Abstract
Lysosomal storage diseases are a group of inherited and acquired disorders. They are characterized by interruption of recycling of cellular and extracellular molecules. Clinically, they are presented as developmental and neurological symptoms similar to other inherited and acquired disorders. This article reviews the function of lysosomes, the current mechanisms that cause the interruption of recycling, the consequences that are manifested clinically, and the methods to diagnose these disorders.
The lysosome is the primary disposal and recycling center of cells. Macromolecules of intracellular and extracellular origin are metabolized in the lysosome. This provides cells with amino acids, fatty acids, nucleic acids, and carbohydrate residues for reuse in cellular synthesis. 1 For example, in fibroblasts, 90% of the glucosylceramide are derived from recycling of sphingolipid bases and only 10% are synthesized de novo.
Lysosomes are present in all cell types. They are membrane-bound organelles containing about 25 lysosomal membrane proteins
2
as well as vacuolar ATPase (v-ATPase) proton pump. They also have 50 known hydrolytic enzymes
2
that participate in the turnover of macromolecules with their associated accessory proteins that also participate in the turnover of the macromolecules. They include glycosidase, lipase, nuclease, phosphatase, protease, and sulfatase that are active at acidic pH and lysosomal pH in the 4.5 to 5 range. Similar to the secretory proteins, lysosomal hydrolases are glycoproteins. Lysosomal enzymes are synthesized in the rough endoplasmic reticulum (RER; Figure 1) following which they undergo a series of posttranslational modifications involving protein and carbohydrate recognition signals, allowing them to reach their final destination in the lysosome. The hydrophobic amino-terminal signal peptide on the nascent protein directs their transport into the lumen of the RER, where they undergo glycosylation of selected asparagine residues. The signal peptide is then cleaved by the removal of 3 glucose residues and 1 mannose residue. The hydrolases are then transported to the Golgi apparatus and acquire a terminal mannose-6-phosphate on the oligosaccharide side of hydrolase via 6-phospho-
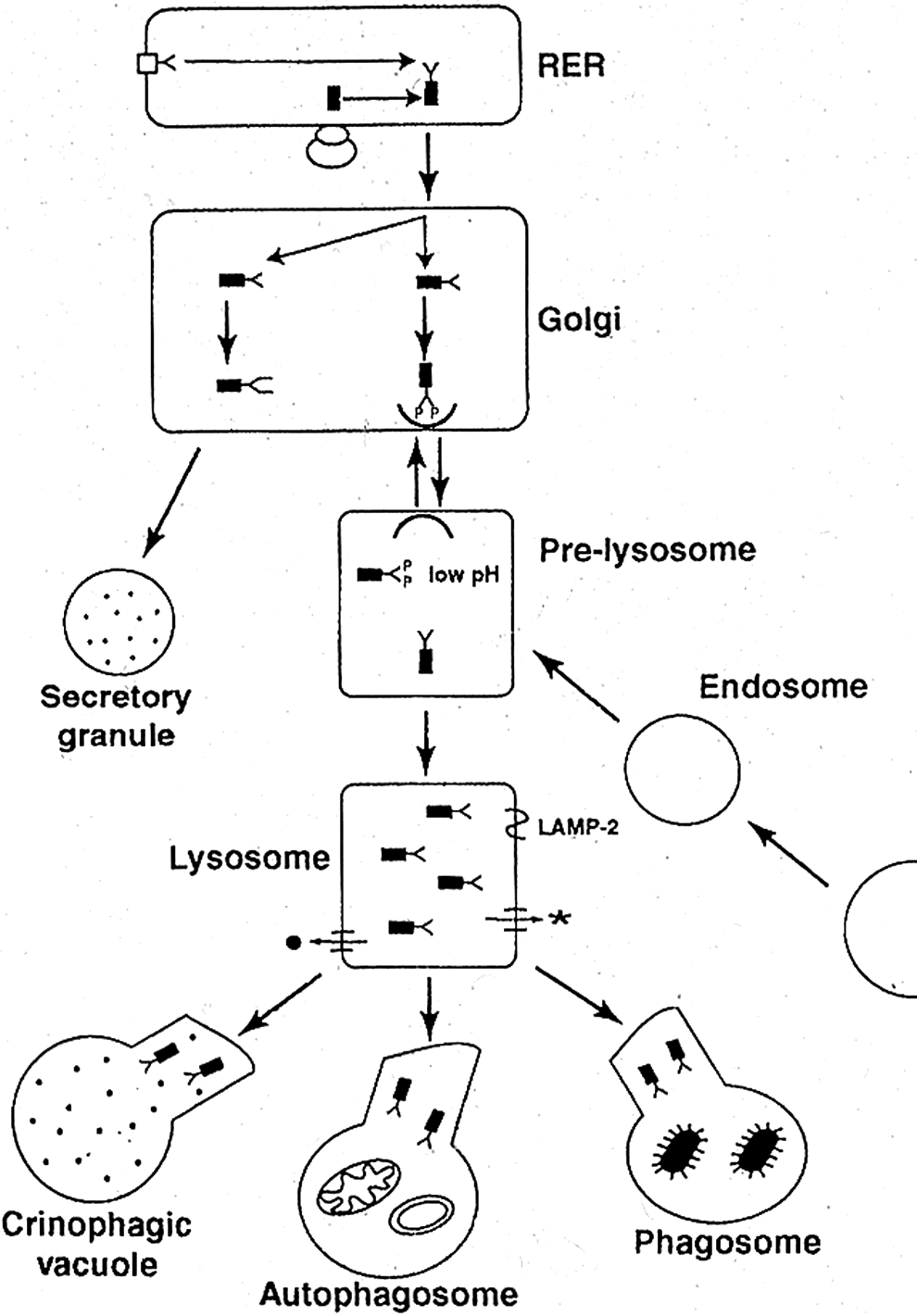
Schematic illustration of synthesis and targeting of lysosomal hydrolases to lysosomes, the mechanisms for transferring substrates to lysosomes for degradation, and the efflux of cystinosin and sialin. Nascent lysosomal hydrolases, secretory, and membrane proteins are glycosylated in the rough endoplasmic reticulum (RER) by transference of preformed oligosaccharides from dolichol-P-P-oligosaccharides. These glycosylated glycoproteins are translocated to the Golgi apparatus, where secretory and membrane glycoproteins are further modified and oligosaccharides of lysosomal hydrolases are phosphorylated (P). All but 2 lysosomal enzymes bind to the M-P receptors and are transported to a prelysosome compartment, where the enzymes are released and the receptor shuttles back to the Golgi apparatus. In the primary lysosome, the prohydrolases are cleaved. The secondary lysosome is formed by fusion of a phagosome with the primary lysosome via lysosomal-associated membrane protein 2 (LAMP-2). The catabolic products exit via transporter proteins such as sialin and cysteine (*).
Lysosomal membrane proteins are classified into several different groups of small metabolite exporter proteins including cystinosin, sialin, and cobalamin transporters. These exporter proteins participate in the efflux of the degradation products such as cysteine, free sialic acid, and cobalamin from the lysosome to the cytosol hybrid enzyme/transporter protein, a defect in 4 different genetic transmembrane enzymes that are defective in mucopolysaccharidosis (MPS)-III (MPS-III) and result in the storage of heparan sulfate. Defect in ion transporter proteins, such as proton/chloride transporter
Lysosomal storage diseases are a group of inherited and acquired disorders. To date, 51 genetically determined lysosomal storage diseases have been described. Since there can be different mutations of the same gene, they result in different clinical manifestations and are classified as infantile and adult types (Table 1). The acquired disorders are the result of inhibition of α-mannosidase II by ingestion of plant materials of
Lysosomal Storage Diseases.
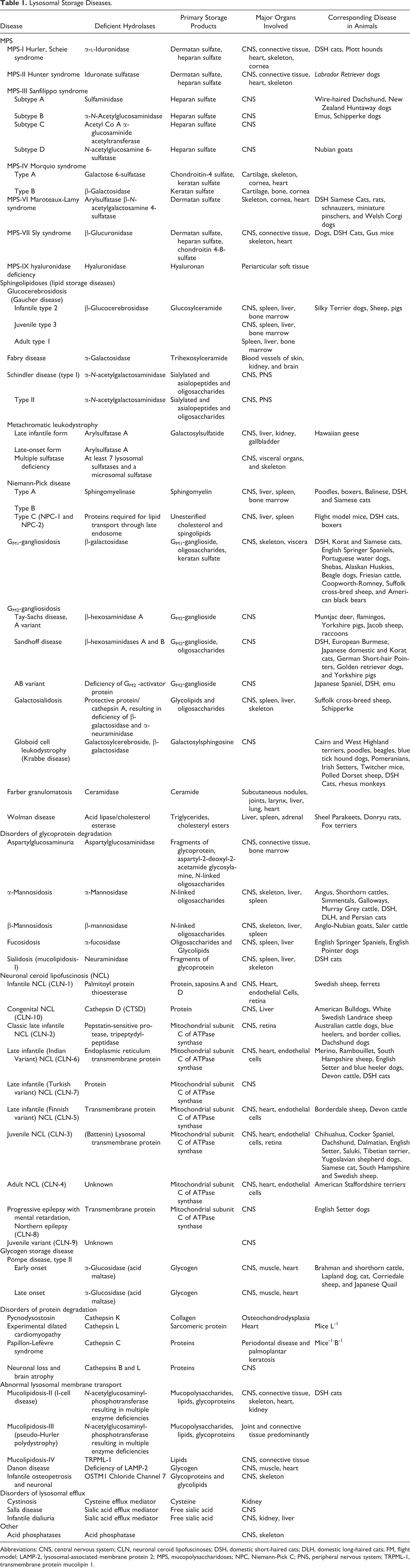
Abbreviations: CNS, central nervous system; CLN, neuronal ceroid lipofuscinoses; DSH, domestic short-haired cats; DLH, domestic long-haired cats; FM, flight model; LAMP-2, lysosomal-associated membrane protein 2; MPS, mucopolysaccharidoses; NPC, Niemann-Pick C; PNS, peripheral nervous system; TRPML-1, transmembrane protein mucolipin 1.
