer et al., 2010). The rationale for applying VNS is based on its therapeutic application in difficult-to-treat epilepsy starting already 20 years ago. The noninvasive variant, tVNS, aims at delivering the same effect as invasive VNS without the need for an implanted stimulator. tVNS targets the auricular branch of the vagus nerve (ABVN) in the outer ear. It has been demonstrated by functional magnetic resonance imaging (fMRI) and electroencephalography (EEG) recordings that tVNS activates the central vagal pathways similar to VNS implants (Dietrich et al., 2008; T. Kraus et al., 2007; Polak et al., 2009). Indeed, in retrospective studies (Clancy et al., 2014a; Ylikoski et al., 2017), it was shown that tVNS can induce a shift in ANS function from sympathetic preponderance toward parasympathetic predominance. Recent studies have observed a positive association between tinnitus-related distress and sympathetic tone (Datzov et al., 1999) and a negative association between tinnitus-related distress and parasympathetic tone (Choi et al., 2013). Also, successful suppression of tinnitus has been associated with an increase in parasympathetic tone (Matsushima et al., 1996). tVNS might offer a targeted, patient-friendly, and low-cost therapeutic tool for tinnitus patients with sympatho-vagal imbalance. Others have shown that invasive VNS at clinically significant levels increases the resting pupil diameter (Desbeaumes Jodoin, Lespérance, Nguyen, Fournier-Gosselin, & Richer, 2015). We were interested in whether tVNS could induce measurable, acute changes in pupil responses.
There are some existing studies that have investigated tinnitus using functional imaging and affective stimuli. For example, Durai, O’Keeffe, and Searchfield (2017) studied patients’ responses to short-term emotional changes along valence and arousal and they revealed that the negative valence dimension of emotion is associated with an increased tinnitus magnitude judgment specifically when given as an auditory stimulus. Schlee, Weisz, Bertrand, Hartmann, and Elbert (2008) found that unpleasant tones stimulated the tinnitus network to a greater extent than the control sounds. Mirz (2000) used an unpleasant tinnitus-like tone to investigate how tinnitus may alter brain function in healthy adults. They found increased activation of limbic regions including the amygdala, parahippocampus, and insula for the unpleasant tinnitus-like tones in healthy volunteers. Carpenter-Thompson et al. (2014) observed differential levels of neural activation within auditory, frontal, and limbic networks among individuals with tinnitus compared with nontinnitus controls upon presentation of affective sounds. Similar results were obtained in a follow-up study by Carpenter-Thompson et al. (2015), where they were able to show differences in the same regions between subjects with recent-onset tinnitus, and subjects with long-lasting tinnitus. Also Davies et al. (2017) found a trend for decreased amygdalar activation in response to emotional sounds, when comparing tinnitus and non-tinnitus groups. The differential response in tinnitus patients in these studies was given an objective correlate using brain imaging, which has indicated functional differences in the autonomous nervous system in tinnitus patients.
Effects of hedonic valence and emotional arousal on pupillary responses have been studied indicating that the pupil diameter may reflect a sensitive parameter for emotional arousal and autonomic activation (Bradley et al., 2008), similarly to fMRI. The choice of utilizing pupillometry instead of fMRI is a trade-off between ease of administration and the degree of specificity in terms of the underlying neural processes. Thus, we sought to make use of the existing knowledge and the widely used International Affective Digitized Sounds (IADS) database (Bradley & Lang, 2007) comprising of sounds of varying emotional content. Here, we hypothesized that tinnitus patients would show larger pupil dilation to negative and positive sounds compared with neutral sounds, as shown by Partala and Surakka (2003), and that tVNS could induce measurable, acute changes in pupil responses in tinnitus patients. Behavioral responses and pupillometry were collected from tinnitus patients in order to subjectively and objectively evaluate affective sound perception in this group. Our data show that affective processing is indeed altered in tinnitus patients, and that the same effect can be seen both in the subjective ratings of the sounds, as well as in the pupil responses.
Methods
Subjects
Patient Characteristics.

Note. THI = tinnitus handicap inventory; N/A = not available, see section Subjects, under Methods.
