Abstract
Background:
Patellar tendon rupture is uncommon but can severely impair knee extension and functional recovery. Most injuries occur during sports involving eccentric quadriceps loading, often with valgus stress. Traditional repair using transosseous sutures reinforced with steel wire cerclage provides initial stability but carries drawbacks, including hardware irritation, stiffness related to immobilization, and the frequent need for secondary removal. Biological augmentation with hamstring tendon autograft has emerged as an alternative that avoids hardware-related morbidity while potentially improving tendon healing.
Hypothesis:
Hamstring tendon autograft augmentation would yield superior functional outcomes and knee motion without increasing complications or rerupture, and it would reduce reoperations by avoiding routine hardware removal compared with steel wire cerclage reinforcement for acute patellar tendon rupture.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A retrospective multicenter analysis across 5 sports medicine centers identified patients treated between February 2011 and May 2023 for isolated patellar tendon rupture or inferior pole patellar avulsion. A total of 81 patients met inclusion criteria. Patients underwent either hamstring tendon autograft augmentation (autograft augmentation group [AAG]; n = 43; semitendinosus ± gracilis through transosseous tunnels) or steel wire cerclage (synthetic augmentation group [SAG]; n = 38). The primary outcome was the Lysholm Knee Score. Secondary outcomes included the Knee injury and Osteoarthritis Outcome Score (KOOS) subscales, the University of California Los Angeles Activity Scale, the Forgotten Joint Score–12, knee range of motion, return to preinjury sport and work, complications, reoperations, and patellar height (Caton-Deschamps). Minimum follow-up was 24 months.
Results:
AAG demonstrated higher Lysholm and KOOS scores (most domains, P < .05), greater knee flexion (129.0° vs 119.7°; P < .01), and a higher return to preinjury sport (82.8% vs 54.5%; P = .03) than SAG. Complications were fewer with AAG than SAG (7.0% vs 28.9%; P = .02). Reoperations were markedly reduced (7.0% vs 71.1%l P < .01), largely because of planned hardware removals in SAG. Rerupture rates were low and similar (4.7% vs 0%; P = .50). Patellar height was preserved after AAG but decreased after SAG.
Conclusion:
In this multicenter cohort with ≥2-year follow-up, hamstring tendon autograft augmentation produced superior functional outcomes, greater flexion, higher return to sport, and fewer complications and reoperations compared with steel wire cerclage, without increasing rerupture risk. These findings support autograft augmentation as a preferable reinforcement strategy for active patients with acute patellar tendon rupture.
Patellar tendon rupture is uncommon (0.48-1.09/100,000 person-years) yet can markedly impair knee function.7,21,24,30 Injuries typically occur during sports involving eccentric quadriceps loading, with valgus stress, seen in sports such as basketball, volleyball, and football.10,23,28,33 Two main morphological patterns predominate: avulsion of the inferior pole of the patella and midsubstance tears involving the tendon body.17,18 Both disrupt the continuity of the extensor mechanism and require prompt surgical repair.7,27
Transosseous suture repair reinforced by steel cerclage remains the traditional gold standard technique.5,13,14 This technique affords immediate stability, yet often requires planned hardware removal and may be associated with postoperative stiffness due to prolonged immobilization during the early rehabilitation phase.3,14,29 Steel wire augmentation can also lead to patella baja, correlating with increased risk of knee flexion contracture and stiffness.13,19
Recently, biological augmentation with semitendinosus (± gracilis) tendon autograft has gained popularity.8,11,20 Cadaveric studies indicate strength comparable with steel wire cerclage while offering greater elasticity and avoiding hardware-related morbidity; early clinical series suggest improved outcomes with biologic augmentation. 14 However, comparative clinical evidence remains limited.
We compared steel wire cerclage reinforcement with hamstring tendon autograft augmentation for acute patellar tendon rupture in active patients. We hypothesized that autograft augmentation would yield superior functional outcomes and range of motion (ROM) without higher complications or rerupture and would reduce reoperations by avoiding routine hardware removal. The primary outcome was the Lysholm Knee Score. Secondary outcomes included the Knee injury and Osteoarthritis Outcome Score (KOOS) subscales, the University of California Los Angeles (UCLA) Activity Scale, the Forgotten Joint Score–12 (FJS-12), knee ROM, return to preinjury sport/work, complications, reoperations, and patellar height (Caton-Deschamps index).
