Abstract
Mitochondrial dysfunction results in the production of an abnormally high amount of reactive oxygen and nitrogen species, which results in redox imbalance and glutathione deficiency. Therapeutics such as EPI-743 (α-tocotrienol quinone) and RP103 (cysteamine bitartrate) have the theoretical potential to improve redox imbalance by increasing intracellular glutathione and are currently under investigation in multiple clinical trials. This review provides an update on the use of these compounds in clinical trials related to primary and secondary mitochondrial disorders. These clinical trials have not only provided hope to affected patients and their families and caregivers, but also will serve as important stepping stones for further studies as our understanding of mitochondrial disease pathogenesis continues to improve.
Mitochondrial respiratory chain dysfunction results in decreased energy production and an increase in the generation of intracellular reactive oxygen and nitrogen species (RONS). 1,2 Mitochondrial impairment occurs in primary genetic disorders affecting the mitochondrial respiratory chain but is also apparent in a number of other conditions, such as organic acidemias and other inborn errors of metabolism, neurodegenerative diseases, diabetes mellitus, and cancer. 3,4 Although RONS are generated by normal function of the mitochondrial electron transport chain and are involved in critical intracellular processes, such as mitochondrial biogenesis and apoptosis, an abnormal increase in RONS damages cellular lipids, proteins, and nucleic acids as intracellular antioxidant systems become overwhelmed. 5 -7
Glutathione (
EPI-743
EPI-743 is a para-benzoquinone analog that is approximately 1000- to 10 000-fold more potent than coenzyme Q10 or idebenone in protecting mitochondrial and Friedreich ataxia patient fibroblasts in an oxidative stress assay. 15 This beneficial effect may be related to an ability for EPI-743 to bind to and modulate oxidoreductases, such as nicotine adenine dinucleotide phosphate [NAD(P)H]: quinone oxidoreductase 1 (NQO1, T-diaphorase), resulting in increased reduced glutathione production and improvement in redox status. 15 -17 Interestingly, pretreatment of fibroblasts derived from a polymerase γ deficiency patient with EPI-743 before exposure of cells to a strong oxidant resulted in a blunting of antioxidant response element gene expression in genes under direct control of nuclear factor-erythroid 2 p45-related factor. Heme oxygenase 1 (HMOX1), aldo-keto reductasae (AKR1C1), thioredoxin reductase 1 (TRXR1), NAD(P)H: quinone oxidoreductase 1 (NQO1), sulfiredoxin (SRXN1), and genes related to glutathione synthesis were especially affected by EPI-743 pretreatment. 16 Therefore, EPI-743 may also potentially have effects related to the transcription of genes involved in antioxidant defense.
Currently, EPI-743 is in use in a number of clinical trials and an emergency access program as described below and has been tolerated well, without evidence of dose-limiting toxicity in over 400 000 patient dosing days (M. Klein MD, email communication, December, 2016). Although the majority of studies involving EPI-743 to date are exploratory, open-label trials involving relatively few patients, more recent clinical trials related to the treatment of Leigh syndrome or Friedreich ataxia involve more patients and are randomized, double-blind, and placebo-controlled (Table 1).
EPI-743 and RP103 Clinical Trials in Mitochondrial Disorders and Conditions With Secondary Mitochondrial Involvement.