Sound Stimuli
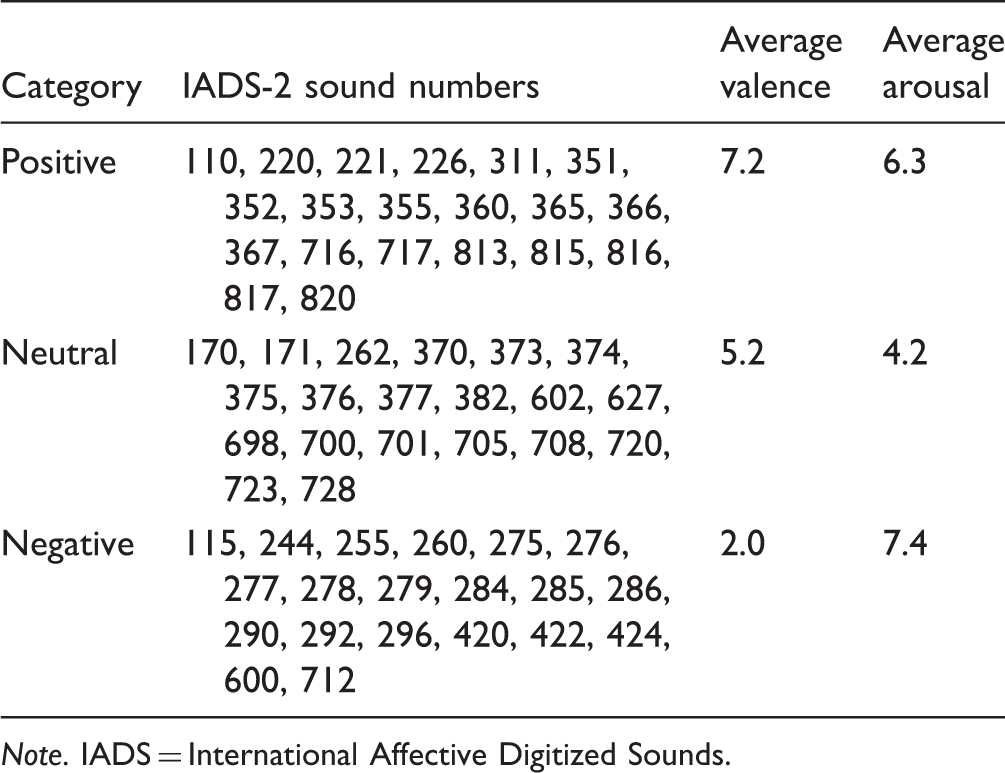
In total, 60 sounds from the IADS-2 database (Bradley & Lang, 2007; Stevenson & James, 2008) were used in the study. The samples were chosen so that they would make up three affective categories based on the normative ratings of the sounds. The three categories were: negative (20 sounds with the lowest valence rating among sounds with an arousal rating below 5), positive (20 sounds with the highest valence ratings amount sounds with an arousal rating above 5), and neutral (20 sounds with the lowest arousal ratings among sounds with a valence rating between 4 and 6). Erotic sounds were excluded from this study. Table 2 shows the selected sounds and average normative ratings for the three categories. These average values are also shown in Figure 2 in gray. Sounds were presented at a comfortable level via AudioTehnica ATH-M50 headphones connected to the headphone output of an Apple MacBook Pro.
Schematic of the experimental set-up. Average SAM ratings for valence (left) and arousal (right), for each sound category in green. The normative values for the stimuli used in this study are shown in gray. Data shown as mean ± SD (n = 10). IADS Sounds Used in This Study and the Average Normative Values for Valence and Arousal, as Reported in the IADS Reference Document (Bradley & Lang, 2007). Note. IADS = International Affective Digitized Sounds.

Eye Tracking
Pupil diameter, expressed in number of raw pixels, from both eyes was measured with a head-mounted SMI 3D VOG eye tracking system at a sampling rate of 50 Hz. Eye tracking was performed continuously throughout the experiment, simultaneously with the experimental procedures explained in the next section.
