Abstract
More as a decade before aniline and phenol were isolated from coal tar by Runge in 1834 and used for the fabrication of a number of important dyes, such as Aniline Black, Mauveine, and Fuchsine, the German naturalist von Reichenberg observed the formation of a red dye from beech tar, which he called pittakal. In this report, both the history of discovery and the laborious structure elucidation of this first tar dye were discussed and the subsequent efforts to obtain further usable dyes from phenol outlined. A decisive contribution for that came from Kolbe and Schmitt, who better known as the inventors of the first technical synthesis of salicylic acid.

In 1834, the German chemist F. F. Runge made a momentous discovery when he found that coal tar, which was produced on a large scale in the course of industrialization at the time, contained some substances that were little or not at all known but could easily be isolated from it by treatment with aqueous bases or acids. 1 These were two acid-soluble compounds, one of which Runge named Leukol and the other Kyanol, and a base-soluble compound, which he named Carbolic Acid. 2 The name for the first-mentioned compound resulted from the fact that it showed no color change when exposed to oxidizing agents, while the second was thereby converted into a deep-colored product, which soon acquired greater practical importance as Aniline Black. 3 Similarly, the Carbolic Acid could be converted into a red-colored reaction product, which Runge named Rosolic Acid. However, the exact structures of these two compounds, and of the starting materials used to produce them, could not be determined at the time. Interestingly, it soon became apparent that the three compounds isolated from the tar by Runge had already been obtained from another authors, but were given different names. This led to them later being referred to by the current names quinoline (for Leukol), aniline (for Kyanol), and phenol (for Carbolic Acid) when their respective identities were recognized. 4
However, since Runge made no further effort to characterize the compounds he had discovered in more detail or to find a practical use for them, this task was left to other chemists. One of them was H. Kolbe, who worked at the University of Leipzig and investigated, among other things, the chemical properties of phenol, such as its reaction to various acids and their derivatives. In 1856, by reacting the sodium salt of phenol with carbon dioxide, he found a simple synthesis for salicylic acid, which was of great practical importance because of its fiber depressing and antipyretic effects and was made, up to that time, only in a complex manner from plant material. 5 His student R. Schmitt was also significantly involved in the development of this synthesis. He received his doctorate from him in 1861 and was appointed as a Professor of organic chemistry at the Royal Saxon Polytechnic in Dresden, today’s Technical University of Dresden, in 1870. Here, together with his students R. Seifert and F. von Heyden, he improved the implementation known in chemistry as the Kolbe–Schmitt reaction 6 and developed a technical process for synthesizing salicylic acid, which was then produced on an industrial scale in the chemical factory von Heyden founded in 1882 7 and used for the fabrication of acetylsalicylic acid (Aspirin). 8
In course of their investigations on the chemistry of phenol, Kolbe and Schmitt made another discovery, which, however, became less well known and seemed to be less important than the synthesis of salicylic acid. Both scientists found in 1861 that by the reaction of phenol with oxalic acid in the presence of sulfuric acid, unlike the reaction of sodium phenolate with CO2, a red dye was formed. This dye appeared to be identical with a product obtained by F. F. Runge that he obtained by oxidation of phenol and had named as Rosolic Acid. 9 However, neither Runge nor Kolbe and Schmitt recognized that this compound could be used as a textile dye; a mistake that was quickly refuted because this dye was soon produced industrially under the name Aurin or Yellow Corallin 10 [SI-1]. This was only a few years later, after W. H. Perkin discovered Mauveine 11 in 1856 and E. Verguin discovered Fuchsine 12 almost simultaneously with A. W. Hofmann in 1858 as part of their research into the chemistry of aniline isolated from tar. Therefore, these dyes are referred usually as the first Tar Dyes produced. 1
The practical interest that these new dyes aroused at the time stimulated various chemists also dealing with alternative methods of their production and with the investigation of their chemical properties, because at the time of their discoveries, their exact structures could not be determined. Information on their structure could only be given from their elemental composition. Kolbe and Schmitt had already determined the formula C20H14O3 for Aurin and concluded, therefore, that it contains three phenol units and a C2 fragment, whereby they postulated that this fragment comes from oxalic acid, which was used as starting material.
9
However, sometimes later