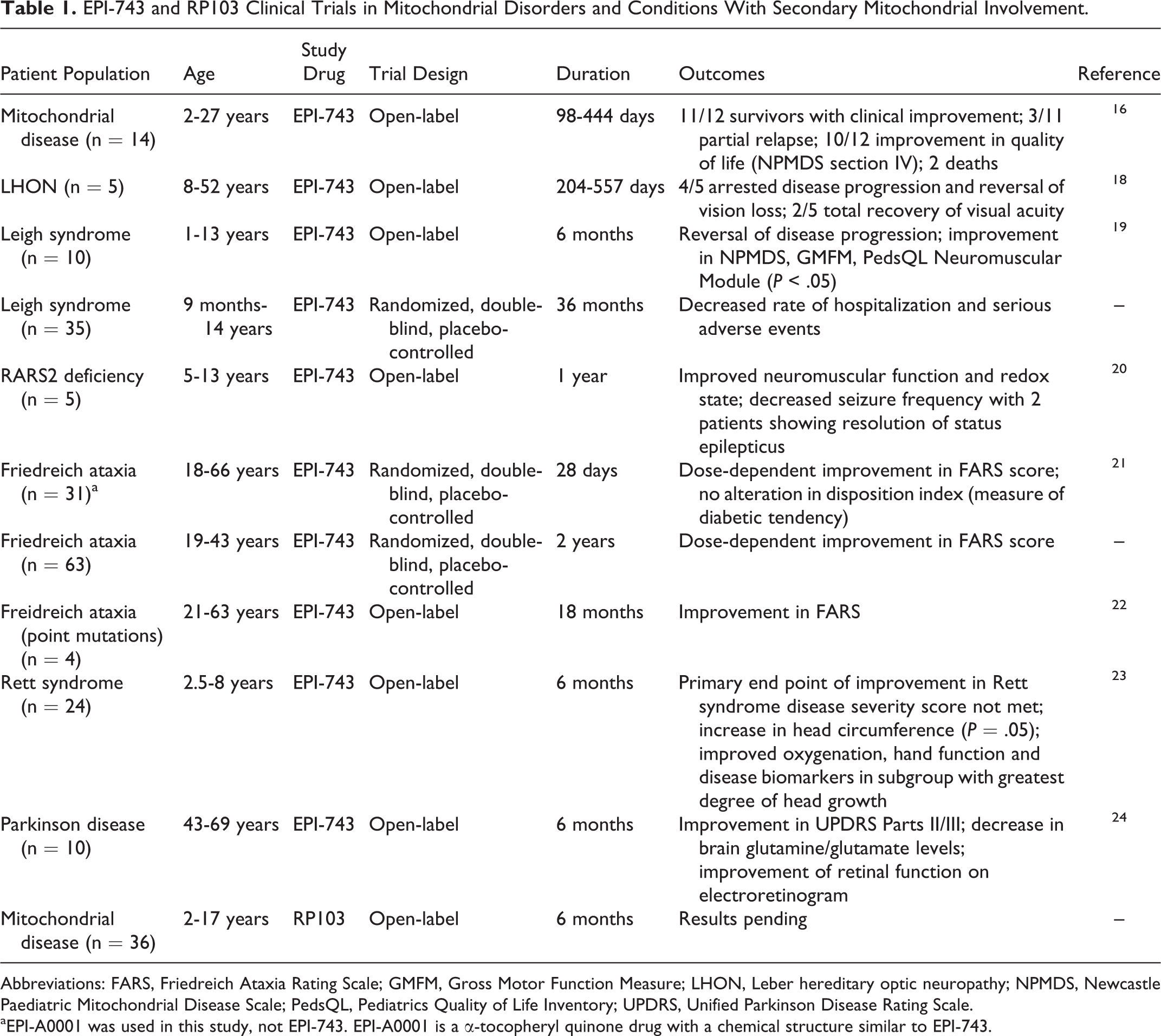
Abbreviations: FARS, Friedreich Ataxia Rating Scale; GMFM, Gross Motor Function Measure; LHON, Leber hereditary optic neuropathy; NPMDS, Newcastle Paediatric Mitochondrial Disease Scale; PedsQL, Pediatrics Quality of Life Inventory; UPDRS, Unified Parkinson Disease Rating Scale.
aEPI-A0001 was used in this study, not EPI-743. EPI-A0001 is a α-tocopheryl quinone drug with a chemical structure similar to EPI-743.