Classification of Mechanism of Lysosomal Storage Disorders

Abbreviation: NPC, Niemann-Pick C.
Most lysosomal storage diseases affect different cell types, tissues, and organs. The brain lesions are particularly prevalent, which comprises two-thirds of all lysosomal diseases. 5 The amount of substrate to be catabolized is influenced by the composition of the cell membrane, the rate of membrane recycling, the rate of cellular catabolism, the life span of the cells, the phagocytic ability of the affected cells, and the nature of phagocytosed substrates. The catabolism of macromolecules in the lysosomes can be regarded as a linear sequence of reactions, in which the product of one reaction serves as substrate for the next. Therefore, the normal and abnormal concentration of any substrate is determined by the influx rate (Vi) of the substrate and the kinetic parameters of the degrading enzymes. They include Michaelis constant (Vm), which is the enzyme–substrate affinity, that is, the binding ability and maximal velocity of the reaction (Vmax), which is obtained when all the enzyme molecules are saturated with substrate.
The majority of these diseases are due to the deficient activity of the lysosomal enzymes caused by mutations of genes coding specific hydrolases. However, the complexity of lysosomal storage disease is seen in mutations of nonenzymatic proteins. 5 The clinical heterogeneity of any one of the lysosomal enzymes is caused by the multiplicity of mutant alleles of a given gene, which leads to difference in the properties of residual enzymes or absence of enzymes.
Clinically, early and severe manifestations are due to the absence of enzyme activity and early death. In cases of low enzyme activity, there is late presentation and mild to moderate manifestations. Normally, most lysosomal hydrolases are present in sufficiently high amount that their respective substrate does not accumulate. Accumulation of undegraded substrate occurs only when the residual enzyme activity is less than the critical threshold of 10% to 15% of the normal enzyme activity. Usually, the residual enzyme activity results in juvenile or adult onset of the disease, and absence of enzyme activity is manifested as severe infantile onset. The third mechanism is due to mutation that results in a defective or absence of the key enzyme, 6-phospho-
Generally, 2 types of changes are observed in lysosomal storage diseases. The primary changes include the direct consequence of abnormal accumulation of substrates or catabolites in the lysosomes, leading to severe impairment of cellular structures and functions and abnormal extracellular matrix. They include alterations of signaling pathways, of intracellular Ca2+ homeostasis, of intracellular trafficking, of lipid biosynthesis, dysmyelinogenesis, formations of ectopic dendrites, retarded bone formation, and cloudy corneas. In some diseases, abnormal accumulation of galactosylsphingosine, that is psychosine, leads to apoptosis. 9 The secondary changes are due to disrupted recycling and are manifested in abnormal or shortage of cellular and extracellular products. In the brain, there is a compromised recycling of neurotransmitter receptors, altered signaling, 10 and dysmylinogenesis. 11 For example, disarray of the collagen fibers leading to cloudy cornea was demonstrated in MPS-I and -VI and retarded bone formation, 12 as was reported in GM1-gangliosidosis and MPS.
The clinical manifestation of some lysosomal storage diseases is very similar, and they are often similar to other developmental and neurological disorders. Therefore, the diagnosis of a specific lysosomal storage disease requires a combination of clinical, morphological, biochemical, and molecular biological techniques.
13
The preliminary diagnosis of most of the lysosomal storage diseases could be made by microscopic examination of the blood smear (Figure 2). If there is an indication of storage disease in paraffin and frozen sections, lectin histochemistry is a useful tool.
14
Electron microscopy of skin biopsies is a useful diagnostic tool for the diagnosis of lysosomal storage diseases, since the skin contains all the body cell types and by observing the morphological features of the storage material that can be identified. Diagnosis of MPS and diseases with interrupted catabolism of oligosaccharides may be determined by urine analysis. Different diagnostic methods such as quick spot test, electrophoresis of urinary glycosaminoglycans, and fluorophore-labeled carbohydrate polyacrylamide gel electrophoresis are used.
13
Specific diagnosis of the majority of lysosomal storage diseases can be achieved by means of assay of the activity of lysosomal hydrolases in white blood cells, cultured fibroblasts, serum, and molecular biology. Postmortem diagnosis is established by both biochemical and morphological examinations. The former includes enzyme assays and biochemical characterization of the storage material, both of which can be done on fresh or frozen tissues. Enlargement and vacuolization of the affected cells (Figure 3A) may be indicators of lysosomal storage disease. Positive staining of the vacuolar membrane with LAMP-2 antibodies will identify the vacuoles as lysosomes. Staining of paraffin and frozen sections with
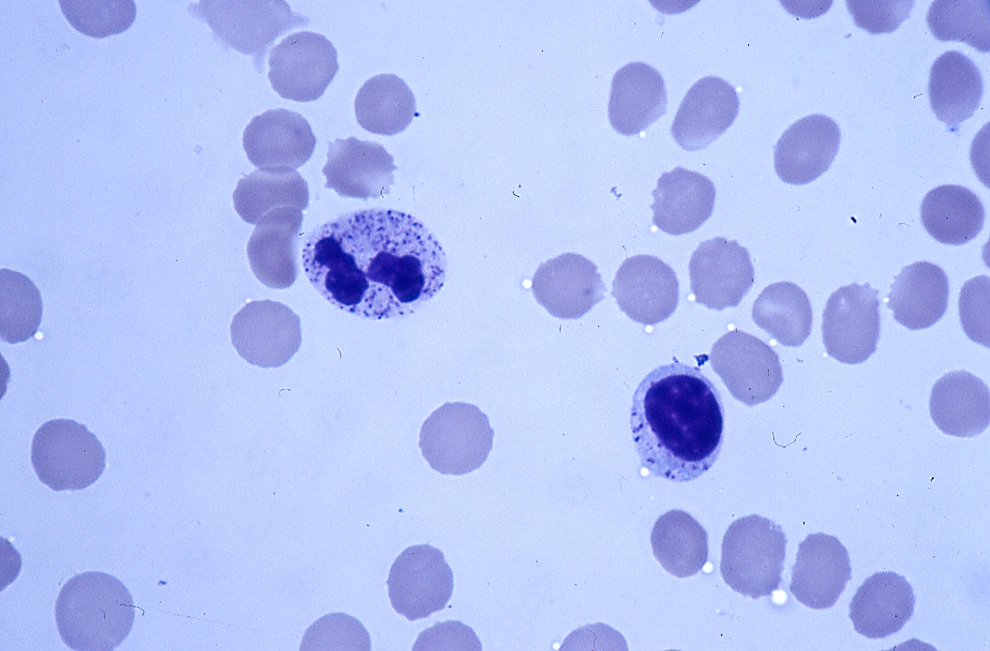
Blood smear of a patient with multiple sulfatase deficiency showing a lymphocyte and neutrophil stained with Wright-Giemsa. The cytoplasm of both cells contains metachromatic granules.
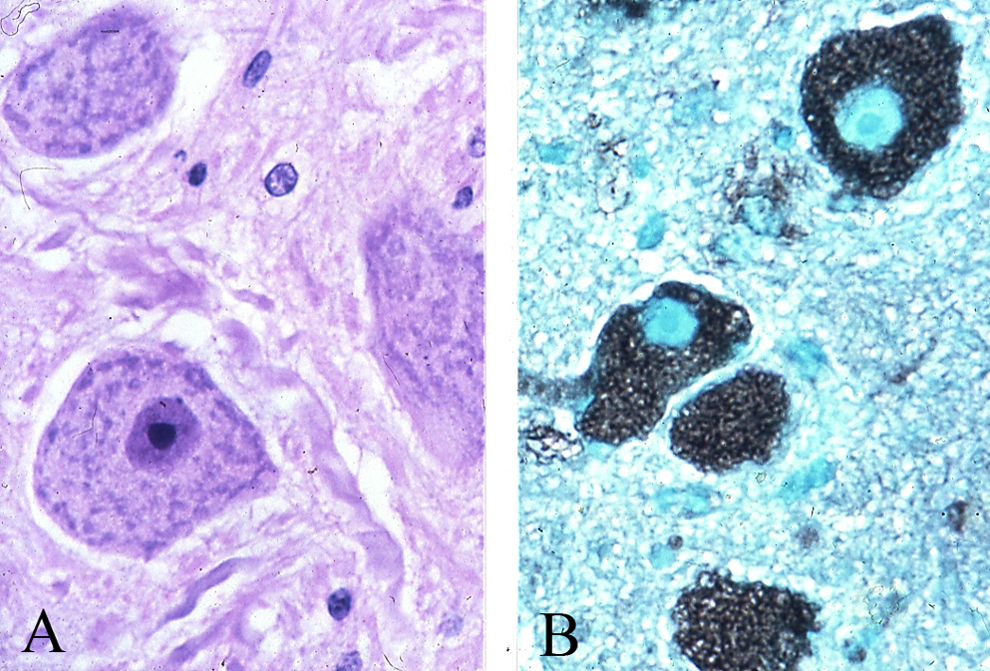
Photomicrographs of a cow cerebrum with α-mannosidosis illustrating (A) enlarged neurons with vacuolated cytoplasm, hematoxylin and eosin (H&E). B, Staining with Con A demonstrating the storage of oligosaccharides with terminal α-mannosyl residues.
