Abstract
The Fabry Outcome Survey (FOS) is an international long-term observational registry sponsored by Shire for patients diagnosed with Fabry disease who are receiving or are candidates for therapy with agalsidase alfa (agalα). Established in 2001, FOS provides long-term data on agalα safety/efficacy and collects data on the natural history of Fabry disease, with the aim of improving clinical management. The FOS publications have helped establish prognostic and severity scores, defined the incidence of specific disease variants and implications for clinical management, described clinical manifestations in special populations, confirmed the high prevalence of cardiac morbidity, and demonstrated correlations between ocular changes and Fabry disease severity. These FOS data represent a rich resource with utility not only for description of natural history/therapeutic effects but also for exploratory hypothesis testing and generation of tools for diagnosis/management, with the potential to improve future patient outcomes.
Introduction
Fabry disease is an X-linked lysosomal storage disorder caused by deficiency of the hydrolytic enzyme α-galactosidase A (α-Gal A). 1 The natural history of Fabry disease includes progressive accumulation of the ganglioside globotriaosylceramide (Gb3) in cells and organs throughout the body, leading to multisystem pathology and ultimately premature death in the fourth or fifth decade from renal, cardiac, or cerebrovascular complications. 1 -6 The specific inborn error of glycosphingolipid catabolism is due to one or more of numerous possible mutations of the GLA gene, which results in reduced or absent enzyme activity. 7 Historically in the general population, classic Fabry disease has an estimated regional prevalence between 1:37 000 8 in male newborns in northwestern Italy and 1:117 000 9 among all newborns in Australia. However, recent publications reporting results from newborn screening for Fabry disease show a much higher prevalence when “atypical” Fabry disease mutations are included, ranging from 1:2900 to 1:3900 8,10 -12 and up to 1:1400 in some regions. 13 Fabry disease was considered to primarily affect male hemizygotes, virtually all of whom exhibit progressive kidney dysfunction 14 ; however, female heterozygotes are not merely carriers of Fabry disease but also can portray all the signs and symptoms of Fabry disease, albeit with a later onset and more variable phenotype than that observed in males. 4,5
The Fabry Outcome Survey (FOS) is an international, observational, physician-directed registry sponsored by Shire for patients diagnosed with Fabry disease who are either candidates for or are receiving enzyme replacement therapy (ERT) with agalsidase alfa (agalα; Replagal, Shire Human Genetic Therapies, Inc., Cambridge, Massachusetts), 15,16 a human form of α-Gal A produced in a cultured human cell line by gene activation. 17 FOS was established in 2001 with the aim of broadening the understanding of the nature of Fabry disease and improving clinical management of affected patients. 16 As of June 1, 2016, FOS includes data from 2962 patients with Fabry disease. Since its inception 15 years ago, FOS has yielded 47 peer-reviewed publications and made key contributions to the understanding and treatment of Fabry disease (Table 1).
Key Contributions of FOS.
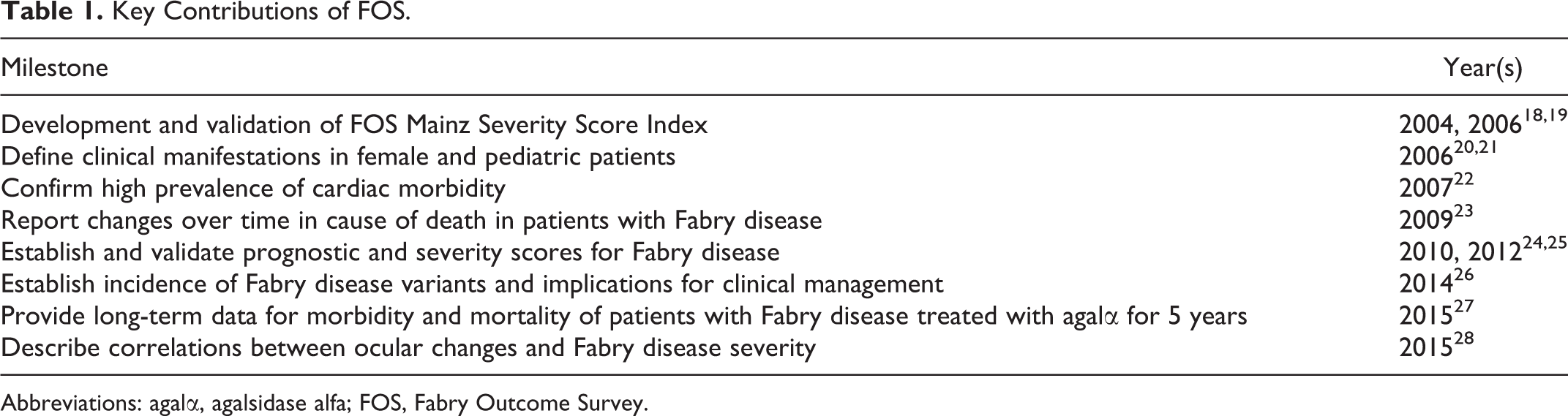
Abbreviations: agalα, agalsidase alfa; FOS, Fabry Outcome Survey.
The aim of this review is to summarize the key attributes of FOS and describe the major contributions to the understanding and treatment of Fabry disease from FOS over its first 15 years.
Evolving Design of FOS
Since its inception in 2001, FOS has grown to include a total of 24 countries represented in the registry in 2016, with 22 countries actively participating (Figure 1). The FOS design factored in knowledge of the natural history of Fabry disease at the time of its inception, such as multiregional information on the genetic component, epidemiology, and diversity of signs and symptoms. 1,2,4,5 For instance, sequential patient-level data on specific clinical outcomes (such as kidney function and quality of life [QOL]) from different FOS specialist centers were pooled to attain a sufficient sample size. 16 In patients receiving agalα ERT, treatment response is assessed at least every 6 months, whereas untreated patients are reviewed annually to monitor disease progression. Hence, FOS facilitates study of the natural history of this rare disease in a large and diverse group and provides baseline data against which the effects of agalα ERT can be gauged. Further, the international distribution of participating FOS centers allows comparison of outcomes between different areas to potentially identify geographic differences in the delivery, effectiveness, and safety of treatment. In addition, FOS supports agalα ERT efficacy shown in short-term clinical trials by providing evidence for long-term effectiveness and safety of agalα in a real-world setting. 29
Countries with centers participating in the Fabry Outcome Survey.
