Abstract
Purpose:
To compare braided and nonbraided hamstring tendon grafts in anterior cruciate ligament (ACL) reconstruction, specifically examining graft thickness, rerupture rates, and clinical outcomes, in order to assess whether braided grafts offer measurable advantages.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
This retrospective study included 154 patients who underwent single-bundle ACL reconstruction between January 2017 and October 2022. Patients were divided into braided (n = 78) and nonbraided (n = 76) hamstring autograft groups. Graft diameter was measured intraoperatively at the midportion using calibrated sizing tubes after standardized preconditioning. The cohort was predominantly male (92.2%). Primary outcomes included graft diameter, rerupture rates, and Knee injury and Osteoarthritis Outcome Score (KOOS), International Knee Documentation Committee (IKDC) score, and Lysholm score at baseline and 6, 12, and 24 months. Comparative analysis used the Mann-Whitney U test for continuous data and Fisher exact test for categorical data. Kaplan-Meier curves were used descriptively to illustrate rerupture-free survival.
Results:
Braided grafts showed a greater mean diameter (9.39 ± 0.63 mm) compared with nonbraided grafts (8.87 ± 0.74 mm) (P < .001) and a numerically lower rerupture rate (2/78 [2.6%] vs 6/76 [7.9%]); however, this difference did not reach statistical significance (Fisher exact test, P = .16; fragility index = 1). Functional outcomes (KOOS, IKDC, Lysholm) improved over time in both groups, with no statistically significant between-group differences at 2 years.
Conclusion:
Braided hamstring tendon grafts reliably achieved increased graft thickness, whereas the lower rerupture rate was numerical only, was not statistically significant, and was statistically fragile. Braiding can be considered an intraoperative option to optimize graft diameter without compromising clinical outcomes, but the findings should be interpreted as exploratory.
Keywords
Hamstring tendon grafts are among the most commonly used options for anterior cruciate ligament (ACL) reconstruction, favored for their biomechanical properties and lower risk of donor site morbidity compared with patellar and quadriceps tendon grafts.2,31,35 However, recent meta-analyses have shown that quadriceps tendon autografts provide at least comparable clinical outcomes and, in some series, lower rerupture rates than hamstring grafts, particularly in active populations.3,4,7,16,20,31,35
Graft diameter is a critical factor in ACL reconstruction success, with evidence suggesting that thinner grafts are associated with elevated rerupture rates, particularly among younger and more active populations.9,28,34 Although the minimum acceptable graft diameter for optimal durability is still debated, achieving an adequate thickness is widely recognized as essential to minimizing failure rates, as graft failure can lead to compromised knee stability, the potential need for revision surgery, and reduced patient quality of life.1,10,19 Although various preoperative strategies to estimate hamstring graft size have been described in the literature, preoperative graft sizing is not universally practiced in routine clinical settings. Consequently, intraoperative decision-making becomes critical when the harvested graft diameter is smaller than desired. In such cases, surgeons may need to modify their plan by augmenting the graft, switching to an alternative graft source, or harvesting from the contralateral limb.12,18,21 These options, however, come with potential morbidity risks, increased operative time, and possible complications at donor sites. Given these limitations, techniques that increase graft thickness without requiring additional harvesting have been explored.5,6,14,33
One such technique is braiding, which increases the apparent graft diameter but does not increase the collagen cross-sectional area.21,23 The apparent diameter gain primarily reflects fiber reorientation and interfascicular spacing rather than an increase in load-bearing tissue. Biomechanical studies on human hamstring tendons have shown that twisting or braiding reduces linear tensile strength and stiffness, as the oblique fiber alignment converts axial load into shear forces.15,19,22 These findings indicate that braiding does not confer a proven mechanical advantage. Although braiding may allow surgeons to obtain a larger appearing graft when intraoperative diameter is insufficient, its biomechanical implications remain uncertain, and clinical effects should be interpreted cautiously.25,26
This lack of clinical evidence limits surgeons’ ability to make informed decisions regarding graft selection based on both biomechanical advantages and patient-specific outcomes. Therefore, the purpose of this study was to compare graft diameters, functional outcomes, and rerupture rates between braided and nonbraided hamstring tendon autografts in ACL reconstruction over a 2-year follow-up period. We hypothesized that braided hamstring grafts, by achieving greater apparent diameter, would be associated with at least comparable functional outcomes and a possible reduction in rerupture rates, although any biomechanical advantage of braiding remains unproven. 27
Methods
Study Design and Participants
This study was approved by the institutional review board of Kutahya Health Sciences University (Approval No. 2025/01-22; January 13, 2025) and was conducted in accordance with the principles of the Declaration of Helsinki. The study followed the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for reporting observational research.