Synthetic routes used for the synthesis of aurin.
Another chemist who concerned with the investigation of properties and the practical use of aniline and phenol was the already mentioned
However, this statement could only be made by comparing the elemental analysis of this sample with a sample that
However, this statement could only be made by comparing the elemental analysis of this sample with a sample that
The difficulties in establishing structural formulas for the aforementioned dyes in the middle of the 19th century resulted from the fact that even the exact chemical structures of the raw materials used to produce them were not known. This problem was solved by

Proposed structures for benzene C6H6 (above) and actual structural formula for benzene and some of its derivatives.
With knowledge of the structural formula for benzene, it was then possible to establish the structural formulas for all tar dyes produced up to that point, including those of Aurin and Pittakal as well as Fuchsine and Parafuchsine as a starting compound suitable for the production of Aurin (Scheme 3).

Structural connections between Parafuchsine, Aurin, and Pittakal.
Definite confirmations of the structures for the compounds considered were recently become possible primarily through the use of nuclear magnetic resonance (NMR) spectroscopy. With their help, it is possible not only to elucidate the structure of special dyes but also to detect the presence and structure of byproducts that may be generated during its synthesis. While this was demonstrated some time ago soon, for example, for Mauveine, 20 Fuchsine, and Parafuchsine, 21 such evidences were missing for Aurin and Pittakal to date. This was the reason for us to carry out analytical studies on these compounds, whereby in the case of aurin, we were able to access dye samples that are stored in the historical dye collection at the TU Dresden (Figure 1). 22 They all come from industrial production in earlier years and were sold under the name Corallin (C.I. 43800). 10 Alternative names are p-Rosolic acid or Rosolic acid.

Probes of Corallin, deposited in the Historical Dyestuff Collection of the Technical University of Dresden.
It is worth mentioning that these dyes are currently no longer used because they were blamed for causing allergy, although it was later discovered that this was caused by the phenol, which was rather toxic 23 and apparently not completely removed from the dye used prior to its application as colorant (Figure 2).

Report in the German Magazine “Die Gartenlaube” on the toxicity of stockings dyed with Corallin and a small explosion of clothes dyed with picric acid; https://epilog.de/giftige-struempfe-und-explodierende-kleider.gartenlaube.1869.
Before monitoring the NMR spectra of these dyes, their purity was checked by means of thin layer chromatography (TLC). As can be seen from Figure 3, left part, these dyes are not pure compounds. Therefore, a purification using Column Chromatography was used. The 1H NMR spectra of purified Corallin are depicted. As can be seen, in the purified probe, no signals for phenyl-linked methyl groups were detected indicating that the checked Corallins were produced accordingly to the Kolbe–Schmitt procedure.

TLC of Corallin (left) and colors of solutions of Corallin and Pittakal in methanol at different pH values.
Unfortunately, there was no sample of Pittakal in the dyestuff collection, so an NMR spectrum of this compound could not be measured. Therefore, we prepared the desired Pittakal according to a method by
Characteristic features of Aurin and for Pittakal are their indicator properties, which have already been examined in more detail for Aurin in the work by Shokrollahi et al. 24 Owing to the hydroxy groups present in this compound, it has a yellow color in both neutral and acidic solutions, while in basic solution, it is converted into a red-colored anion. The Pittakal, however, behaves differently. Although it has also a yellow color in neutral solution, it is converted into a purple-colored cation by dissolving in mineralic acids and into a blue-colored anion in strongly alkaline solution (Scheme 4 and Figure 1, right).