A key challenge when collecting data via a patient registry is ensuring comprehensive data capture. Several steps were initiated in 2005 to improve the completeness of FOS data capture, namely (1) a focus on centers enrolling ≥20 patients, (2) concentration on a limited number of clinically relevant core variables (ie, Fabry disease–related signs and symptoms, pain, QOL, height, weight, blood pressure [BP], serum creatinine, urinary protein, and left ventricular mass), and (3) the introduction of clinical research associates to facilitate data collection and handling by participating treatment centers. 15
The physician assessments and patient questionnaires used within FOS are reflective of the multisystem involvement of Fabry disease and designed to capture multiple aspects of the disease. Participating center staff are trained on the use of the database, and guidelines produced by FOS working groups promote uniform data collection. Adults and children participating in FOS undergo thorough and regular health checks, including physical examination, vital sign measurements, echocardiography, and clinical laboratory testing. Patient-reported outcomes used in FOS by adult patients include the Brief Pain Inventory, 30 EuroQol 5-Dimensions questionnaire, 31 and the general FOS questionnaire. In addition, women are provided the Female FOS questionnaire, which aims to identify potential early predictive factors for disease progression and severity in this patient group. Child health and social effects are assessed with the FOS Paediatric Health and Pain questionnaire (FPHPQ; adapted from the Brief Pain Inventory and the general FOS questionnaire), 32 whereas health-related QOL is measured by the KINDL questionnaire. 33
Fabry Outcome Survey Analyses and Publications: Highlights of the Last 15 Years
Diagnosis, Disease Severity, and Mortality
A major attribute of FOS is the capacity to perform different types of observational analyses. The first publication emanating from FOS reported on a cross-sectional analysis of baseline data from a cohort of 366 patients across 11 European countries. 16 The analysis revealed long delays from onset of symptoms to a correct diagnosis in both male and female patients (13.7 and 16.3 years, respectively), demonstrated that the disease affects females as well as males, and showed that signs and symptoms may be present from early childhood. 16 These findings were confirmed in a more recent cross-sectional analysis of 262 FOS patients residing in Austria, Germany, and Switzerland, which also found that Fabry disease remained underdiagnosed in these countries. 34 Furthermore, this analysis linked disease severity measured by the FOS Mainz Severity Score Index (MSSI) to patient age (P = .0001). 34 A subsequent analysis of Spanish FOS patients found that they were diagnosed at a slightly earlier age of 11 years. Spanish patients had a smaller proportion of organs involved, irrespective of sex, although FOS-MSSI scores were worse in male patients. 35
Before FOS, it was difficult to define genotype–phenotype relationships because of extreme variability in the clinical phenotype, age of onset, and course of Fabry disease, even within the same family. By extracting data from male patients in FOS (to minimize nongenetic factors and avoid the large phenotypic variability associated with Fabry disease in women), positive correlations were found between age at FOS entry and FOS-MSSI score, as well as the number of affected organs. 36
In the 2007 cross-sectional analysis by Cybulla et al, involvement of the kidneys or heart was the main cause of morbidity and death in 75% of patients with Fabry disease. 34 A cohort study compared the causes of death in 42 FOS patients between 2001 and 2007 with that of their 181 non-FOS relatives who had Fabry disease and had died previously (most before 2001). 23 Among the 42 FOS patients, cardiac disease was the main cause of death in both male (34%) and female (57%) patients. In contrast, the principal causes of death among Fabry-affected relatives before 2001 were renal failure in males (42%) and cerebrovascular disease in females (25%). These data suggest that renal disease is less important as a cause of mortality in patients with Fabry disease now than in the past and that the relative importance of cardiac disease has increased, possibly reflecting recent improvements in the management of renal disease with supportive care and ERT. 23
