Abstract
Carbamoyl phosphate synthetase 1 (CPS1) is a key gene in the first step of urea cycle and has been correlated with nitric oxide level and vascular smooth muscle activity. A functional single-nucleotide polymorphism C/A at position 4217 in CPS1 (National Center for Biotechnology Information SNP database no. rs7422339, T1405N) was reported to be associated with high homocysteine (Hcy) plasma values. Although genetic variants of methylenetetrahydrofolate reductase (MTHFR) gene are known to influence Hcy concentration, other genetic determinants of Hcy remain largely unknown. The association between the CPS1 rs7422339 and the risk of hyperhomocysteinemia in Latin American populations is unknown. Here, we study this association in 100 patients having hyperhomocysteinemia without MTHFR c.677C>T polymorphism and 100 controls. CPS1 rs7422339 was studied using polymerase chain reaction and enzymatic restriction. Comparisons of the CPS1 rs7422339 genotype distributions revealed a significant difference between groups (P = 2.3 × 10−3). Patients carrying polymorphic allele showed almost 3 times higher risk (odds ratio [OR] = 2.47) of hyperhomocysteinemia than wild-type allele, suggesting that rs7422339 SNP is associated with high Hcy levels in the Argentine population.
Introduction
Homocysteine (Hcy) is a nonprotein-forming sulfur amino acid produced during the catabolism of methionine. The small amount of Hcy found in plasma is the result of a cellular export mechanism that complements the remethylation and transsulfuration pathways in maintaining low intracellular concentration of this potentially cytotoxic and pro-oxidant amino acid. 1 –3 Plasma Hcy levels are influenced by genetic as well as environmental factors, such as age, sex, smoking status, intake of folate, and B vitamins. The concentration of Hcy has been epidemiologically correlated with the risk of cardiovascular diseases, 4 neural tube defects, 5 and loss of cognitive functions. 6
Despite heritability estimates of hyperhomocysteinemia (HHcy) ranging from 25% to 44%, 7,8 relatively little is known of the genetic determinants or whether the same variants influence Hcy levels across populations. Rare homozygous defects in genes encoding for enzymes of Hcy metabolism, as cystationine β synthase (CBS) and methylenetetrahydrofolate reductase (MTHFR), lead to dramatically increased Hcy concentration and premature occlusive vascular disease. 1 However, few common polymorphisms have been unequivocally associated with Hcy concentration. Among these, the strongest is the MTHFR single-nucleotide polymorphism (SNP) rs1801133 (c.677C>T) correlated with reduced enzymatic activity and higher Hcy levels. 9 Previous genome-wide associations reports for Hcy levels in Caucasian women 10 and non-European women from the Philippines 11 observed significant evidence for an association (P = 1.9 × 10−11 and P = 4.7 × 10−13, respectively) between Hcy levels and a novel locus, rs7422339 (c.4217C>A), which encodes p.Thr1405Asn in carbamoyl phosphate synthetase 1 (CPS1).
The protein encoded by CPS1 catalyzes the first committed step of the hepatic urea cycle. 12 CPS1 SNP rs7422339 was estimated to explain a greater proportion of the variability in log-Hcy levels in the Filipino mothers (R 2 = 3.0%) than in the Caucasian group (R2 = 0.3%) despite a lower frequency (0.24 vs 0.31) of the minor allele in Filipino mothers.
Previously, we studied in Argentine population different polymorphisms commonly associated with HHcy in MTHFR and CBS. The study of c.677C>T-MTHFR showed a high prevalence of this substitution in individuals with HHcy. However, the c.1298A>C-MTHFR and c.844ins68-CBS did not correlate with high levels of Hcy, leaving a significant number of patients having HHcy without known genetic determinant.
13
Therefore, in order to find genetic determinants that could explain high plasma values of Hcy in our population, we studied T1405N
Materials and Methods
Study Population
Pathological group
This group consists of 100 patients derived from state and/or private services of cardiology, angiology, neurology, and hematology, with defined cardiovascular, cerebral, or peripheral disease. The patients were selected according to the following criteria: (i) over 18 years old, (ii) high levels of Hcy in plasma considering normal fasting values <15 µmol/L and post methionine load test <30 µmol/L, and (iii) normal genotype for c.677C>T MTHFR polymorphism to discard a possible increase in Hcy because of this SNP.
