Abstract
Contamination of the environment directly impacts animal, plant, and human health, as well as agricultural production. A potential solution tested in this work incorporates the use of enzymes from fungi, manganese (Mn) peroxidase (MnP) and laccase, to degrade pollutants. Unfortunately, the MnP is too expensive to produce (more than US$24,000 per gram) to be used in remediation applications. MnP from a corn kernel biofactory can be produced in large amounts, and at <US$5 per gram, would be cost-effective in this application and resolve pollution issues. Two formulations of recombinant MnP from the corn kernel production system and commercial fungal laccase have been tested for degradation of synthetic dyes in an aqueous environment. Numerous concentrations of four synthetic azo or anthraquinone dyes were used. All dyes were partially or completely degraded by both enzymes.
Introduction
Pollution is the unfortunate byproduct of human progress. Agricultural development itself has used fungicides, herbicides, and insecticides that increase crop productivity, but also exposed the ecosystem to pollutants. Plastics have become a prominent component of marine debris. 1 Synthetic dyes became an environmental presence after their discovery by W. H. Perkins in 1856. 2 Phenolic compounds in wastewater from the petroleum industry and wood treatments are hazardous to public health. 3 Shifting economies and globalization have created opportunities for waste dumping and environmental contamination. For example, industry relocation to China has led to abandoned factory sites and contaminated soil with a combination of heavy metals and organic molecules like polycyclic aromatic hydrocarbons (PAHs). 4 The ripple effect of many human achievements has thus created a need for research into mitigating the destructive effects of pollution. While all these pollutants need to be removed, this study focused on textile dyes that pollute water.
Synthetic dyes, including azo, anthraquinone, triphenylmethane, and heterocyclic dyes are used in the food, paper, cosmetics, printing, and textile industries. 5 These dyes fulfill an important role in maintaining the level of technological development and convenience in modern societies. However, the use of dyes inevitably creates waste. Azo dyes contain one or more azo bonds, which are bonds containing two adjacent nitrogen atoms between carbon atoms, contributing to their toxicity (Figure 1). They are the most used dyes in textile processing industries. 6 Worldwide industry uses about 7105 tons of dye every year from 10,000 different types. 7 Approximately 15% of the dyes are discharged in industrial liquid waste or sewage. 7 There is a textile mill in India that has a concentration of 3600–6540 mg/L of total dissolved solids in the effluent from its dyeing unit. 2
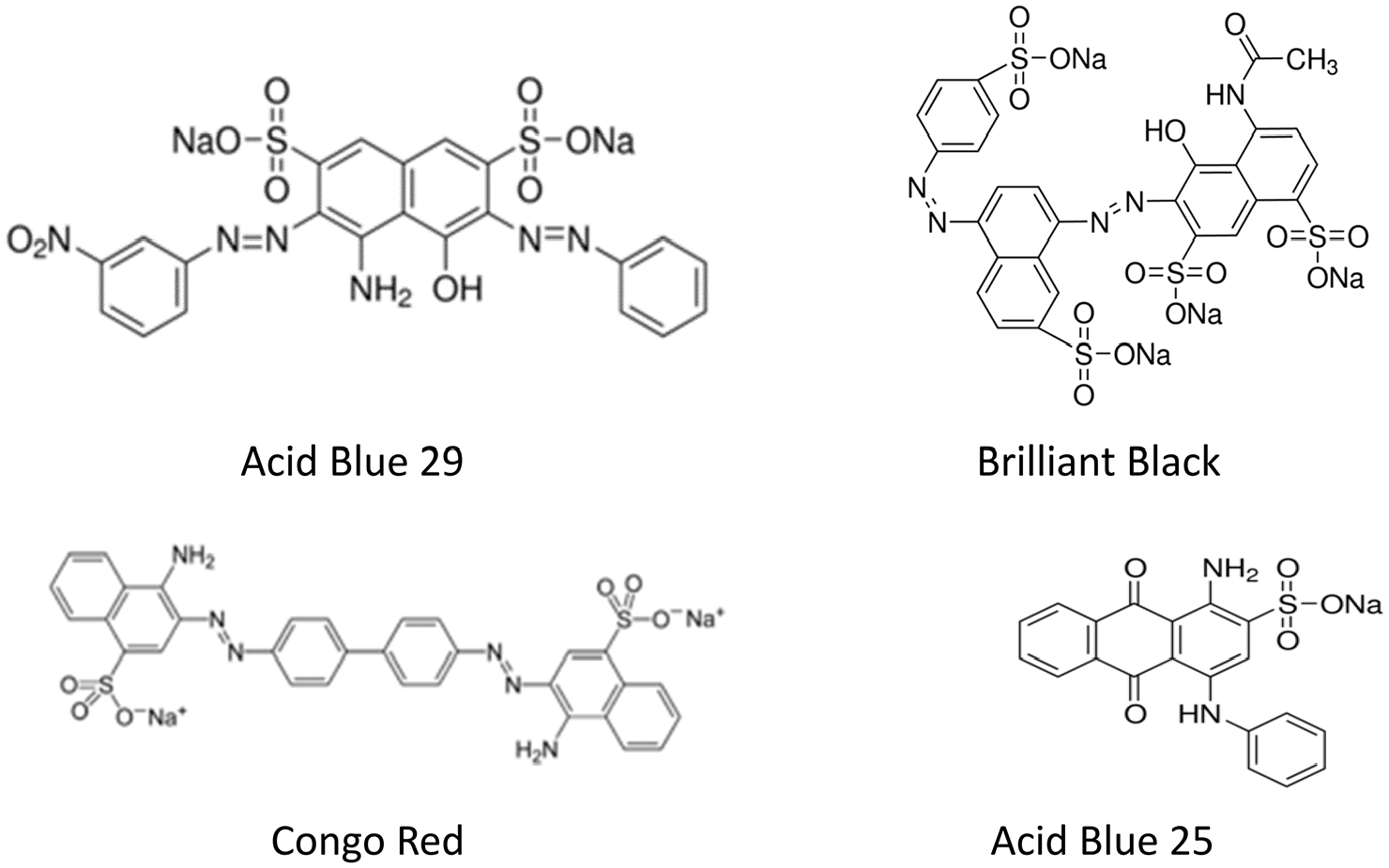
The structures of the four synthetic dyes used in the study.
Dyes have negative effects on the organisms that come into contact with them, while also indirectly affecting plant life by blocking photosynthesis. 8 Dyes released into bodies of water can be carcinogenic, mutagenic, and toxic to aquatic life. 9 The high solubility of many dyes makes them difficult to remove from water. 9 Azo dyes and their byproducts can cause tumors in humans and animals. 10 These dyes are now in major water systems, and a method of removing them is critical.
