Abstract
Since Christian de Duve first described the lysosome in the 1950s, it has been generally presented as a membrane-bound compartment containing acid hydrolases that enables the cell to degrade molecules without being digested by autolysis. For those working on the field of lysosomal storage disorders, the lack of one such hydrolase would lead to undegraded or partially degraded substrate storage inside engorged organelles disturbing cellular function by yet poorly explored mechanisms. However, in recent years, a much more complex scenario of lysosomal function has emerged, beyond and above the cellular “digestive” system. Knowledge on how the impairment of this organelle affects cell functioning may shed light on signs and symptoms of lysosomal disorders and open new roads for therapy.
Lysosomal Composition and Biogenesis
Lysosomes are membrane-bound compartments formed by a lipid bilayer that contains a number of characteristic proteins, such as lysosome-associated membrane proteins (LAMPs) 1 and 2, lysosome integral membrane protein (LIMP2), and tetraspanin CD63. 1 Their biogenesis and functions are shared by lysosome-related organelles (LROs) whose composition and denomination vary according to cell type. Such organelles include melanosomes in melanocytes, lytic granules in lymphocytes, and delta granules in platelets, among others. Both lysosomes and LRO participate in many cellular processes such as signal transduction, antigen presentation and immune response, cell death, autophagy, and membrane repair. 2 Lysosome-associated membrane protein cytosolic tails interact with microtubules, thus having an important role in lysosome exocytosis, dynamics, and protein translocation. 1
Lysosomal biogenesis relies on a complex trafficking mechanism from the trans-Golgi network (TGN) that is responsible for sorting the intraluminal and membrane-bound proteins that form the lysosomes. Acid hydrolases are modified with mannose 6-phosphate (M6P) residues, allowing their recognition by M6P receptors in the TGN and ensuing transport to the endosomal/lysosomal system. On the other hand, other soluble enzymes and nonenzymatic proteins are transported to lysosomes in an M6P-independent manner by LIMP-2 or sortilin. Sorting of cargo receptors and lysosomal transmembrane proteins requires sorting signals present in their cytosolic domains. 3
Lysosome function is heavily dependent on its fusogenic and acidic properties. The first makes it possible for the organelle to merge not only with the endocytic vesicle but also with the autophagosome and the plasma membrane. The latter is responsible for regulating the optimal pH for substrate degradation, a measure to ensure that the lytic pathway is only activated at the precise moment. Both characteristics are due to the presence of families of lysosome membrane proteins such as proton-pumping ATPases or vesicle-associated membrane proteins (VAMPs) and other soluble N-ethylmaleimide-sensitive factor attachment protein receptors (SNARE). It is important to recognize that the addition of such proteins changes the properties of the compartment itself. For instance, acidity can be altered by the delivery of vesicular type H+ ATPase complex, whereas fusogenicity can be altered by the delivery of VAMP7. Therefore, the multiple delivery pathways must act together to allow the regulated and sequential deposition of lysosomal components. 1 The increased acidic pH is a transition marker from early to late endosomal vesicles (multivesicular bodies) and then to lysosomes. 4
Gene Expression
A body of work led by Andrea Ballabio group identified transcription factor EB (TFEB) as a master regulator of genes related to lysosome biogenesis, acidification, and function. 5 Moreover, they characterized a network of coregulated genes involved in many cellular processes directly or indirectly related to lysosomes (including autophagy, vesicle-mediated transport, and cell response to stress). This Coordinated Lysosomal Expression and Regulation (CLEAR) network includes over 470 genes with a consensus TFEB-binding site. 6 TFEB is a basic helix–loop–helix (bHLH) leucine zipper transcription factor that also regulates autophagy-related genes. Phosphorylated TFEB is retained at the cytoplasm, whereas nuclear translocation is regulated by sensing mechanisms via mammalian (or mechanistic) target of rapamycin complex 1 (mTORC1, see below). 7,8 Another transcription factor from the same family as TFEB, TFE3, was also shown to regulate the expression of genes associated with autophagy and lysosomal biogenesis, even though not binding to CLEAR motifs, but still regulated by mTORC. 9 Evidence suggests that increasing TFEB expression and nuclear translocation may result in reduced storage in lysosomal diseases and increased autophagic activity that is beneficial for a variety of neurodegenerative diseases. 10 Similar preliminary results were also shown for TFE3. 9
The TFEB and the TFE3 are part of a family of 4 closely related bHLH leucine zipper transcription factors also composed by Microphtalmia-associated transcription factor (MITF) and TFEC, which can bind target DNA both as homodimers or heterodimers. 11 Transcription factor E3 or TFEB proteins also regulate several molecular pathways in carcinogenesis, other than metabolism regulation, including the activation of transforming growth factor β, E-cadherin expression, and retinoblastoma-dependent cell cycle arrest. 12 The MITF was recently reported as able to increase expression of late endosomal proteins, such as Rab7, LAMP1, and CD63 in a cell model of melanoma. These late endosomes, however, were not functional lysosomes as they were less active in proteolysis. However, the accumulation of multiple vesicular bodies led to enhanced Wnt signaling generating a positive-feedback loop that contributes to the proliferation of melanoma. 13
Brignull et al 14 using expression correlation analyses found that signal transducer and activator of transcription 6 (Stat6)/interleukin (IL)-4 are novel regulators of lysosome-specific genes. The Stat6 is itself regulated by IL-4, and these results were further confirmed in primary mouse macrophages derived from Stat6 deficient or wild-type animals. The authors suggest that in cells cultured with IL-4, the expression of 103 lysosomal genes was dependent on Stat6, reflecting 40% of the known lysosomal proteome and 54% of lysosomal genes expressed in this cell type. Furthermore, Stat6, under IL-4 induction, seems to control 14 of the 15 different subunits of vacuolar H+ ATPase thus proving fundamental for lysosomal pH maintenance and therefore lysosomal function.