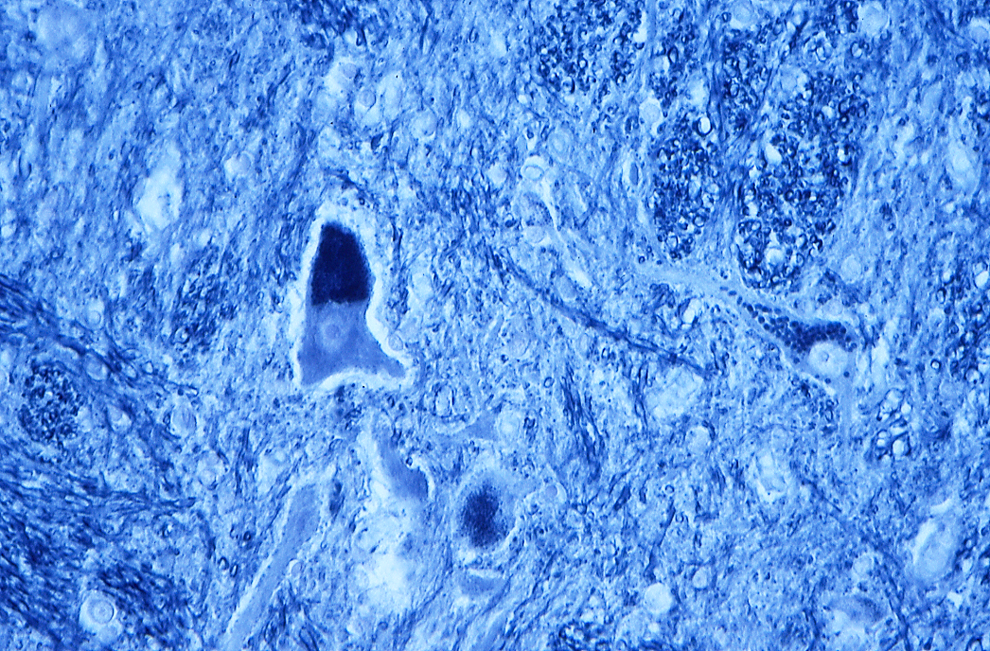
Photomicrograph of frozen section of spinal cord of a 1½-year-old Australian Blue Heeler dog with neuronal ceroid lipofuscinosis in spinal cord stained with Sudan black revealing the storage of ceroid lipofuscin within the neurons.
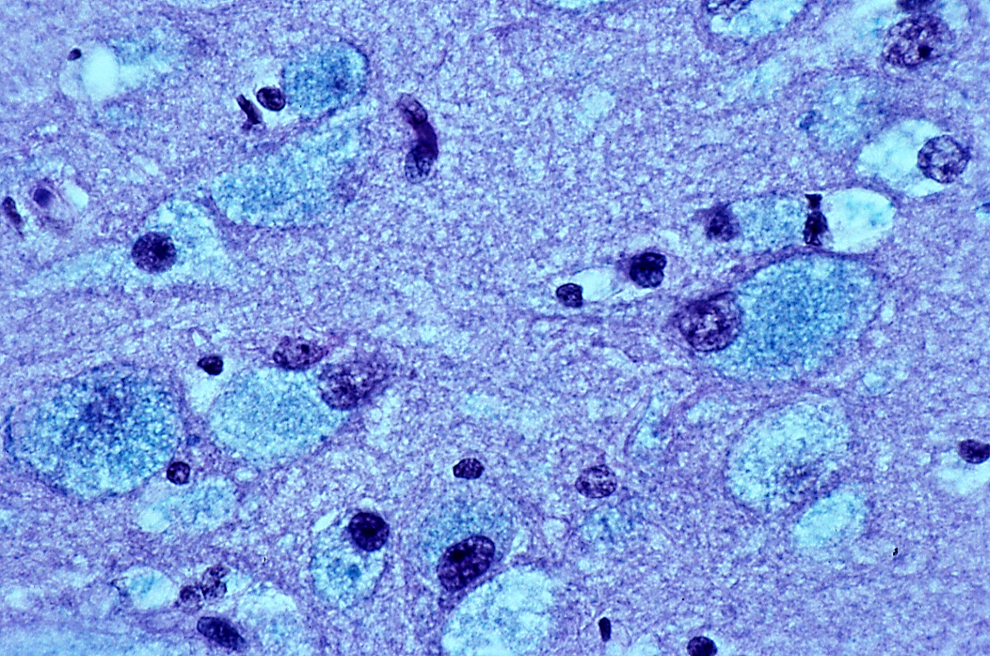
Paraffin section of the brain of a 7-month-old English Springer Spaniel dog stained with Luxol Fast Blue demonstrating the storage of GM1-gangliosides.
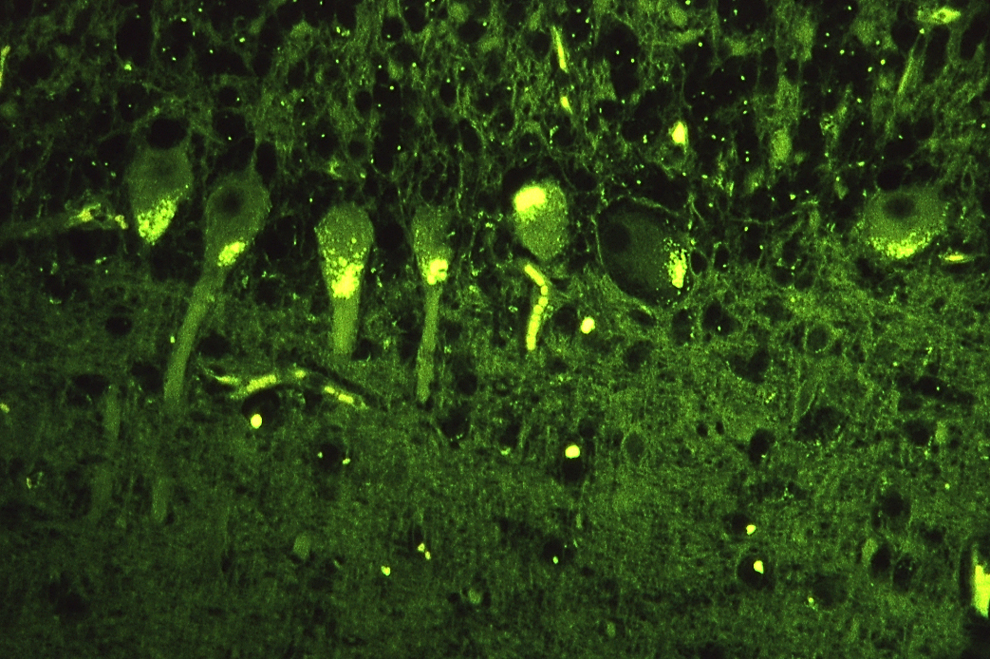
Photomicrograph of the cerebellum of New Zealand sheep with neuronal ceroid lipofuscinosis revealing the presence of autofluorescent neurons.

Electron micrographs of the cerebellum from a 5½-month-old Portuguese water dog. A, Low magnification illustrating on the left neuronal cytoplasm packed with enlarged secondary lysosomes, at the center is a section through an endothelial cell, and on the right is an axon. B, High magnification reveals lysosomes packed with gangliosides. C, Close-up view of the endothelial cell that has enlarged electron-lucent lysosomes containing oligosaccharides. D, On the right, there is high magnification of an axon packed with spheroids.
