Abstract
Succinyl-CoA:3-oxoacid CoA transferase (SCOT) deficiency is an autosomal recessive disorder of ketone body utilization that is clinically characterized with intermittent ketoacidosis crises. We report here the second Turkish case with SCOT deficiency. She experienced 3 ketoacidotic episodes: The first ketoacidotic crisis mimicked diabetic ketoacidosis because of the associated hyperglycemia. Among patients with SCOT deficiency, the blood glucose levels at the first crises were variable, and this case had the highest ever reported blood glucose level. She is a compound heterozygote with 2 novel mutations, c.517A>G (K173E) and c.1543A>G (M515V), in exons 5 and 17 of the OXCT1 gene, respectively. In patient’s fibroblasts, SCOT activity was deficient and, by immunoblot analysis, SCOT protein was much reduced. The patient attained normal development and had no permanent ketosis. The accurate diagnosis of SCOT deficiency in this case had a vital impact on the management strategy and outcome.
Introduction
Succinyl-CoA:3-oxoacid CoA transferase (SCOT) deficiency (OMIM 245050) is an autosomal recessive disorder affecting ketone body utilization. In SCOT deficiency, ketone bodies produced from fatty acids and ketogenic amino acids in the liver cannot be used by the extrahepatic tissues. Patients with SCOT deficiency develop severe ketoacidotic episodes triggered by stress conditions, such as infection and fasting, within the neonatal period or around 6 to 24 months of age. 1 Because of the absence of specific urinary organic acids, except for large amounts of 3-hydroxybutyrate and acetoacetate, biochemical diagnosis using urinary organic acid analysis or blood acylcarnitine analysis is impossible. If present, permanent ketosis or ketonuria is a characteristic feature for SCOT deficiency. Enzyme assay or mutation analysis is necessary to confirm the diagnosis. 2
Marked hyperglycemia together with ketoacidosis is a common feature of diabetic ketoacidosis. However, only a few patients with some organic acidemias have manifested with episodes of hyperglycemic ketoacidosis. 2 -8 To our knowledge, severe hyperglycemia has not been reported in SCOT deficiency. We herein describe a Turkish SCOT-deficient patient whose first episode mimicked diabetic ketoacidosis because of the associated hyperglycemia.
Case Report
An 8-month-old female girl was admitted to our hospital with rapid breathing and lethargy preceded by 2 days of upper respiratory tract infection. She was born to nonconsanguineous Turkish parents. Her birth weight was 2780 g, and she did not have any remarkable health problems. Her growth and development parameters were appropriate to age. Family history revealed that her elder sister died of an episode of metabolic acidosis at the age of 11 months. Initial laboratory investigations revealed severe metabolic acidosis with an arterial pH of 7.08, pCO2 of 11.7 mm Hg, bicarbonate level of 3.4 mmol/L, and base deficit of −24.5 mmol/L. The blood glucose was high at 26.5 mmol/L. Urinary ketones were highly positive (++++). Serum lactate and ammonia were normal. Both the clinical and the laboratory data were suggestive of diabetic ketoacidosis. However, an organic acidemia was suspected rather than diabetic ketoacidosis based on her sister’s history and the normal HbA1C level. Hence, the patient was started on glucose infusion (8 mg/kg/min), and she successfully recovered. At that episode, we could not make a correct final diagnosis. At the age of 10 months, the patient developed a second episode. Apart from normal blood glucose level (5.5 mmol/L), the clinical and laboratory data of the second episode were similar to the first one. Deficiency of SCOT was suspected at that time. Urinary organic acid analysis revealed increased 3-hydroxybutyric acid, 4182.9 mmol/mol creatinine (upper limit: 11.1 mmol/mol creatinine). Mutation analysis of SCOT gene revealed 2 novel mutations. After the diagnosis was made, the maintenance therapy consisted of a high-carbohydrate, low-protein, and low-fat diet, including 3 g/kg of body weight per day of cornstarch and 2 ml/kg of body weight Shohl’s solution (bicarbonate). A third milder ketoacidotic episode occurred at 31 months of age, after which the patient has not developed any further episodes till now, the age of 5 years. Her blood 3-hydroxybutyrate level after overnight fast and 2 hours after breakfast were 0.4 to 0.7 mmol/L and 0.1 to 0.2 mmol/L, respectively, and urine ketone was negative during nonepisodic condition. This indicates that she has no permanent ketosis or ketonuria. Her height and weight were 104.5 cm (10-25p), 16.3 kg (10-25p), respectively. Her development was normal.