Longitudinal examination of FOS data also has demonstrated real-world effectiveness and safety of agalα 0.2 mg/kg every other week in adults and children, 3,37 -40 its beneficial effects on pain scores/QOL, 41 and its effectiveness in patients with Fabry nephropathy. 42 In addition, findings from a retrospective FOS cohort analysis showed that 5 years of agalα ERT in FOS resulted in delayed onset of morbidity (estimated risk of a composite morbidity event: ∼16% [∼26% in males] in patients treated with agalα 0.2 mg/kg every other week vs ∼45% for patients not treated with ERT at 24 months) and mortality (estimated median survival for males: 77.5 vs 60 years, respectively), compared with a well-described and closely matched external cohort of untreated patients. 27
Cardiac Manifestations
Although there are characteristic signs and symptoms of Fabry disease such as acroparesthesia, pain, gastrointestinal symptoms, and angiokeratomas that are commonly seen in patients with Fabry disease, cardiac involvement is important because of its association with premature death. 16 Given its progressive nature, all patients with classic Fabry disease will develop characteristic signs and symptoms. These include cardiac manifestations such as left ventricular hypertrophy (LVH) with cardiomyopathy, myocardial fibrosis, and symptoms like angina, palpitations, and dyspnea, which were reported frequently among untreated men and women in the seminal cross-sectional analysis of FOS data. 16 A larger analysis of cardiac manifestations in 714 FOS patients from 11 countries showed that untreated men and women had hypertrophic cardiomyopathy reflecting the progressive nature of the disease over time, with ongoing disease-related alterations such as myocardial fibrosis and clinical symptoms including angina, dyspnea, arrhythmias, and syncope. 22 The presence of hypertrophic cardiomyopathy was associated with a statistically significantly higher frequency of cardiac symptoms, arrhythmias, and valvular heart disease. Multivariate logistic regression analysis in untreated patients demonstrated that sex, age, and renal function, but not BP, were independently related to LVH. Cardiac disease accounted for 29% of all deaths and was the most frequent reported cause of death in females. 22 In patients with Fabry disease not treated with ERT, LVH progression is rapid and may be detected even within a relatively short follow-up period of 6 months. 43
Overall in FOS, onset of any cardiac symptom occurred earlier in males than in females (31.5 vs 39.9 years, respectively), 22 as did documentation of LVH (38.0 vs 55.4 years, respectively). 16 Similarly, in a subanalysis of Taiwanese FOS patients with LVH, cardiac symptoms appeared at an earlier age in men with classic Fabry disease mutations than in women, respectively (30.0 vs 49.6 years; P < .05). 44 However, baseline analysis of the late-onset cardiac variant GLA splicing Fabry mutation, IVS4 +919G>A, revealed that, unlike in classic Fabry disease, cardiac symptoms appeared at the same age in males and females (55.9 and 57.4 years, respectively). 44
Treatment with agalα in FOS has shown that several cardiac outcome measurements can improve and the progression of reductions in cardiac function may be slowed (Table 2). In a longitudinal analysis of 5 years of agalα ERT in FOS, patients with baseline LVH (defined as left ventricular mass index [LVMI] ≥48 g/m2.7) had a sustained reduction in LVMI from 71.4 to 64.1 g/m2.7 (P = .0111; Figure 2) and a significant increase in midwall fractional shortening (MFS; a measure of systolic function) from 14.3% to 16.0% (P = .02) after 3 years of treatment. 3 From baseline to 5 years, 80.7% of patients receiving agalα ERT had a stable LVMI and 76.9% had stable or improved MFS. 3 Stable LVMI and MFS were also noted in agalα-treated patients without baseline LVH; this suggests that when ERT is initiated before LVH has developed, these patients are unlikely to develop cardiomyopathy. 3 These results were corroborated by a separate 5-year analysis that compared FOS patients with an external cohort of untreated and closely matched patients with Fabry disease (Table 2). 27
Effects of Agalα ERT on Cardiac and Renal Manifestations in FOS Analyses.
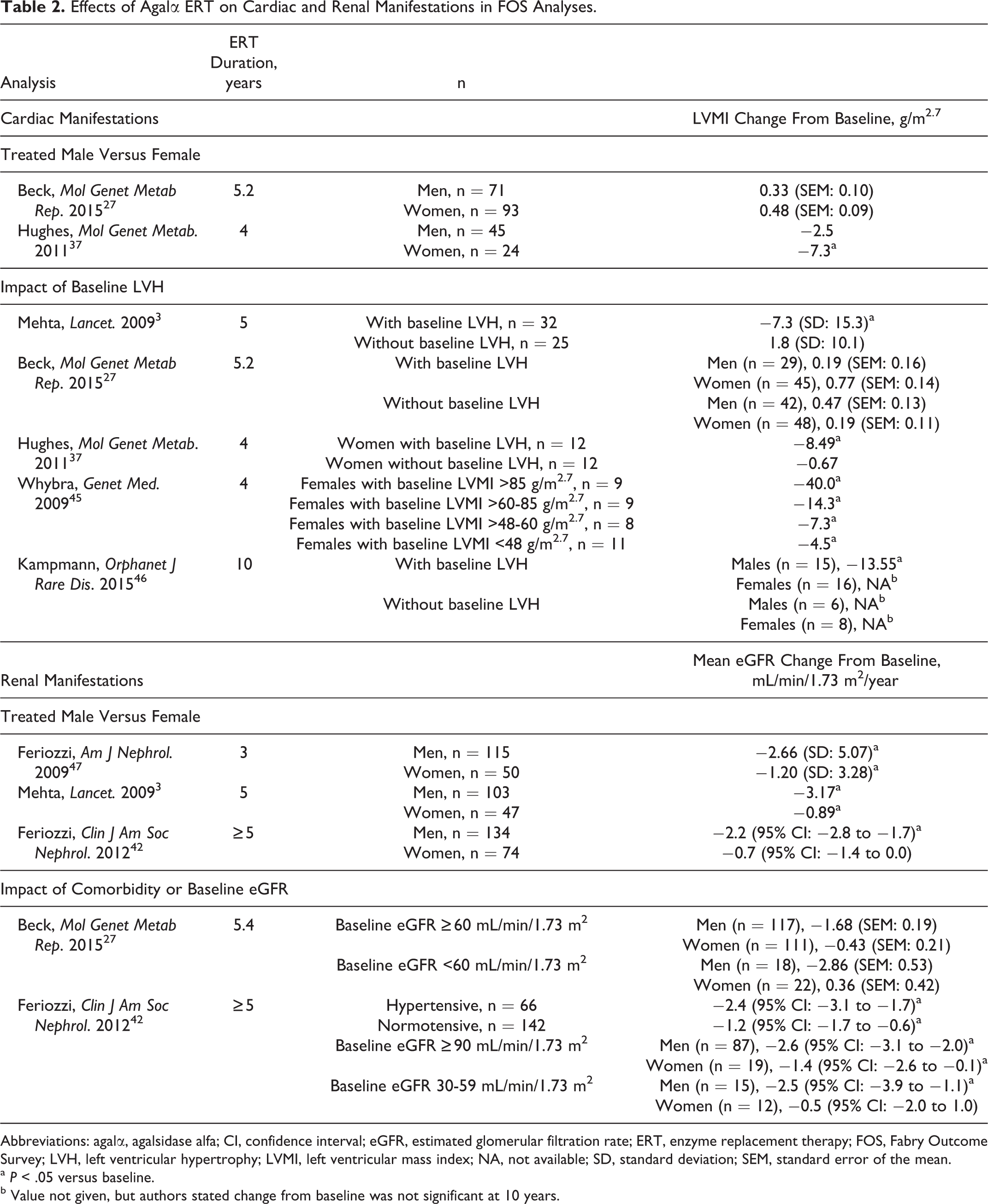
Abbreviations: agalα, agalsidase alfa; CI, confidence interval; eGFR, estimated glomerular filtration rate; ERT, enzyme replacement therapy; FOS, Fabry Outcome Survey; LVH, left ventricular hypertrophy; LVMI, left ventricular mass index; NA, not available; SD, standard deviation; SEM, standard error of the mean.
a P < .05 versus baseline.
b Value not given, but authors stated change from baseline was not significant at 10 years.