Remediation is defined as the removal of pollutants from the environment. Conventional means of physical and chemical remediation of organic compounds have limitations. The high cost of remediation, low efficiency of methods, and limitations in the concentration range of the pollutants that can be addressed by these methods are accompanied by the possible production of hazardous byproducts and incomplete removal. 11 Calcination is an example of a physical form of remediation that involves heating soil to extreme temperatures to remove pollutants, 4 a process that uses great amounts of energy and converts the pollutant from one physical form to another. However, progress in cost-efficient physical removal was discovered by using silylated clays. 12 Chemical remediation involves the use of bases, acids, inorganic compounds, and chelating agents in the removal of hazardous substances from the environment through leaching and/or precipitation. 13 The chemical remediation of soils commonly involves the stabilization, solidification, and immobilization of the pollutant in the soil. The stabilization and solidification technique is defined by the compound used to achieve the solidification. These include a cement-based method, a thermoplastic method, pozzolan-based, organophilic-clay-based, an organic polymerization methods, and an encapsulation method. 14 Immobilization involves the reduction of potential solubility, toxicity, or mobility of the contaminant by adding stabilizing agents to contaminated soil. Each of these methods is ineffective in removing the contaminants from the environment in large amounts with cost-effective amounts of input. These methods also have the potential of rendering the soil unusable for agricultural purposes, and introducing toxic molecules to remove the original contaminant.
Living organisms are often used in remediation today. For example, because of their ability to degrade lignin, a complex phenolic compound, white rot fungi are used to degrade polluting phenolic compounds, a process which is facilitated by the production of manganese peroxidase (MnP) by the fungus. 15 The continued presence of aromatic pollutants after application of potentially effective organisms demonstrates the technique’s limited effectiveness. 11 Another limitation inherently involved with the use of living organisms is keeping the organism alive while remediation takes place. 15 Progress in this area is also being made using Phanerochaete chysosporium under specific conditions.16,17 The use of living organisms can be replaced through the direct use of the enzymes they produce. However, the cost of enzymes useful in remediation such as laccases (Lcc) and peroxidases is too high when produced by traditional fermentation methods. A more cost-effective method of enzyme production would be ideal.
Fungi and bacteria are traditionally used for producing commercial enzymes and proteins and indeed produce most of today’s industrial enzymes, but this method has some limitations. Fermentation uses these microorganisms to produce compounds through the biological conversion of complex substrates. 18 Fermentation systems require large-scale facilities, water, and nutrients. In addition, traditional techniques have production challenges with oxidation/reduction enzymes because of the need to keep the bacteria or fungi alive in the presence of these highly reactive enzymes in these single-cell systems. Another system of commercial enzyme production is justified for these important enzymes.
Plants have been used to produce proteins from different species for some time now.19,20 The use of enzymes made in transgenic plants can cut the cost of many processes like the bleaching of paper. 21 The advantages of this method are that the plants can be grown in fields, which is less costly than the millions of dollars needed to build facilities for fermentation. The enzyme can be produced in biodegradable vessels (i.e., in a seed) that pose little threat in the form of industrial waste and do not require the introduction of construction material like cement or steel into the environment.
Corn has been a very useful system for protein production due to its high yield, stable grain storage, and large production potential. 22 The first commercially produced proteins made from genetically engineered corn were avidin and β-glucuronidase.23–25 MnP is an important example of a redox enzyme that has been produced by the transgenic corn system. 26 MnP degrades lignin and bleaches paper pulp. 27 A new bioremediation method could be developed by using the corn-based, less expensive source of this enzyme. However, testing the enzyme from this new source, corn kernels, is important to prove this application.
Trametes versicolor is a fungus that produces Lcc and has been used in alternative pulp bleaching research. 28 Lcc is involved in the fungal biodegradation of lignin by catalyzing the removal of a hydrogen atom. 29 This enzyme’s capacity as a redox reaction catalyst also could facilitate bioremediation. The goal of this project was to use recombinant MnP produced in genetically engineered corn and commercial laccase to degrade synthetic dyes.
Materials and Methods
Protein Preparation
Transgenic corn lines containing the MnP enzyme were obtained from Infinite Enzymes LLC (Jonesboro, AR, USA). The corn was ground to a fine flour and extracted using 50 mM sodium tartrate (pH 4.5) at a ratio of 1.5 L per 1 kg of corn. Extraction was conducted for 1 h in an ice bath with vigorous mixing. A filtering aid (Dicalite, Ideal Chemical, Memphis, TN, USA) was added to the slurry and filtered through a 15-cm Whatman 1 filter in a Buchner funnel using a vacuum pump, and the filtered solids were discarded. The filtrate was then concentrated to half of its volume using a Pelicon-2 Tangential Flow Filtration apparatus (TFF; Millipore, Burlington, MA, USA) with a molecular weight cut-off (MWCO) of 10 kDa on a PES filter. Solid ammonium sulfate was gradually added to 95% saturation and the slurry was mixed for ~1 h. Dicalite was added and the slurry was filtered through a Buchner funnel, and the filtrate was discarded, while the precipitate was saved. Sodium tartrate buffer (50 mM) at pH 4.5 was added to the precipitate at a concentration of 1 mg of precipitate per 1 mL of buffer and mixed for 30 min. The slurry was filtered (with Dicalite), the resulting filtrate was saved, and the precipitate was discarded. The filtrate was concentrated and desalted using the TFF as above. The filtrate was either used directly as the concentrated formulation or purified. For purification, the resulting filtrate was applied to a GigaCap S-650M (Tosoh Bioscience LLC, King of Prussia, PA, USA) column, and the flow-through contained the MnP. The combined fractions containing the MnP were concentrated using TFF and were lyophilized on a Labconco FreeZone Benchtop freeze dryer. Lcc was obtained from Millipore-Sigma (38429, St Louis, MO, USA) and dissolved in 50 mM sodium tartrate at pH 4.5.
MnP Characterization
A 12% polyacrylamide protein gel (Invitrogen, ThermoFisher Scientific, Waltham, MA, USA) was run in MOPs buffer and stained with a Silver Staining kit (ThermoFisher Scientific), as per the manufacturer’s directions. Protein quantification (mg) was calculated from the lyophilized purified material and activity assay. Specific activity (419 DMP units mg−1 min−1) was determined using a known quantity of protein in the dimethoxyphenol assay. The protein assay used a 96-well microplate reader (BioTeK, Winooski, VT, USA) and Gen5 software, version 2.09. Materials used include sodium tartrate (50 mM, pH 4.5), manganese sulfate stock (50 mM), and 2,6-dimethoxyphenol (DMP) stock (100 mM). The assay was activated with 0.003% H2O2.
