Abstract
Guanidinoacetate methyltransferase (GAMT) deficiency is an autosomal recessively inherited disorder of the metabolism of creatine that leads to depleted levels of creatine and excessive concentrations of guanidinoacetate (GAA). Patients affected develop neurological symptoms during childhood, such as muscular hypotonia, involuntary extrapyramidal movements, convulsions, slurred speech, and even autism. Although the pathophysiology of GAMT deficiency is unclear, neurological dysfunction is commonly found in this disease, and it has been mainly attributed to a reduction in creatine or/and an increase in GAA levels. Reports from literature suggest that GAA may interfere with neuronal γ-aminobutyric acid (GABA) receptors type A and cause epilepsy in human. Preclinical studies show that GAA increases free radical formation and decreases brain antioxidant defenses, inducing alteration in oxidative status. Guanidinoacetate also impairs energy metabolism in brain. The discussion of this review focuses on various and latest studies addressing GAMT deficiency and creatine metabolism, as well as addresses the question of neurotoxicity GAA.
Guanidinoacetate and Creatine Metabolism—An Overview
Guanidinoacetate (GAA) is a metabolite of glycine in which the amino group can be converted into a guanidine. It is highly involved in the metabolism of creatine, being its direct precursor. 1
Creatine has been known for its essential functions in muscle, contributing to a transient intracellular storage of metabolic energy mainly in the form of phosphocreatine that is nothing more than creatine with a phosphate group.
1
Mammals take about half of their creatine from diet (mostly from meat and fish) and synthesize the other half through a relative simple reaction that occurs mainly in the kidney and liver involving a 2-step pathway with 2 enzymes and 1 membrane carrier.
2
The enzymes involved are
Guanidinoacetate Methyltransferase Deficiency
In 1994, Stockler et al described a new inborn error of metabolism (IEM) that was addressed as the first IEM of creatine. The new disease was named after the affected enzyme: GAMT deficiency. 15 This metabolic disease is an autosomal recessively inherited disorder caused by mutations in the GAMT gene that leads to depleted levels of creatine and excessive concentrations of GAA in skeletal muscle, blood, brain, and other tissues. 16 -18 It is a rare disease with approximately 110 cases reported so far, mostly from Europe and the Middle East. 19 The cerebral tissue relies on a continuous energy supply in order to maintain its functions properly. Therefore, with creatine in low levels, the brain becomes the most affected organ in GAMT-deficient patients. Neurological symptoms are common and variable among individuals affected by GAMT deficiency, including intractable epilepsy, intellectual impairment, autism, automutilating behavior, extrapyramidal syndrome, slurred speech, and hypotonia. 16,17,20,21
Supplementation with creatine has been used as pharmacological treatment in GAMT deficiency for a long time. 17 Nonetheless, one of the major issues surrounding GAMT deficiency and the brain is the difficulty that creatine has to penetrate the blood–brain barrier (BBB) due to the small amount of SLC6A8 transporter present in the microcapillaries, the absence of the same transporter in astrocytes lining them, and the limited diffusion of creatine through extracellular matrix surrounding BBB. 22 To provide sufficient levels of creatine in healthy individuals, the brain has its own synthesis expressing AGAT and GAMT locally. In the cortex and other brain regions, AGAT and GAMT are dissociated, and the intermediate GAA has to be transported between AGAT- and GAMT-expressing cells to complete the synthetic pathway. 23 The presence of this endogenous creatine synthesis is supported by studies that demonstrated that despite the inability to capture creatine from periphery, patients with SLC6A8 deficiency still have normal levels of this molecule in their central nervous system (CNS), whereas GAMT-deficient patients presented a dramatic reduction in comparison. 24 The phenotypic diversity in patients with GAMT deficiency may be partially explained by a variety of functions that creatine seems to play in the brain. Apart from its functions in energy regeneration, researchers proposed that creatine may act as a neurotransmitter in the CNS modulating GABAergic and/or glutamatergic neurons. 25 This theory is supported by studies that show that rat brain synaptosomes express creatine transporter SLC6A8, 26 implying the existence of a creatine reuptake mechanism in axon terminal membrane. 27
