Abstract
Fabry disease is a rare, X-linked lysosomal storage disorder resulting from deficient α-galactosidase A activity and globotriaosylceramide accumulation throughout the body. This accumulation leads to various clinical disorders, including inner ear lesions, with sensorineural hearing loss and dizziness. Although hearing loss is recognized in these patients, its incidence and natural history have not been characterized. Hearing disorders develop mainly in adulthood, and tinnitus may be an earlier symptom in Fabry disease. A significant incidence of mid- and high-frequency sensorineural hearing loss in affected males is commonly reported, whereas in female carriers, it is much less frequent. In addition, a high incidence of vestibular disorders with dizziness and chronic instability is also observed in these patients. The few studies about the effects of enzyme replacement therapy (ERT) on cochleovestibular symptoms show controversial results. Based on the model of densely stained material accumulation in the inner ear, stria vascularis cell, and organ damage, an early indication of ERT may prevent hearing loss due to the reduction in substrate accumulation.
Introduction
Fabry disease (FD) is an inborn error of glycosphingolipid catabolism and results from the enzymatic deficiency of the α-galactosidase A. It produces a progressive accumulation of globotriaosylceramide (Gb3) and globotriaosylsphingosine (lyso-Gb3) in multiple tissues and organs, including the ear. 1 The otorhinolaryngologic manifestations such as hearing loss, tinnitus, vertigo, and instability express inner ear damage, in both cochlear and vestibular system. Clinical and instrumental evaluation allows diagnose cochleovestibular symptoms in patients with FD. A few studies about the effects of enzyme replacement therapy (ERT) on cochleovestibular symptoms showed controversial results.
Epidemiology
Hearing loss occurs in the second and fourth decade of life in males and females, respectively. 2 Otherwise, subclinical changes in hearing can occur in childhood. Hemizygous male patients had earlier and more severe hearing loss than heterozygous female patients. 3 -5 Adult patients (mainly males) with renal failure and/or cerebrovascular insufficiency and no residual enzyme activity are high-risk population for developing hearing loss. The residual enzyme activity, greater than 1.5%, provides a protective effect on the inner ear.
Histopathology
The underlying mechanism of auditory and vestibular symptoms remains unclear. Histopathological changes in cochleovestibular system appear to be responsible for such symptoms or can explain the otological symptoms associated with FD. Schachern et al 1 described histopathological findings in temporal bones of patients with FD. In this report, cochleovestibular damage was the result of moderate loss of outer hair cells, areas of tectorial membrane collapse, reduction in number of fibers in spiral ganglia in the basal turn with normal cell morphology, atrophy of the stria vascularis and the spiral ligament in each turn, new bone formation and fibrotic