Dye Degradation
Dry dye powder was mixed with deionized water. Dye concentrations used were 500, 750, 1000, 1500, and 2000 mg/L. The dye solution was autoclaved prior to degradation experiments.
Purified MnP and Lcc were used in degradation assays of dyes and the reactions were analyzed by absorbance. The dyes used were Acid Blue 25 (Millipore-Sigma; St Louis, MO, USA), Acid Blue 29 and Brilliant Black (MP Biomedicals, Irvine, CA, USA), and Congo Red (Fisher Scientific). See Table 1 for dye details.
Detailed characteristics of the dyes used in this study.
The enzymes were stored at −20°C. All reactions were conducted at 22°C. Hydrogen peroxide (H2O2) is the activator in any reaction with MnP and is added last to the reaction. The reactions occurred at pH 4.5 in 50 mM sodium tartrate buffer. The concentrated formulation of MnP was used in degradation assays parallel to those run with the purified MnP. The total volume of the reactions varied from 200 µL in 96-well plates to 12.5 and 25 mL in conical-bottom centrifuge tubes with constant rotation. Pure MnP was added at concentrations between 0.005 and 0.04 mg/mL. The reactions were incubated up to 24 h after the initial application of H2O2. The final concentration of buffer was 41.5 mM sodium tartrate, 1 mM MnSO4, 8 µg of MnP in 200 µL, and 20 µL of dye. The reaction was scanned from 200 to 700 nm at 10 nm intervals in a BioTek Synergy HT scanner (Winooski, VT, USA), prior to adding H2O2 (negative control). H2O2 (0.003%) was added every 30 min for 2 h.
Lcc reactions contained 30 ng of enzyme in sodium tartrate pH 4.5 and various concentrations of dyes. The reactions were incubated at room temperature (RT, 20–25°C) overnight. For double enzyme degradation, 30 ng of Lcc was added, and the reaction ran for 16 to 24 additional hours before or after MnP. The reaction was then scanned with the Synergy HT to observe the change in absorbance of the reaction. For the sequential reaction that began with the use of Lcc in 41.2 mM sodium tartrate, 20 µL of dye at a concentration of 1000 mg/L was added to the plate well, the reaction scanned, and 30 ng of laccase was subsequently added to the reaction. The reaction ran from 16 to 24 h. Subsequently, 1 mM MnSO4 and 8 µg of MnP were added. The reaction was activated with 0.003% H2O2, which was added every 30 min for 2 h. The reaction was scanned for absorbance. All reactions were run in triplicate.
Thin Layer Chromatography
Acid Blue 25 (AB25) was treated for 60 min with MnP as previously described. The resulting mixtures containing 20 mg (20) and 50 mg (50) were separated on a silica gel thin layer chromatography (TLC) plate in a solution of ethanol:butanol:water (10:20:1). Spots were visualized after heating for 5 min on a hot plate at 100°C.