The energetic reservoir provided by the system creatine/CK also helps to prevent overload of the mitochondrial respiratory chain, reducing generation of reactive species that have the power to cause oxidative stress and induce cytochrome C dissociation from the inner mitochondrial membrane, initiating early apoptotic triggering events. 28 In fact, several studies claim that creatine can not only prevent the generation of reactive species but also act as a direct scavenger of a range of radicals, including superoxide anion and peroxynitrite. 29,30 This effect happens regardless if creatine is in or out a mammalian cell. 31 A recent study revealed that creatine is able to prevent damage induced in vitro by oxidizing agents in both nucleated and nonnucleated cells, suggesting that creatinine may possess similar properties. 32 These antioxidant properties presented by creatine appear to be beneficial not only to GAMT 33 but also to other innate errors of metabolism where creatine was able to prevent lipid peroxidation and imbalance of redox homeostasis. 34,35
Neurotoxicity of GAA
Despite the fact that decreased levels of creatine play a key role in the physiopathology of GAMT deficiency, the evidence shows that elevated levels of GAA in the brain are the major responsible for triggering the neurological symptoms of patients affected by this disease, particularly the epileptogenic action. 21 Under physiological conditions, no transport of GAA occurs at the BBB, but there is a way out via taurine transporters (TauT) in the blood–cerebrospinal fluid barrier. 22 Under GAMT deficiency, there is an uptake of GAA from the periphery at BBB, and the exit of GAA from CSF to blood may be increased using both TauT and SLC6A8, that is also capable of transporting GAA. 22,29 The accumulation of GAA also happens via the brain endogenous AGAT activity that is plainly functional. It is suggested that high concentrations of GAA can interfere with neuronal γ-aminobutyric acid (GABA) receptors type A, but not with GABA receptor type B, and cause the neurological dysfunction behind the symptoms presented by GAMT-deficient patients. 36 The GAA seems to act as a direct agonist of GABAA receptors with a less efficient GABA mimetic potential than GABA itself. However, at high concentrations, like the ones found in patients with GAMT, GAA is able to cause chronic stimulation in GABAA, and consequently, receptors undergo desensitization. This effect causes an increase in neuronal excitability due to internalization of GABAA receptors and a decreased response to GABA. 37,38 The basal ganglia output nuclei globus pallidus and substantia nigra pars reticulata are involved in both movement and seizure control through GABAergic projections that appear altered in experiments conducted in vitro. 37 Therefore, the motor dysfunctions and particularly seizure activity in patients with GAMT deficiency may be explained by interference of GAA in these interconnections present in the basal ganglia.
In addition, studies have shown that an intrastriatal injection of GAA in rats leads to a series of biochemical alterations and abnormalities in behavioral tests. First, the intrastriatal injection appeared to cause a decrease in antioxidant defenses at the same time that inhibit the activity of crucial enzymes for the generation of energy in the CNS. These enzymes are Na+, K+-ATPase, complex II in the mitochondrial respiratory chain, and CK. 39,40 Nonetheless, the cholinergic system plays a crucial role in cognitive function, including memory, and results showed that GAA significantly increased acetylcholinesterase activity and impaired retention of an inhibitory avoidance task, implying that GAA accumulation affects all steps of memory processing following distinct time-dependent mechanisms. 41 Furthermore, another study showed that GAA markedly inhibited glutamate uptake by striatum, what can be linked to the neurological dysfunction characteristic of GAMT-deficient patients, especially the generalized convulsions. 42 These series of heterogenic alterations appear to be involved in the pathophysiology of the neurological features present in patients with GAMT deficiency. Suggested mechanisms of neurochemical effects of GAA accumulation in brain are summarized in Figure 1.