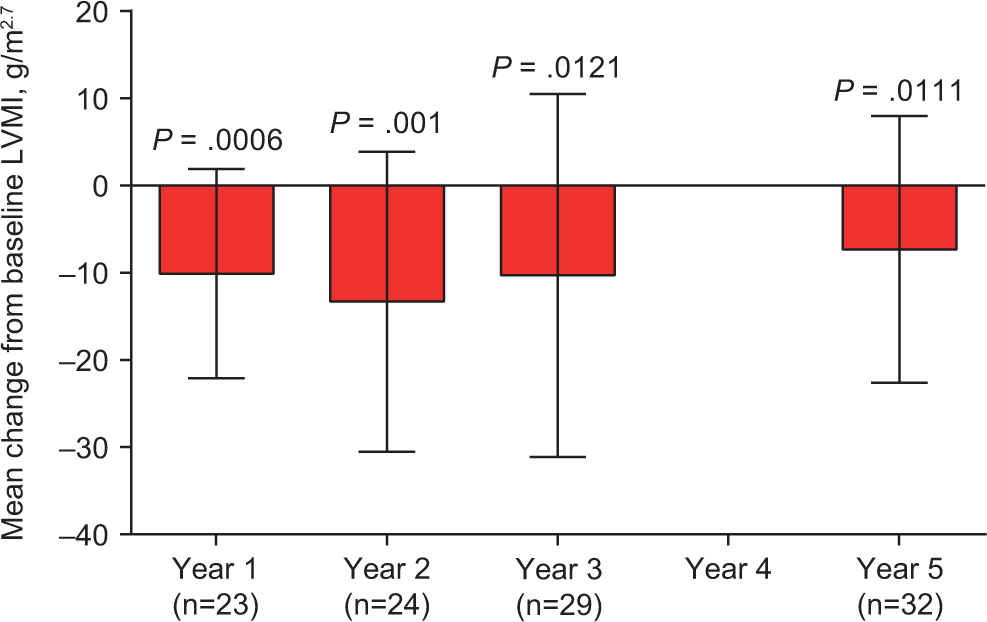
Mean (standard deviation) change in LVMI in FOS patients with baseline cardiac hypertrophy who were treated with agalα. 3 agalα indicates agalsidase alfa; FOS, Fabry Outcome Survey; LVMI, left ventricular mass index.
Other analyses from FOS suggest that treatment-related effects associated with agalα ERT are not affected by the sex of the patient. When FOS data were stratified by sex and examined over a 4-year agalα treatment period, the extent of the mean reduction in LVMI from baseline in 78 females with preexisting LVH was similar to that in 172 male counterparts. 37 These findings are consistent with those from a prospective, single-center, open-label study of 36 women with symptomatic Fabry disease, showing that in the subgroup of women with LVH at baseline, mean LVMI decreased from 89.4 to 66.5 g/m2.7 after 12 months of agalα ERT (P < .001) and remained at this lower level throughout 4 years of agalα ERT. 45 In 10 of the 11 women without LVH at baseline, LVMI remained within the normal range after 4 years of agalα ERT. 45
Subanalysis of FOS agalα ERT data by region has consistently shown effects associated with treatment comparable with those seen in the overall group. 46,48 A retrospective analysis of data prospectively collected from 45 adults who received agalα ERT for 10 years at a single German FOS center showed no progression of cardiomyopathy against a range of structural and functional end points. 46 Agalα ERT was associated with improvements in heart failure classification by ≥1 class (22 of 42 evaluable patients), stable or improved angina scores (41 of 42 patients), no LVH development in patients without LVH at treatment initiation, and significant reduction in LVMI among male patients with LVH at treatment initiation. 46 Another analysis showed that agalα ERT stabilized left ventricular heart size over a 4-year period in 33 Spanish patients participating in FOS. 48
In a 2004 cross-sectional analysis of FOS data, the prevalence of uncontrolled hypertension (defined as BP ≥130/80 mm Hg) was 52.4% among 391 men and women with Fabry disease (60 received agalα ERT), 49 which compares unfavorably with a 23% prevalence of uncontrolled office BP >130/80 mm Hg in 436 patients not from FOS with hypertension and chronic kidney disease but without Fabry disease. 50 As is the case in other hypertensive patient populations, an inverse relationship between BP and estimated glomerular filtration rate (eGFR) in FOS has been described on several occasions. 49,51,52 Over the course of 2 years of agalα ERT, a significant decrease in both systolic and diastolic BP was observed, accompanied by a stabilization of kidney function. 49 In another analysis of 66 FOS patients with hypertension, systolic and diastolic BP were reduced after ≥5 years of agalα ERT, from mean (standard deviation) values of 131.9 (17.2) and 80.7 (12.1) mm Hg to 123.3 (15.7) and 75.9 (11.4) mm Hg, respectively. 