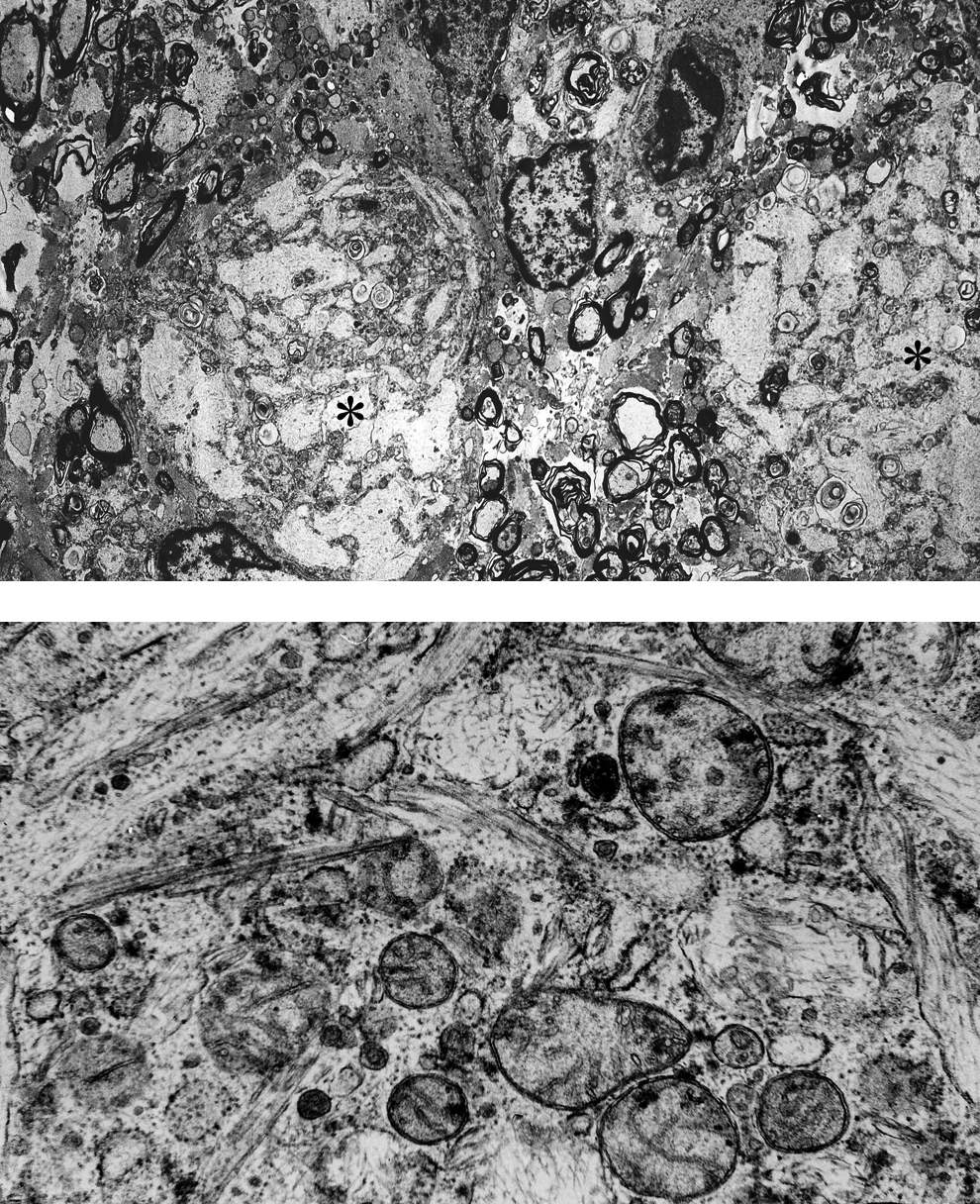
Low (A) and high magnification (B) of a brain from a rhesus monkey with Krabbe disease demonstrating globoid cells (*) that have cleft-like storage material.
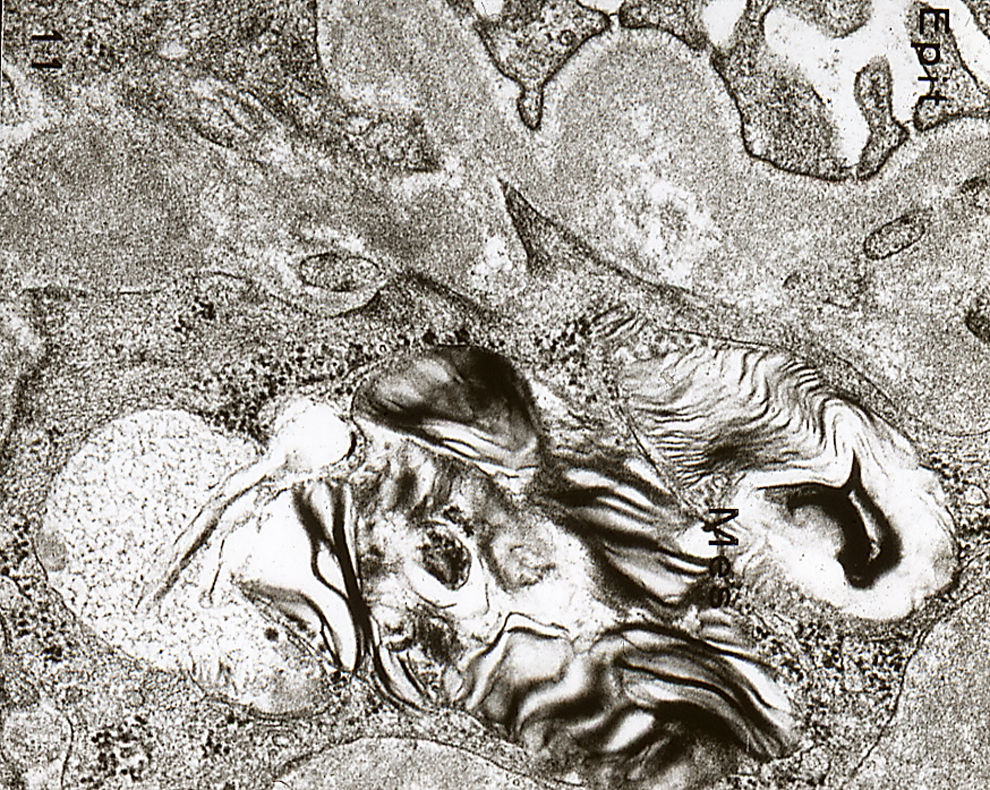
Electron micrograph of glomerular mesangial cell of a Siamese cat with mucopolysaccharidosis (MPS)-IV revealing secondary lysosomes that contains a mixture of lamellated membrane structures and fine fibrillar material.

Low-magnification electron micrograph of myocardial cells from a 2-year-old Australian Blue Heeler dog with neuronal lipofuscinosis that contain secondary lysososmes. Inset: Higher magnification of a lysosome that is packed with curvilinear material.
Mucopolysaccharidosis
Mucopolysaccharidosis is a group of lysosomal storage diseases, which results from the deficiency of specific lysosomal enzymes involved in the degradation of mucopolysaccharides, that is, glycosaminoglycans that are abundant in the extracellular matrix. Chondroitin, dermatan, heparin, and keratan sulfates are the major constituents of the glycosaminoglycan portions of proteoglycans (PGs). The majority of the PGs are present in the extracellular matrix interacting with or binding to collagen, elastin, fibronectin, laminin, and plasma membrane. The hyaluranan is mostly present in soft connective tissue. The inheritance of all these diseases with the exception of MPS-II is autosomal recessive. Mucopolysaccharidosis-II is an X-linked disorder. In all these diseases, except in MPS-IIIC, the deficient enzymes are hydrolases. In these diseases, there is multiple tissue and organ involvement, in particular those that are rich in mesenchymal cells such as the skeletal tissue and cornea. In addition to the primary changes, that is, lysosomal storage, secondary changes such as skeletal development and cloudy cornea are also observed. 10 It has been reported that cloudy cornea as seen in MPS-I and MPS-VI is due to alteration in the corneal stroma (Figure 11). 16 In MPS-IX, the changes are periarticular. In general, the Berry spot test that is done on urine samples is often very useful for early diagnosis of MPS, but enzyme analysis is the gold standard to confirm the diagnosis. The presence of metachromatic granules in leukocytes is indicative of MPS.
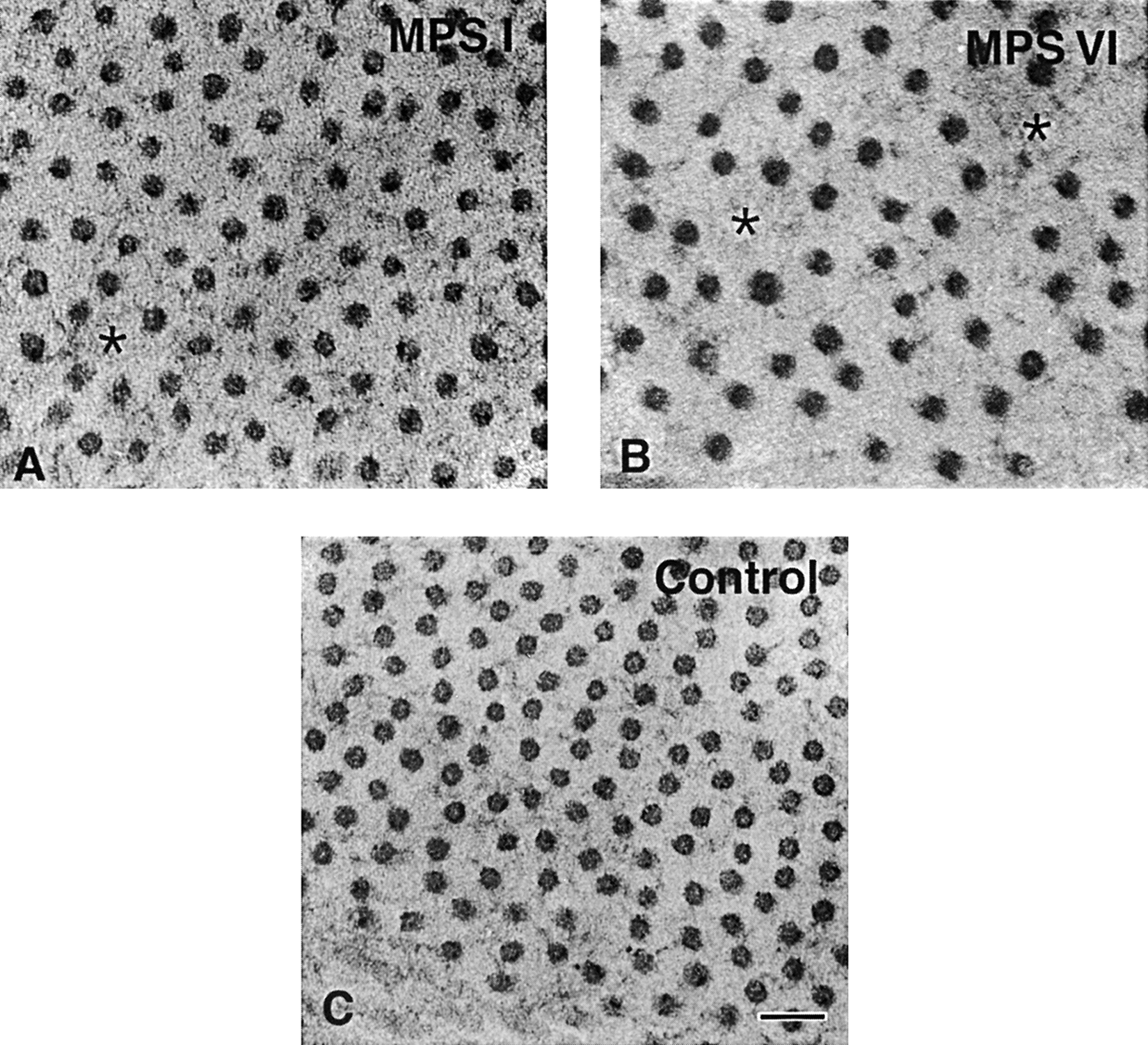
Electron micrograph of the corneal stroma from cats with mucopolysaccharidosis (MPS)-I (A), MPS-VI (B), both with cloudy cornea, and a normal control cat (C). There is an increase in fibril spacing and more irregular packing of fibrils (*) in the affected corneas but not in the control. The collagen fibrils from affected cats are larger and more varied in diameter size. Bar 100 nm. 16
Mucopolysaccharidosis-I
Deficient activity of α-
Mucopolysaccharidosis-II
Mucopolysaccharidosis-II or Hunter syndrome is transmitted as an X-linked recessive disorder that causes the deficiency of iduronate sulfatase activity. This results in primary storage of dermatan sulfate and heparan sulfate. An adult form has been described in Labrador Retriever dogs that are presented with course facial features, visual impairment, progressively neurological, and osteopenia. Similar to MPS-I, neuronal, mesenchymal, and epithelial cells are affected. They are enlarged, vacuolated, and stained with PAS and Alcian blue.