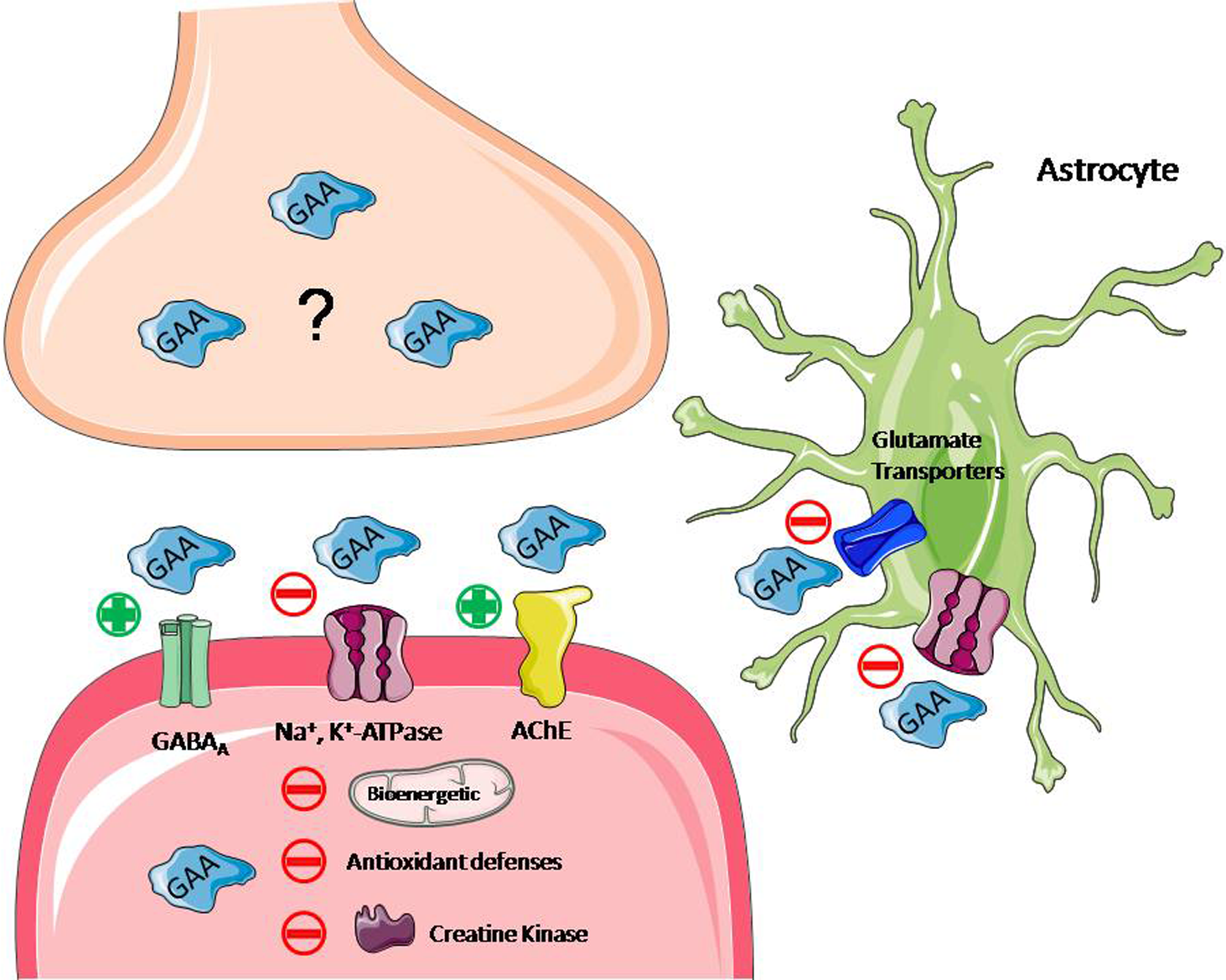
Suggested mechanisms of neurochemical effects of GAA accumulation in the brain. Under GAMT deficiency, there is uptake of GAA from the periphery and GAA synthesis in the neuron occurring mainly in the mitochondria intermembrane space via AGAT. The GAA leaves the neuron through an unknown mechanism, and due to the lack of functional GAMT, it accumulates in the synaptic cliff and within the neuron itself. This accumulation leads to a series of imbalances in enzymes, receptors, and redox state. The GAA seems to act as a direct agonist of GABAA receptors, leading to their desensitization and internalization. It appears to cause a decrease in antioxidant defenses at the same time that inhibits the activity of crucial enzymes for energy generation: Na+, K+-ATPase, complex II in the mitochondrial respiratory chain, and creatine kinase. The GAA significantly increased AChE activity, leading to less AChE available in the synaptic cliff. Furthermore, GAA markedly inhibited glutamate uptake by astrocytes. AChE indicates acetylcholinesterase; AGAT, arginine glycine amidinotransferase; ATP, adenosine triphosphate; GAA, guanidinoacetate; GAMT, guanidinoacetate methyltransferase.