Methods
Following institutional review board approval, a retrospective analysis of a prospectively maintained multicenter database from 5 sports medicine centers was conducted. A total of 145 consecutive patients operated between February 2011 and May 2023 for either an isolated rupture of the patellar tendon or an inferior pole patellar avulsion fracture were identified.
Eligibility Criteria
Inclusion criteria were acute patellar tendon rupture (<6 weeks from injury) in patients aged ≥18 years and complete patellar tendon rupture or inferior pole avulsion injury.
Exclusion criteria were age <18 years, total or unicompartmental knee arthroplasty, chronic ruptures (≥6 weeks), associated patellar fractures, follow-up <24 months, advanced neuromuscular disorders, previous surgery involving the patellar tendon, and any rerupture or contralateral rupture occurring between index surgery and final follow-up.
Patient Flow
Of 145 screened, 44 were excluded per predefined criteria, 14 were lost to follow-up, and 6 sustained a contralateral rupture before final assessment. Ultimately, a total of 81 patients were analyzed (Figure 1).

Patient flowchart. AIPP, avulsion of the inferior pole of the patella; PTR, patellar tendon rupture; TKA, total knee arthroplasty.
Preoperative Assessment
Demographics (age, sex, body mass index [BMI]) and systemic risk factors (smoking, obesity [BMI ≥30 kg/m2], diabetes) were recorded.
Ruptures were classified intraoperatively and radiographically as midsubstance tears or inferior patellar pole avulsions. Standardized lateral knee radiographs were obtained in all cases; in 74 of 81 patients (91.4%), the contralateral knee was used to assess baseline patellar height. Patients were allocated to the autograft augmentation group (AAG; n = 43; semitendinosus ± gracilis through transosseous tunnels) or to the synthetic augmentation group (SAG; n = 38; stainless steel wire looped around the tendon and fixed through patellar and tibial tunnels) based on the operating surgeon's preference. Surgical technique and rehabilitation protocol were not influenced by type of lesion.
Surgical Techniques
All procedures were performed by senior surgeons (C.J., R.P., M.O., V.G., H.P.) at the participating centers, using either the hamstring augmentation technique or stainless steel wire cerclage (Figures 2 and 3). The tendon suture technique (transosseous or Krackow) varied by surgeon.

Intraoperative view in which the gracilis and semitendinosus tendon grafts are prepared and whipstitched at their free end with nonabsorbable sutures.

Hamstring augmentation. (A) The ipsilateral semitendinosus and gracilis tendons are harvested.
Hamstring augmentation involved the ipsilateral semitendinosus (and occasionally gracilis) harvested with the tibial attachment preserved, transosseous tunnels in the tibial tubercle and patella, graft passage, and fixation with the knee at 30° flexion. Grafts were sutured to the native tendon and parapatellar tissues with absorbable sutures; no metal augmentation was used.
Stainless steel wire cerclage followed AO guidelines, 13 with primary repair (Krackow or transosseous) reinforced by an 18-gauge wire passed laterally along tendon borders and fixed between the patella and tibial tubercle at 30° flexion.
Rehabilitation Protocol
All patients generally followed the same staged rehabilitation program:
Phase 1 (0-3 weeks): full weightbearing in extension brace; passive ROM 0°-60°
Phase 2 (3-6 weeks): ROM increased to 0° to 90°; brace discontinued upon quadriceps control
Phase 3 (6-20 weeks): progressive active ROM to 120°
Phase 4 (>20 weeks): sport-specific rehabilitation and return to athletic activities
Follow-up and Outcome Measures
Lysholm Knee Score (0-100 points): this score is used for knee injuries, is designed to evaluate knee stability, function, and pain and is applicable after ligament or extensor mechanism injuries 6
KOOS (0-100 points): this patient-administered questionnaire assesses 5 subscales, including Pain, Symptoms, Activities of Daily Living (ADL), Sport and Recreation (Sport/Rec), and Knee-related Quality of Life (QOL) 26
UCLA Activity Scale (1-10 points): this scale measures the patient's activity level, particularly one's engagement in physical and sports pursuits, with higher scores reflecting greater activity 22
FJS-12 (0-100 points): this questionnaire measures the patient's ability to “forget” his or her operated joint in daily life and is a sensitive outcome tool for functional integration and satisfaction after knee procedures 2
Functional outcomes were collected at final follow-up by independent staff not involved in patient care. Assessors were blinded to the surgical technique whenever possible. ROM was measured using a goniometer, and return to sport/work and complications were recorded from follow-up visits and medical records.