Recently, a database of human lysosomal genes (http://lysosome.unipg.it) and its regulation have been established to collect information on the several genes related to lysosomal biogenesis and function. Genes are included based on the presence of TFEB-binding sites and the presence of the nucleotide motif (GTCACGTGAC) characteristic of genes belonging to the CLEAR network. The database focuses not only on lysosomal genes but also on putative microRNAs that can act as their posttranscriptional regulators. 15
Lysosomal Function in Substrate Degradation
Lysosomal Storage Disorders
In the field of medical genetics, lysosomes are primarily associated to lysosomal storage disorders (LSDs), a group of more than 50 inborn errors of metabolism that has an incidence of about 1:7000. 16 Although most LSDs are due to deficiencies in lysosomal hydrolases, defects in membrane-associated proteins and nonenzymatic lysosomal proteins have also been described. Soluble lysosomal enzymes, such as glycosidases, sulfatases, phosphatases, and lipases, are involved in substrate degradation and are crucial to the turnover of cellular components. Other causes of LSD include errors in enzyme targeting (such as in mucolipidosis II and III), errors in posttranslational processing (multiple sulfatase deficiency), and activator deficiencies (as saposin deficiencies). Also, defects in transport proteins are related to LSDs (such as Niemann-Pick disease type C1 [NPC1], ceroid-lipofuscinosis neuronal 3 [CLN3], and cystinosis). 1,17,18
For many years, a simplistic mechanism of substrate accumulation leading to disturbance of cell homeostasis was applied to LSD. Over the years, however, and especially from the 1990s with the development of animal models, a more complex understanding of these diseases has emerged. Disturbances in lysosomal degradation of macromolecules were viewed not isolated but in conjunction with other lysosomal functions in the cell—and in some instances, the very study of LSD contributed to the knowledge of lysosome biology as well. 19 More than grouping LSD by the type of stored substrate, primary or secondary storage, and enzyme or nonenzyme defects, researchers are recognizing the many common pathogenic cascades elicited by the dysfunction in one or many cellular processes involving the lysosomal compartment. 17,20 Moreover, phenocopies of lysosomal diseases can be made by silencing RagA/B in mice, showing that RagA/B GTPases are key regulators of lysosomal function and cardiac protection. 21
Lysosomal Functions Other Than Substrate Degradation
Signal Transduction
Endocytosis of ligand-bound transmembrane receptors is a general mechanism by which cells control signal transduction. Tyrosine kinase receptors, for instance, are internalized following ligand-induced activation. The specific internalization pathway may vary between clathrin-dependent and clathrin-independent routes, depending on the specific type of receptor. 22 For Epidermal Growth Factor Receptor (EGFR), the canonical process elicited by EGF binding initiates with the receptor migrating to a clathrin-coated pit, where it is internalized by endocytosis in a clathrin-coated vesicle. Afterward, the pathway includes migration through early and late endosomes, with ligand release. Part of EGFR enters the recycling pathway, being shuttled back to the plasma membrane. The remaining receptor molecules and ligands are digested in the lysosomes. Interestingly, decorin, a small leucine-rich proteoglycan found in the extracellular matrix, changes the endocytic pathway upon EGFR ligation. Internalization occurs via caveolae-mediated endocytosis, and EGFR is transferred directly to late endosomes for final degradation. This leads to a decreased number of EGF-binding sites on cell surface, thus promoting growth suppression and anti-oncogenic activities. 23