Results
Purified MnP
MnP is present in corn seed at ~0.4% of dry weight. A silver stained 12% polyacrylamide protein gel run in MOPs buffer demonstrated that the MnP was highly purified (Figure 2). Protein quantification (mg) and specific activity (419 DMP units mg−1 min−1) were determined. This preparation of purified enzyme was used in all degradation reactions.

Silver stained polyacrylamide gel of purified MnP. The gel was a NuPage 4%–12% Bis-Tris gel and it was run in MOPS buffer. The lanes show the molecular weight standards (left) and the MnP at 53 kDa (blue arrow, right).
Synthetic Dye Degradation with MnP
First, the degradation activity of MnP relative to the dye concentration was performed to choose a standard dye concentration for further experiments. Thus, experiments involving multiple concentrations of dye ranging from 500 to 2000 mg/L were conducted. The same amount of MnP (8 m) was used on each dye concentration.
Multiple concentrations of Acid Blue 29 (AB29), an azo dye, were treated with MnP (Figure 3). The relative amount of absorbance of the treated sample was the same for all concentrations. Thus, AB29 appears to be highly susceptible to degradation by MnP. The ratio between the peaks of the treated and untreated dye ranges between 0.21 and 0.27 (Figure 3).
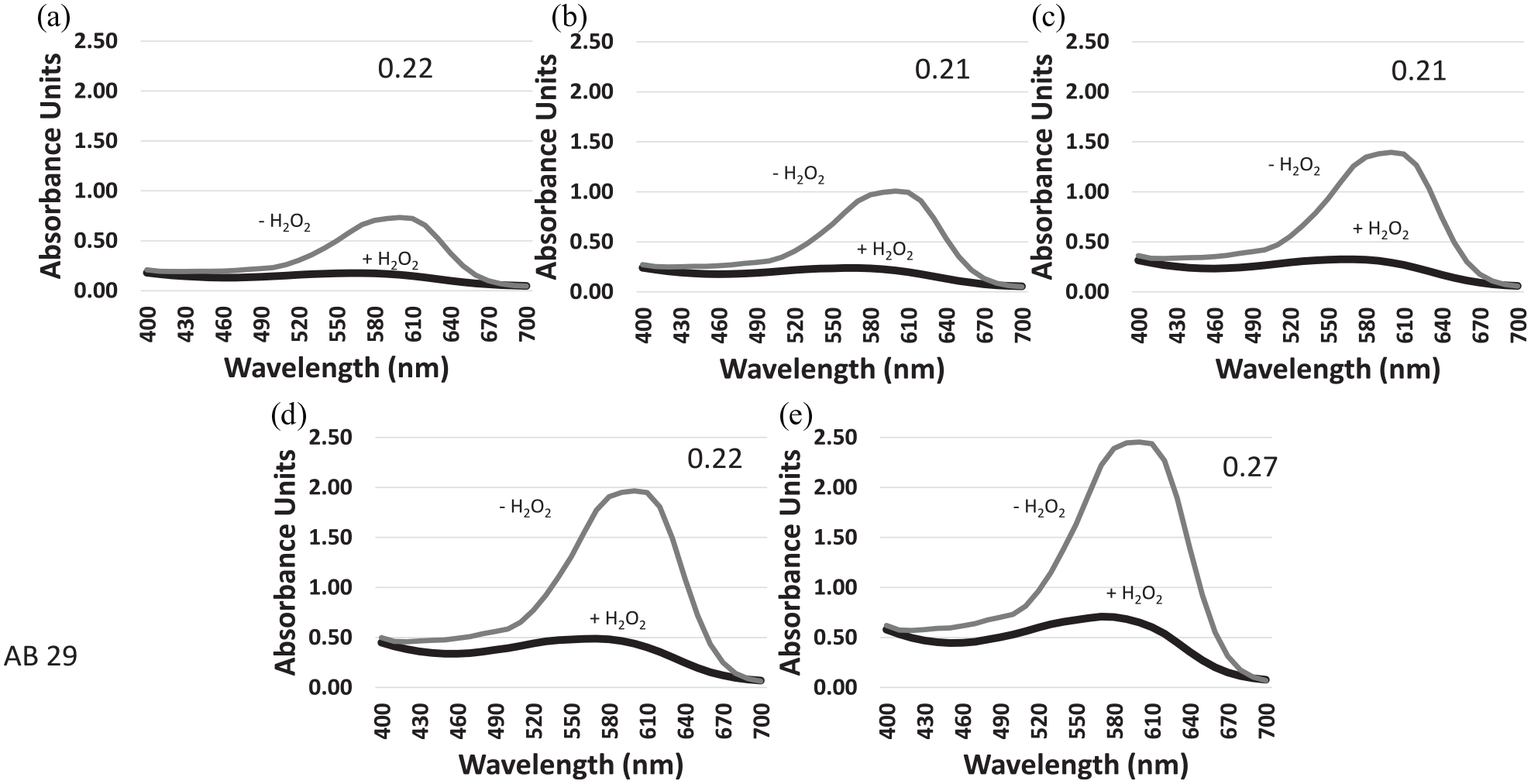
Absorbance comparison of the degradation of AB29 using MnP. The dye was at the concentrations of (a) 500, (b) 750, (c)1000, (d) 1500, and (e) 2000 mg/L. −H2O2 indicates that the reaction has not been started and +H2O2 indicates that the peroxide has been added to start the degradation reaction. Numbers on the graphs indicate the ratio of the absorbance of the degraded dye peak divided by the absorbance of the concentrated dye prior to enzyme treatment.
A single concentration (500 mg/L) of each of the other dyes: AB25, Brilliant Black, and Congo Red (all anthraquinone dyes) was treated with MnP (Figure 4). At 500 mg/L of AB25, it appears that a significant amount of the dye remained. However, TLC of products showed that no intact AB25 remained (Figure 5). At this concentration of Brilliant Black, the amount of dye remaining appeared to be high, with a small decrease in absorbance in the reaction after treatment. Similarly, for 500 mg/L of Congo Red, based on the relative absorbance, it appears that minimal dye degradation occurred (Figure 4).
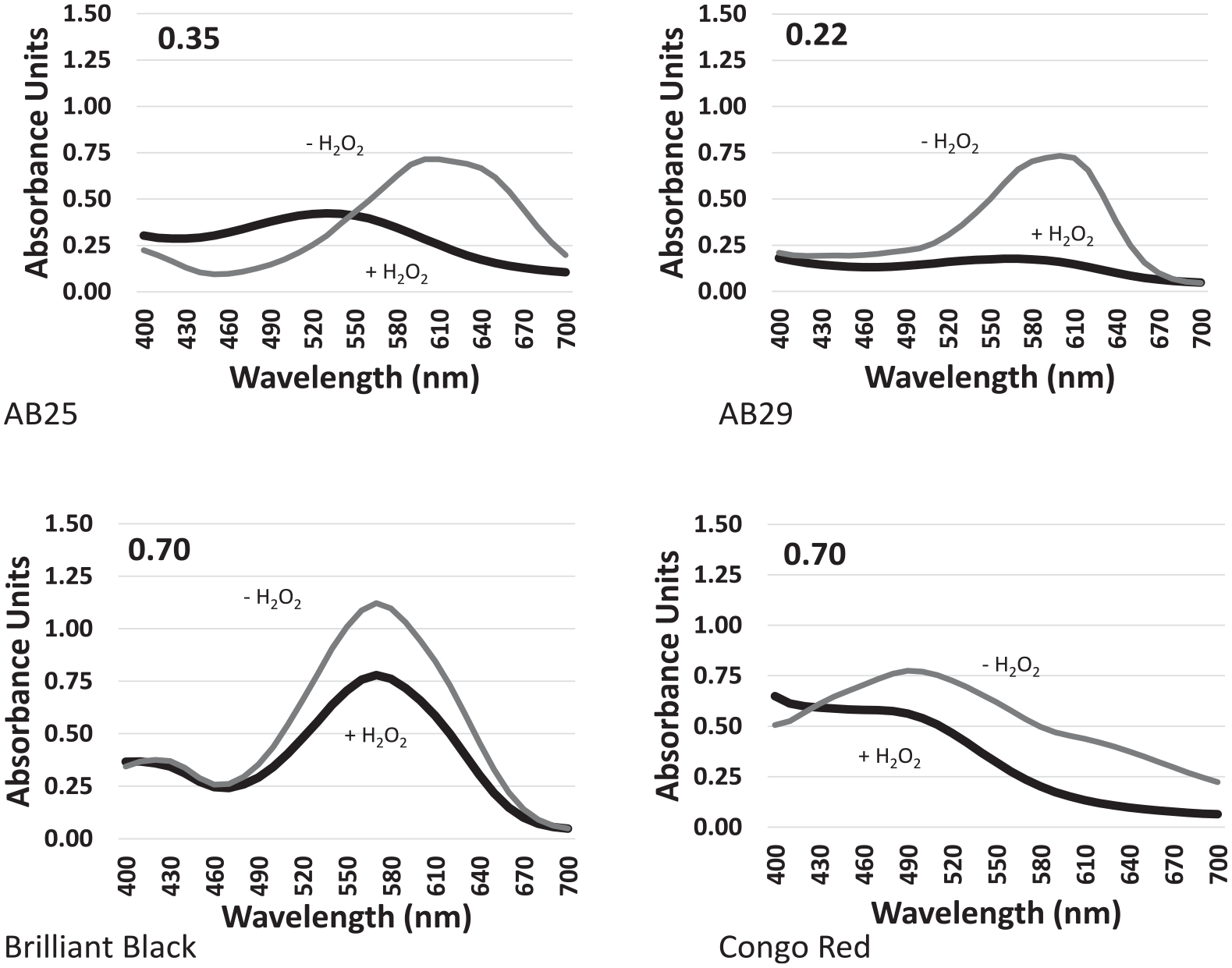
Absorbance comparison of the degradation of various dyes using MnP at 500 mg/L.