Patients were consecutively recruited between January 2017 and October 2022. A total of 154 patients who underwent primary ACL reconstruction using either braided hamstring tendon autografts or conventional nonbraided 4-strand hamstring tendon autografts were initially identified. The final study population was determined based on the recruitment period and inclusion criteria, without the use of an a priori power analysis; however, a post hoc power analysis was performed and is detailed in the statistical analysis section. The cohort reflects our institution's patient demographic characteristics, which predominantly include young, male, competitive athletes undergoing ACL reconstruction.
Inclusion criteria were as follows:
Age between 18 and 45 years
Primary ACL reconstruction with semitendinosus and gracilis autograft
Minimum postoperative follow-up of 24 months
Exclusion criteria included the following:
History of previous surgery on the ipsilateral knee
Multiligamentous knee injuries
Revision ACL surgery
Loss to follow-up
Allocation to the braided or nonbraided group was not randomized. When intraoperative measurement of the harvested hamstring tendons in the standard 4-strand configuration showed a diameter <8.5 mm, the operating surgeon elected to perform braiding to augment graft thickness; otherwise, the standard nonbraided configuration was used.
Surgical Technique
All procedures were performed by a single fellowship-trained sports traumatology and arthroscopy surgeon (N.D.D.) using a standard anatomic single-bundle ACL reconstruction technique. Through an oblique 2.5-cm incision medial to the tibial tubercle, the semitendinosus and gracilis tendons were harvested with an open tendon stripper and cleared of residual muscle tissue.
Braided Graft Preparation
The tendons were mounted onto a cortical suspension device (CorticalButton; Aleza) and manually braided using alternating over-under passes, following the method described by Samitier and Vinagre.25,26 The distal ends were secured with Vicryl No. 2 sutures (Ethicon; Johnson & Johnson). Grafts were stored in a rifamycin-soaked sterile sponge mesh (Rif; 250 mg/3 mL; Koçak Farma) (Figure 1). Graft diameter was measured at the midportion using sterile calibrated sizing tubes.
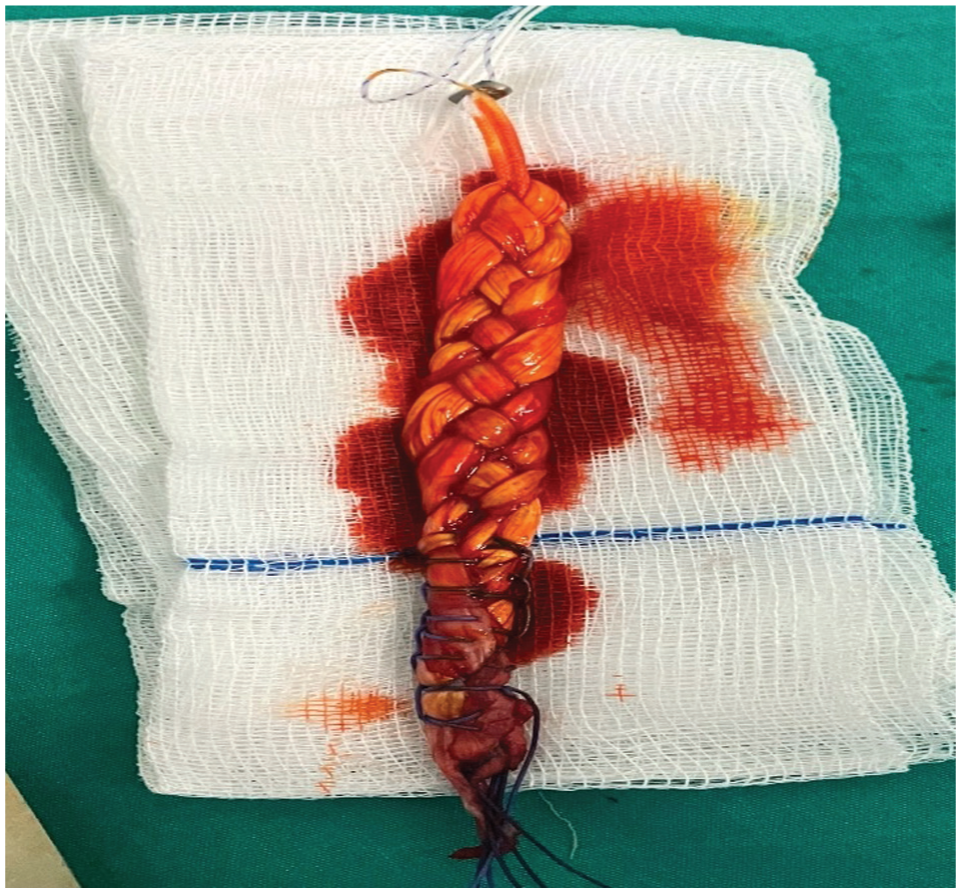
Four-strand braided hamstring graft. Semitendinosus and gracilis tendons were mounted on a cortical suspension device (CorticalButton; Aleza) in a 4-strand braided configuration, secured with Vicryl No. 2 sutures, and stored in rifamycin-soaked sterile sponge mesh.