Antigen Presentation and Immune Response
Antigen presentation and immune response show lysosome involvement. Antigen presentation involves processing foreign peptides for major histocompatibility complex (MHC) class II presentation, and the role of lysosomes spans from epitope liberation to the processing of MHC class II molecules. Antigens that enter the cell via endocytosis move through the endolysosomal pathway by a series of pH changes and vesicle aggregation. As the antigen is processed in the early endosome, MHC class II molecules are transferred from the TGN into the nascent late endosome/early lysosome vesicle. There, proteases perform a stepwise maturation of the invariant chain of MHC class II that prepares the molecule for receiving the foreign peptide. Finally, epitopes are mounted on the MHC molecules for antigen presentation. The whole vesicle then moves along microtubules toward the cell membrane where MHC class II molecules bound to epitopes are exposed for CD4+ T cells in the cell surface. A very rigid mechanism of control based on low levels of protease expression and slow acidification kinetics has a profound impact on antigen presentation. 24
In addition, cells of the immune system, such as T cells and Natural Killer (NK) cells, possess secretory lysosomes or LROs, which are also called lytic granules. These contain not only MHC class II molecules but also Fas ligand, granzyme A, and perforin. Upon activation, secretory lysosomes move along microtubules toward the plasma membrane in direction to the target cell. The lytic granules are then released to the immunological synapse that is formed between the lymphocyte and the target cell. 25
Autophagy
Lysosomes are also involved in autophagy. Autophagy is a vacuolar self-digesting mechanism responsible for the removal of long-lived proteins and damaged organelles. It has a role in quality control of cellular components, degrading aberrant protein aggregates, and defective organelles, as depolarized mitochondria. 26 It is also essential in metabolic stress situations, as nutrient deprivation, providing energy by recycling components for protein synthesis. In mammalian cells, autophagy can be classified in 3 types of events—microautophagy, chaperone-mediated autophagy (CMA), and macroautophagy. Chaperone-mediated autophagy involves direct engulfment of proteins with KFERQ motifs contents by the lysosomal membrane or interaction with specific proteins, such as heat shock protein (hsp)70 and LAMP2A. 27 Macroautophagy, commonly referred to simply as autophagy, is the most well-studied type and requires the formation of an autophagosome and its subsequent fusion to endosomes–lysosomes. 28,29
The autophagy mechanism itself may be directly involved in the pathogenesis of lysosomal disorders. In cystinosis, caused by defects in cystine transporter cystinosin (CTNS), there is a decrease and mislocalization of LAMP2A and consequent impairment of CMA. Correction of CMA is only achieved by the expression of
One of the major suppressors of autophagy is the mTORC1 signaling pathway. In normal cellular conditions, in the presence of sufficient nutrients, mTORC1 is recruited to the lysosomal membrane and activated by another complex formed by v-ATPase, Ragulator, and Rag GTPases. It then phosphorylates TFEB at Ser142 and Ser211 and TFE3 at Ser321, which induces ligation with 14-3-3 proteins and retention at the cytoplasm. 8,9,30 In situations of starvation and lysosomal stress, mTORC1 is released from lysosomes. The unphosphorylated TFEB and TFE3 are able to translocate to the nucleus and directly induce transcription of the CLEAR network genes, including several genes related to LSDs, as discussed above.