Radiographic assessments were performed by a single independent observer. Blinding to treatment allocation was not feasible because steel wire hardware is visible in the SAG images. The Caton-Deschamps index was measured on standardized lateral radiographs in 30° of flexion. The index is defined as the ratio of the distance between the anterior tibial plateau angle and the inferior patellar articular surface to the length of the patellar articular surface and is considered normal between 0.6 and 1.3 9 (with lower values indicating patella baja and higher values indicating patella alta).
Statistical Analysis
Continuous variables were expressed as mean ± SD and compared using independent-samples t tests or 1-way analysis of variance, depending on variance homogeneity and normality of distribution (Shapiro-Wilk test). Categorical variables were reported as frequencies and percentages, with between-group comparisons performed using Fisher exact test or chi-square test, as appropriate. A P value < .05 was considered statistically significant. Statistical analyses were performed using JMP software. No a priori sample size calculation was performed because of the retrospective nature of this study.
Results
Baseline injury patterns and follow-up duration differed between groups. Mean follow-up was longer in the SAG (58.1 ± 33.9 vs 42.2 ± 27.0 months; P = .02). Patellar tendon ruptures were the most common injury type in both groups (72.1% in AAG vs 73.7% in SAG; P = .93), with remaining cases consisting of inferior pole patellar avulsions (Table 1).
Patients Characteristics a

Data are presented as mean ± SD (range) or n (%). Age, sex, and medical history refer to the patients, whereas the other variables correspond to the operated knees. Medical history refers to the patients’ comorbid conditions reported at the time of each patient's admission for surgery. Bold P value indicates statistical significance (P < .05). AAG, autograft augmentation group; AIPP, avulsion of inferior part of patella; BMI, body mass index; PTR, patellar tendon rupture; SAG, synthetic augmentation group.
The AAG had higher Lysholm scores than the steel wire cerclage group (84.8 ± 17.8 vs 74.1 ± 18.8; P < .01); KOOS domains were also higher in AAG (most domains, P < .05), whereas KOOS Symptoms and FJS-12 did not differ (Table 2; Figure 4). UCLA Activity scores were higher in AAG (8.2 ± 1.8 vs 7.2 ± 2.0; P = .03). Mean knee flexion was greater with AAG (129.0°± 15.3° vs 119.7°± 12.4°; P < .01).
Postoperative Outcomes a

Data are presented as mean ± SD, mean ± SD (range), or n (%), unless otherwise indicated. Sample sizes vary across variables because of missing data. AAG, autograft augmentation group; ADL, Activities of Daily Living; FJS-12, Forgotten Joint Score–12; KOOS, Knee injury and Osteoarthritis Outcome Score; QOL, Quality of Life; SAG, synthetic augmentation group; Sport/Rec, Sport and Recreation; UCLA University of California Los Angeles.

Functional outcomes comparing the autograft augmentation group (AAG) and the synthetic augmentation group (SAG) at final follow-up. AAG, autograft augmentation group; ADL, Activities of Daily Living; FJS-12, Forgotten Joint Score–12; KOOS, Knee injury and Osteoarthritis Outcome Score; QOL, Quality of Life; SAG, synthetic augmentation group; Sport/Rec, Sport and Recreation; UCLA University of California Los Angeles.
There was no statistically significant difference in time to return to sport between groups (8.5 ± 5.8 vs 10.6 ± 6.3 months; P = .26) but a larger proportion returned to their preinjury level (82.8% vs 54.5%; P = .03). Return-to-work times were similar (5.3 ± 2.7 vs 5.5 ± 2.6 months; P = .74).
Radiographically, patellar height was assessed using the Caton-Deschamps index. At final follow-up, the operated knee did not differ from the contralateral knee in the AAG (1.05 ± 0.36 vs 1.06 ± 0.18; P = .98), whereas the SAG showed a lower index on the operated side (0.93 ± 0.24 vs 1.05 ± 0.18; P < .01).
The overall reoperation rate was lower in the AAG (7.0% vs 71.1%; P < .01), driven predominantly by planned hardware removal in SAG. Rates of unplanned reoperation were not significantly different (7.0% vs 21.1%; P = .20). In the synthetic group, hardware removal occurred at a mean of 7.4 ± 5.6 months (1.9-21.9 months).