42 Hypertension and other cardiovascular diseases have a shared epidemiology with chronic kidney disease due to pathophysiologic connections between these disease states, and their coexistence portends cardiovascular events and death. 53 Data from FOS also suggest that Fabry disease may also accelerate the development and progression of coexisting heart and chronic kidney disease via the accumulation of Gb3 in each organ type. 54 Retrospective data collected over 14 years in 25 men with Fabry disease indicated that stage 5 chronic kidney disease (ie, glomerular filtration rate category G5 [kidney failure]) was a strong indicator of cardiovascular disease progression irrespective of the use of ERT. 54 In contrast, ERT initiated before kidney failure stabilized both cardiac and renal disease in this cohort. 54
Renal Manifestations
Renal manifestations are another major area of interest in Fabry disease, because renal impairment is an important cause of morbidity and mortality. 14,34 Progressive decline in renal function occurs in approximately half of the patients in FOS; requirement for renal transplantation or dialysis was reported in 17% of men and 1% of women in a cross-sectional study. 16 One analysis reported renal disease in 84% of FOS patients. 41 Renal involvement typically includes proteinuria, seen in 44% to 54% of males and 33% to 41% of females. 16,41 Among 366 FOS patients (241 were receiving ERT), progression to renal failure requiring renal transplant or dialysis was reported in 17% of male and 1% of female patients. 16
Data from FOS (Table 2) have provided supporting evidence that agalα ERT can stabilize or slow the progressive decline in renal function typically seen in patients with untreated Fabry disease, 3,27,42,47,55,56 particularly in patients without severe renal impairment before treatment. 41 Over 3 years of agalα ERT, male and female patients had mean (standard deviation) changes in eGFR of −2.7 (5.1) and −1.2 (3.3) mL/min/1.73 m2/year, respectively, with no significant change in proteinuria. 42,47 During longer periods of treatment (mean [range]: 7.4 [5.0-11.2] years), mean (95% confidence interval) changes from baseline in eGFR were −2.2 (−2.8 to −1.7; P < .01 vs baseline) mL/min/1.73 m2/year in men and −0.7 (−1.4 to 0.0; P = .05 vs baseline) mL/min/1.73 m2/year in women (Figure 3), with minimal change in proteinuria levels and irrespective of therapy with angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers. 42 One FOS analysis of patients who received 5 years of treatment showed mean eGFR changes in men and women of −3.2 and −0.9 mL/min/1.73 m2/year, respectively. 3 A separate analysis retrospectively compared outcomes in FOS patients who received 5 years of agalα ERT with published outcomes in an external cohort of untreated and closely matched patients with Fabry disease and found that treated patients experienced slower declines in renal function (Table 2). 27
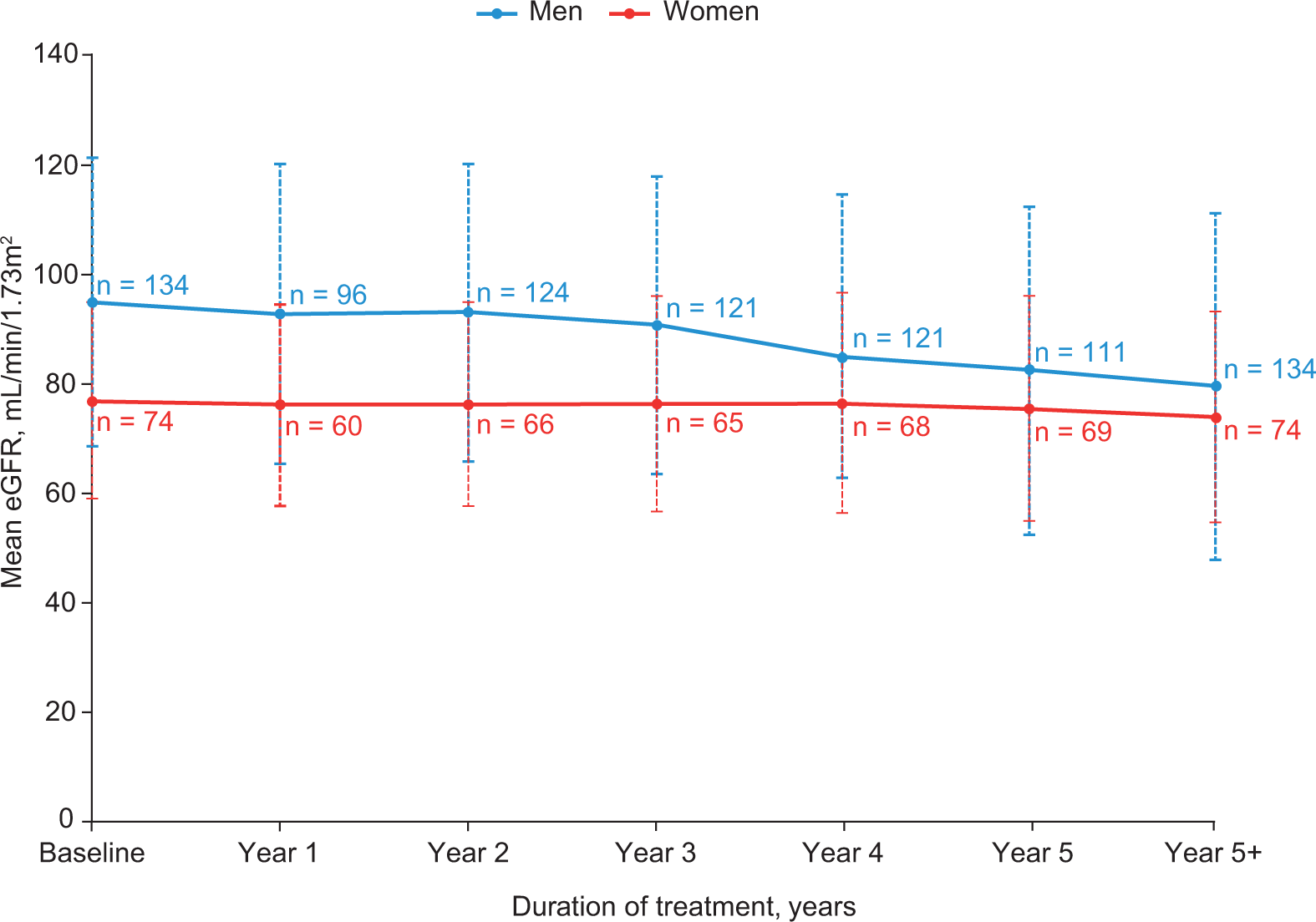
Mean (standard deviation) eGFR in men and women from FOS during ≥5 years of agalα treatment. 42 agalα indicates agalsidase alfa; eGFR, estimated glomerular filtration rate; FOS, Fabry Outcome Survey.