Mitochondrial Disorders
The initial experience with EPI-743 in mitochondrial disease was reported in 13 children and 1 adult who were enrolled in a 13-week emergency treatment protocol, followed by an extension protocol, for individuals who were considered to be at risk of progressing to end-of-life care within 90 days. The patients had a wide range of genetically-confirmed mitochondrial diseases (polymerase γ deficiency, n = 4; Leigh syndrome, n = 4; mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes [MELAS], n = 3; mitochondrial DNA deletion syndrome n = 1; and Friedreich ataxia, n = 1), and surviving patients were treated with EPI-743 for between 98 and 444 days. Of the 14 patients, 12 survived initially; a 7-year-old girl with Alpers syndrome died of pneumonia 9 days after starting EPI-743, and a 2-year-old boy with Leigh syndrome secondary to mutations in SURF1 died suddenly after developing respiratory insufficiency secondary to brain stem dysfunction. Of the 12 survivors, 11 demonstrated clinical improvement, with 3 showing partial relapse with time. The Newcastle Paediatric Mitochondrial Disease Scale (NPMDS) 25 was performed before and after treatment with EPI-743; NPMDS scores were unchanged or minimally different for sections I to III (current clinical function, system-specific involvement, and current clinical assessment), whereas 10 patients showed improvement in section IV (quality of life). 16
Of the 14 patients, 12 also underwent serial brain imaging with technetium-99m-hexamethylpropyleneamine oxime (HMPAO) single-photon emission computed tomography (SPECT). The 2 patients who did not undergo HMPAO SPECT scanning included a 7-year-old girl with severe Alpers syndrome who died 9 days after starting EPI-743 and a 2-year-old girl with Leigh syndrome. A 2-year-old boy with Leigh syndrome died suddenly after developing respiratory symptoms associated with brain stem disease but was able to undergo serial HMPAO SPECT scan analysis. Hexamethylpropyleneamine oxime is a lipophilic radionuclide tracer that is redox sensitive; the tracer is retained intracellularly in the presence of sufficient reducing equivalents generated by functional mitochondria. 26 At baseline, all 12 patients showed decreased brain HMPAO uptake compared to normal controls. After 3 months of EPI-743 therapy, whole-brain HMPAO uptake increased significantly. 16
An additional study of 22 mitochondrial disease patients enrolled in the EPI-743 emergency treatment protocol demonstrated a significant linear correlation between the increase in cerebellar uptake and improvement in the Newcastle score (r = 0.623; P = .00161). Furthermore, the MELAS subgroup (n = 5) in this study showed a significant relationship between whole-brain uptake of HMPAO and the Newcastle score (r = 0.917; P = .028). 27
Leber Hereditary Optic Neuropathy
Subacute, painless vision loss, more commonly in males in adolescence or early adulthood, secondary to optic nerve and retinal ganglion cell dysfunction is the primary manifestation of Leber hereditary optic neuropathy (LHON). Patients may also have extraocular manifestations such as cardiac arrhythmias and neurological involvement, including dystonia, movement disorders, tremor, peripheral neuropathy, and myopathy. 28,29 Three common mitochondrial DNA pathogenic variants (m.3460G>A in MT-ND1, m.11778G>A in MT-ND4, and m.14484T>C in MT-ND6) are responsible for causing LHON in about 90% of cases. 30 Interestingly, the m.14484T>C pathogenic variant is associated with the most favorable outcomes and even spontaneous recovery of vision in some cases. 31
A case report of 2 siblings harboring the m.14484T>C variant in a homoplasmic state noted a divergent clinical course, with a younger brother receiving EPI-743 and showing a rapid and complete recovery of vision soon after the start of therapy, whereas the older sibling did not receive specific treatment and underwent slow, partial visual recovery over years. The authors speculated about the possible etiology of the divergent clinical course in the siblings, considering (1) different nuclear genetic backgrounds, (2) environmental factors (without obvious difference between the 2 brothers), and (3) EPI-743 therapy very early after conversion as potential factors influencing the observed outcomes. 32
An earlier report of a small, open-label trial using EPI-743 included the m.14484T>C case of complete recovery discussed above and 4 additional patients, 3 of whom harbored the m.11778G>A variant and 1 the m.3460G>A variant. EPI-743 arrested disease progression and reversed vision loss in all but 1 of the 5 consecutively treated patients. No drug-related adverse events were noted. 18 Another small series evaluated EPI-743 in 3 Italian patients (a female with the m.3460G>A variant and 2 males with the m.11778G>A variant) at the stage of monocular vision loss. Following 3 days of EPI-743 therapy, all 3 patients showed improved visual function in the affected eye, with absence of disease in the second eye. 33
Leigh Syndrome
Leigh syndrome is a severe mitochondrial encephalopathy, primarily affecting the basal ganglia, thalamus, and brain stem, that is associated with significant neurological impairment in affected individuals. Multiple pathogenic variants, in both nuclear and mitochondrial DNA, cause Leigh syndrome. 34