Mucopolysaccharidosis-III
Mucopolysaccharidosis-III or Sanfilippo disease is a group of diseases that are caused by mutations of 4 different enzymes but have a common primary storage material, that is, heparan sulfate. Unlike the other components of glycosaminoglycans, which are the major components of the extracellular matrix, heparin sulfate is a component of transmembrane syndecan. As a result, unlike other MPS, neurological signs are the major clinical manifestation. However, it is noteworthy that in a few cases of different types of MPS-III in humans and in a colony of Nubian goats with MPS-IIID, only 1 goat had both cloudy corneas due to irregular packing of the collagen fibrils and skeletal abnormalities, indicating relatively low activity of another enzyme in the pathway, which contributes to those lesions.
Mucopolysaccharidosis-III A is caused by the deficiency of heparin
Mucopolysaccharidosis-IV
Mucopolysaccharidosis-IV or Morquio syndrome is a group of diseases that are caused by mutations of 2 different genes but with similar clinical manifestations and morphological changes. Mucopolysaccharidosis-IVA is caused by the deficiency of galactose 6-sulfatase activity and results in lysosomal storage of chondroitin-4-sulfate and keratan sulfate. Mucopolysaccharidosis-IVB is caused by mutations of β-galactosidase and leads to the storage of keratan sulfate. Clinically, the deficient activity of both hydrolases is manifested by corneal clouding and skeletal abnormalities.
Mucopolysaccharidosis-VI
Mucopolysaccharidosis-VI or Maroteaux-Lamy syndrome is due to the deficiency of
Mucopolysaccharidosis-VII
Mucopolysaccharidosis-VII or Sly syndrome is caused by the deficiency of β-glucoronidase activity and results in lysosomal storage of dermatan sulfate, heparin sulfate, and chondroitin-4-6 sulfates. Clinical manifestations include severe skeletal abnormalities, hepatosplenomegaly, and thickening of atrioventricular heart valves. It has been described in DSH cats, mixed breed dogs, and Gus mice.
Mucopolysaccharidosis-IX
Mucopolysaccharidosis-IX is due to the deficiency of lysosomal hyaluronidase-1 activity that results in the storage of hyaluronan. Hyaluronan is a major component of extracellular matrix. Patients present with short stature, cutaneous swelling, and periarticular swelling.
Mucolipidosis-I
Mucolipidosis-I, or sialidosis, is the result of deficiency of lysosomal α-neuraminidase (sialidase) activity. Its deficiency results in lysosomal storage of sialilated oligosaccharides and sialilated glycolipids. Clinical manifestations include neurological signs, organomegaly, and skeletal abnormalities. 17 It has been described in DSH cats.
Mucolipidosis-II and Pseudo-Hurler Polydystrophy
The ML-II or I-cell disease and Pseudo-Hurler polydystrophy (ML-III) are the consequence of a primary deficiency of phospho-
Mucolipidosis-IV
Mucolipidosis-IV is due to the mutation of the transmembrane protein ML-1, a transient receptor potential, also known as TRPML1. 19 It plays a major role in the fusion of autophagosomes with late endosomes/lysosomes and serves as a cation channel. Gene mutations result in defective chaperone-mediated autophagy. The clinical manifestations include psychomotor retardation, blood iron deficiency, and gastric achloridia.
Sphingolipidosis (Lipid Storage Diseases)
Sphingolipidosis refers to a group of disorders caused by deficient activity of lysosomal hydrolases that participate in the degradation of lipid that contains sphingosine as well as by mutations of the corresponding sphingolipid activator proteins (SAPs), including saposins and GM2 activator protein, and the protective protein/cathepsin A. The SAPs and GM2 activator protein are required for binding the glycolipids to their corresponding hydrolase. 20,21
GM1-Gangliosidosis
GM1-gangliosidosis is caused by deficient activity of β-galactosidase, resulting in lysosomal storage of glycolipids, oligosaccharides, and glycosaminoglycans, with a nonreducing terminal β-galactoside.
22
These molecules are major constituents of cell membrane and extracellular matrix. Therefore, almost all cell types are affected, but they differ in the nature of the storage materials. Neurons contain numerous lamellated membrane structures that are often described as fingerprints or zebra bodies (Figure 7A and B), and on frozen sections they stain with the lectin
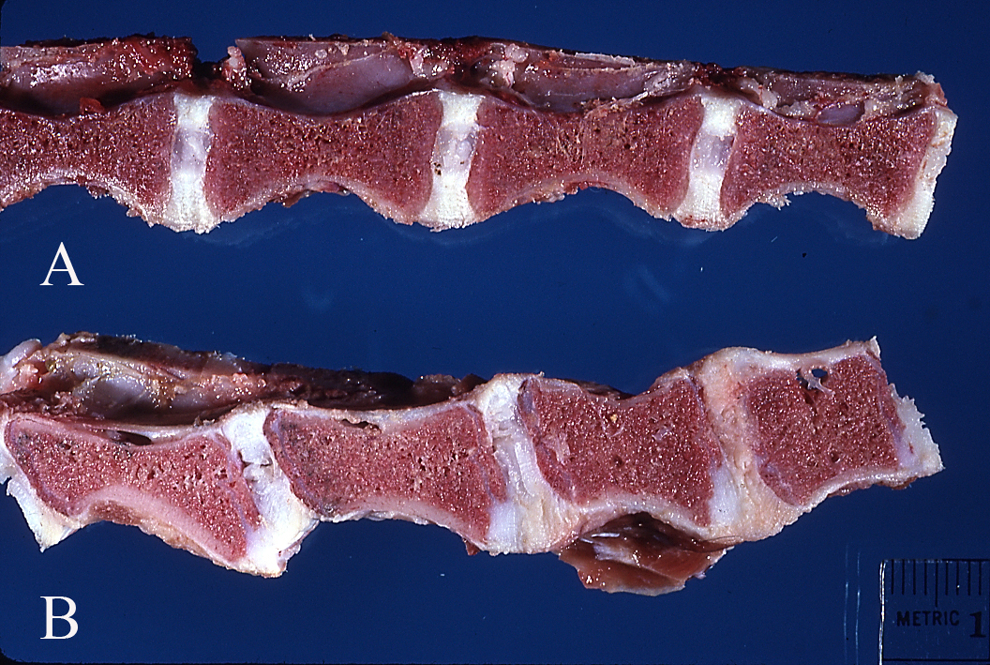
Photograph of lumbar vertebrae from a 3-month-old normal Portuguese water dog (A) and his brother with GM1-gangliosidosis (B). In (B) the vertebrae are shorter and the intervertebral disks are irregular and wide.