After 5 years of agalα ERT, 66 patients with hypertension in FOS showed a greater decline in renal function than 142 normotensive patients (mean [95% confidence interval] change in eGFR: −2.4 [−3.1 to −1.7] vs −1.2 [−1.7 to −0.6] mL/min/1.73 m2/year, respectively; P < .01). 42 In another analysis of outcomes following 5 years of agalα ERT, patients with better-preserved renal function at baseline (eGFR ≥60 mL/min/1.73 m2) had a mean (standard error of the mean) eGFR change of −1.7 (0.2) mL/min/1.73 m2/year in males and −0.4 (0.2) mL/min/1.73 m2/year in females; among patients with more severe baseline renal impairment, mean (standard error of the mean) eGFR change was −2.9 (0.53) mL/min/1.73 m2/year in males, whereas eGFR showed little change in females, with a minimal increase of 0.36 (0.42) mL/min/1.73 m2/year. 27 This advantage for patients on ERT with better-preserved eGFR is consistent with the hypothesis that earlier therapy is better and that this may be due to less renal sclerosis or scarring.
Among FOS kidney transplant recipients, those not receiving agalα ERT had more severe renal impairment and proteinuria than those receiving agalα ERT; over 2 years of ERT, there was a decrease in eGFR (from 59.2 to 51.1 mL/min/1.73 m2) and proteinuria remained stable. 57 A sensitivity analysis by Beck et al that included patients who had received a renal transplant or dialysis before FOS entry showed no substantial differences from the results in the overall renal cohort, which showed a slower decline in renal function over 5 years of agalα ERT than that seen in an untreated population. 27
Neurologic Manifestations
The most frequently reported signs and symptoms of Fabry disease are neurological, seen in 84% of male and 79% of female FOS patients in Europe. 16 The most common neurological symptom is neuropathic pain, occurring in 76% of male and 64% of female patients and beginning at a mean age of 9.4 years in males and 16.9 years in females. 16 One of the common early symptoms of Fabry disease is acroparesthesia (burning, tingling, or pricking sensations), which, along with small fiber peripheral neuropathic pain, can substantially reduce QOL. 5,58 -60 Another FOS analysis reported that mean age of onset for pain was 14.8 years in males and 19.8 years in females. 60 Pain is an important dimension affecting QOL; data from untreated men and women in FOS showed significantly lower QOL than in an age- and sex-matched population without Fabry disease. 58 The impact of agalα ERT has been favorable; significant 12% to 32% improvements in measures of QOL and pain, as well as significant decreases in pain prevalence and severity, have been reported after 1, 2, 3, and 5 years of agalα ERT. 3,58,60
Fabry disease manifests in the central and peripheral nervous system; perhaps the most devastating neurologic consequence is stroke, which occurs at an increased prevalence and at a younger age in the Fabry disease patient population compared with the general population. 61,62 Stroke is frequently seen along with cardiovascular abnormalities including valvular heart disease, LVH, arrhythmia, or hypertension. 61,63 One analysis found that cerebrovascular events, including stroke and transient ischemic attack/prolonged reversible ischemic neurologic deficit, occurred more often in women than men (27% and 12%, respectively), although the mean age at onset of cerebrovascular events was higher in female than in male patients (43.4 and 28.8 years, respectively). 16 Characteristic white matter changes in magnetic resonance imaging scans of the brain have been described in patients from FOS who experienced ischemic stroke, as well as subcortical gray matter lesions and the pulvinar sign (bilateral hyperintensities around the pulvinar thalamic nuclei in the region of the posterior thalamus). 61,64 To date, no definitive evidence of a reduced incidence of stroke in patients receiving agalα ERT has been published, although significant improvements in cerebral vascular function have been noted. 65 -67
Auditory symptoms, including tinnitus and hearing loss, have been described in 57% of male and 47% of female FOS patients. 16 Hearing was described as significantly worse among patients with Fabry disease than in an age-matched general population, although clinically relevant hearing impairment was noted in only 16% of patients. 68 In a study of children in the FOS database, subjective hearing impairment was reported by 33%, although this was confirmed by audiometry in only 19% of children; tinnitus was reported by 44% and the presence of tinnitus was correlated with overall Fabry disease severity. 69 This pattern of hearing loss in patients with Fabry disease is similar to age-related hearing loss seen in non-Fabry populations, but it starts at a younger age and progresses more rapidly; it is more severe in males than in females. 68 Hearing loss was classified as sensorineural in most patients, although a conductive component also was described in some; acute hearing loss has also been described. 68 Hajioff et al examined the effects of agalα ERT on hearing and concluded that a median of ∼1 year of treatment resulted in stabilized or improved hearing among patients who had not already progressed to severe hearing loss. 70 Significant improvements were noted among patients with mild or moderate hearing loss before treatment, whereas no significant changes were seen in patients who had normal hearing or severe hearing loss before treatment. 70
Other Manifestations
Gastrointestinal manifestations, including abdominal pain and diarrhea, have been reported in 57% of male and 47% of female patients in FOS 16 ; another FOS analysis found gastrointestinal involvement in 52% of patients (50% of adults and 61% of children). 71,72 The most common symptoms were abdominal pain in 33% and diarrhea in 21% of patients. 71,72 Roughly similar to acroparesthesia, the median age at onset of earliest gastrointestinal symptoms was ∼13 years. 72 Gastrointestinal symptoms were seen at a lower prevalence after 1 and 2 years of agalα ERT, with the greatest improvements noted in children. 71,72 Furthermore, among children who did not have abdominal pain before starting treatment, there were no reports of abdominal pain as a new symptom during 2 years of ERT. 71,72
Anemia is considered an important additional risk factor for morbidity and mortality in patients with various comorbidities, including renal, 73 cardiac, 74 and cerebrovascular 75 disease. A cross-sectional study by Kleinert et al found that anemia with hemoglobin <11 g/dL was present in 34% of FOS patients (47% of men and 20% of women). 76 Anemia in patients with Fabry disease is more common than in the general population and is often seen in association with impaired renal function, heart failure, and evidence of systemic inflammation (eg, elevated C-reactive protein levels in blood), 76 as well as in association with use of ACEIs. 77 Anemia was more prevalent among patients with eGFR <60 mL/min/1.73 m2 (61%) versus patients with eGFR ≥60 mL/min/1.73 m2 (30%; P < .001). 76 Among those patients who received agalα ERT, the median hemoglobin concentration was stable over 2 years of therapy. 76 Information regarding the use of ACEIs was not provided in this study.