An open-label study of EPI-743 therapy in 10 children with genetically-confirmed Leigh syndrome caused by pathogenic variants in a number of different genes showed stabilization and even reversal of disease progression. Outcome measures included the Newcastle Paediatric Mitochondrial Disease Scale, Gross Motor Function Measure, and PedsQL Neuromuscular Module. Overall, a statistically significant improvement was noted for each of the primary end points (P < .05). Adverse events related to taking EPI-743 were not observed. 19 Enrolled patients also had glutathione levels measured before and after treatment with EPI-743 in order to determine the effect of therapy on redox imbalance. Not surprisingly, Leigh syndrome patients had decreased total and reduced glutathione levels in peripheral blood lymphocytes as well as high levels of oxidized glutathione at baseline. Following treatment with EPI-743, a marked increase in reduced glutathione (P < .001) and a 96% decrease in the ratio of oxidized to reduced glutathione (P < .001) was noted. 17
More recently, EPI-743 has been used in a randomized, double-blind, placebo-controlled clinical trial in children with Leigh syndrome (NCT01721733; NCT02352896). Enrolled patients first participated in a 6-month placebo-controlled phase, which was followed by a 30-month extension phase to assess the long-term drug safety and impact on disease morbidity. In the initial 6-month placebo-controlled phase, treatment with EPI-743 was associated with fewer patients requiring hospitalization or experiencing serious adverse events as compared with those patients who received placebo (11.8% vs 42.8%). Analysis of extension phase data showed that there was a progressive decline in hospitalizations and serious adverse events from the first 6 months of EPI-743 treatment to months 19 to 24. When adjusted for the number of patients, the number of hospitalizations per patient declined by 40%. On further examination of the hospitalization rate in the cohort initially randomized to receive placebo, there was a 73% decrease in per-patient hospitalizations following EPI-743 treatment when compared to the initial period during which patients received placebo (M. Klein, MD, email communication, December, 2016; manuscript in preparation; data presented in part at the United Mitochondrial Disease Foundation Annual Meeting, Seattle, Washington, USA, 2016).
RARS2 Deficiency
Autosomal recessive pathogenic variants in the RARS2 gene, which codes for the mitochondrial arginyl-transfer RNA synthetase, have been associated with an early onset encephalopathy characterized by microcephaly, profound developmental delay, intractable seizures, spastic-dystonic quadriplegia, and, in most cases, pontocerebellar hypoplasia. Affected children have decreased levels of oxidative phosphorylation components, with variably decreased mitochondrial respiratory chain activities in multiple complexes, and biochemical evidence of mitochondrial impairment. 35 -37 To date, approximately 30 patients have been reported with pathogenic variants in RARS2. 35 In an open-label study, 5 children with RARS2 deficiency were given EPI-743 over a 12-month treatment phase, followed by an ongoing extension phase. All patients demonstrated reversal of disease progression regardless of the severity of clinical involvement at baseline. Two children showed resolution of status epilepticus, and the other 3 patients demonstrated a reduction in seizure frequency and duration. 20 All patients remain active on EPI-743 with continued improvement in seizure frequency and duration (M. Klein, MD, email communication, December, 2016).
Friedreich Ataxia
Friedreich ataxia is an autosomal recessive neurodegenerative disorder caused by abnormal GAA trinucleotide repeat expansion in FXN, the gene-encoding frataxin. During childhood, affected individuals typically present with progressive ataxia, dysarthria, weakness, absent lower limb reflexes, and a decrease in proprioception and vibratory sensation. Diabetes mellitus and cardiomyopathy are other common clinical findings. 38 An initial study involving a drug related to EPI-743 in chemical structure (EPI-A0001, α-tocopheryl quinone) in 31 adults with Friedreich ataxia used the disposition index, a measure of diabetic tendency, as the primary clinical trial outcome measure (NCT01035671). The Friedreich Ataxia Rating Scale (FARS) was used as a secondary neurological outcome measure. 39 No significant difference was observed in the disposition index between treated patients and controls after 4 weeks of therapy. However, a dose-dependent improvement in the FARS score was observed; patients on low-dose A0001 improved by 4.9 points (P = .4), whereas those on high-dose therapy improved by 6.1 points (P < .01). 21
In a phase 2 double-blind placebo-controlled trial of EPI-743 in adults with Friedreich ataxia (NCT01728064), EPI-743 showed a statistically significant improvement in neurological function and disease progression when compared to an age-, gender- and disease severity–matched natural history cohort as measured by the FARS. There was an overall improvement of 1.8 points on the FARS score following 24 months of treatment, compared to a 4.8 point worsening in the natural history cohort (P < .0001). The improvement in FARS score was dose dependent; patients who received EPI-743 at 400 mg 3 times daily for the entire 24-month study period registered the greatest degree of improvement (cf. 200 mg 3 times daily). Furthermore, there was an association with delay in starting EPI-743 and treatment effect (M. Klein, MD, email communication, December, 2016; manuscript in preparation; data presented at the Friedreich’s Ataxia Research Alliance Annual Meeting, Tampa, Florida, USA, 2016).