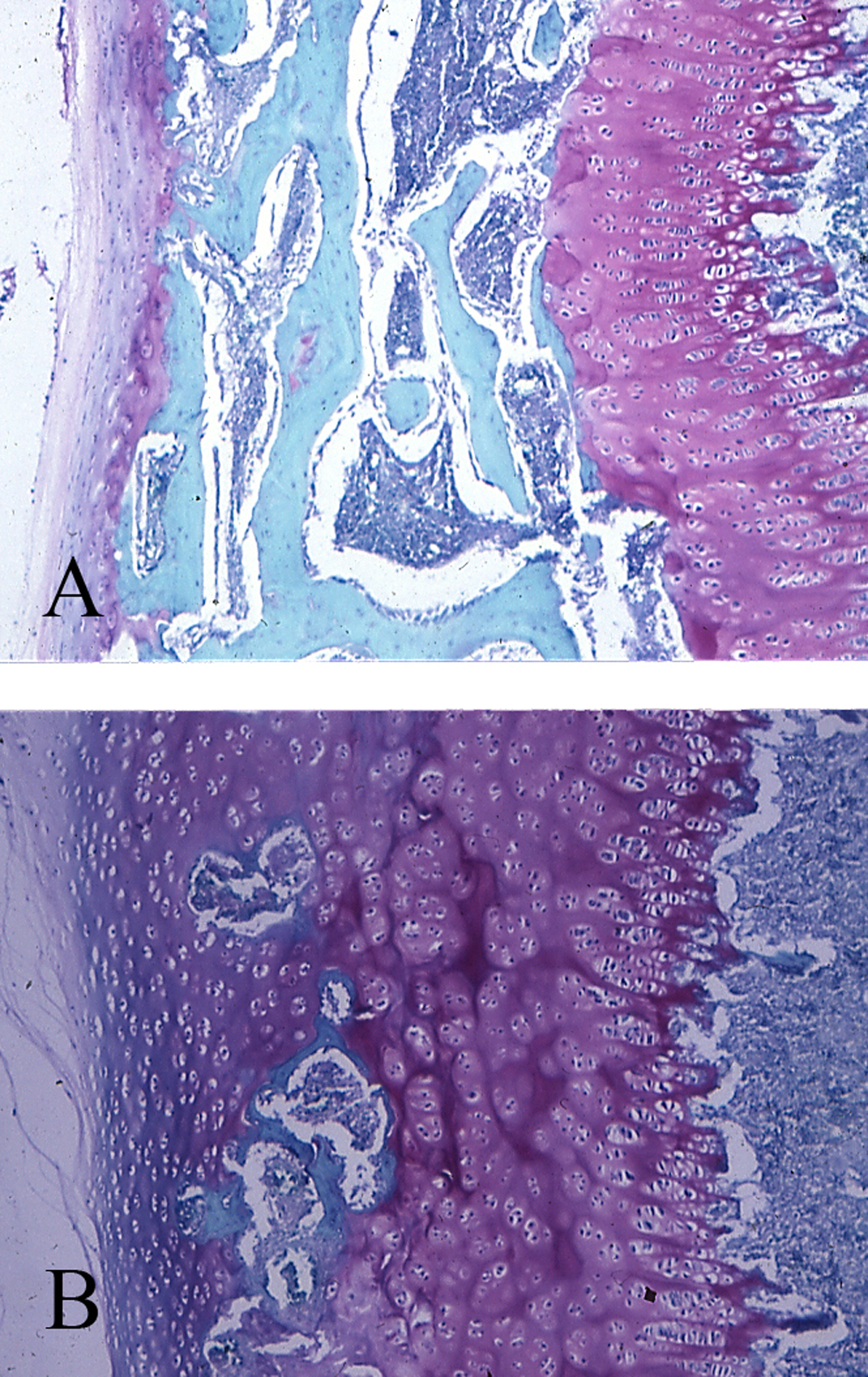
Longitudinal section of part of a lumbar vertebra of a 2-month-old normal Portuguese water dog and his affected sex- and age-matched brother. B, In the normal puppy (A), the ossification is advanced and the primary spongiosa are well developed. In the affected puppy (B), the ossification is retarded, the primary spongiosis is poorly developed and there is metaphysical osteoporosis. Safranin-O. 12
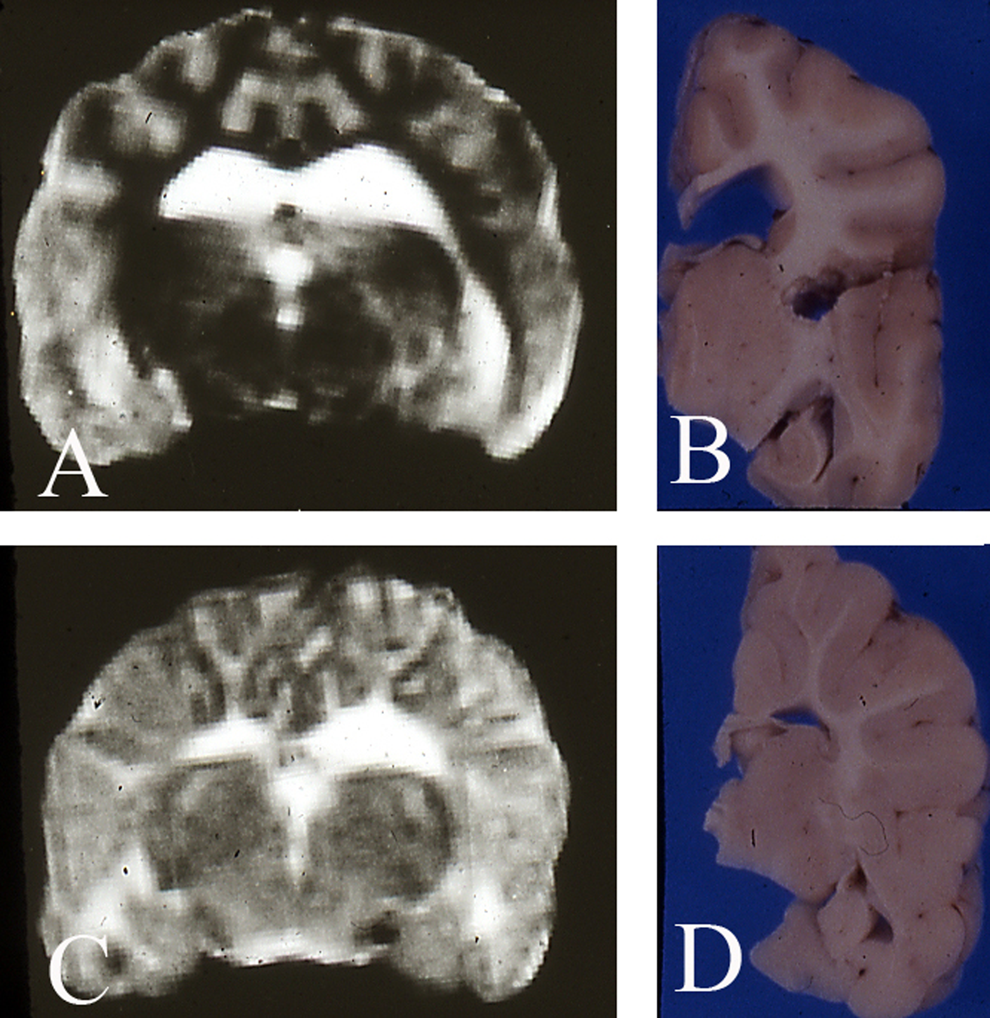
A, T2-weighted magnetic resonance imaging (MRI) of a 9-month-old normal control English Springer Spaniel, illustrating a normal ratio between gray and white matter. B, Corresponding coronal section from the hemisected fixed brain demonstrating prominent gray and white matter. C, MRI of an affected 9-month-old littermate illustrating increased volume of gray matter and poor development of low signal intensity of the white matter. D, Corresponding coronal section revealed increased gray matter and reduced white matter, in particular in the corona radiate. 11
GM2-Gangliosidosis
GM2-gangliosidosis is a group of 3 different disorders characterized by primary or secondary deficiency in the activity of lysosomal β-hexosaminidase. There are 2 isoenzymes of β-hexosaminidase, Hex A and Hex B. Three genes are coding proteins that are required for the degradation of GM2-ganglioside. They include the genes that encode α and β subunits of β-hexosaminidase and the GM2 activator protein. As a consequence, these 3 disorders differ in their clinical manifestations, in the type of the affected cells, and in the nature of the storage material. 23
Tay-Sachs Disease
Tay-Sachs disease, often referred to as B variant, is due to a defect in α subunit of Hex A, which results in lysosomal storage of GM2-gangliosides in neural tissue. Clinically, it presents with neurological signs. It has been described in Muntjac deers and in the American flamingos. Ultrastucturally, the neurons contain enlarged lysosomes filled with lamellated membrane structures. Tay-Sachs disease had been described in American flamingo, European Burmese cast, Muntjak deers, Yorkshire pig, Jacob sheep, and raccoons.
Sandhoff Disease
Sandoff disease, often referred to as O variant, is caused by a defect in β subunit that is present in both Hex A and Hex B. This results in lysosomal storage of GM2-gangliosides in neural tissue and oligosaccharides and glycosaminoglycans in mesenchymal and epithelial tissues. Clinically, it is manifested by neurological, visceral, and skeletal changes. Neurons contain numerous lysosomes packed with lamellated membrane structures that often are described as fingerprints or zebra bodies, while other cell types such as mesenchymal and epithelial contain fine fibrillar material or the lysosomes appear empty. It has been reported in DSH, Japanese domestic and Korat cats, German Shorthair, Golden Retriever, Pointer dogs, and in Yorkshire pigs. Four different mutations of the β subunit have been identified in cats.
AB variant
AB variant is due to mutations of the GM2 activator protein that is transported to the lysosome via sortilin. The GM2 activator protein is required for binding the GM2 gangliosides to β-hexoasminidase. Therefore, similar to Tay-Sachs, there is lysosomal storage of GM2 gangliosides in neural tissue. Since GM2 activator protein is required for binding the GM2 gangliosides to β-hexoasminidase, its mutation results in neurological signs. It has been described in Japanese Spaniel dogs and DSH cats.