TLC of AB25 after treatment with MnP. 5 mL of each of 20 and 50 m reactions of treated AB25 were spotted onto a silica gel TLC plate. Spots were visualized after heating and show original AB25 (left) and the breakdown product (right). Samples on the left are untreated, whereas samples on the right are enzyme treated.
A summary of the ratio of the peaks from the reactions before and after treatment for the other dye concentrations of AB25, Congo Red, and Brilliant Black is shown in Table 2. The ratio of each reaction represented the peak of the treated dye divided by the value of the untreated dye at the maximum wavelength of the untreated dye. The comparison between the peak ratios from the various concentrations gives a comparative estimate of enzyme activity on the different dyes. This allowed a determination of the activity of the enzyme with respect to a certain dye concentration.
MnP degradation of multiple concentrations of AB25, Congo Red, and Brilliant Black Dyes a .
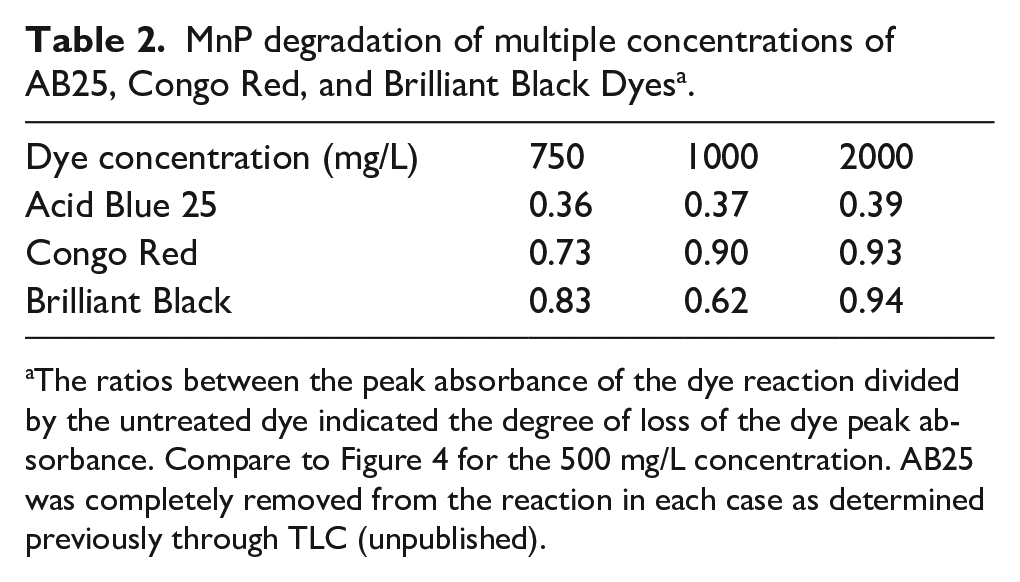
The ratios between the peak absorbance of the dye reaction divided by the untreated dye indicated the degree of loss of the dye peak absorbance. Compare to Figure 4 for the 500 mg/L concentration. AB25 was completely removed from the reaction in each case as determined previously through TLC (unpublished).
The use of MnP did not appear to have much activity against Congo Red or Brilliant Black at any concentration. The ratio between the peaks of the treated and untreated dye never fell below 0.62 and was as high as 0.94. At the highest concentration of 2000 mg/L, the dye seemed to not be affected at all. However, AB25 and AB29 appeared to lose most or all absorbance at 600 nm, their peak of absorbance prior to treatment (Figure 3 and Table 2).
Synthetic Dye Degradation using MnP and Lcc