replacement tissue in the nonampulated end of the superior semicircular canal, and the ballooning of ganglion cells in Scarpa’s ganglia. At middle ear level seropurulent effusions, hyperplastic mucoperiosteum in mastoid and normal ossicles aspect with the exception of hyperostosis of the stapedial crura was described. Finally, foamy vacuolated deposits (Gb3) in endothelial cells and smooth muscle cells were evident in the internal auditory artery, modiolar vessels, and vas spirale.
Cochleovestibular Manifestations
The cochleovestibular symptoms in FD include progressive or sudden hearing loss, tinnitus, vertigo, and dizziness. These symptoms are due to histopathological changes in cochleovestibular structures as well as the result of the accumulation of glycosphingolipids, mainly in the endothelium and smooth muscle cells of the microvasculature with subsequent stenosis and thrombosis of small arteries and arterioles with degeneration in the cochlea, otolithic organs, and semicircular canals. 1
Computerized tomography of the brain and temporal bones and magnetic resonance with angiography 2 respectively showed enlargement of intradiploic vascular channels of the temporal bones and elongation and ectasia of the middle cerebral arteries and the arteries of the circle of Willis, especially the internal carotid and basilar arteries (Figure 1). 6
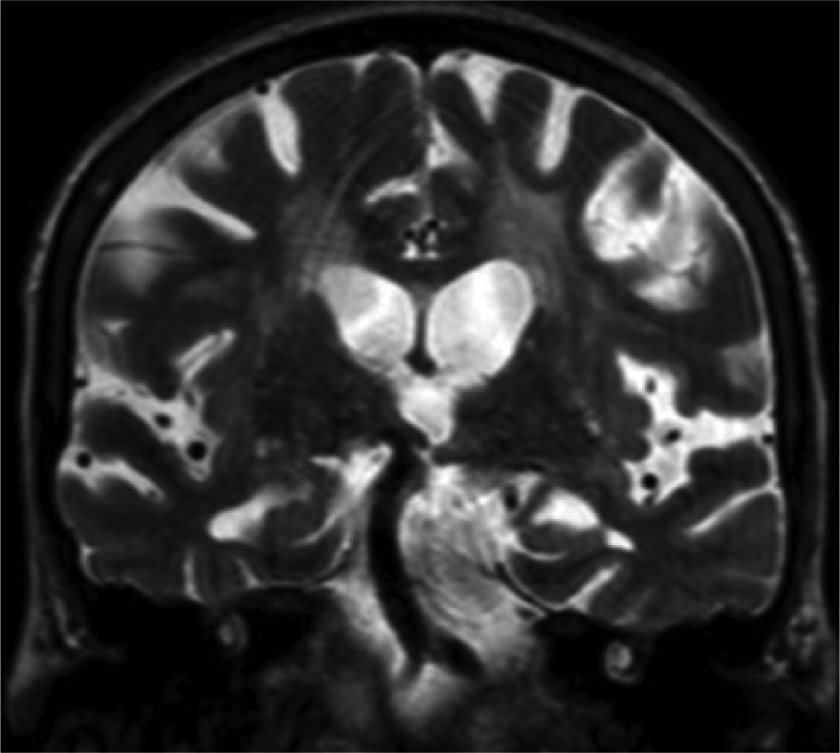
Vertebrobasilar dolichoectasia in a 42-year-old male with Fabry disease.
The internal auditory artery, which usually originates from the anterior inferior cerebellar artery, even though it can also branch directly from the basilar artery, is the main arterial that gives supply to the vestibular system and the cochlea. Due to the frequent occurrence of sudden hearing loss, it was suggested that ischemic repeated injury of the organ of corti (due to stenosis or occlusion of cochlear small vessels) 1,4,7 rather than neural hearing pathways could be the leading explanation for this manifestation. 8 However, progressive hearing loss is frequently reported as a result of Gb3 accumulation in the audiovestibular ganglia and vessels of the cochlea. 1
Nevertheless, other authors correlate lyso-Gb3 levels in plasma with hearing loss and postulated that elevated levels have a neurotoxic effect. However, there is little evidence that supports this hypothesis. 9 According to the literature, hearing loss in patients with FD seems to be the most important otorhinolaryngologic manifestation and accounts for 50% of morbidity, being sensorineural the most common type. Most adult patients (18%-55%) 9 show a bilateral progressive sensorineural hearing loss that affects mainly the higher frequencies (Figure 2), 10 although other studies showed deterioration in all frequencies, 11 which is associated with reduced word recognition. These findings are related to cochlear rather than retrocochlear deficit. 4
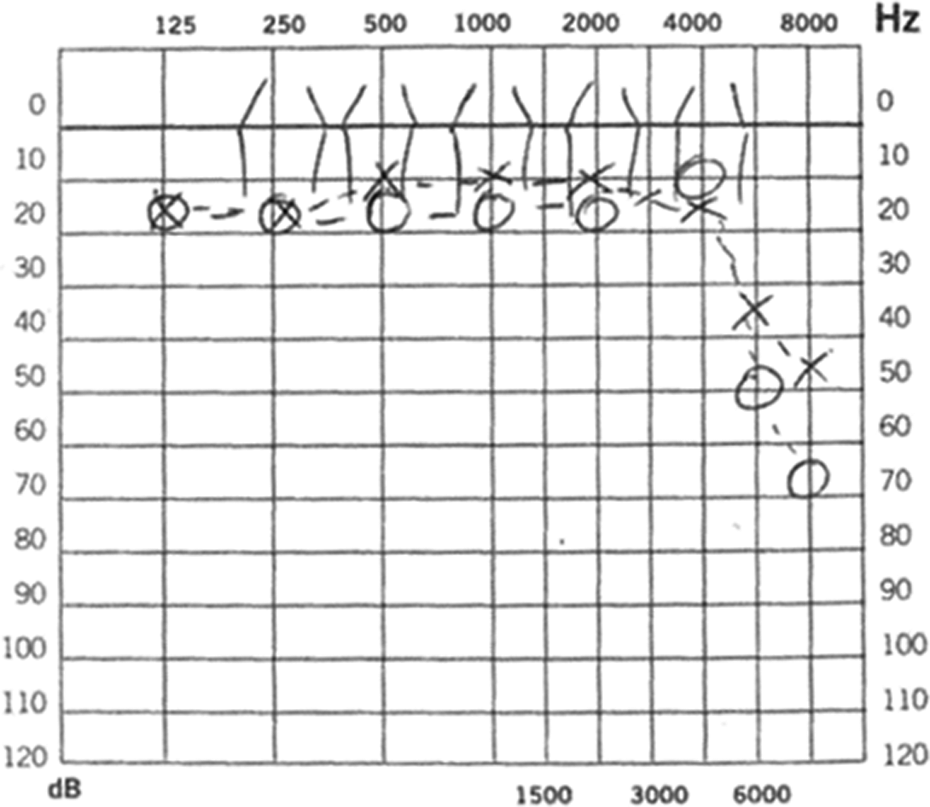
Pure tone audiogram: an 8-year-old boy with Fabry disease. Bilateral normal hearing between 125 and 4000 Hz and high-frequency (6000-8000 Hz) hearing loss.