Clinical Cases and Possible Treatments to GAMT Deficiency
The diagnosis of GAMT deficiency can be made based on a series of tests that include creatinine excretion in 24-hour urine, Sakaguchi staining reaction of guanidino compounds in urine samples, GAA excretion measured quantitatively, and magnetic resonance spectroscopy to detect accumulation of GAA and depletion of creatine in the brain. 43 Measurements of GAMT activity are now available for some cell types. 44 Once the pathology is confirmed, the treatment of GAMT deficiency aims for both repletion of cerebral creatine and reduction of GAA concentrations.
Oral supplementation with creatine (administered as creatine monohydrate) at high doses can restore brain levels and result in improvements to the patients mostly in the involuntary extrapyramidal movements and convulsions.
16,17
The benefits of this supplementation can be reached only with long-term treatment due to the slow cross of creatine through BBB. The positive effects may take months or even years to appear. The doses used are normally around 400−670 mg per kg per day. This dose appears to be safe, since excess of creatine may cause oxidative stress.
32
A presupplementation investigation of kidney function might be considered for reasons of safety but in normal healthy subjects appears unnecessary.
45
A recent study performed by Peters et al showed that the supplementation with creatine (3 g/d) decreases GAA concentrations in plasma of Bangladeshi adults without GAA deficiency.
46
However, it is important to highlight that creatine supplementation per se does not fully normalize GAA levels in patients with GAMT deficiency. In order to achieve an even better clinical outcome, studies suggest that lowering the ingestion of one of the precursor substrates of GAA, arginine, as well as high-dose
This evidence-based treatment using creatine, ornithine, and sodium benzoate supplements along with dietary protein restriction was able to improve seizures and development in 5 patients with GAMT deficiency. 50 The average age of diagnosis in this study was 25.8 ± 26.2 months, but one of the subjects was diagnosed earlier, at 8 days from birth. The remarkable fact is that this subject treated at birth remains developmentally normal at 12 months of age, whereas the others still face neurological dysfunctions. This is in agreement with adult and child cases of GAMT deficiency, where diagnosis and treatment later in life correlates with persistency of motor and neurological impairments, despite minor improvements. 49,51,52
A larger study collected data on 48 patients with GAMT deficiency worldwide
48
and showed that the clinical presentation of this innate IEM is indeed highly variable and best treated with supplementation with creatine and
Although early treatment seems to prevent permanent damage from occurring during CNS development, it is important to note that treatment should continue through lifetime, as we can see in the case of one of the patients in the study conducted by Stockler et al. Treatment was started within the first 3 weeks of life. At 32 months, her development was normal, while her late-treated brother (who also have GAMT deficiency) faced more difficulties. However, at 4 years, the treatment was discontinued, and she was affected by episodes of febrile seizures. She recommenced the treatment, and even though her current status is not as good as in the beginning of treatment, it is still better than her late-treated brother. This provides evidence of the importance of searching for new prenatal diagnosis of GAMT, such as measurements in the amniotic fluid and direct sequencing of the GAMT gene in newborns. 53,54
Conclusion
In conclusion, GAMT deficiency is a rare innate IEM of creatine that results in severe neurological effects. It is a treatable disorder, since supplementation with creatine, depletion of arginine in the diet, and supplementation with ornithine considerably ameliorate the symptoms. The seizures do not respond to conventional epileptic treatment, and there is still controversy about the effectiveness of using sodium benzoate supplementation. Moreover, early diagnosis and start of treatment improve the outcome and may even lead, if begun presymptomatically, to normal development. Ideally, treatment should start before the creatine pool supplied from maternal body during gestation ends. This makes GAMT deficiency an important target to prenatal diagnosis. The treatment must be uninterrupted and always guided by health professionals in order to avoid nutritional deficits and low compliance. Nevertheless, a lot of study is still needed to better understand the mechanism behind all the neurological dysfunctions presented by patients, and finding a better way to remove brain GAA is the main goal of researches in the field.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