Experimental Procedure
The subjects were seated in an upright position in front of a computer screen. The auditory stimuli and visual instructions were presented using the PsychoPy toolbox (Peirce, 2007) running on an Apple MacBook Pro, to which the external screen and keyboard for response collection were connected.
For each individual sound, the following trial structure was employed (see also Figure 1):
A get ready for the next sound text was shown on the screen for 3 s. A fixation cross was shown in the middle of the screen. After 4 to 6 s (random interval, uniform distribution), the sound sample was played. The fixation cross was shown up to 5 s after the end of the sound. A valence self-assessment manikin (SAM) scale (Bradley & Lang, 1994) was presented and the subject was instructed to enter their rating (a value from 1 to 9). An arousal SAM scale was shown and the subject was instructed to enter their rating (a value from 1 to 9). A blank screen was shown for 3 s before moving to the next trial.
The experiment was split into two separate blocks, both containing 10 sounds from each category (in total 30 sounds in one block). The sounds in each category were randomly split into the two blocks, individually for each subject. In other words, the order in which the sounds were presented to each subject was completely randomized, with the constraint that the number of sounds from each category was balanced between the two experimental blocks. This step was taken to avoid any systematic bias between the recording blocks.
During one of the blocks, the subjects received transcutaneous vagus nerve stimulation (tVNS), administered to the left tragus at a level exceeding the tactile threshold but below nociceptive threshold. There was no electrical stimulation in the other block. The block in which tVNS was applied was chosen randomly for each subject.
Analysis
For each response, the subject’s own SAM rating was subtracted from the normative value from the IADS-2 database to get a measure of deviation from normative values. Averaging over individual responses for each rating scale (arousal and valence) and sound category (positive, neutral, and negative) resulted in six values of average deviation per subject. On a group level, a separate t statistic was calculated for each of these six variables to test the null hypothesis that the average deviation from normative values was zero, the alternative hypothesis being that the tinnitus patients’ ratings differed from normative ratings.
Pupil size time series was averaged across both eyes, after which the signal was smoothed using a 15-sample median filter. Eye blinking appeared in the data as missing values, and imputation was performed by replacing missing values with the last valid value. Trials with more than 20% of blinking were rejected and not included in further analysis. Only 1.8% of all trials were rejected, and the amount of blinking did not differ between sound categories. The continuous pupil recording was split into 13-s epochs spanning from 2 s prior to sound onset to 5 s after the 6-s stimulus offset. As a final preprocessing step, pupil dilation was expressed as a relative value with respect to the average of a 2-s baseline period prior to sound onset. Pupil responses were compared across the three sound categories by calculating an average dilation between 2- and 8-s poststimulus. For each subject, the averaging was performed across time and across all sounds in belonging to the same category, in total resulting in three values for each individual. Pairwise t tests were performed between the different categories to test the null hypothesis that the average pupil dilation did not differ between sound categories, and the alternative hypothesis being that the pupil responses would be larger or smaller depending on the sound category.
As there was no effect of tVNS either on the subjective ratings (valence: t = −0.76, p = .47; arousal: t = −0.55, p = .60) or pupil response characteristics (baseline: t = −0.30, p = .77; mean response dilation: t = −1.24, p = .25), results from the two experimental blocks were pooled for each subject.
Results
Analysis of the SAM ratings indicated that the tinnitus patients deviated from normative values (valence: −0.54, t = −3.6, p < .01; arousal: −1.6; t = −4.1, p < .01). Looking at valence ratings by sound category, only one difference was observed for sounds in the “positive” category, which were rated lower than the normative values (mean deviation: −1.45; t = −5.37, p < .001). For arousal, the sounds were rated below the normative values across all categories (“positive”-mean deviation: −2.16; t = −5.04, p < .001; “neutral”-mean deviation: −1.32; t = −3.86, p < .01; “neutral”-mean deviation: −1.19; t = −2.57, p < .05). The group-level average ratings and their relation to the normative values are illustrated in Figure 2. The distribution of the average individual deviation from normative scores is shown in Figure 3.