Control group
The population sample taken as control consisted of 100 unrelated and healthy individuals, of which 58 were women and 42 were men (mean age: 33.8 ± 10.8 years, range 11-72).
This study was duly conducted in compliance with the Helsinki Declaration and the approval of the Institutional Ethics Review Board for Health Research on Adult and Child of Córdoba government. Written consent of all participating individuals was obtained.
Blood samples and DNA isolation
A blood sample of 2 mL was collected from each individual in sterile anticoagulant tubes. Genomic DNA was extracted using the protocol of Wizard Genomic’s purification DNA Promega Purification Kit (Promega, Madison).
Genotyping of CPS1 T1405N rs7422339 polymorphism
DNA samples from patients and controls were genotyped for rs7422339 SNP using polymerase chain reaction (PCR) and enzymatic restriction assay. Primer pair amplified the SNP region used for forward primer (5′-ATGGCACTATACTACTTCTC-3′) and for reverse (5′-TAGCTGGTCATCTTATACTC-3′). Polymerase chain reaction amplification was carried out in a total volume of 50 μL containing 1× GoTaq Buffer, 0.5 μL each deoxynucleotide, 1 pmol each primer, 1 U GoTaq DNA Polymerase (Promega), and 20 ng genomic DNA. The PCR cycles involved initial denaturation at 96°C for 5 minutes, 30 cycles with denaturation at 95°C for 1 minute, annealing at 52°C for 1 minute, extension at 72°C for 1 minute, and final extension at 72°C for 7 minutes. The PCR product (571 base pairs [bp]) was digested by 0.5 μL (10 U) of AleI restriction endonuclease (New England Biolabs) at 37°C overnight. AleI recognition site is represented by the presence of C allele (A
DNA sequence analysis
DNA sequence analysis was performed in 2 different genotype samples to confirm the integrity of the area surrounding the restriction site. DNA sequence analysis was made to the PCR product using forward primer. The samples were analyzed using ABI 3500xl automated DNA sequencer at INTA Institute (Buenos Aires, Argentina) .
Statistical analysis
Statistical analysis was performed with INFOSTAT software v2008. 14 Comparison between distributions of categorical variables was performed using chi-square (χ2) test. In addition, variables were described as odds ratio (OR) with 95% confidence interval (95% CI) where appropriate. The data were considered significant if P values were <.05 and highly significant if P < .01.
Results and Discussion
DNA Sequence Analysis for 2 Different Genotype Individuals
Successful sequencing of CC genotype was proved by detection of a single peak corresponding to C nucleotide, while AC genotype sequence was confirmed by the presence of 2 overlapping peaks as expected.
Genotype and Allele Frequencies of CPS1 T1405N SNP
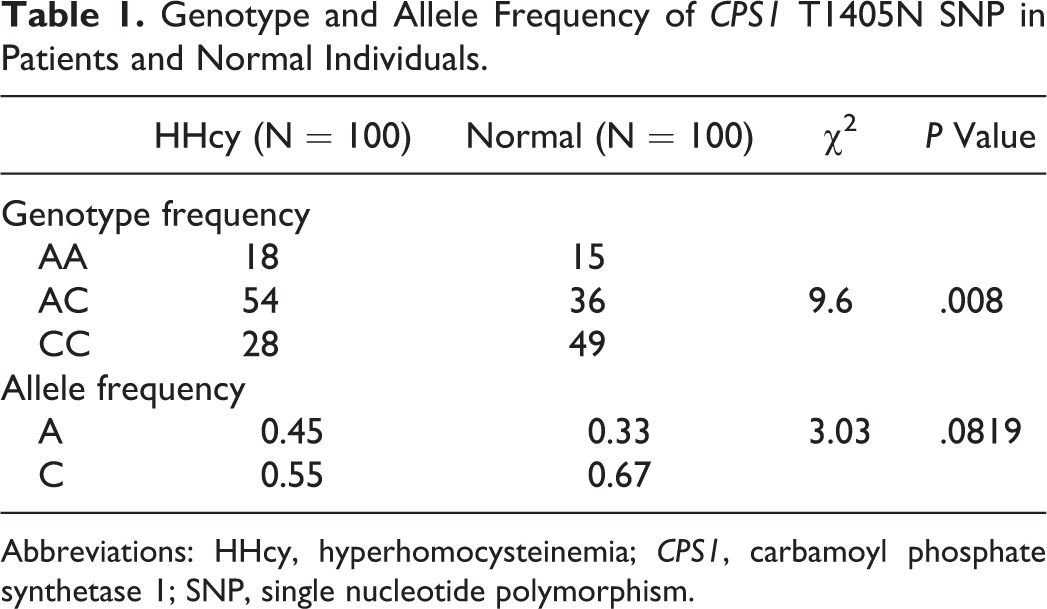
Genotype frequencies of CPS1 rs7422339 showed a statistically significant differential distribution within patients with HHcy and control individuals as shown in Table 1. The percentage of individuals observed within each genotype with respect to HHcy or normal condition was as follows: for AA genotype, 54.5% HHcy and 45.5% normal; for AC genotype, 60% and 40%; and for CC genotype, 36.4% and 63.6%, respectively. Thus, it is to be noted that the ratio of CC carrying control individuals is about to be twice that for HHcy, reflecting an impact of the other genotypes (AA and AC) on the elevation in Hcy levels. This effect replicated only when the dominant model was applied, where 63.6% of patients with CC genotype were normal versus 41.4% of normal individuals with AA+AC genotypes as shown in Table 2.