Methods
Mutation Analysis
Genomic DNA and RNA were extracted from peripheral white blood cells using the RTA Genomic DNA Isolation Kit from BLOOD and the RNeasy Mini Kit (250; Qiagen). Total RNA obtained from the patient is converted to complementary DNA (cDNA) by reverse transcription polymerase chain reaction (RT-PCR) using the Ipsogen RT Kit (Qiagen). Primers of DNA and cDNA were designed using the Primer3 software (http://bioinfo.ut.ee/primer3/). The cDNA complementary to the OXCT1 messenger RNA is then amplified by PCR by forward primer-1 5′- c.-79CGGGTTTCCTTCGCAGTC -3′, reverse primer-1 5′- c.874CTTCCCCATCTCCCTCTTTC -3′ and forward primer-2 5′- c.705CAAAGCTGCAGAAACCACAG -3′, reverse primer-2 5′- c.1703TGGCTGCATAAAGTCTGAAACA -3′. It was analyzed on an ABI Prism 3130 DNA Analyzer using the ABI Big Dye Terminator v3.1 Cycle Sequencing Kits. These mutations were confirmed by genomic DNA sequencing. Exons 5 and 17 of the OXCT1 gene sequences were amplified by PCR by forward primer-1 5′- g.20432 GGAACCACGTCTAAATGTTTGTG –3′, reverse primer-2 5′- g.20716AAAATCCCACGTGGAAAGAG -3′ and forward primer-2 5′- g.138858AAGGAGAGAGATTTATATCATGC AG -3′, reverse primer-2 5′ g.139028TGTCCTTTCAATTAAATCTTGTGTG -3′, respectively (GenBank accession no. NG_011823).
Enzyme Assay and Immunoblots
Assays for mitochondrial acetoacetyl-CoA thiolase (T2) and SCOT were done as described previously. 9 An immunoblot analysis was performed using rabbit polyclonal antibodies and the ProtoBlot Western blot AP system (Promega, Madison, Wisconsin). The first antibody was a mixture of an anti-human T2 antibody and an anti-human SCOT antibody. 9