Structural formula and colors of Pittakal in acid, neutral, and basic solutions.
However, after confirming the structural formulas for Aurin and Pittakal as well as for the before-mentioned Parafuchsine by the NMR measurements, the question arose as to how these compounds are formed if only phenol or pyrogallic acid or aniline is used by the oxidation procedure. These educts do not carry any additional carbon atom, which is necessary for linking the three aromatic rings. However, this carbon atom undoubtedly comes from corresponding methyl derivatives of the staring materials used, such as p-cresol (1b) or p-toluidine (

Origin of the central carbon atom in the compounds
To remain historically accurate, it must be mentioned that before the discovery of Pittakal, another organic dye had already been discovered and was used to dye textiles. This is picric acid, which was obtained in 1771 by P. Woulfe when treating indigo with nitric acid, and who also recognized that it was suitable for dyeing silk. 27 At the end of the 19th century, picric acid was widely used to color baked goods as Welter’s bitter also. 28 However, after numerous cases of poisoning occurred, the use of picric acid as dye was banned. The most spectacular event was a devastating explosion of 2300 t (!) picric acid in Halifax in 1917, 29 so that this dangerous compound is now nearly not produced in large scales and only permitted for special purposes, such as indicator in fluorescent microscopy, 30 for etching metals 31 or as non-linear optics (NLO) materials. 32
Nevertheless, it was later found that picric acid can be produced much more easily by reacting phenol with a mixture of nitric acid and sulfuric acid, the so-called nitrating acid, so that it can be counted among the dyes that can be obtained from phenol. It is therefore also listed in Scheme 5 together with some other dyes accessible from phenol. This applies furthermore to a whole series of azo dyes that could be obtained based on phenol or salicylic acid, and phenolphthalein, which is formed by heating of phenol with phthalic anhydride and, in contrast to the azo dyes mentioned, still has a certain importance as acid–base indicator (Scheme 6). 33