Galactosialidosis
Galactosialidosis is a lysosomal storage disease that is caused by a defect in the protective protein/cathepsin A that results in the deficiency of β-galactosidase and neuraminidase. The deficiency of both enzymes leads to lysosmal storage of GM1-gangliosides in neural tissue and oligosaccharides in mesenchymal and epithelial tissues. Clinically, the infantile form presents with neurological visceromegaly and skeletal abnormalities. 24 It has been reported in sheep.
Glucocerebrosidosis (Gaucher Disease)
Gaucher disease is primarily characterized by deficient activity of glucocerebrosidase and lysosomal storage of glucocerebrosides 25 and polylactosaminoglycans. 26 Rarely, deficient activity of activator protein (saposin C) causes a similar condition. Saposin C is transmitted to the lysosome via sortilin. 3 Since the major stored substrate originates from the plasma membrane of erythrocytes and leukocytes, reticuloendothelial cells are the most severely affected. The characteristic feature of Gaucher disease is the presence of lipid-laden reticuloendothelial cells (Gaucher cells) in the spleen, liver, and bone marrow. The Gaucher cells have enlarged striated cytoplasm. Ultrastructurally, Gaucher cells have enlarged lysosomes that contain twisted tubular structures. Gaucher disease has been reported in Silky terrier dogs, pigs, and sheep.
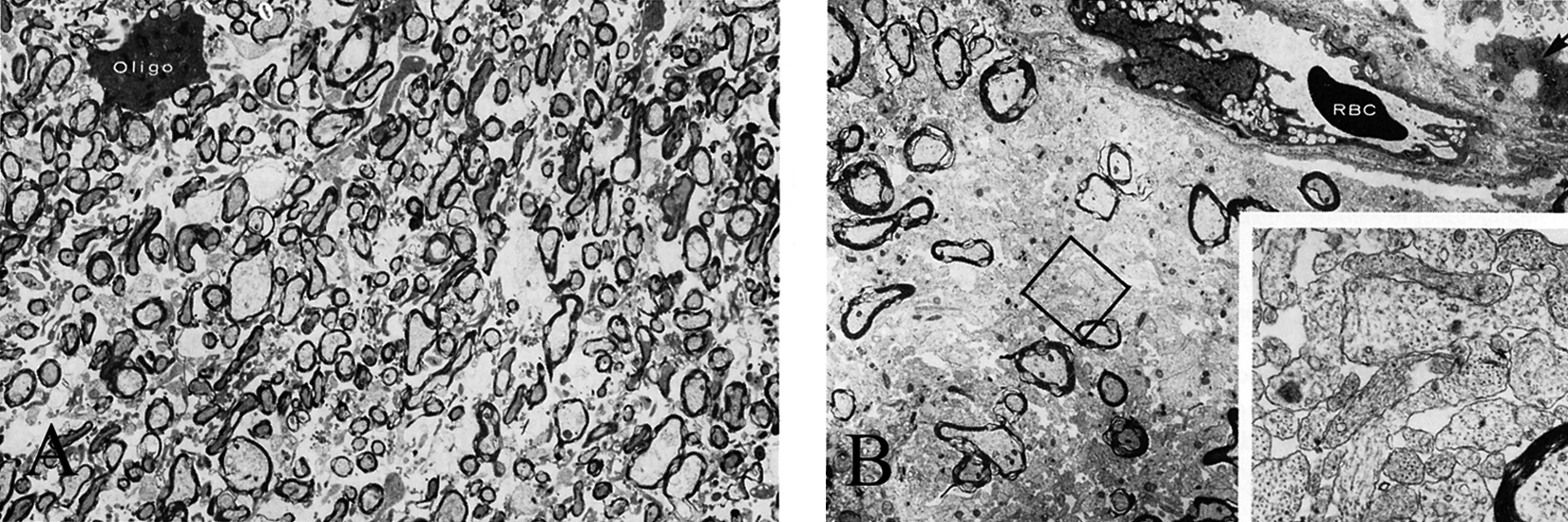
A, Electron micrograph of the corpus callosum from a 2-month-old normal Portuguese water dog (PWD) puppy, illustrating an oligodendrocyte (oligo) and normal myelinated axons. Corresponding electron micrograph from an age-matched affected PWD litter mate demonstrating erythrocyte (RBC), vacuolated endothelial cells, and only few myelinated axons. Inset: Higher magnification of the square revealing normal appearing unmyelinated axons. 11
Gaucher-like cells are often observed in disorders in which there is oversupply of substrate, such as myelogeneous leukemia, lymphoma, and various types of hemolytic anemia. In these conditions, there is an oversupply of plasma membrane from white or red cells, which must be catabolized by the reticuloendothelial cells.
Galactosylceramide Lipidosis (Krabbe Disease)
Krabbe disease, also known as globoid cell leukodystrophy, is caused by deficient activity of lysosomal galactosylcerebroside β-galactosidase and lysosomal storage of galactosylsphingosine (psychosine). 27 Rarely, deficient activity of activator protein (saposin A) results in a similar manifestation. Myelinated tissues in the central and peripheral nervous system are most severely affected since galactosylceramide is found almost exclusively in myelin. Morphologically, it is characterized by destruction of myelin and oligodendroglia, glial scarring, and proliferation of globoid cells throughout the white matter. The globoid cells are enlarged vacuolated macrophages typically localized around blood vessels. Ultrastructurally, globoid cells contain enlarged lysosomes packed with twisted tubules (Figure 8). Krabbe disease has been described in Cairn and West Highland white terriers, beagles, blue tick hounds, Pomeranians, Irish setters, miniature poodles, DSH cats, Polled Dorset sheep, “twitcher” mice, and rhesus monkeys. In frozen sections, the storage material in the globoid cells is stained with RCA-I lectin that binds to the terminal β-galactosyl residues.
Niemann-Pick Disease Types A and B
Niemann-Pick types A and B are lysosomal storage diseases that result from deficient activity of sphingomyelinase and lysosmal storage of sphingomyelin. 28 Sphingomyelinase is internalized to the lysosome via sortilin. 29 Type A is presented early in life. It is a fatal disorder with severe progressive neurodegeneration and hepatosplenomegaly. In contrast, type B presents later in life and has mild to moderate manifestation. They are presented with hepatosplenomegaly and they have mild or no neurological signs. Morphologically, the affected cells have an enlarged foamy cytoplasm that contains lysosomes packed with lamellated membrane structures. It has been described in poodles and boxer dogs and in Balinese, DSH, and Siamese cats.
Niemann-Pick Type C Disease
Niemann-Pick type C is due to mutations of 2 different genes
Fabry Disease
Fabry disease is an X-linked recessive disorder caused by deficient activity of lysosomal α-galactosidase in males and often in heterozygous females. This enzyme cleaves the α-galactosyl residues mostly from globotriaosylceramide and to a lesser extent from galactosylceramide.
31
It is thought that the major mechanism that leads to the clinical manifestations is partly due to poor blood perfusion caused by lysosomal storage in vascular endothelium, particularly in the kidneys, heart, nervous system, and skin. It is noteworthy that other tissues are also involved. The clinical manifestations in heterozygous females are milder. Electron microscopic examination of affected cells reveals enlarged lysosomes containing lamellated membrane structures. On frozen sections, the storage material is stained with
Farber Lipogranulomatosis
Farber disease is due to the deficient activity of lysosmal acid ceramidase and accumulation of ceramide in lysosomes. It is noteworthy that saposin D is needed to activate the lysosomal ceramidase. Ceramide is an important component in the ganglioside and myelin metabolism. It is characterized by the presence of subcutaneous nodules that are most prominent around joints and also by liver, spleen, and lung granulomas. The granulomas consist of enlarged foamy macrphages and multinucleated giant cells that contain lysosomes. Ultrastructure of the storage material appears as banana-shaped or tubular-shaped vacuoles. 32 Storage material is present in other cell types including neurons and even chondrocytes.
Wolman Disease and Cholesteryl Ester Storage Disease
Both conditions are due to the deficient activity of lysosomal acid lipase and storage of cholesteryl esters and triglycerides in most tissues. 33 The onset of Wolman disease is in infancy. It is severe and is manifested by hepatosplenomegaly, steatorrhea, and adrenal calcification. In contrast, the onset of cholesteryl ester storage disease is later in life; it is milder, and hepatomegaly maybe the only sign. Morphologically, the affected cells contain lysosomes filled with lipid droplets and cholesterol clefts. Wolman disease has been reported in the Sheel parakeet, Donrou rats, and Fox terriers.
Metachromatic Leukodystrophy
Metachromatic leukodystrophy (MLD) is mainly due to the deficient activity of arylsulfatase A and rarely due to the deficiency of activator protein (saposin B). 34 Saposin B is transmitted to the lysosome via sortilin. 3 Arylsulfatase A desulfates the 3-0-sulfogalactosyl from the glycolipids containing 3-0-sulfogalactosyl. These sulfated glycolipids are mostly present in myelin sheaths in the central and peripheral nervous system and less so in the kidney, gallbladder, and liver. Severe neurological defects are manifested in the infantile form; these are moderate to mild in the juvenile and adult forms. Morphologic findings include demyelination in the central and peripheral nervous systems and storage of metachromatic material in neurons and Schwann cells. Metachromatic storage material is present in bile ducts, gallbladder epithelium, hepatocytes, and in segments of the renal tubules. Ultrastructural studies reveal the lysosomal storage of lamellated membrane structures. It had been reported in Hawaian geese.