Results
Mutation analysis was conducted. The direct sequence analysis of the cDNA that spans the full-length coding sequence revealed the patient was a compound heterozygote for the mutation c.517A>G (Lys173Glu) and c.1543A>G (Met515Val) in exons 5 and 17 of the OXCT1 gene, respectively (Figure. 1). Furthermore, c.517A>G (Lys173Glu) and c.1543A>G (Met515Val) mutations were found in a heterozygous state in her mother and father, respectively (Figure. 1). Since both mutations were novel, enzyme assay on patient’s fibroblasts was performed to confirm the diagnosis of SCOT deficiency. Activity of SCOT in her fibroblasts was decreased to 0.3 mmol/min/mg protein (control 6.7 ± 2.1 mmol/min/mg protein). The SCOT protein was hardly detected in extracts from her fibroblasts in the immunoblot analysis (Figure. 2). These analyses confirmed the diagnosis of SCOT deficiency.
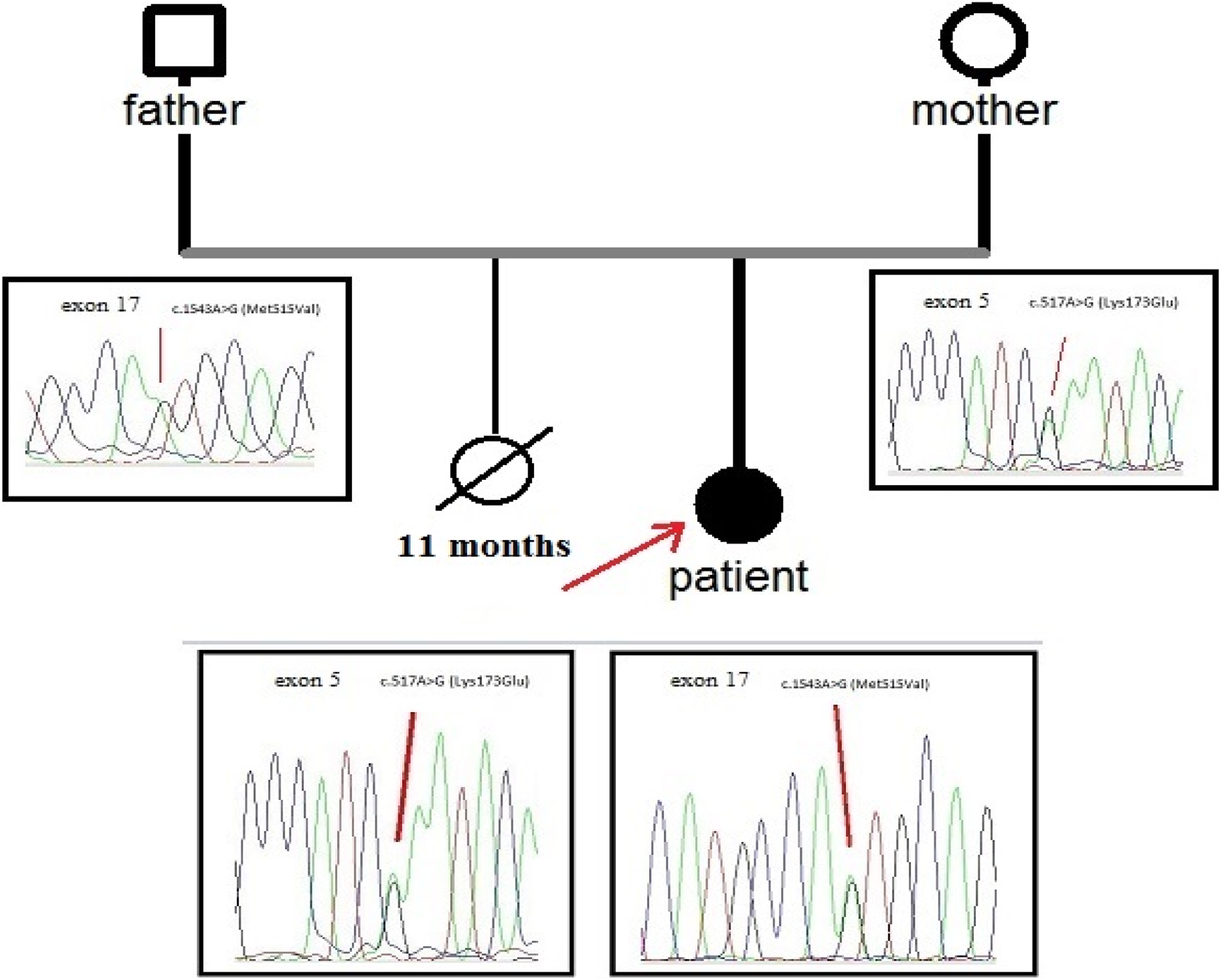
Compound heterozygous detected in terms of c.517A>G (Lys173Glu) and c.1543A>G (Met515Val) in exons 5 and 17 of the OXCT1 gene in the patient. Furthermore, c.517A>G (Lys173Glu) and c.1543A>G (Met515Val) mutations were found to be heterozygous mutations in the mother and father, respectively.
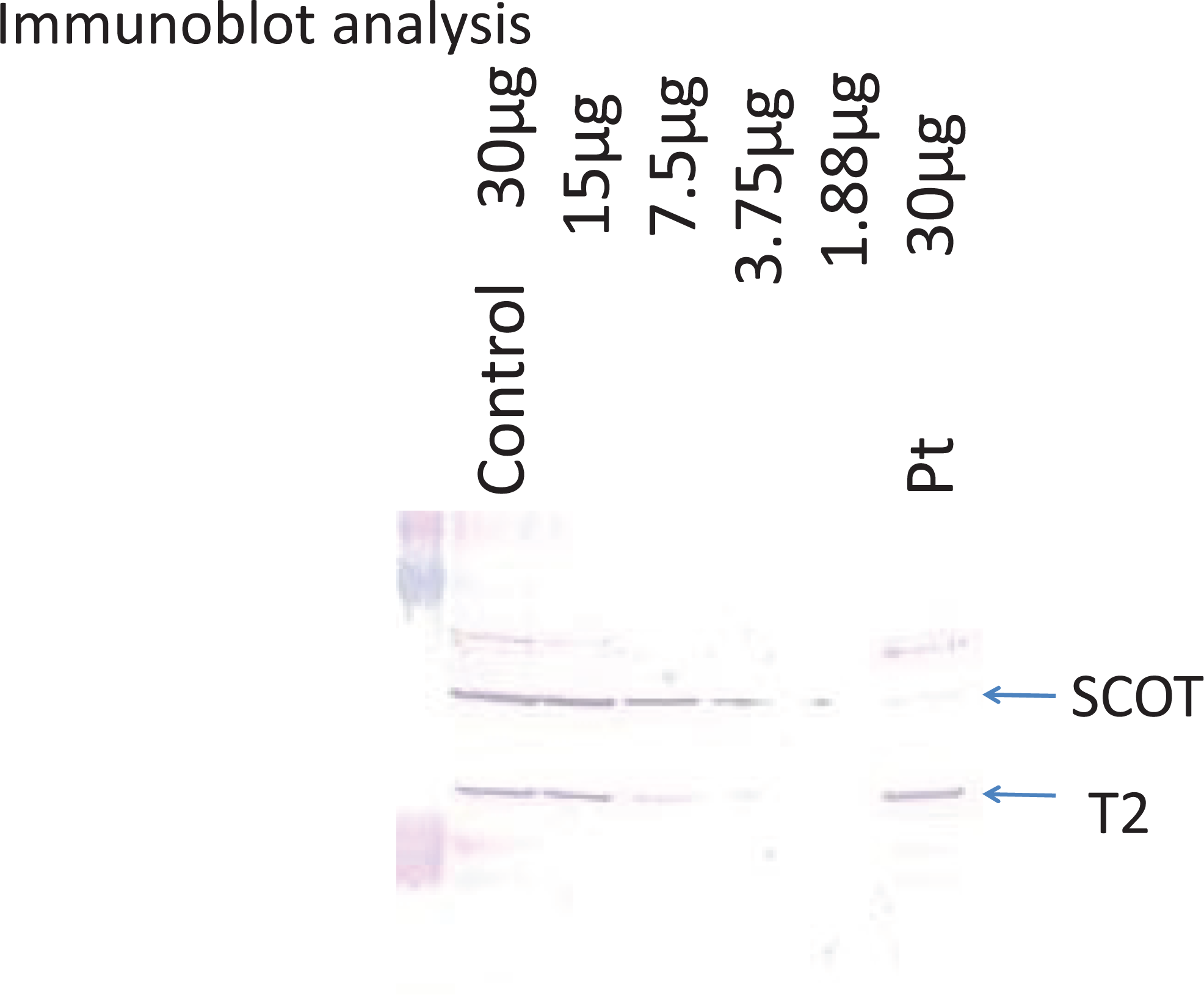
Immunoblot analysis. The protein amounts applied are shown above the lines. The first antibody was a mixture of an anti-human mitochondrial acetoacetyl- CoA thiolase (T2) antibody and an anti-human SCOT antibody.
Discussion