Rarely, Friedreich ataxia may be caused by a point mutation in FXN on 1 allele in combination with a typical GAA trinucleotide repeat expansion on the other allele. Four patients treated with EPI-743 in an open-label study of Friedreich ataxia patients who harbor a point mutation on 1 FXN allele (NCT01962363) showed clinical improvement as assessed by FARS score over 18 months of therapy. After 6 months of EPI-743 treatment, total FARS score improved an average of 9%, with bulbar and upper limb coordination subscales showing the most improvement. No serious adverse events were observed. 22
Rett Syndrome
Rett syndrome is a progressive neurodevelopmental disorder primarily affecting girls and characterized by a period of normal development followed by acquired microcephaly and neurological regression. Mitochondrial dysfunction, with resulting redox imbalance and increased oxidative stress, has been implicated in the pathogenesis of Rett syndrome. 40,41 In a 6-month, randomized, double-blind, placebo-controlled trial involving 24 Rett syndrome patients aged 2.5 to 8 years (NCT01822249), those who were treated with EPI-743 showed a statistically significant increase in head circumference relative to placebo patients. Head growth in the treatment group was 0.75 cm, compared to 0.29 cm in the placebo group (P = .05). In a subgroup of children with the greatest degree of head growth, improvements in oxygenation, hand function, and disease biomarkers were also observed. 23
Parkinson Disease
Parkinson disease is a common neurodegenerative disorder that is characterized by a loss of dopaminergic neurons of the substantia nigra and subsequent rigidity, bradykinesia, resting tremor, and postural instability. Mitochondrial dysfunction, free radical generation, and redox imbalance appear to play a role in disease pathogenesis. 42,43 A phase 2a open-label pilot study was performed in order to determine whether EPI-743 might have potential benefit in the treatment of Parkinson disease (NCT01923584). Ten patients with Parkinson disease were enrolled and 3 discontinued the study after 3 months for reasons unrelated to EPI-743 therapy. The Unified Parkinson Disease Rating Scale (UPDRS), 44 electroretinography, and brain metabolite levels as measured by magnetic resonance spectroscopy (MRS) were used as the clinical outcome measures. Six of 7 patients with follow-up MRS studies showed improvement in an objective marker of brain oxidative stress, namely, a decrease in glutamine/glutamate levels in the basal ganglia opposite the side most severely affected by Parkinson disease. In addition, there was a statistically significant improvement in retinal function on evaluation by electroretinogram. Clinically, patients demonstrated an improvement in UPDRS scores that approached statistical significance. 24
N-Acetylcysteine and Cysteamine Bitartrate Delayed-Release (RP103)
N-acetylcysteine (NAC) and cysteamine are other compounds that have the potential to increase intracellular reduced glutathione, but there is limited clinical trial experience to date. 45,46 NAC is well-known for its therapeutic effects in acetaminophen-induced liver failure, and as a mucolytic for the treatment of cystic fibrosis. N-acetylcysteine has also been used to treat a variety of other conditions, such as chronic obstructive pulmonary disease, diabetes mellitus, and patients infected with HIV. 46 N-acetylcysteine has the ability to improve markers of oxidative stress in cell lines derived from patients with mitochondrial respiratory chain disorders or Huntington disease and in a Huntington disease animal model. 47 -49 There have been case reports in which NAC was used to treat primary mitochondrial disorders. For example, 2 mitochondrial disease patients who had significant liver dysfunction, including elevated transaminases and coagulation abnormalities, showed normalization of laboratory studies within 3 to 7 days after starting NAC. 50 However, the evidence base for the use of NAC in treating mitochondrial disorders is limited. To our knowledge, controlled clinical trials that explore the efficacy of NAC in these conditions have not yet been performed but are clearly needed. N-acetylcysteine has been used in controlled trials in various conditions with likely secondary mitochondrial involvement, including Alzheimer disease, amyotrophic lateral sclerosis, and autism. 51 Although no improvement in survival or disease progression was noted in those with amyotrophic lateral sclerosis, patients with Alzheimer disease showed improvement in some measures of cognitive ability after NAC supplementation. Patients with autism have shown improvement in some aberrant behaviors, especially irritability, following treatment with NAC. 51