Postoperative complications occurred in 7.0% (3/43) of patients in AAG vs 28.9% (11/38) for the SAG (P = .02). Fixed flexion deformity ≥10° was more common in the SAG (P = .02). Severe stiffness (<90° flexion) was similar (2.3% vs 7.9%; P = .34), as was rerupture (4.7% vs 0%; P = .50) (Table 3).
Reoperations and Postoperative Complications a
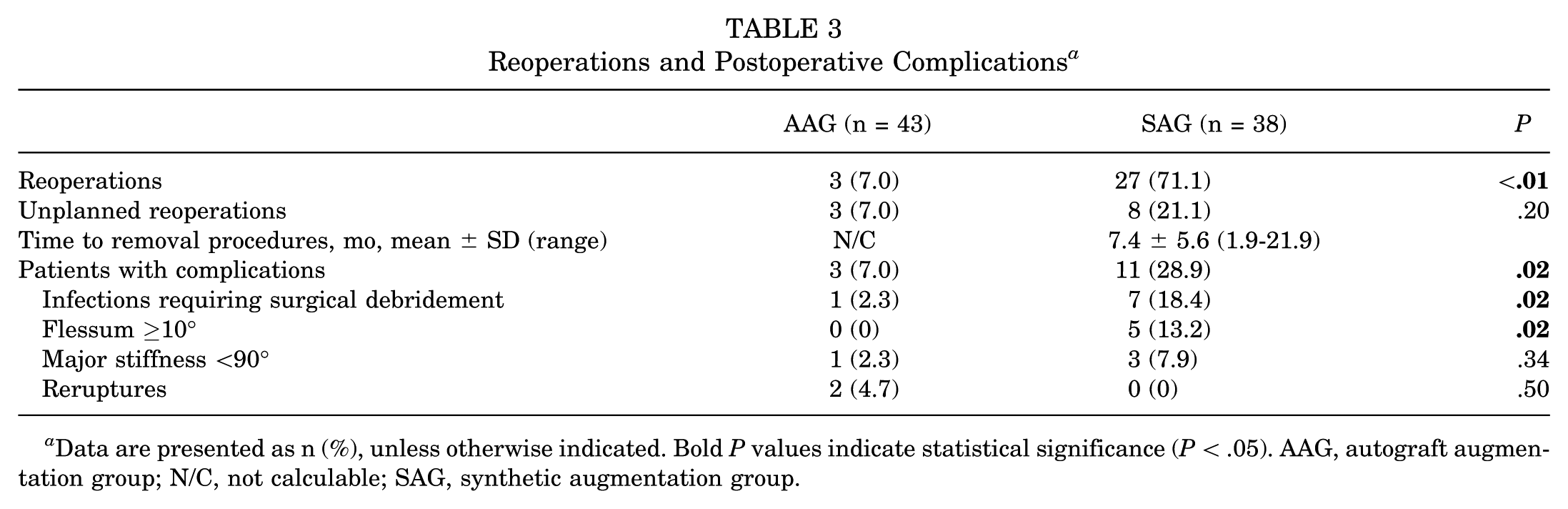
Data are presented as n (%), unless otherwise indicated. Bold P values indicate statistical significance (P < .05). AAG, autograft augmentation group; N/C, not calculable; SAG, synthetic augmentation group.
Discussion
This study evaluated between-group differences in clinical outcomes, return to sport, and complication rates between autograft augmentation and synthetic cerclage. The results show statistically significant superiority of the autograft augmentation technique across multiple key clinical parameters, supporting the initial hypothesis. Functional outcomes (Lysholm, KOOS, UCLA) were significantly improved without higher complication or rerupture rates. In addition, the need for secondary surgery was reduced by avoiding hardware removal.
Black et al 4 conducted a cadaveric study to evaluate the mechanical strength of patellar tendon repairs, highlighting the biomechanical benefits of augmentation: resistance to gap formation was improved and ultimate load to failure increased. These findings support the augmentation principle, 15 with augmented repairs demonstrating superior biomechanical performance to simple repairs, with notably reduced gap formation and improved structural integrity under cyclic loading.
However, metallic augmentation techniques carry specific risks, including hardware failure, soft tissue irritation, and the need for secondary removal.19,29 Our findings corroborate this, with a 71.1% reoperation rate in the SAG primarily related to scheduled hardware removal procedures.
The use of hamstring tendon autografts has emerged, applying the same biologic principles used in knee ligament reconstruction8,11,18,27 This technique employs autologous tissue, avoids complications related to foreign material, and provides robust mechanical support. Valianatos et al 31 reported excellent outcomes with this approach, with a mean Lysholm score of 93, aligning with our AAG results.