Ketone body utilization disorders, T2 and SCOT deficiency, usually present with severe ketoacidotic attacks out of proportion to the triggering illness. While T2 deficiency usually has a characteristic profile of urinary organic acids, only a nonspecific increase in 3-hydroxybutyrate and acetoacetate could be detected in SCOT deficiency. Unless a high index of suspicion and a meticulous interpretation of clinical and laboratory data, SCOT-deficient cases might be missed, especially in presence of apparently reasonable and more relevant explanations of the ketoacidosis as diabetic ketoacidosis. 1,2
Marked hyperglycemia together with ketoacidosis was reported in some organic acidurias as multiple carboxylase deficiency,3 methylmalonic acidemia, 4,5 propionic academia, 6 and isovaleric acidemia. 7,8 Such hyperglycemia may be attributed to a response to metabolic stresses. The blood glucose level at the presenting ketoacidotic attack in the reported 15 SCOT-deficient patients ranged from 1.3 to 10.5 mmol/L (Figure. 3). 10 -20 Among them, most patients showed normal or high-normal glycemia, and only 3 patients showed hypoglycemia at the first crises. This leads to the general rule that ketoacidosis precedes of the appearance of hypoglycemia in SCOT deficiency. On the other hand, our case, at her first crisis, had the highest ever reported hyperglycemia among SCOT-deficient patients. Hence, she was initially misdiagnosed as diabetic ketoacidosis. The accurate diagnosis had a vital impact on the management strategy and outcome.