Dermatologic manifestations of Fabry disease have been described in 78% of male and 50% of female patients in FOS. Angiokeratomas have been reported in 66% of males and 36% of females in FOS, 78 appearing at a mean age of 17.9 years in males and 29.1 years in females. 16,78 Telangiectasias generally appeared later and were reported in 23% of males and 9% of females. 78 Along with neurological pain and gastrointestinal symptoms, angiokeratomas are common in children with Fabry disease and may represent a dermatologic hallmark sign to aid in the diagnosis. 16 The presence of angiokeratomas along with telangiectasias is considered characteristic although not specific for the disease. 78 Other dermatological signs associated with Fabry disease are sweating abnormalities (hypohidrosis, hyperhidrosis, anhidrosis), peripheral edema, and lymphedema. 78 Further, the presence of cutaneous vascular signs has been shown to correlate with the severity of systemic involvement of Fabry disease, including pain, renal impairment, and cardiac and cerebrovascular abnormalities. 78 Hypohidrosis is well recognized as a hallmark feature of Fabry disease, but hyperhidrosis is also seen, especially in girls and women. 78,79 Lidove et al examined FOS data and found hyperhidrosis at an overall prevalence of 9.2% (6.4% in males and 11.9% in females). 79 The mean (range) age at onset of hyperhidrosis was 34.6 (3.0-58.0) years in males and 32.1 (3.4-66.6) years in females. 79 The authors suggested that hyperhidrosis, when present, may be a helpful symptom to improve diagnosis, particularly in females.
Ocular manifestations of Fabry disease have been described in patients from the FOS database; these include cornea verticillata (whorl-like epithelial-to-subepithelial corneal opacities), tortuosity and occasional aneurysms of conjunctival and retinal vessels, and a pathognomonic spoke-like Fabry posterior cataract. 28,80 These ophthalmological manifestations do not result in visual impairment or symptoms, but the presence of cornea verticillata in particular is a suggestive sign for Fabry disease. 80 Sodi et al described cornea verticillata in 73.1% of males and 76.9% of females (across an age range of 3-71 years), tortuous ocular vessels in 48.7% of males and 21.9% of females (P < .01 for males vs females; overall age range: 4-69 years), and Fabry cataract in 23.1% of males and 9.8% of females (P = .02; age range: 5-68 years). 80 There were significant associations between the presence of ocular vessel tortuosity and disease severity (as measured by FOS-MSSI), eGFR deterioration, and cardiac enlargement. 80 An analysis of a larger sample of adult FOS patients by Pitz et al reported somewhat lower prevalences of eye findings: cornea verticillata, 50.8% in men, 51.1% in women; tortuous ocular vessels, 24.6% in men, 16.0% in women; Fabry cataract, 7.9% in men, 5.7% in women. 28 Eye manifestations were also more prevalent among patients with null or missense Fabry mutations versus patients with mild missense mutations or the p.N215 S mutation associated with late-onset cardiac variant disease. 28 In the adult FOS population, Pitz et al found that the overall severity of Fabry disease was associated with the presence of each of the 3 types of ocular changes (cornea verticillata, tortuous ocular vessels, and Fabry cataract) as well as with null and missense mutations; thus, the presence of ocular signs may be useful in identifying patients at risk for more severe disease. 28
Special Populations
Our expanding knowledge has led to increasing recognition that women are not merely asymptomatic carriers of the mutations that cause Fabry disease; instead, most females who are heterozygous for Fabry-associated mutations display clinical manifestations of the disease. Although symptom onset generally occurs at an older age than in males, female patients typically experience signs and symptoms of Fabry disease by their third and fourth decades and severe manifestations may be seen in girls and women. 16,20 In an analysis of the FOS female patient population, Deegan et al found that 77% had neurologic features, 59% had cardiac manifestations, and 40% had self-reported indications of renal involvement. 20 As well as being the most common, neurologic signs were the earliest ones to be seen in female patients, at ∼16 years of age, whereas cardiac and renal features were noted at an average age of 33.5 and 37.3 years, respectively. 20 The most common specific signs and symptoms were acute pain attacks in 57% of patients, angiokeratoma in 40%, cornea verticillata in 40%, proteinuria in 35%, vertigo in 32%, chronic pain in 32%, and LVH in 26%. 20
The Fabry disease phenotype in girls and women is variable. Barba Romero et al examined the relationship in FOS patients between phenotype and geographic location across Europe and found that women living in northern European countries had significantly higher α-Gal A enzyme activity, but also significantly higher disease severity scores, than women in southern European countries. 81 These severity score differences were significant for most of the general, neurological, and renal FOS-MSSI subscores, but not for any of the cardiovascular subscores. 81 Women in northern Europe also had higher levels of proteinuria than those in southern Europe. 81 The authors speculated that these differences may be attributable to the effects of epigenetic or extragenetic factors (such as diet or other environmental factors) on Fabry disease expression in women. 81
An analysis of FOS data by Hughes et al demonstrated that the effectiveness of 4 years of agalα ERT in women was comparable with that in men, with similar improvements in a variety of signs and symptoms, including cardiac structure and function, and for a number of measures of pain, health-related QOL, and renal function. 37
Ramaswami et al examined data from children (<18 years of age) in FOS and found that mean age at Fabry diagnosis was ∼10 years, with a mean delay between symptom onset and diagnosis of ∼3 years. 21 The most common manifestations were neurological and gastrointestinal signs and symptoms, reported in ∼80% and ∼60% of children, respectively. 21 Symptoms were noted at a similar frequency in boys and girls, although symptom onset occurred 2 to 5 years later in girls than in boys. 21 Recently, the FPHPQ has been developed and validated, which allows quantitative assessment of patient-reported Fabry disease symptoms in children, making it a useful instrument to monitor disease progression and treatment effects. 32
Data from FOS have provided evidence of the effectiveness and safety of agalα in children, including safety in those <7 years of age. 38 A good response to agalα has been demonstrated in children, with benefits including improvements in pain scores and QOL measures, as well as stabilization of cardiac and renal parameters. 39,40 Improvements in measures of pain and QOL were seen in a 23-week open-label study of children 3.5 to 18.0 years of age. 40 It is hoped that long-term follow-up of children in FOS will provide evidence regarding whether early initiation of ERT in childhood can inhibit the development of serious Fabry disease complications later in life.