Individual averages of deviation from the normative scores, shown for each sound category. (N.S.: p > .05, *p < .05, **p < .01, ***p < .001.).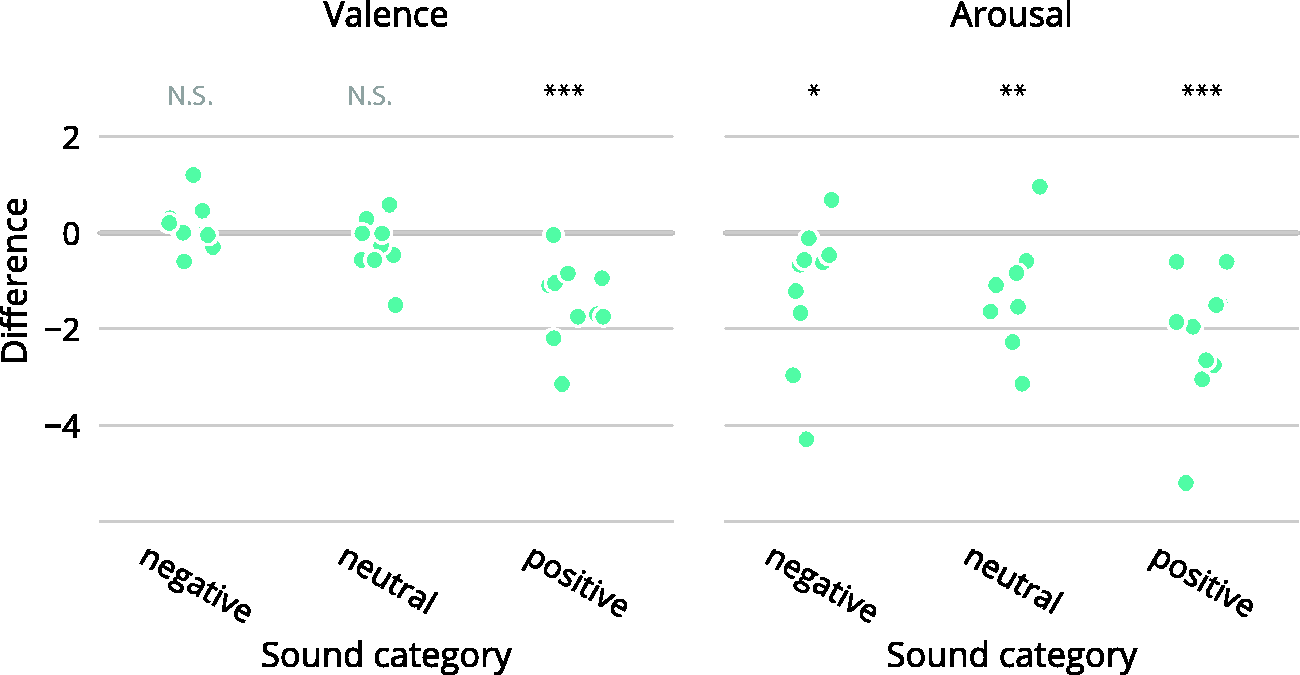
The average pupil response traces are shown in Figure 4 for each sound category. Comparing the average values for the interval between 2 and 8 s (Figure 5), sounds in the negative category elicited a significantly larger response compared with neutral (t = 4.6, p < .01) and positive sounds (t = 3.0, p < .05). No statistically significant difference was found between responses to neutral and positive sounds.
Average time courses of sound-elicited pupil dilation for the three sound categories. Pupil size is shown relative to the 2-s prestimulus baseline. Sound onset is at time point 0. Lines represent mean values (n = 10). Individual average pupil dilation for the interval between 2 to 8 s poststimulus for the three sound categories. (*p < .05, **p < .01).

Discussion
The current results show that tinnitus patients differ from normative ratings in their subjective responses to positive emotion-carrying sounds but not for neutral or negative sounds. This finding is also supported by pupillometry, which shows the same response attenuation for positive sounds. In addition to collecting the responses to the sound samples, we also attempted to measure the influence of tVNS on the stimulus-related activity of the autonomic nervous system, but no effect of this stimulation was found in subjective ratings or pupillometry.