Genotype and Allele Frequency of CPS1 T1405N SNP in Patients and Normal Individuals.
Abbreviations: HHcy, hyperhomocysteinemia; CPS1, carbamoyl phosphate synthetase 1; SNP, single nucleotide polymorphism.
Dominant Model of Inheritance of CPS1 T1405N SNP in HHcy Versus Normal individuals.

Abbreviations: HHcy, hyperhomocysteinemia; CPS1, carbamoyl phosphate synthetase 1; SNP, single nucleotide polymorphism.
Regarding the allele frequency, a differential distribution was observed in CPS1 T1405N SNP in HHcy versus control, as shown in Table 1. Notably, the percentage of patients harboring the A allele who had HHcy (45 of 78: 57.7%) is higher than that of C allele carriers (55 of 122: 45%).
Influence of CPS1 T1405N SNP Stratified According to Gender Parameter
The HHcy study group was constituted by 55 females of which 12 were AA, 28 were AC, and 15 were CC and 45 males of which 6 were AA, 26 were AC, and 13 were CC. Analysis of the effect of CPS1 genotypes according to gender showed that in both males and females, those carrying the AA and AC genotypes were more likely to have HHcy than those carrying CC; as for CC male carriers, 28.9% (13 of 45) were HHcy compared to 71.1% (32 of 45) of AA and AC male carriers. In females, 27.3% (15 of 55) were HHcy versus 72.7% of AA and AC female carriers. Results of studying each genotype separately also showed no statistically significant difference between distribution of genotypes in males and females with a P value of .857. Also, allele frequency of CPS1 1405 T/N SNP in males (A: 0.42; C: 0.58) and females (A: 0.47; C: 0.53) with HHcy showed no significant difference, giving a P value of .999. Thus, we can show that there is no difference in genotype distributions between males and females in the studied group.
Conclusion
This study identified for the first time CPS1 T1405N polymorphism in a Latin American population. Frequency calculation proved to be one of the highest described worldwide, emphasizing the importance of identifying the effects of this change on the susceptibility to HHcy condition.
We demonstrated a significant statistical association between CPS1 T1405N polymorphism and HHcy condition, regardless of gender, developmental status, or another SNP interaction. These data reveal that functional variants within the urea cycle are significant not only within the pathway but also in other physiologic systems. We hypothesize that this polymorphism affects the functional efficiency of CPS1 and thus the entire urea cycle and, as such, the availability of the nitric oxide substrates, one of the regulators of 1 carbon metabolism.
Our findings suggest that in our population at least one of the main genetic factors that are known to influence plasma Hcy levels is shared with Asian and European populations, regardless of their particular diet and genetic background. This knowledge should allow better intervention strategies to either predict or modify the associated adverse outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by grants from Secretaría de Ciencia y Técnica, Universidad Nacional de Córdoba (no. 124/13; 203/14), Consejo Nacional de Investigaciones Científicas y Técnicas and Secretaría de Ciencia y Tecnología, Gobierno de Córdoba, Argentina.