The results from MnP degradation were encouraging and a decision was made to test other methods of enzyme application. Dyes at a 1 g/L concentration were used in these experiments because that level of dye was moderately degraded by MnP. Individually, the enzyme-treated dyes showed varying amounts of absorbance reduction. The hypothesis was that using Lcc and MnP sequentially might degrade the dyes better. These experiments tested the use of the MnP protocol followed by the Lcc protocol and the reverse.
Applying Lcc alone to the dyes was tested first to determine its impact. Mixed results with Lcc, similar to the results of using MnP alone, were obtained (Figure 6). AB25 and AB29, the blue dyes, were highly degraded by the Lcc enzyme alone in an overnight reaction. Congo Red was also susceptible to Lcc degradation. Brilliant Black was again fairly resistant to degradation, and as shown in Figures 4 and 5, Lcc performed similarly to MnP.
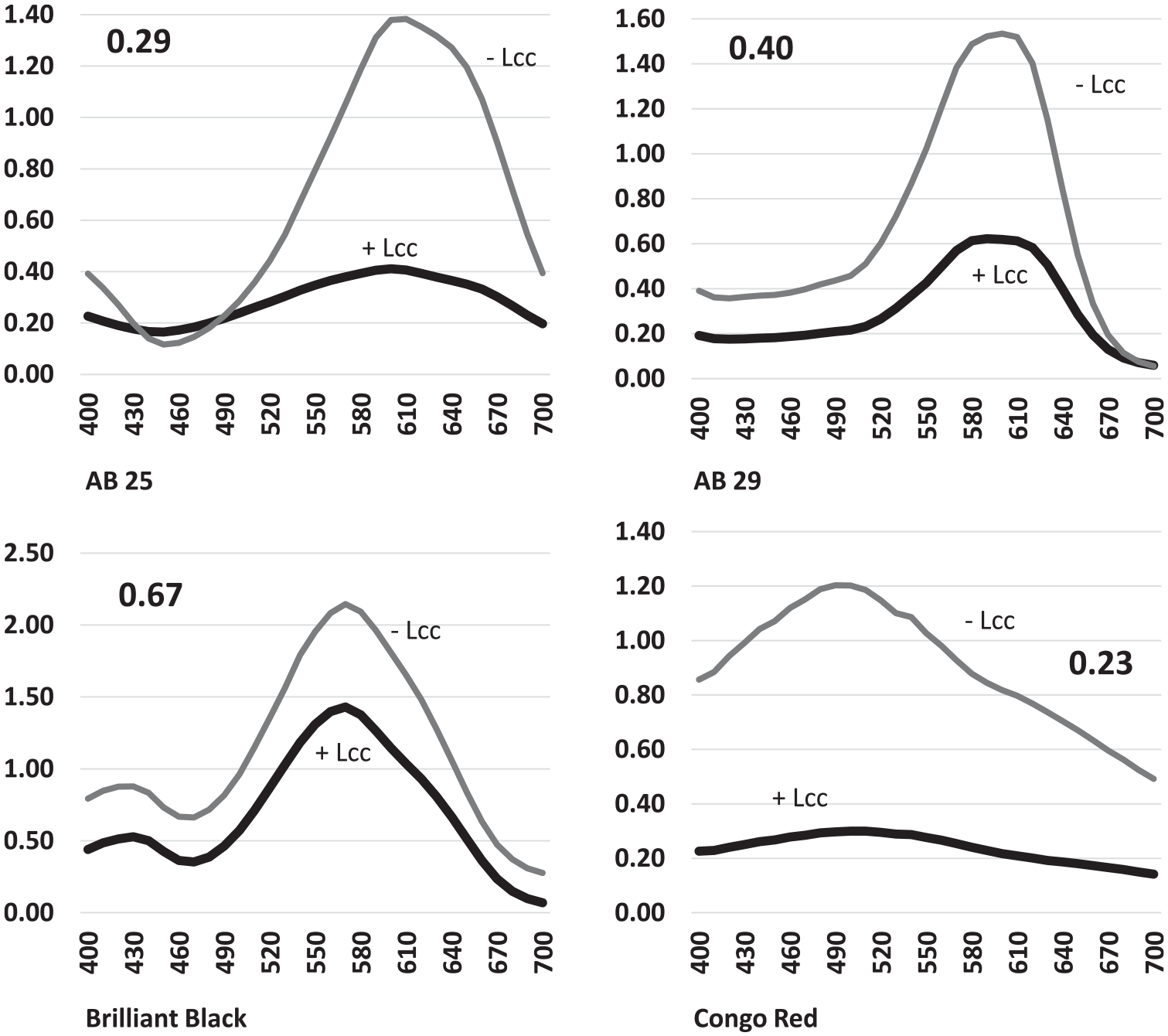
Laccase reactions with all dyes. Numbers on the graphs indicate the ratio of the absorbance of the degraded dye peak divided by the absorbance of the concentrated dye prior to enzyme treatment.
Brilliant Black did not show much degradation when treated with either Lcc or MnP alone (Figure 7). The MnP individually showed a decrease in relative absorbance with a ratio of 0.64, which was similar to the ratio 0.67 from the individual degradation using Lcc. The double digests that utilized the protocols from both enzymes showed a greater decrease in relative absorbance than the use of the enzymes individually. The double digest that used the MnP protocol followed by the Lcc protocol showed a much greater decrease in absorbance of the dye (0.23) than the reverse (0.59). The results of this series of degradation reactions suggested that the double-enzyme treatment could have great utility for this apparently recalcitrant dye.
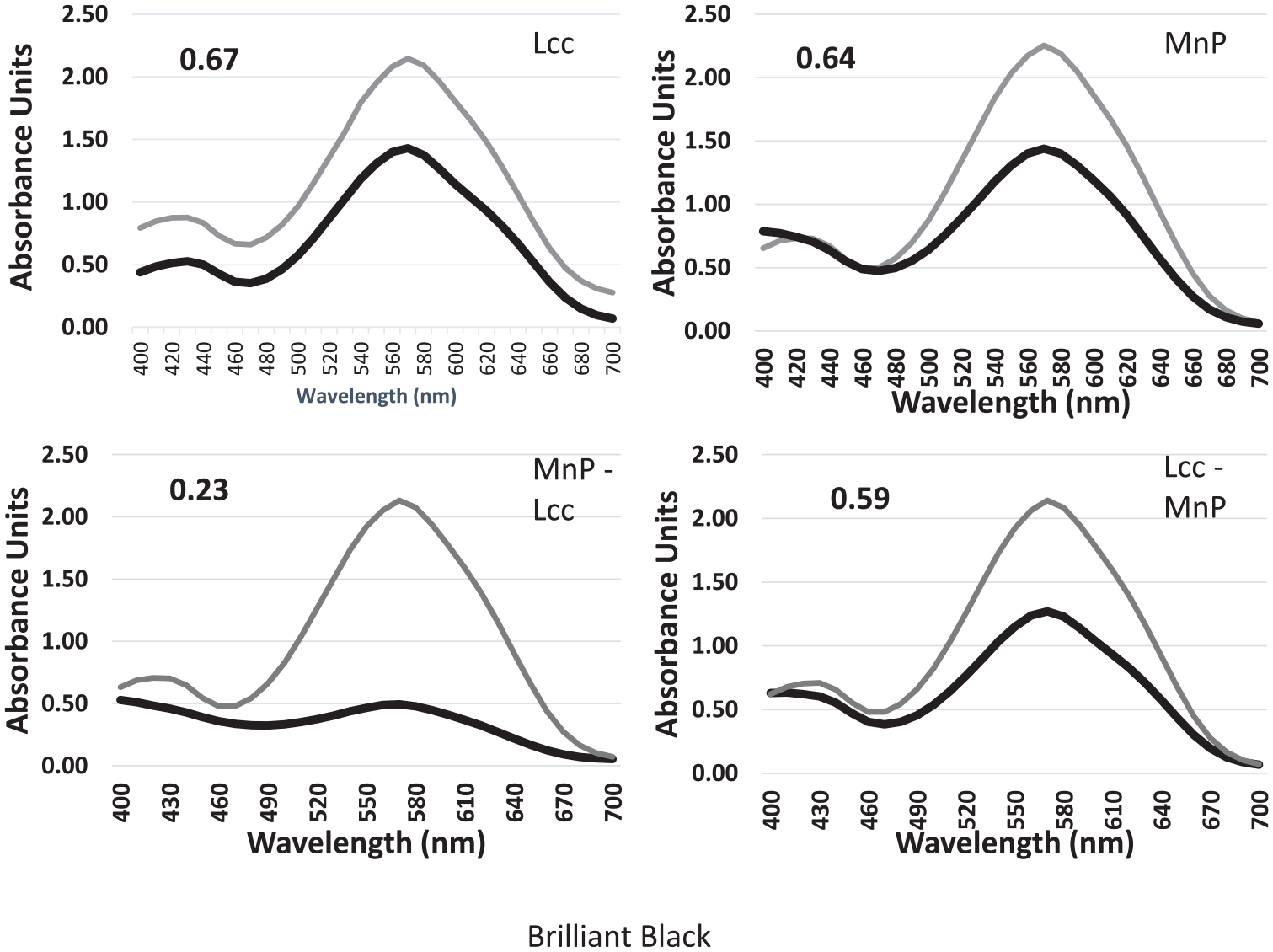
Single and double degradation reactions with Brilliant Black. Numbers on the graphs indicate the ratio of the absorbance of the degraded dye peak divided by the absorbance of the concentrated dye prior to enzyme treatment.