Important products obtained from phenol.
Finally, it should be noted that phenol also serves as the starting material for Bakelite as the first duroplastic material, which was discovered by
Experimental
The 1H NMR spectra were recorded with a Bruker DRX 500 P instrument at 500.13 MHz and the electrospray ionization (ESI) mass spectrum with an Esquire-LC-00084 instrument.
Preparation of pittakal (sodium salt of 4-[bis(4-hydroxy-3,5-dimethoxyphenyl)methylene]-2,6-dimethoxycyclohexa-2,5-dien-1-one)
To a solution of 2,6-dimethoxyphenol (3.9 g, 0.02 mol) in 25 mL, nitromethane NaH (1.0 g, Sigma-Aldrich) was added. After 5 min stirring at room temperature, tetrachloromethane (2.0 mL) is added and the resulting mixture is heated in a water bath at 100°C. The mixture turns violet after some time and solidified. After 6 h, the mixture was cooled at room temperature and diethylether (50 mL) was added. The product formed was isolated by suction, washed with some diethylether and dried. The pittakal was obtained as violet solid that is soluble in methanol after the addition of some acetic acid with orange color. After evaporation of the solvent, a pink-colored product remains.
Mass spectrum of Pittakal
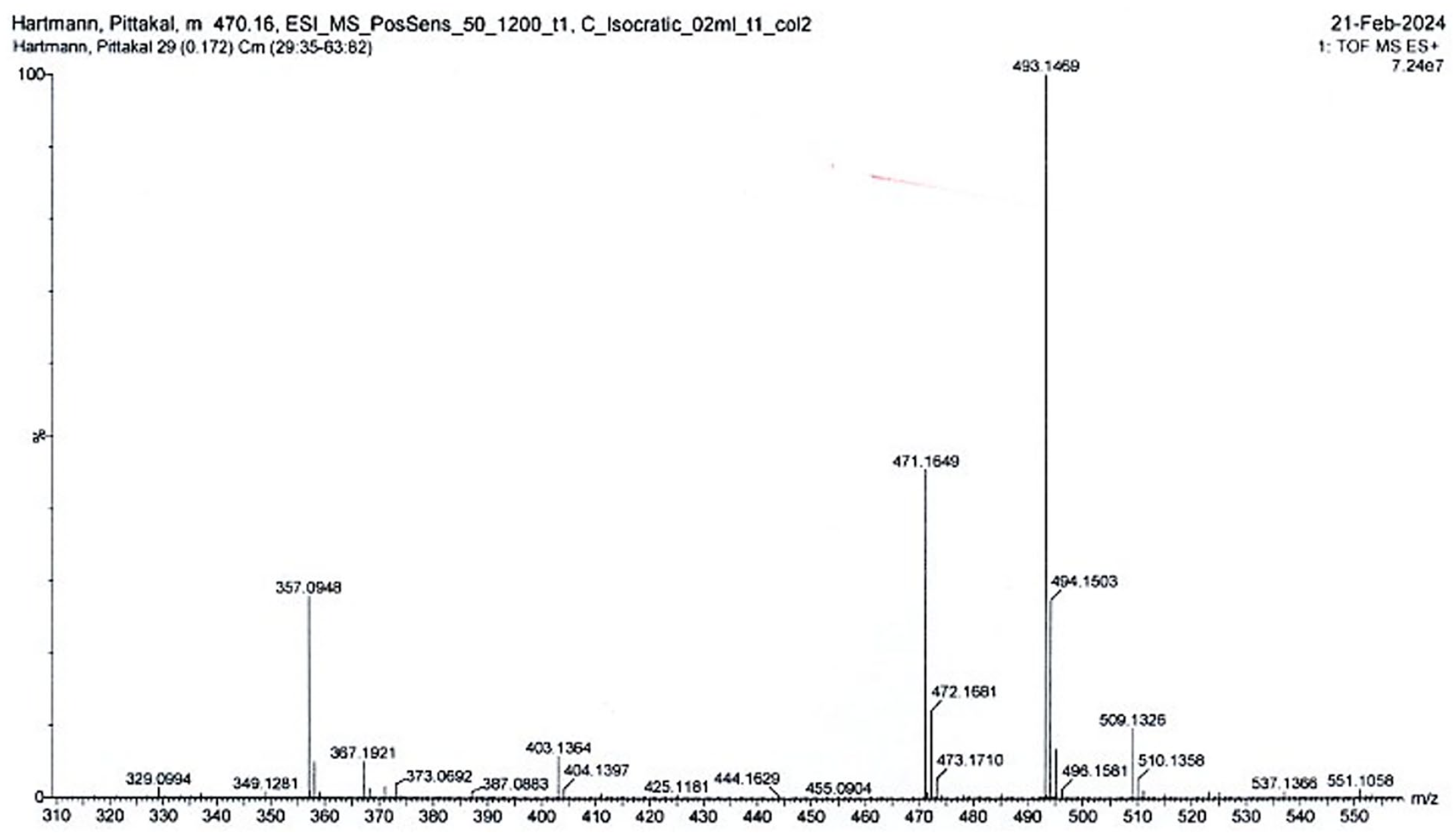
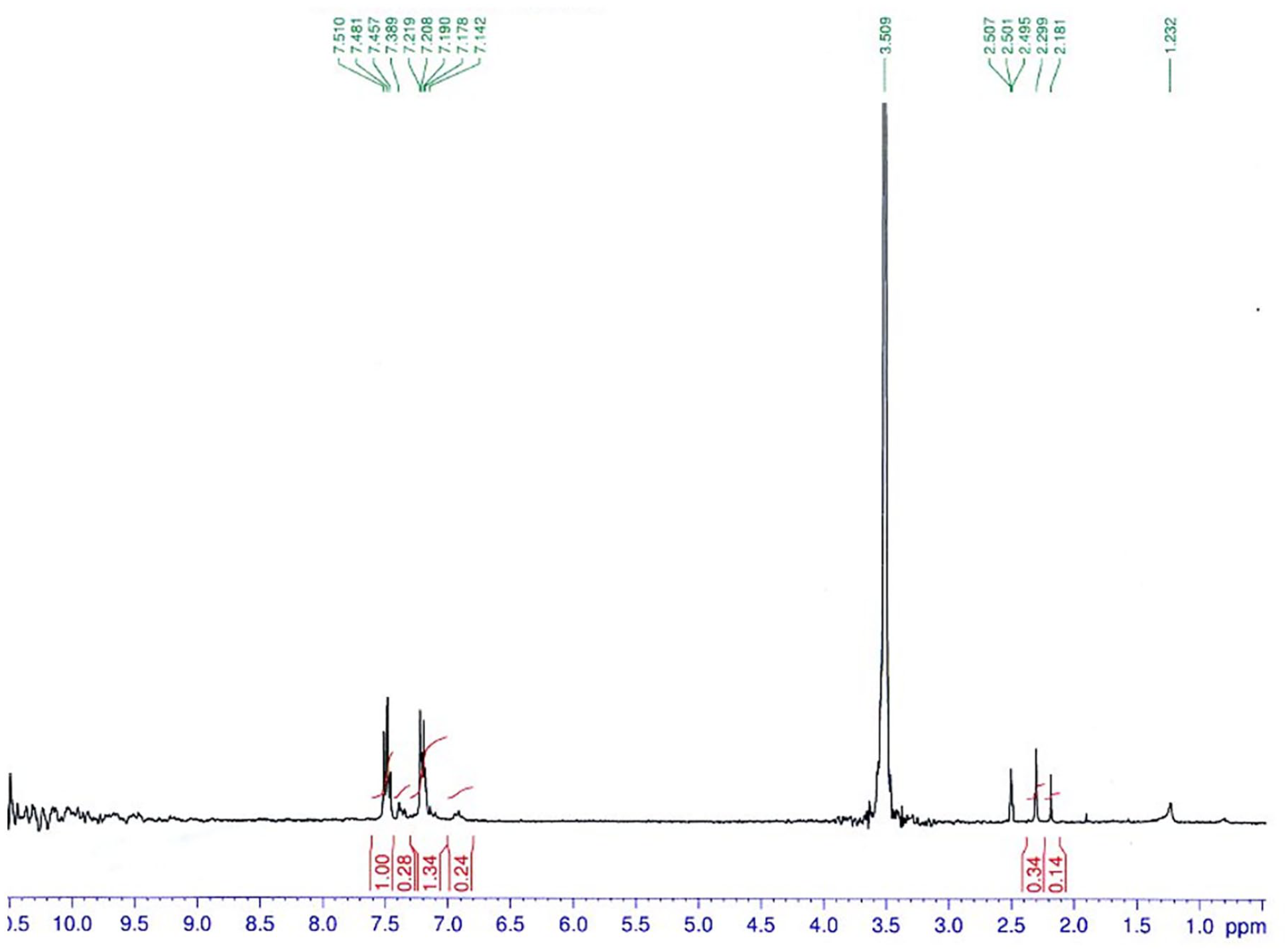
1H NMR spectrum of Leuko-Pittakal, measured in CDCl3, synthesized accordingly to Ref. [35].