The subjective ratings by the tinnitus patients differed from normative ratings especially in the positive sound category, where the valence of sounds was rated significantly below those reported in the IADS reference document. This finding is in full accordance with the results reported by Fournier, Cormier, Rainville, and Hébert (2014), who compared responses between a tinnitus group and a nontinnitus control group. They found that tinnitus sufferers rated positive emotion-carrying sounds and images as less positive compared with the control group, whereas ratings of negative and neutral stimuli did not differ between the groups. The sound samples used by Fournier et al. (2014) were also from the IADS library and rated with the SAM scales. Thus, this study was able to replicate the finding related to the subjective ratings by tinnitus patients in their work. Fournier et al. also conducted heart rate variability (HRV) and skin conductance recordings as physiological measures of affective processing, whereas in this study pupillometry was used for the same purpose. They reported reduced HRV in the tinnitus group when listening to emotion-carrying sounds, but this result was presented as an average over all sound categories. Therefore, it is not possible to directly compare our findings regarding reduced pupil responses in the positive sound category with the physiological results from Fournier et al. (2014).
The current results are also in line with the study by Carpenter-Thompson et al. (2014) who found that pleasant sounds were most successful in differentiating between tinnitus and nontinnitus groups, when analyzing sound-evoked brain activity with fMRI. They also collected behavioral responses and found that both tinnitus and control groups categorized more sounds as unpleasant than as neutral or positive, even though the normative ratings of the presented sounds were equally distributed across the three categories. However, while in this study we collected numeral ratings with the SAM scale, Carpenter-Thompson et al. instructed the participants to use three discrete categories (pleasant, neutral, or unpleasant), and thus the behavioral responses are not directly comparable. Nevertheless, both studies report a decrease in the average valence of all sounds, although the interpretation of which sounds might be driving this effect differs between the studies, likely because of the methodological differences in the response collection. The underlying similarity in the reactions is better reflected in the electrophysiological findings: Carpenter-Thompson et al. found that the response of the limbic system differed between tinnitus and control subjects for the positive sounds, which is also where this study demonstrates differences between tinnitus patients and normative data, as well as a deviation from expected pupil responses (Partala & Surakka, 2003).
The diminished valence ratings of positive sounds are similar to those seen in subjects with depressive symptoms. When viewing images of positive and negative content, depressive subjects have repeatedly been shown to differ from control groups only in the positive and not in the negative or neutral categories (Berenbaum & Oltmanns, 1992; Dunn, Dalgleish, Lawrence, Cusack, & Ogilvie, 2004; Sloan, Strauss, Quirk, & Sajatovic, 1997; Sloan, Strauss, & Wisner, 2001). These previous findings have been interpreted as supporting the so-called positive attenuation hypothesis of depression, which suggests that depression is best characterized by a lack of positive affect instead of excessive negative affect (Bylsma, Morris, & Rottenberg, 2008). A dampened response to positive valence has even been shown to predict depression prospectively in a nonclinical sample (Raes, Smets, Nelis, & Schoofs, 2012). Tinnitus and depression also often cooccur (Baguley, McFerran, & Hall, 2013; Kim et al., 2015; Lockwood, Salvi, & Burkard, 2002; McKenna, Handscomb, Hoare, & Hall, 2014; Nondahl et al., 2011), and it is possible that the current results reflect the comorbid depression rather than any mechanism that would be specific only for tinnitus. Unfortunately, we did not collect data regarding the depressive symptoms of the subjects in this study, so it is not possible to disentangle the individual effects of these two phenomena in the results. However, even if this information was available, the observation that the dampening of positive affect can precede depressive symptoms could mean that the link between depression questionnaire scores and valence ratings might not be so straightforward. Nevertheless, future studies in tinnitus involving the use of affective stimuli could benefit from controlling for depression symptoms.