Table 3 shows the comparative absorbance of the peak of the double-enzyme treatment divided by the untreated dye peak for AB25, AB29, and Congo Red. It illustrates the comparative effectiveness of the enzyme treatments on the various dyes. Based on the ratios in Table 3 and the various figures, no single enzymatic treatment of the dyes appeared to work in all cases. In some cases, such as AB29, more than one enzyme treatment appeared to be effective. In other cases, such as Congo Red and Brilliant Black, only one method of treatment showed a major decrease in absorbance after treatment. For AB25, all treatments appeared to work effectively.
A comparison of the peak ratios from the sequential degradation of AB25, AB29, and Congo Red using two enzymes a .
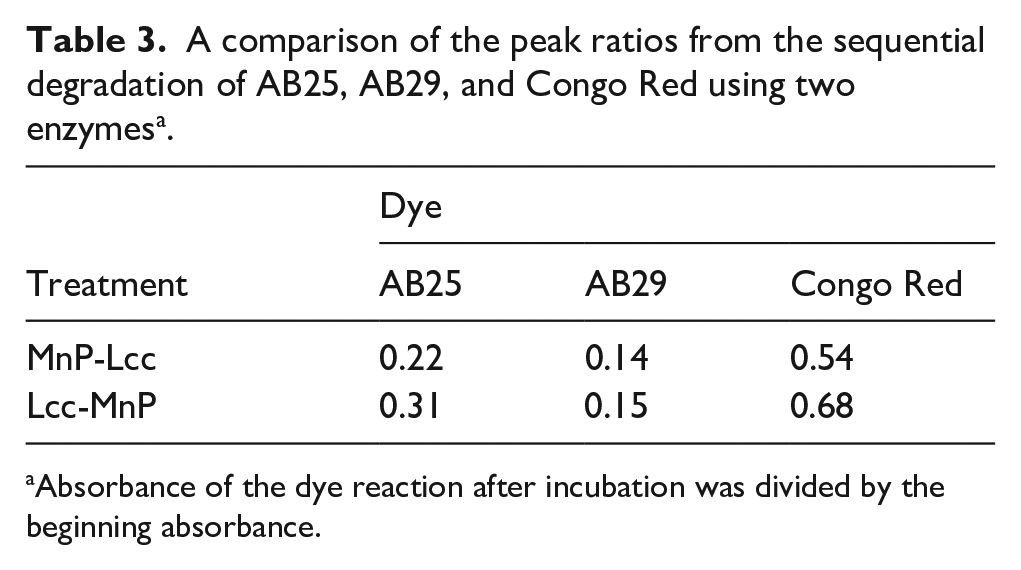
Absorbance of the dye reaction after incubation was divided by the beginning absorbance.
When AB25 was the substrate, the various methods of enzyme application all showed a decrease in dye absorbance after treatment (Table 3 and Figures 4 and 6).
Congo Red did not appear to be very susceptible to these redox enzymes. In fact, the ratio of absorbance after treatment was nearly the same in all cases. The use of Lcc individually showed the greatest decrease out of all the methods with a ratio of 0.23 (Figure 6). The double-digest reactions showed greater decrease in absorbance than MnP individually, but the ratios were still higher than those of Lcc alone (Table 3). Out of the two double digests, the protocol that used MnP first showed a greater decrease in absorbance, with a ratio of 0.54, than the protocol that used Lcc first. The products of the reaction were not analyzed for structural changes.
Finally, the enzymatic degradation of AB29 indicates that Lcc individually (0.4, Figure 6) was not as active against the 1 g/L AB29 as MnP (0.21, Figure 3). The double digests using the protocols of both enzymes showed a similar level of activity and decrease in absorbance (Table 3). Either double digest showed slightly improved reactivity when the enzymes were used in tandem rather than when they were used individually.
When using MnP for these dye degradation reactions, hydrogen peroxide is required to activate the enzyme. This could be a problem in large-scale applications because H2O2 is volatile and explosive. An application method for H2O2 that would allow for a consistent supply, but not too much at any one time, was needed. The enzyme glucose oxidase produces H2O2 as a product when it is provided with glucose to metabolize. Thus, glucose oxidase was tested to determine whether it was able to produce enough H2O2 to allow MnP to perform its redox reaction. After glucose oxidase was proven to be able to deliver enough H2O2 when given glucose, testing was done to find the correlation between the amount of glucose given and the performance of the MnP. To do this, multiple concentrations of glucose were tested (Figure 8). High levels of glucose (10%–50% stocks) had inhibitory effects on MnP degradation of AB25 when using glucose oxidase as the peroxide provider (data not shown). Thus, lower concentrations of glucose were tested. The use of glucose oxidase and glucose at 0.1% mimicked the direct addition of H2O2 to the reaction.