Discussion and a Look to the Future
Some of the key challenges in maintaining FOS over the last 15 years and beyond arise from the importance of facilitating data quality and the evolution of the FOS governance system. The FOS steering committee is well balanced across medical specialties and geographic regions, enabling a broad approach to the rich data generated by FOS. Additionally, task forces are established to address specific topics and generate publications representing valuable scientific contributions in defined time frames, thus translating the FOS data collection work into tangible benefits. As described in detail by others, 15,82 there are inherent limitations of any registry data that create challenges for working with these data. These limitations include patient enrollment bias (patients with more severe symptoms or who are receiving treatment are more likely to be enrolled in a registry), lack of data completeness, and uncertain quality of data (because assessments may not be standardized across participating sites). 15,82 As described in the Evolving Design of FOS section earlier in this article, several measures aimed at improving FOS data capture and handling have been undertaken.
However, particularly with a rare disease affecting relatively small numbers of patients, FOS data offer a resource with utility not only for description of natural history and therapeutic effects but also for exploratory hypothesis testing and the generation of tools for diagnosis and management. Disease heterogeneity and understanding of the relative contribution of underlying mutations in the α-Gal A gene and other genetic and environmental factors to clinical severity require sophisticated scoring techniques. The Fabry International Prognostic Index, a prognostic severity score, was developed using data from FOS patients and allows the use of early features to differentiate patients with different probabilities of clinically significant events. 24 Age-adjusted FOS-MSSI severity scores have been developed and validated, which correct for the effect of age and sex on disease severity; this allows meaningful comparison of disease severity between population subgroups (eg, different genotypes) without confounding by factors such as age and sex. 25 Mean scores for genotypes can be generated and outliers interrogated for additional factors contributing to their presentation. As described in the Special Populations section earlier, the FPHPQ is another instrument that has come out of FOS. 32 A recent report by Terryn et al describing the phenotype and implications of the GLA p.Ala143Thr mutation is an example of patient management learning from FOS data. 83 Observations from FOS patient data were compared with information from patient charts and the published literature and allowed the investigators to characterize this mutation as possibly pathogenic. 83
Hypothesis generation by FOS may help direct future research (for example, in areas such as genotype-phenotype correlations and the growing but still poorly defined use of biomarkers). FOS and other interventional data can also support the concept of timely initiation of ERT before the onset of irreversible organ involvement in Fabry disease to prevent or stabilize organ deterioration before irreversible escalation of tissue damage and the expanding use of newborn screening to identify patients, as well as helping to further define the correlation between classic Fabry disease and that associated with late-onset or oligosymptomatic mutations, which may account for 86% to 92% of Fabry disease cases identified by newborn screening. 8,84,85 Unresolved questions remain around issues such as patients who show an apparent lack of response to therapy (nonresponders) and the detection of alterations in survival.
The past 15 years of FOS typify a successful international cooperative effort, whereas the future of FOS offers additional opportunities for value in site-specific or country-specific performance metrics, the potential to explore mutation-specific information, the power of serial longitudinal measurements, and the ability to track an evolving natural (and treated) history over time. Although we recognize that there are limitations associated with a registry database such as FOS, in particular regarding data completeness and accuracy, this volume of real-world data from a critical mass of participating centers can function to stimulate and direct future areas of research.
Footnotes
Authors’ Note
Under the direction of the authors, Malcolm Darkes and Margit Rezabek, employees of Excel Scientific Solutions, provided writing assistance for this publication. Editorial assistance in formatting, proofreading, copyediting, and fact checking was also provided by Excel Scientific Solutions.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Roberto Giugliani has received consulting fees from Amicus, BioMarin, Genzyme, Shire, and Synageva/Alexion; participated in clinical trials sponsored by Amicus, BioMarin, Genzyme, GlaxoSmithKline, Shire, and Synageva/Alexion; and assisted in the design of and/or participated in clinical studies using products manufactured by Actelion, BioMarin, Genzyme, and Shire.
Dau-Ming Niu has received research and travel support from Genzyme and Shire.
Uma Ramaswami has received travel and research grants, as well as honoraria for lectures and consulting, from Amicus, Alexion, Genzyme, Protalix, and Shire.
Michael West has received research grants, consulting fees, speaker fees, and/or travel support from Actelion, Alexion, Amicus, Excelsior, GlaxoSmithKline, Sanofi-Genzyme, Shire, Sumitomo, and Synageva.
Derralynn Hughes has received travel and research grants and honoraria for speaking and consulting from Amicus, Protalix, Sanofi-Genzyme, and Shire.
Christoph Kampmann has received research support from BioMarin and Shire and consulting/speaker fees from BioMarin, Gore, and Shire; participated in clinical studies sponsored by Actelion, BioMarin, Genzyme, Gore, and Shire; and served on advisory committees for BioMarin and Shire.
Guillem Pintos-Morell has received travel grants and honoraria for consulting and lectures from BioMarin and Shire.
Kathleen Nicholls has received research support and/or travel support from Amicus, Sanofi-Genzyme, and Shire, and has served on advisory committees for Amicus, FOS, Sanofi-Genzyme, and Shire.
Jörn-Magnus Schenk is an employee of, and holds stock options in, Shire.
Michael Beck has received unrestricted grants, travel support, and honoraria from Actelion, BioMarin, Genzyme, Shire, and Synageva.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shire. The Fabry Outcome Survey is sponsored by Shire Human Genetic Therapies, Inc. Shire International GmbH provided funding to Excel Scientific Solutions for support in writing and editing this manuscript.