The normative scores for the IADS samples have been collected from a group of college students. Although statistics about the age of that study group has not been reported explicitly (Bradley & Lang, 2007), it is clear that this study group is older, and that this can have an effect on the ratings and reactions to the emotion-carrying sounds. However, previous research has shown that increasing age would be expected to have the opposite effect on valence ratings than what is demonstrated here; that is, older subjects tend to rate positive stimuli as more positive than younger subjects do (Grühn & Scheibe, 2008; Mather & Carstensen, 2005). Therefore, due to the higher age of the study group, the current results may underestimate the attenuation of positive valence.
Another interesting finding from the subjective ratings is that the arousal ratings were below normative values across all sound categories. It is not surprising that the relative decrease in arousal ratings was greatest in the positive category, where sounds were also rated lower on the valence scale. Since valence and arousal have a nonlinear relationship—high arousal for sounds rated as very pleasant or unpleasant—this effect underlines the dampening of the response to positive sounds. It is not clear, why also negative and neutral sounds were rated as less arousing, although also here the older age of this study group may have an effect through improved emotion control (Mather & Carstensen, 2005). This finding, however, suggests that tinnitus patients are not generally more reactive to affective sounds, and that possible emotional sensitization might only occur specifically in relation to the tinnitus sound. In this respect, the distress dimension of tinnitus could more resemble misophonia than hyperacusis, evidencing itself as decreased tolerance toward only certain—but not all—sounds (Jastreboff & Jastreboff, 2015).
We were not able to show an effect of tVNS on the subjective ratings or on the pupillometric results. It is possible that the duration of the applied stimulation was not sufficient, or other stimulation parameters not optimal to induce notable changes in pupil function (Badran, Mithoefer, et al., 2018). Previously, invasive VNS has been shown to induce a pupil dilation, although in that study there were no auditory or other stimuli presented during measurement (Desbeaumes Jodoin et al., 2015). In this study, we decided to study sound-evoked changes in the pupil diameter, since the onset of tVNS also induces a cutaneous sensation which during piloting was found to result in a pupil response by itself. Another plausible explanation for finding no effect of tVNS is that tVNS and pupillometry may have been targeting different parts of the autonomic nervous system, and therefore no effect could be found. The auricular branch of the vagus nerve, targeted in tVNS, is part of the parasympathetic system (Badran, Brown, et al., 2018; Badran, Dowdle, et al., 2018; De Couck et al., 2017; T. Kraus et al., 2007), whereas pupil size is controlled by both the sympathetic and parasympathetic systems (Eckstein et al., 2017; Laeng, Sirois, & Gredeback, 2012). It has been suggested that the pupil dilation in response to affective sounds mostly reflects the sympathetic nervous system activity (Bradley et al., 2008). Therefore, the possible effects of tVNS may not be directly reflected in the pupil response. Other measures of autonomic system activity with a more direct link with the parasympathetic system, such as HRV, might be better suited for monitoring the acute effects of tVNS (Clancy et al., 2014b; De Couck et al., 2017; Ylikoski et al., 2017).
To our knowledge, this is the first study investigating the pupil responses to affective sounds in tinnitus patients. Pupillometry is a relatively inexpensive and easy approach to be administered in the armamentarium of evaluation methods in this patient population. This study demonstrates that pupillometry can be considered a viable option for studying autonomic activation in tinnitus subjects. In addition to affective processing, pupillometry—by itself or combined with eye gaze information—can also be utilized in experiments designed to probe the working memory, attention, or cognitive control (Eckstein et al., 2017), which are also important lines of investigation in tinnitus (Andersson & McKenna, 2006; Roberts, Husain, & Eggermont, 2013). Furthermore, to the best of our knowledge, the study by Fournier et al. (2014) and this study are the only ones reporting behavioral responses in a series of tinnitus patients obtained with the same SAM scale as in the original IADS reference document and multiple later psychological studies in different fields. The use of a well-established scale allows comparison of findings across different studies and fields of research and has now highlighted the known association between tinnitus and depression. The robustness of the effect, shown by the replication of the result of Fournier et al. (2014), along with the strong link to earlier findings in depression make a case for further utilization of affective sounds in probing the mechanisms of tinnitus.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work has been supported by the Helsinki University Hospital Research Fund. PH has received financial support from Pivikki and Sakari Sohlberg Foundation.