1H NMR spectrum of Pittakal, measured in CD3COOD

Mass spectrum of Corallin (C.I. 43800)
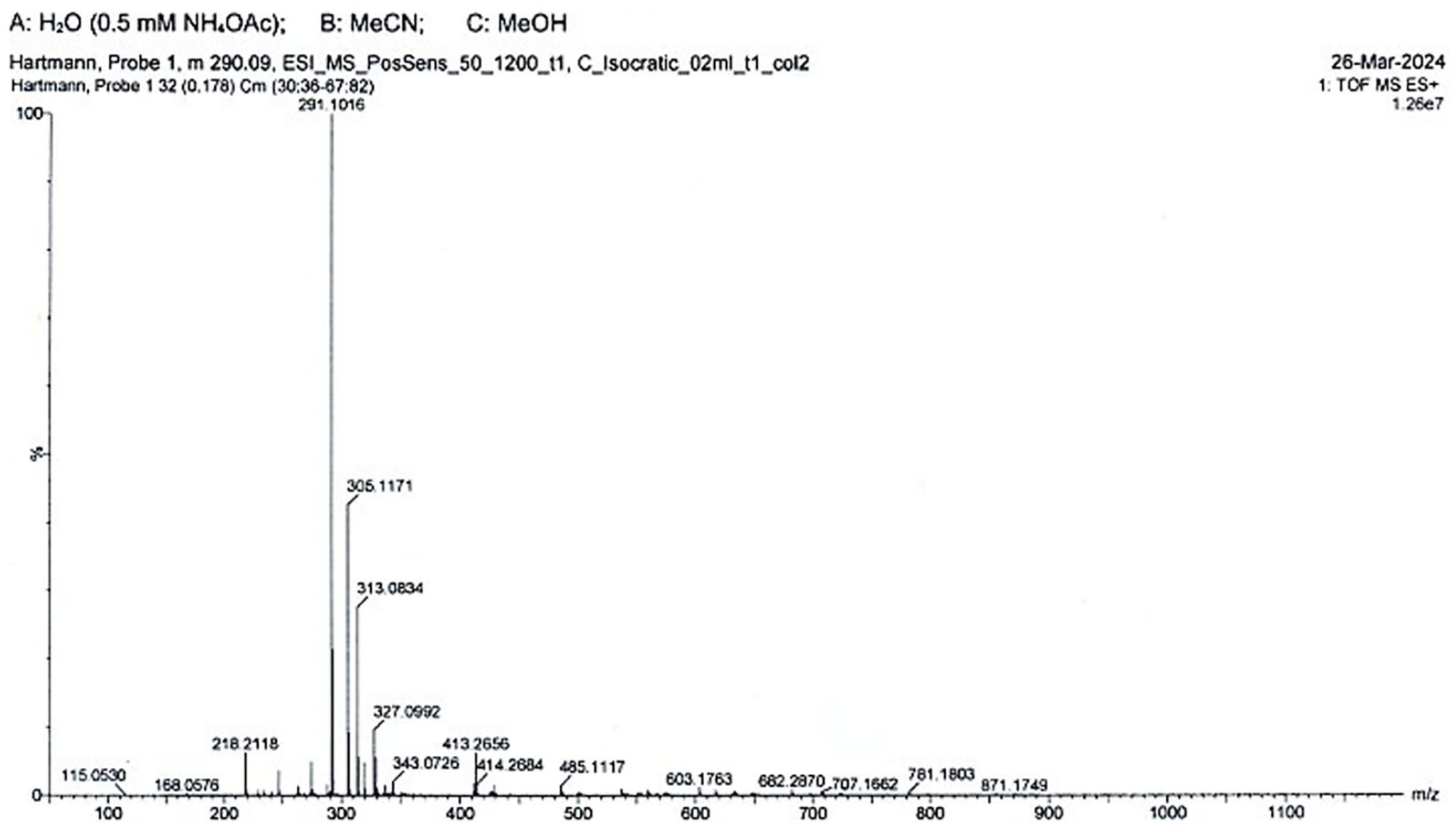
A probe of the dye purchased from R. Graesser, Ruabon, North Wales (https://www.gracesguide.co.uk/Robert_Graesser) was dissolved in ethyl acetate, added to a column filled with silica and eluated with toluene/ethyl acetate (1:1).
1
Supplemental Material
sj-docx-1-chl-10.1177_17475198241262008 – Supplemental material for On the discovery of the first synthetic dyes prepared from phenolic tar ingredients
Supplemental material, sj-docx-1-chl-10.1177_17475198241262008 for On the discovery of the first synthetic dyes prepared from phenolic tar ingredients by Horst Hartmann in Journal of Chemical Research
Footnotes
Acknowledgements
The author thanks Mrs Anett Rudolph for measuring the 1H NMR spectra and Dr I. Bauer for recording the mass spectrum of Pittakal.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author received no financial support for the research, authorship, and/or publication of this paper.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