Prevalence of sudden hearing loss in FD ranges from 5% to 30% (Figure 3). 2 The diagnostic criteria for sudden hearing loss are a decrease in the hearing acuity ≥30 dB for at least 3 consecutive frequencies, occurring over a 72-hour period. Despite the high prevalence of sensorineural hearing loss, other studies reported cases of conductive impairment. 10 Also, one case report described sudden hearing loss associated with otosclerosis (stapes fixation and cochlear capsule resorption), but the link between otosclerosis and FD is not clear. 12
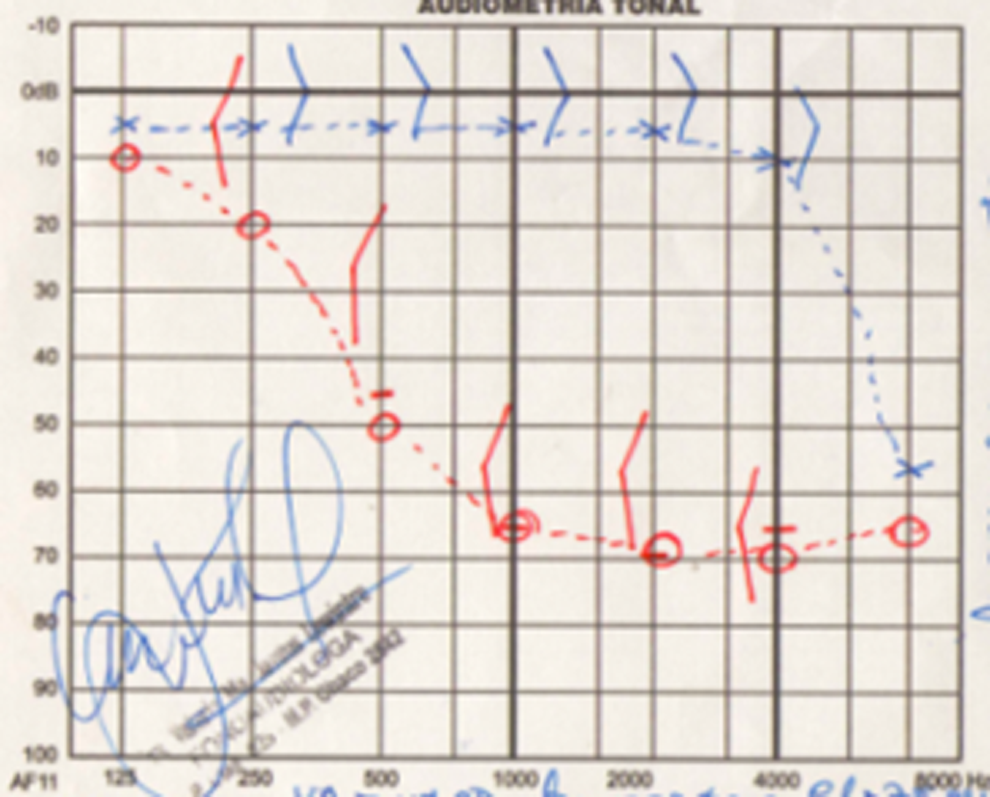
Pure tone audiogram: 14-year-old male patient with Fabry disease. Left ear (blue line): normal hearing (125-4000 Hz) and high-frequency (8000 Hz) hearing loss. Right ear (red line): sudden hearing loss and high-frequency tinnitus (8000 Hz).