Multiple Sulfatase Deficiency
Multiple sulfatase deficiency (MSD) is due to mutations of sulfatase-modifying factor 1 (SUMF1). 35 Sulfatase-modifying factor 1 encodes the enzyme responsible for posttranslation modification of a cysteine residue that is essential for the activity of sulfatases. Multiple sulfatase deficiency is characterized by a deficiency of 12 sulfatases and leads to lysosomal storage of sulfated lipids. The clinical manifestations resemble those seen in MLD and MPS-I to MPS-VII. Morphological examinations of neuronal tissue reveals lysosomal storage of lamellated membrane structure, whereas in mesenchymal and some epithelial cells there is storage of a fine fibrillar material.
Glycoprotein Storage Diseases
In this group of storage diseases, oligosaccharides are the major storage compounds, although in some disorders such as fucosidosis there is also storage of glycolipids. 17 Glycoprotein molecules contain oligosaccharide chains covalently attached to a protein. There are 2 major types of glycoproteins, the O-linked glycoproteins that are attached through hydroxyl groups of serine or threonine and the N-linked glycoproteins that are linked through the free amino acid group of asparagines. Glycoproteins are major constituents of cellular membranes and extracellular matrix, including proteoglycan and collagen. Deficient activity of α-fucosidase, α-neuraminidase (sialidase), α-mannosidase, β-mannosidase, β-endo-N-actylglucosaminidase, β-hexosaminidases A and B, which participate in the degradation of oligosaccharides, is manifested in most cell types.
α-Mannosidosis
Deficient activity of lysosomal α-mannosidase results in lysosomal storage and elevated urinary excretion of α-mannosyl-rich
β-Mannosidosis
Deficient activity of lysosomal β-mannosidosis results in lysosomal storage and increased urinary secretion of β-mannosyl-rich
α-Fucosidosis
Fucosidosis is due to the deficient activity of α-fucosidase that cleaves the terminal residues of glycoproteins and oligosaccharides and also cleaves the glycolipids in neural tissues. It had been reported in humans and as an infantile form in English Pointers and as a juvenile form in English Springer Spaniel dogs. Clinically, in both breeds they presented with progressive neuromuscular signs. Morphological studies of dogs revealed vacuolization of neural, epithelial, and mesenchymal cells. The lysosomes in all tissues are electron-lucent/or with very fine fibrils; while in neurons and Schwann cells, the lysosomes contain lamellated membrane structures. Paraffin sections of human tissue are stained with
Aspartylglucosaminuria
Aspartylglucosaminuria is due to the lack of activity of lysosomal aspartylglucosaminidase (AGU) hydrolase that cleaves the link between aspargine and glycosamine, which is the final step in the breakdown of glycoproteins and oligosaccharides. Deficient activity of AGU results in lysosomal accumulation of aspartylglucosamine. Clinically, it is manifested by developmental delay, skeletal abnormalities, and neurological signs. 37 Morphologically, there is cytoplasmic vacuolation in majority of the cells, and ultrastuctural studies reveal electron-lucent lysosomes or very fine fibrils
Sialidosis
Sialidosis often referred to as mucolipidosis-I is a lysosomal disorder caused by deficient activity of neuraminidase, an enzyme that cleaves α2→3 and α2→6 sialyl linked with several oligosaccharides, glycopeptides, and glycolipids. Clinically, it presents as a severe infantile form with neurovisceral and skeletal abnormalities. 17 Slightly moderate symptoms are manifested later in childhood, while neurological symptoms are characteristic of the adult form. Lysosomes containing lamellated membrane structures are seen in neural tissue while electron-lucent lysosomes or very fine fibrils are seen in mesenchymal and epithelial cells. The storage material stains with wheat germ agglutinin. It has been described in DSH cats.
α-N-acetylgalactosaminidase Deficiency: Schindler Disease
Deficient activity of α-
Glycogen Storage Disease Type II: Acid α-Glucosidase Deficiency
Glycogen storage disease type II, also referred to as Pompe disease or acid maltase deficiency, is caused by the deficient activity of acid α-glucosidase. This enzyme degrades glycogen and maltase. Deficient activity of acid α-glucosidase results in impaired degradation of glycogen and its lysosomal storage in many tissues, but the largest amounts occurs in cardiac and skeletal muscle and in hepatocytes in severe infantile form. The infantile early-onset form presents with hypotonia, cardiomegaly, rapid progressive weakness, and less marked hepatomegaly. The juvenile and adult onsets are milder and characterized by muscle weakness. Morphologically, the cells are enlarged and vacuolated. 39 On frozen sections, the stored glycogen is stained by PAS and removed by treatment with diastase. Electron microscopy reveals lysosomes packed with fine granular material that is characteristic of glycogen. This disorder had been described in Brahman and Shorthorn cattle and Lapland dogs, cats, Corriedale sheep, and Japanese Quails.
Neuronal Ceroid Lipofuscinosis
The NCL is a heterogenous group of 10 different inherited neurodegenerative lysosomal storage diseases. 40 –42 Their clinical manifestations include visual impairment, seizures, progressive psychomotor retardation, and premature death. Histologically, they are characterized by accumulation of autofluorescent material that stains with prostate-specific antigen and Sudan Black in neurons and by a wide variety of other cell types.
Infantile NCL
Infantile NCL (INCL) and late NCL are due to mutations of the
Late-Infantile NCL
Late-infantile NCL is a disorder caused due to mutations of
Juvenile NCL Batten Disease
Juvenile NCL (JNCL) is due to the mutations of
Adult NCL (NCL-4)
Adult NCL or Kufs disease is caused by the mutation of gene
Finnish Variant Late Infantile NCL (NCL-5)
The NCL-5 disorder is due to the mutations of the
Late Infantile Early Juventile NCL (NCL-6)
The NCL-6 disease is caused by mutations of the
Turkish Variant Late-Infantile NCL (NCL-7)
The product of the
Northern Epilepsy
This condition is due to the mutations of the
Juvenile Variant of NCL (NCL-9)
A new variant of JNCL was recently identified and characterized.
42
It is due to the mutation of
Cathepsin D deficiency (NCL-10)
Cathepsin D deficiency (NCL-10) is due to the mutation of the cathepsin D gene (
Pycnodysostosis: Cathepsin K Deficiency
Cathepsin K is a cysteine protease and is a member of the family of lysosomal proteases that catabolized organic bone matrix and collagen in other tissues. Cathepsin K is internalized into the lysosome via sortilin. Deficient activity of cathepsin K results in defective bone growth and remodeling. It is manifested by short stature, dysmorphism, osteopetrosis, pathologic fractures, and dental abnormalities. Osteopetrosis is the major histological feature. 43 Ultrastructural examinations reveal vacuolated osteoclasts that contain lysosomes with collagen fragments.
Infantile Osteopetrosis and Neuronal Storage Disease
Infantile osteopetrosis and neuronal storage disease is due to mutation of a gene that codes chloride channel 7 (ClC-7). Ostoclasts degrade bone matrix by secretion of acid by vacuolar H+ adenosine triphosphatase (V-ATPase) and ClC-7. 44 It is followed by the degradation of the bone matrix, mostly collagen type I by cathepsin K. Neuronal storage of PAS and stainable and autofluorescent LFB material is observed. Electron microscopic findings revealed lysosomes containing lamellated membrane structures and electron-dense material and axons packed with spheroids.
Disorders of Membrane Transport
Free sialic acid storage
Lysosomal storage of free sialic acid is due to mutations of a gene that codes for sialin, a lysosomal membrane protein. Sialin is an anion transporter protein that transports free sialic acids to the cytosol. Mutations of sialin result in lysosomal storage of free sialic acids. Clinically, it is manifested as a severe infantile form with severe neurological signs, hepatosplenomegaly, bone dysplasias, and nephrosis. An adult form is milder and is characterized by neurological signs. Morphologically, most cell types have enlarged vacuolated cytoplasm, that is, electron-lucent lysosomes. 45
Cystinosis
Cystinosis is due to mutations of the gene that codes cystinosin, a lysosomal membrane protein. Cystinosin is a transporter protein that carries the amino acid cysteine from the lysosome to the cytosol. Storage of cysteine occurs in multiple organs including brain, eyes, kidneys, liver, spleen, and thyroids. This results in neurological, visual, thyroid, and renal dysfunction. Light microscopic studies demonstrate cytoplasmic storage of crystals that are birefringent on polarized frozen sections. Electron micrographs demonstrate angular, needle-shaped, and hexagonal crystalline spaces in the cytoplasm. 46
Danon Disease: Glycogen Storage Disease With Normal Acid Maltase
Danon disease is an X-linked dominant disorder due to mutations of
Disorders of Lysosomal-Related Organelles
Genetic, biochemical, and structural studies have revealed that certain specialized cell types have lysosome-related organelles that share features with lysosomes but have distinct morphology, composition, and/or function. They include melanosomes in melanocytes, lytic granules and delta granules in platelets, lamellar bodies in type II pneumocytes, and variants of acidic granules. The most common disorders in the biosynthesis of lysosomal-related organelles is Chediak-Higashi syndrome that have been described in different breeds of cattle, including Brangus, Hereford, and Japanese black, in Persian cats, Killer whales, Aleutian strain of mink and Beige mice. It results in partial albinism, mild bleeding, immune dysfunction, and interstitial pneumonia. Spontaneous mutations in pale ear mice and in cappuccino mice are analogous to the Hermansky-Pudlak syndrome that is associated with severe albinism and very mild bleeding.
Footnotes
Acknowledgments
The authors wish to acknowledge the contribution of Susan Marino for her technical help.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