Patients who underwent biological reconstruction showed higher KOOS and Lysholm scores, with differences considered clinically important. The Lysholm score, considered the gold standard for knee function, was >10 points higher in the biological group (84.8 vs 74.1; P < .01). Using established minimal clinically important difference (MCID) thresholds (10 points for Lysholm; 8-10 points for KOOS subscales; 10-14 points for FJS-12; and 8°-10° for knee flexion), the differences observed for Lysholm, KOOS Pain, KOOS QOL, and knee flexion exceeded these thresholds, while KOOS ADL, KOOS Sport/Rec, and FJS-12 approached them. KOOS Symptoms did not reach the MCID.1,2,6,25
Return to sport is a key goal in the young and active population; we observed a significantly higher rate of return to preinjury level with hamstring augmentation (82.8% vs 54.5%; P = .03). These results are consistent with previous data, such as Fredericks et al, 12 who show the importance of surgical technique in optimizing return-to-sport outcomes after patellar tendon rupture. 16
Radiographic analysis using the Caton-Deschamps index showed a significant patellar height reduction with the SAG technique (1.05 ± 0.18 vs 0.93 ± 0.24; P < .01), indicating iatrogenic shortening. In contrast, patellar height was preserved with the autograft augmentation technique (1.06 ± 0.18 vs 1.05 ± 0.36; P = .98). The literature also suggests an increased risk of patella baja with metallic cerclage. Kuo et al 19 reported patella baja in 5 out of 20 patients in the augmentation group versus none in the tendon-only group; this difference did not reach statistical significance.
Complication rates differed between groups, with 3/43 (7.0%) in AAG versus 11/38 (28.9%) in SAG (P = .02). ROM was significantly greater in the AAG (129.0° vs 119.7°; P < .01), reflecting improved joint mobility. This ~10° difference is clinically important because a flexion beyond 120° is generally needed for many daily and sporting activities.
The reoperation rate was also higher in the SAG (71.1% vs 7.0%; P < .01), mainly due to hardware removal to address stiffness, implant-related discomfort, and infections.
In this study, far more hardware removal procedures were observed than reported by Kuo et al, 19 who noted a 20% hardware removal rate after wire augmentation. This difference can be explained by different surgical habits or health care systems, particularly the accessibility and cost of reoperations. Nevertheless, autograft reconstruction also involves specific morbidity, notably a longer surgical incision and the need for hamstring tendon harvesting
Two reruptures occurred in the AAG (<5%) versus none in the SAG (P = .50). This aligns with the literature, which reports variable rerupture (2% to 50%) across both hamstring and synthetic augmentation methods.14,32
A key strength of this study is the 2-year minimum follow-up, providing a solid view of midterm outcomes after healing and rehabilitation. In addition, standardized assessments during a dedicated follow-up visit helped minimize potential bias. The multicenter design supports generalizability, and this cohort (N = 81) represents the largest comparative series of these 2 techniques to date.
Limitations
Limitations include the retrospective, nonrandomized design, which may introduce selection bias, although the groups were demographically comparable. Multiple surgeons were involved, but independent standardized assessments may mitigate this effect. In spite of some variability in suture techniques across surgeons, this remains a minor limitation. Differences in rehabilitation protocols, while following the same principles (full weightbearing and immediate flexion), may represent a confounder. Moreover, the choice of technique depended on the operating surgeon, introducing a selection bias. Finally, follow-up duration differed between groups (AAG vs SAG: 42.2 ± 27.0 vs 58.1 ± 33.9 months, respectively), reflecting the more recent adoption of biologic augmentation, though outcomes appear stable beyond the second postoperative year. Future studies should be prospective and randomized, include longer follow-up, and assess cost-effectiveness, including reoperation costs.
Conclusion
In this multicenter cohort with ≥2-year follow-up, hamstring tendon autograft augmentation produced superior functional outcomes, greater flexion, higher return to sport, and fewer complications and reoperations compared with steel wire cerclage, without increasing rerupture risk. These findings support autograft augmentation as a preferable reinforcement strategy for active patients with acute patellar tendon rupture.
Footnotes
Final revision submitted January 18, 2026; accepted January 27, 2026.
One or more of the authors has declared the following potential conflict of interest or source of funding: M.O. is an educational consultant for New-Clip, Stryker, and Arthrex. J.-N.A. receives royalties from Zimmer. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the local ethics committee (No. PADS23-173).