N-acetylcysteine, in combination with metronidazole, has been used in an open-label clinical study to treat ethylmalonic encephalopathy, a disorder caused by mutations in ETHE1. Disease pathogenesis relates to a secondary inhibition of cytochrome c oxidase, short chain fatty acid oxidation, and other enzymes. Ethe1-deficient mice treated with NAC had a prolonged life span, and 5 patients with ethylmalonic encephalopathy demonstrated marked clinical improvement following combined therapy. Improvements consisted of increased body weight and reduction, followed by virtual disappearance, of important clinical features of this disease, including diarrhea, petechiae, and acrocyanosis. Furthermore, plasma ethylmalonic acid and serum thiosulfate, a toxic marker of disease, decreased during combined therapy. 52
Cysteamine bitartrate (cysteamine), an established therapy for cystinosis, works by decreasing the abnormal lysosomal storage of cystine. Cysteamine also promotes the transport of cysteine into cells, which has the potential to increase intracellular glutathione levels. 46 Cysteamine has been shown to improve symptoms in mouse models of Huntington disease and has been used in a small open-label clinical trial in patients with Huntington disease. 53 Clinical efficacy was not demonstrated, although the trial established a safe cysteamine dosage regimen in patients with Huntington disease. 53,54 Cysteamine bitartrate delayed-release (RP103) is a microsphere formulation associated with decreased gastrointestinal symptoms. 55 RP103 is currently being used in a randomized, controlled, double-blind multicenter trial for Huntington disease (NCT02101957) and an open-label study in children with mitochondrial disease (NCT02023866) but results have not yet been published. The multiinstitutional mitochondrial trial included children aged 2 to <18 years with molecularly genetic confirmation of a mitochondrial disease. The primary endpoint of the study was the changes in the NPMDS (sections I-IV), with secondary endpoints including safety, pharmacodynamics, pharmacokinetics, and glutathione studies. As additional secondary endpoints, investigators also chose 2 standard assessments related to the predominant symptoms present in a given subject, including myopathy, dystonia, ataxia, retarded motor development, reduced activities of daily living, and vision. Children were treated in an open-label fashion, with dose escalation based on their tolerance of the drug up to a total dose of 1.3 g/m2/d. A total of 53 patients were screened and 36 enrolled in the study. Twenty-five patients completed the full 24 weeks of study. Reasons for early withdraw included noncompliance (n = 2), adverse event (n = 3), and withdraw of consent (n = 1). Patients completing the initial study were allowed to enroll in a continuation study (NCT01197378). The study was terminated by the sponsor and data analysis is in progress.
We are still at a relatively early stage in the development of therapies for mitochondrial disorders. However, multiple clinical trials related to improving cellular redox status in patients with primary or secondary mitochondrial disorders are certainly encouraging. Such clinical trials not only have provided hope to the patients and their families and caregivers, but also will serve as important stepping stones for further studies as our understanding of mitochondrial disease pathogenesis continues to improve.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: GME and BHC report receiving funding for being an investigator in clinical trials related to EPI-743 (BioElectron Technology Corporation, previously known as Edison Pharmaceuticals, Inc) and RP-103 (Horizon Pharmaceuticals, Inc, Raptor Pharmaceuticals, Inc), and GME has received unrestricted gift research funds from Edison Pharmaceuticals, Inc.