Other kinases also interfere with TFEB signaling, and extracellular signal-regulated kinase 2 (ERK2) has a similar effect to mTORC1 on TFEB. However, in osteoclasts, ERK2 phosphorylation of TFE3 induces its promoter activity on target genes. 9,29
The binding of Rag GTPases to the lysosomal vacuolar-type H+-ATPase is thought to be a way for the Rags to sense the amino acid content inside lysosomes and, by extension, the nutritional state of the cell. 31 It is also known that TFEB and MITF interact directly with Rags, and that the binding is dependent on the nucleotide binding state of Rags. 32 This mechanism is important in many disease-related situations, such as cancer. Pancreatic ductal adenocarcinoma, for instance, heavily depends on MITF/TFE family of transcription factors (MITF, TFE3, and TFEB) that induce high levels of lysosomal catabolic function essential for tumor growth. 33
Cell Death
Besides participating in the autophagic cell death, lysosomes are associated with the 2 other major types of cell death—apoptosis and necrosis. 34 It has been shown that in response to endogenous or exogenous stress, the lysosomal membrane can leak, leading to the release of catabolic hydrolases (cathepsins) that mediate caspase-dependent apoptosis, caspase-independent apoptosis-like cell death, or even necrosis 35 (Figure 1). Apparently, the extent of lysosomal membrane permeabilization (LMP) is related to the ensuing type of cell death, with a limited release of lysosomal contents triggering apoptosis or apoptosis-like cell death, whereas generalized lysosomal rupture results in rapid cellular necrosis. 36
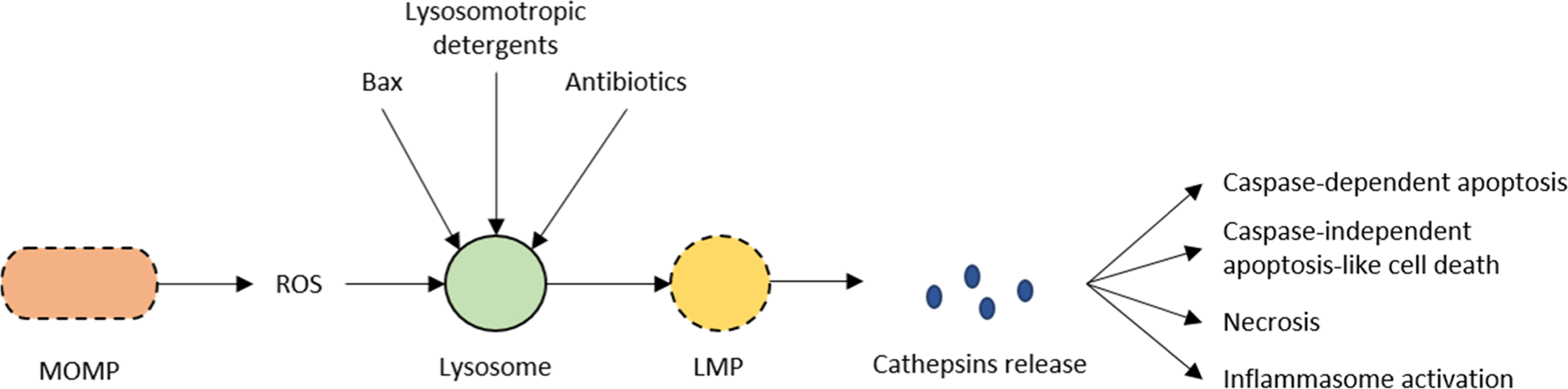
Lysosomes and cell death. Proapoptotic molecules (as Bax, lysosomotropic detergents, and some antibiotics) directly target the lysosomal membrane, causing lysosomal membrane permeabilization (LMP). The mitochondrial outer membrane permeabilization (MOMP) and consequent reactive oxygen species (ROS) generation also affect the lysosomal membrane, being the major physiologically relevant route to late-onset LMP. It leads to the release of cathepsins, which mediate caspase-dependent apoptosis, caspase-independent apoptosis-like cell death, and necrosis. It can also activate the inflammasome, resulting in caspase 1-dependent pyroptosis.
Different agents are reported to induce LMP. Proapoptotic molecules, such as Bax, directly target the lysosomal membrane as well as lysosomotropic detergents and some antibiotics. 37 Lysosomal membranes are particularly sensitive to reactive oxygen species (ROS)-mediated damage, which can be generated by destabilized mitochondria leading to peroxidation of membrane lipids. The mitochondrial outer membrane permeabilization (MOMP), which is accompanied by ROS generation, seems to be the major physiologically relevant route to late-onset LMP. 35 In addition, osmotic lysis or direct destabilization by surfactant activity is also known to cause LMP. Cathepsin release after LMP results in both caspase-dependent and caspase-independent pathways of apoptosis. 34 It can also activate the inflammasome, resulting in caspase 1-dependent pyroptosis. 37 Moreover, LMP also induces the formation of protein aggregates in classic late-infantile CLN2. 38
Lysosomal cell death occurs in tissue remodeling, immune response to intracellular pathogens, aging, and neurodegenerative diseases. The limitation of neutrophil population during infection and inflammation is mainly achieved by LMP. 37 Aging alters the physical and chemical properties of lysosomes, rendering them more sensitive to stress, and thus to LMP 39 and neurodegenerative conditions such as Parkinson, Alzheimer, and Huntington diseases. 37 It also mediates death of cancer cells, and a target for cancer therapy uses lysosomotropic drugs that enhance LMP. Dielschneider et al 40 showed that patients with chronic lymphocytic leukemia (CLL) are more susceptible to LMP due to increased levels of sphingosine and the overexpression of sphingosine-1-phosphate phosphatase 1 compared to normal B cells. The CLL cells were found to be sensitive to a lysosomotropic agent, despite other prognostic factors. Similarly, in NPC, the storage of sphingosine leads to LMP and cell death, particularly in the cerebellum, with consequent neurodegeneration. 41 Cell death by LMP has also been reported in mucopolysaccharidosis type I. 42 Moreover, since this mechanism can be induced by immunological reactions and ROS-mediated damage, common characteristics of LSDs, it may be more frequent than reported and an important factor in several diseases.