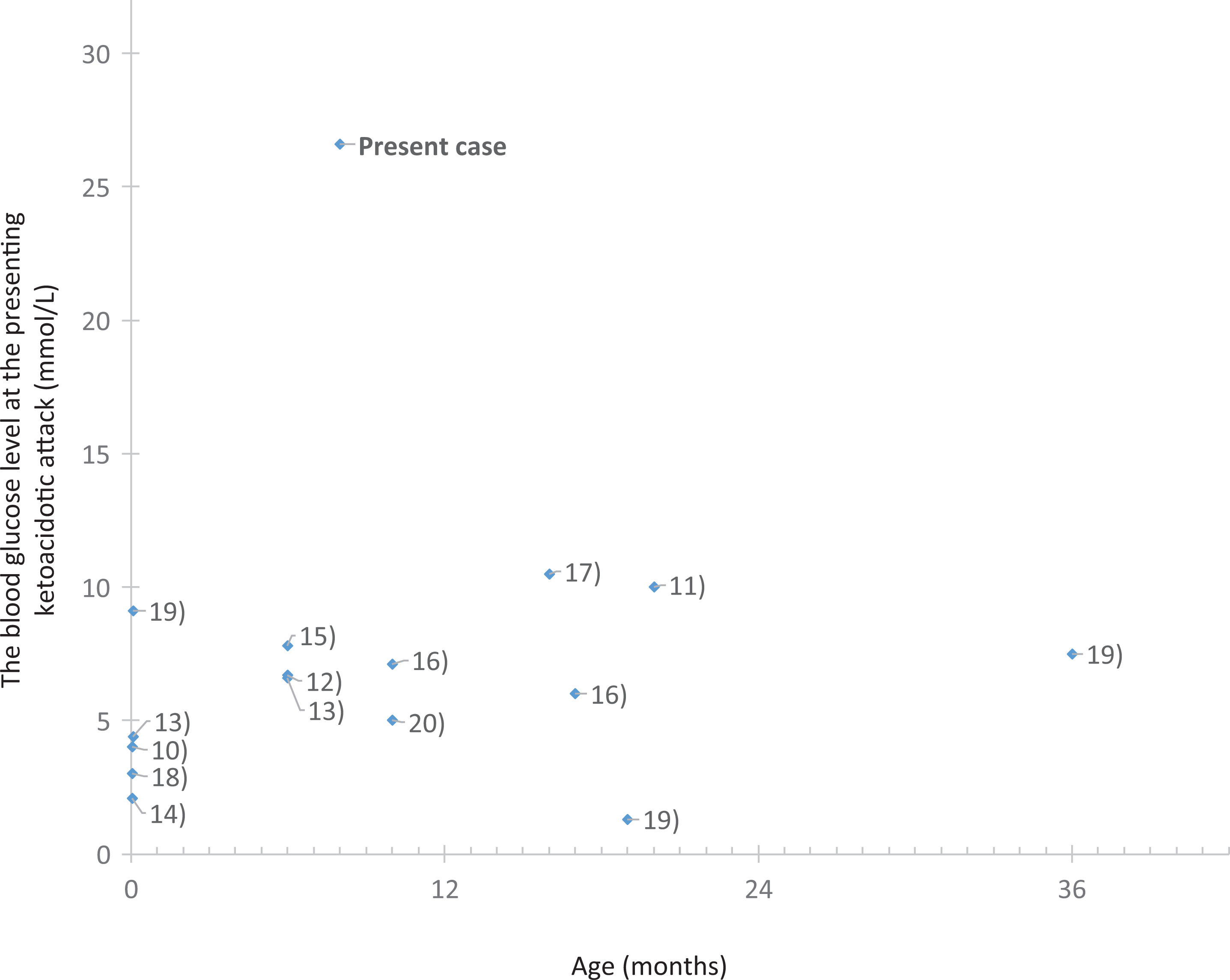
Blood glucose levels at the first metabolic crises in published SCOT-deficient patients. Reference numbers are shown in parenthesis.
In conclusion, we reported herein the unusual presentation of the second Turkish SCOT-deficient patient with a severe hyperglycemia mimicking diabetic ketoacidosis. We detected 2 novel mutations in OXCT1 gene and confirmed the diagnosis by enzyme assay and immunoblot analysis.
Footnotes
Authors’ Note
Sahin Erdol and Halil Saglam contributed to the diagnosis and follow-up of the patient, wrote, and edited the manuscript. Mehmet Ture performed the genetic analysis and contributed to the writing of the genetic part of the manuscript. Toshiyuki Fukao, Hideo Sasai, Hiroki Otsuka, Elsayed Abdelkreem performed enzyme assay and immunoblot analysis using fibroblast and critical editing of the manuscript. Tahsin Yakut contributed to editing of the genetic part of the manuscript.
Compliance with ethics guidelines: All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Written informed consent was obtained from the parents of the patient prior to inclusion in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (26114708, 24591505, 16K09962), Health and Labour Science Research Grants for Research on Intractable Diseases from the Ministry of Health, Labour and Welfare of Japan and the Practical Research Project for Rare/Intractable Diseases (15AeK0109050h0002) from Japan Agency for Medical Research and Development, AMED to T.F.