Tinnitus is another frequent symptom (17%-53%) 9 in patients with FD. Unlike hearing loss, tinnitus is more frequent in younger patients without significant gender differences and frequency increases with age. 5 Independent of the hearing loss or tinnitus, patients can undergo vestibular symptoms such as vertigo, dizziness, and/or chronic instability related to peripheral and central vestibular damage. The abnormalities of the stria and spiral ligament can lead to an endolymphatic hydrops, and thrombotic zones in the cerebellar vermis and vestibular nuclei occasioned by focal infarcts cause disturbance in the balance system. The major cause of dizziness is an alteration of the central balance centers, 13 but Vibert et al 14 have proposed that endolymphatic hydrops due to an obstruction of the semicircular canals could be other reason. The frequency of vertigo episodes is as similar as tinnitus and the incidence increases with age. However, most of these patients are susceptible to have orthostatic hypotension, which can cause dizziness and faintness, instead of true vertigo. 5
Cochlear and Vestibular Evaluation
Audiological tests used to identify and diagnose hearing loss depends on patient age. Audiological evaluation in adults consists of pure tone air and bone conduction thresholds audiometry for 250 to 8000 Hz, logoaudiometry or speech audiometry (which evaluates word recognition ability) with phonetically balanced monosyllabic words, and acufenometry or tinnitometry (it characterizes tinnitus intensity, frequency, and location). 15 These studies determine the type and degree of hearing loss, word recognition failure, and characteristics of the tinnitus (loudness, location).
If these studies show normal results, other audiological tests allow the assessment of cochlear function and study possible hearing impairment not detected with the tests referred. These tests are otoacoustic emissions (evaluates outer hair cells function) and high-frequency audiometry (explores frequency ranges from 8000 to 16 000 Hz). Some patients displayed no otoacoustic emissions without having reported hearing loss. 3
In order to evaluate the middle ear functions, the tympanometry is used for the evaluation of middle ear pressure and compliance, and acoustic stapedius reflexes (impedanciometry) are used for the evaluation of mobility of ossicles. For children under 5 years old, instead of pure tone and speech audiometry, conditioned play audiometry (uses behavioral conditioning to get children to respond to sounds), tympanometry and stapedius reflexes threshold, and otoacoustic emissions are used. 15
In case of suspicion of retrocochlear involvement (auditory pathway), the brain stem evoked response audiometry (BERA) is indicated, both for adults and children. The BERA is an electrophysiological test procedure that studies the electrical potential generated at various levels of the auditory system starting from cochlea to cortex. With regard to vestibular evaluation, it consists of clinical examination and videonystagmography (VNG) with ocular, positional, and caloric tests. The VNG analyzes and reports involuntary eye movements (nystagmus) in response to certain visual cues, positional changes in head and body, and warm and cold water circulated in the ear canal in order to evaluate the oculomotor and vestibular function. This test allows to differentiate between central and peripheral vertigo. Caloric vestibular testing can reveal a peripheral vestibular deficit in patients without symptoms. Other optional tests for dizziness include vestibular evoked myogenic potentials and electrocochleography.
Effects of ERT on Cochleovestibular Symptoms
A few studies reported the effects of ERT on cochleovestibular symptoms and showed controversial results. Some authors reported a slightly hearing loss improvement after long-term treatment (30 months of ERT). This improvement was observed in patients with mild to moderate hearing loss, but no changes were reported in patients with severe hearing loss degree. 5,10 Meanwhile, some studies show evidence that hearing loss remained stable with ERT and suggested that it provides some protection for the ear inner epithelium. 3 Finally, other study proposed no benefits with ERT. 14
Despite the different results described above, ERT has no proven effect on prevention. However, some authors, based on the model of densely stained material accumulation in the inner ear, stria vascularis cell, and organ damage, argue that ERT may prevent hearing loss because it would decrease the accumulation of lyso-Gb3. Therefore, the onset of otorhinolaryngologic symptoms would be delayed. 16
Conclusion
Involvement of the inner ear is frequent in patients with FD, contributing to its morbidity. Ear, nose, and throat symptoms include progressive unilateral or bilateral sensorineural hearing loss, sudden hearing loss, tinnitus, vertigo, and instability. These symptoms occur with greater frequency and severity in male patients in the second decade of life. The cerebrovascular insufficiency and kidney failure increase the risk of hearing loss. The residual enzyme activity, greater than 1.5%, provides a protective effect on the inner ear. It is recommended that all patients (adults and children) diagnosed with FD be submitted annually to an audiological and vestibular evaluation. In this way, it can be discovered the cochlear and vestibular involvement in patients without clinical symptoms of hearing loss and vestibular symptoms. This will allow an early diagnosis and treatment indication.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