Even though lysosomal cell death is being increasingly studied and viewed as a potential alternative for anticancer therapies, many doubts remain about the underlying mechanisms. For instance, it is not known whether LMP results in the selective release of certain cathepsins and whether a subpopulation of lysosomes is especially susceptible to LMP. 36 Moreover, it remains to be elucidated whether LMP is a primary event in the cascade of cell death, upstream of MOMP, and other signals or whether it is an intermediate mechanism. Finally, even though Bid is considered the main physiological substrate of lysosomal cathepsins, there is evidence of cathepsin-independent signaling in lysosomal-associated cell death. 35
Other Functions
Lysosomes are also involved in cell membrane repair. The mechanism of wound repair involves Ca2+ influx that triggers lysosomal exocytosis. In some circumstances, this leads to a patch repair of the wound with lysosomal markers such as LAMP-1 detected at the outer leaflet of the plasma membrane. More recent evidence, though, suggests that lysosomal patching and endocytic events may act simultaneously during membrane repair. 43 In this model, lysosome exocytosis in fact promotes remodeling of the plasma membrane by releasing acid sphingomyelinase (ASM). Acid sphingomyelinase cleaves the phosphocholine head group of sphingomyelin, generating ceramide which, in turn, drives membrane invagination and endocytosis. The simultaneous translocation of lysosomal V1 H+-ATPase to the plasma membrane may help to achieve the low pH necessary for ASM activity. This suggests that, at least for some types of lesions, plasma membrane wound repair involves not only patching but also lesion removal. 44,45 Either way, lysosomes are pivotal players for maintaining cell integrity.
Lysosomes may play a role in tumor invasion and metastasis, as cancer cells show altered lysosomal trafficking and increased expression of cathepsins. In cells located to the edge of the tumor, lysosome localization shifts from a perinuclear to a peripheral pattern, indicating that lysosomal contents are secreted into the extracellular space, where they help degrading the extracellular matrix. 36
This location of lysosomes may be related to the modulation of components of the adhesion machinery by lysosomal proteases, although it remains to be determined if the subcellular localization alters the lysosomal membrane stability and hydrolytic capacity. 46 Recent work from Johnson et al 47 described reduced acidity and proteolytic activity in peripheral lysosomes when compared to juxtanuclear ones in HeLa cells. This findings, however, must be confirmed in other cellular types.
In addition, it has been shown that lysosome exocytosis may represent an important mechanism of drug resistance in tumor cells by which lysosomotropic drugs are extruded to the extracellular compartment. Furthermore, different mechanisms of lysosomal sequestration of chemoterapics have been shown, either passive or mediated by ATP-binding cassette (ABC) transporters. In addition, TFEB-mediated lysosomal biogenesis is triggered by drug sequestration, resulting in enhanced capacity of chemoresistance. 48 The inhibition of lysosomal activity could emerge as a new therapeutic strategy to overcome drug resistance in cancer. 49 Machado et al 50 showed that the inhibition of lysosomal exocytosis reversed invasiveness and chemoresistance in aggressive sarcoma cells.
Finally, lysosomes are involved in double-stranded DNA degradation mediated by Dnase2a, a lysosomal endonuclease. In Dnase2A-deficient cells, damaged DNA originated in the nucleus accumulates in small speckles that colocalize with the autophagy machinery. However, the lysosome-autophagosome fusion fails to happen in the absence of the enzyme, and the DNA fragments accumulated in the cytosol trigger inflammation via the Sting cytosolic DNA-sensing pathway. 51
Conclusion and Future Prospects
This interaction between lysosomal biology and LSD pathophysiology has shown to be mutually beneficial, both for the basic science of cell biology as to the applied field of medical genetics. For example, the role of lysosomes in membrane repair led the observation that cells from patients with Niemann-Pick disease type A (NPA), lacking ASM activity, show signs of reduced injury-induced endocytosis and deficient membrane repair that may account for some aspects of the pathology. 52 The broader understanding of cellular mechanisms involving the lysosomes may lead to secondary therapeutic targets of LSD other than the enzyme replacement/substrate reduction binomium. On the other hand, the implication of lysosomes in pathogenic mechanisms of common diseases, such as Alzheimer and cancer, opened new roads for research in the field of lysosomal biology that eventually will also benefit the LSD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research and/or authorship of this article.