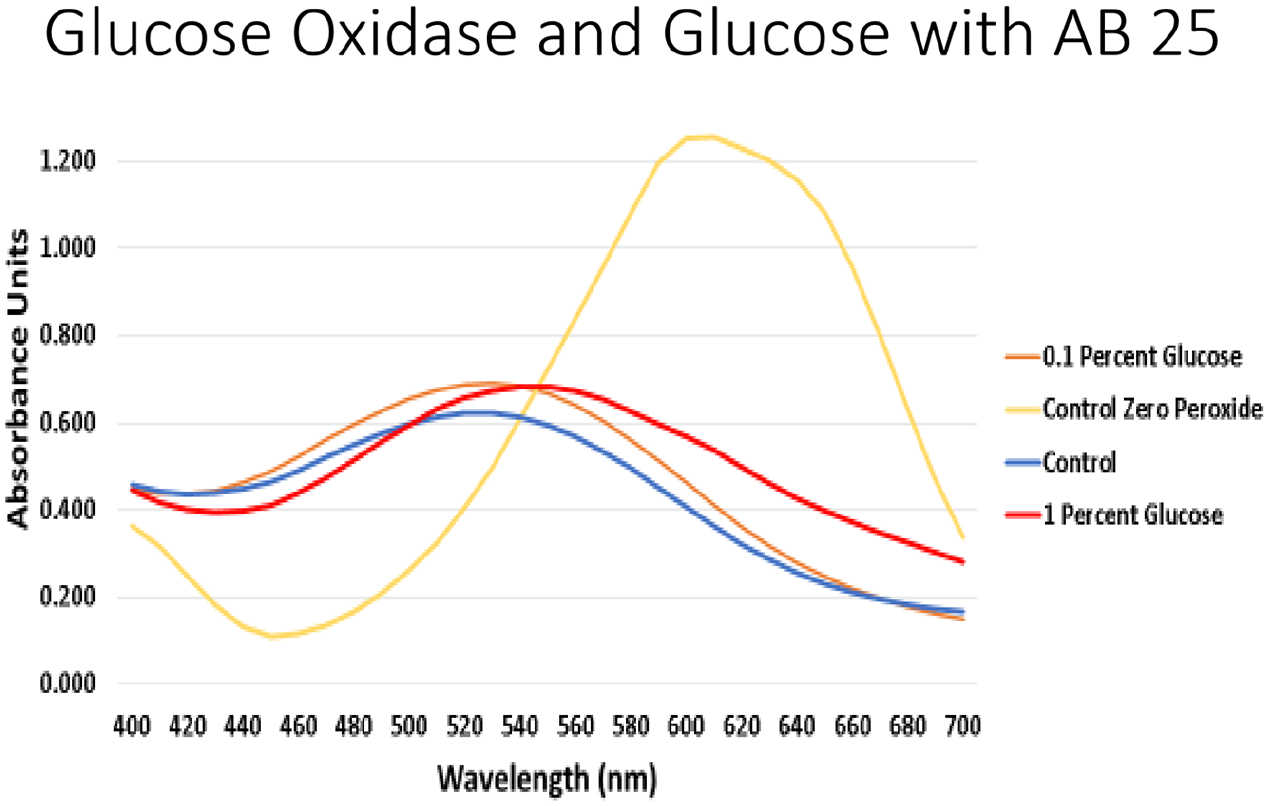
MnP degradation of AB 25 using different sources of H2O2. A comparison between the different concentrations of glucose used in combination with glucose oxidase were tested. Control = direct addition of H2O2.
Discussion and Conclusion
Pollutants, such as herbicides and synthetic dyes, are often released into the environment without treatment. Synthetic dyes are used in paper printing, textile dyeing, color photography, and in the production of petroleum products. 30 The accumulation of these pollutants can cause problems because they are a hazard to animal and plant life. 31 An environmentally sound solution is needed to eliminate these compounds.
Several approaches to minimizing the impact of textile dyes on the environment have been published recently. One interesting study proposed changing the process of indigo dyeing of cotton without sodium hydrosulfite (Chakraborty 32 ). The hydrosulfite produced sulfate, sulfite, and thiosulfate ions with toxic effects. Changing the process to using alkaline catalase and iron removed the sulfate pollution. Silylated bentonite clays were effective at removing Sudan dyes from wastewater streams at low cost, but the dyes were still intact. 12
A method proposed in 2000 was to utilize live fungi, such as Phanerochaete chrysosporium, to degrade a wide range of pollutants. 33 In fact, this method worked on a small scale, but it is impractical at a larger scale because organism survival may be compromised by the toxic environment. More recent studies have focused on the conditions that favor the fungus and analyzed the levels of the redox enzymes in the reaction. 17 Many parameters affected the efficiency of dye degradation in these experiments such as pH, temperature, fungal inoculant, and dye concentration. When the parameters were optimized, dye degradation was very high. The concentrations of enzymes laccase, MnP, and lignin peroxidase were also elevated. A combination of methods using the Fenton reaction and the fungus Aspergillus niger showed near complete removal of dyes from synthetic mixes as well as from actual effluents. 16 In all of the above studies, the dye concentrations ranged from ~130 to 1.2 g/L.
MnP from P. chrysosporium has been shown to break down azo dyes. 34 However, because of its high cost of production, it has not been commercialized. At the same time, Lcc enzyme has also been shown to break down phenolic dyes 35 and is available, but is also expensive for remediation applications because of its low level of production in the native fungus. 33
The present study tested a remediation solution in the form of enzyme-mediated reactions. Several synthetic dyes at high concentrations (1–2 g/L) were degraded using these oxidation/reduction enzymes. MnP, a fungal enzyme produced in transgenic corn, and laccase from a native fungal source were used to test dye degradation. The dyes showed differential responses to enzyme treatments.
Fermentation using bacterial and fungal systems is currently the established commercial production system for industrial enzymes. 36 Multiple parameters can be manipulated to increase the yield and lower the cost. However, fermentation is a mature technology with limited technological improvement. However, plants provide an alternative system with much greater potential for technology development. In fact, the use of transgenic corn provides benefits that affect industry and the environment. The corn production system is a “cradle to grave” green technology. This will leave little to no pollution if the production fails or has to be moved to a new location. The enzyme is easy to store because it is stable within the seed. 19
The MnP enzyme has high levels of accumulation in corn seed. 26 This fact combined with the ease of production of corn allows enzyme production on a large scale. Although in this study, the enzyme was purified, it would not need to be purified for applications in remediation. Many formulations could be used such as ground corn flour or a concentrated extract. The established corn processing infrastructure eliminates the need for major industrial renovation to build more fermentation facilities. The large quantity of enzyme available in the grain (0.5% of grain dry weight) will allow large amounts for bioremediation. Large quantities of recombinant corn enzymes can be applied because corn is an inexpensive production system. Lcc has also been produced in plants. 37 However, it is not easily extracted, and its activity could not be determined for this study. Therefore, commercial Lcc was chosen for this project. Nevertheless, plant-produced laccase is a potential option for bioremediation in the future.
The use of biotechnology to produce foreign proteins in plants is an established practice. 38 This project is a step toward applications testing of plant-produced recombinant proteins. Although the focus of this project is redox enzymes, the possibilities of what can be produced are vast. The enzymes were extracted from the corn in a way that requires little labor or equipment. The conditions required for the individual enzymes differ. For example, the substrate required by Lcc is merely oxygen, but the time needed for the concentration of this enzyme tested was great. The MnP reaction is limited by the amount of H2O2 and manganese available.
The recombinant MnP from corn and commercial Lcc can degrade synthetic dyes either alone or together. Encouragingly, the synthetic dyes tested were partially or wholly degraded by the oxidation/reduction enzymes. This outcome was observed through absorbance analysis before and after treatment and visual observation.
Footnotes
Declaration of conflicting interests
The author, J Byrd declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Author EH owns the company, Infinite Enzymes, that supplied the enzyme.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author received from the USDA NIFA Non-Land Grant Colleges of Agriculture Capacity Building Fund, Grant #2017-70001-25987.