Nonbraided Graft Preparation
Tendons were folded over the same suspension device into a standard 4-strand configuration and sutured similarly. The same storage method was used.
In both groups, anatomic femoral and tibial tunnels were drilled using transportal and tibial aimer techniques, respectively (Figure 2). Tibial fixation was identical in both groups and consisted of a bioabsorbable interference screw (BioSure; Smith & Nephew) and a staple.
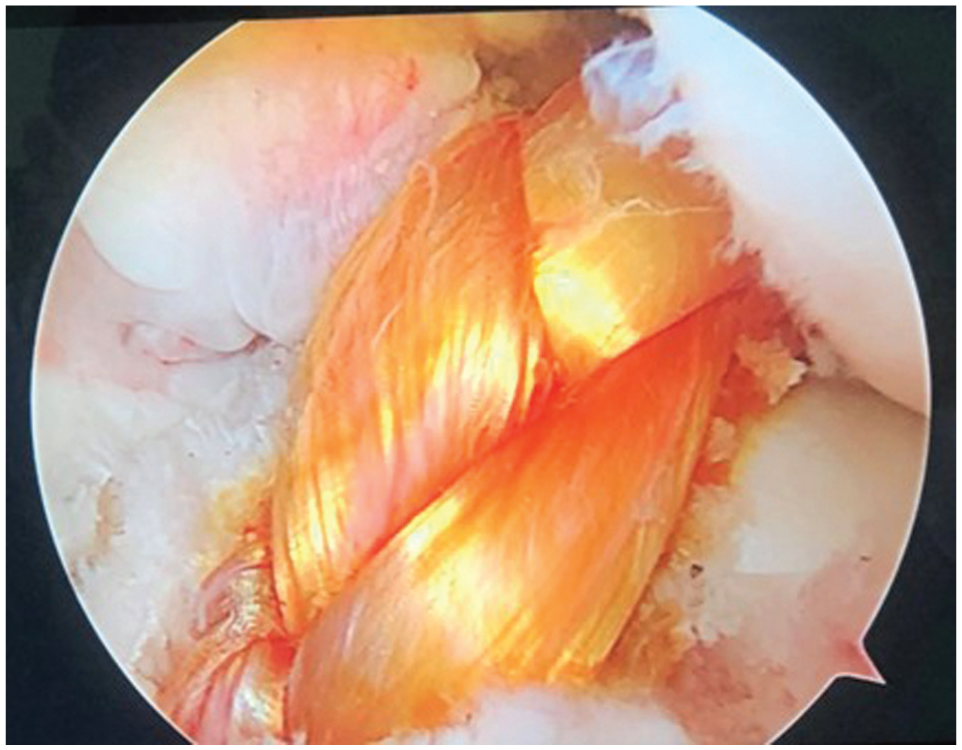
Arthroscopic view of the braided hamstring tendon graft in situ during anterior cruciate ligament reconstruction.
Femoral tunnels were drilled through the anteromedial portal using a transportal guide at the native femoral footprint, and tibial tunnels were created with a tibial aimer at 55°. Tunnel diameter was matched to the measured graft diameter using calibrated sizing tubes (1-mm increments). Measurements were read aloud by the assistant surgeon and recorded on a standardized case report form by a circulating nurse; the operating surgeon did not complete the study forms to reduce observer bias. The graft was tensioned with the knee at 20° of flexion after cycling and fixed with the cortical button on the femoral side and a bioabsorbable interference screw (BioSure; Smith & Nephew) plus a staple on the tibial side.
Postoperative Rehabilitation
All patients followed the same standardized, criterion-based rehabilitation program supervised by the same physiotherapy team, without any graft-specific modifications:
0-2 weeks: knee brace in full extension with partial weightbearing
2-6 weeks: gradual range of motion (ROM) up to 120°, full weightbearing
6-12 weeks: progressive strengthening
3-6 months: jogging permitted
6-9 months: return to pivoting sports if quadriceps strength >90% of contralateral side
Clinical Evaluation and Follow-up
Outcomes were evaluated at baseline and at 15 days and 1, 3, 6, 12, and 24 months postoperatively. Evaluators were 2 independent, fellowship-trained orthopaedic research assistants who were not involved in the surgical procedures and were blinded to group allocation. Patient-reported outcome measures (PROMs) included the following:
Subjective International Knee Documentation Committee (IKDC) score 33
Knee injury and Osteoarthritis Outcome Score (KOOS) 6
Lysholm Knee Score 14
PROMs were administered face-to-face at clinic visits. Graft reruptures were diagnosed based on clinical instability, positive Lachman test, and confirmation via magnetic resonance imaging (MRI). Suspected reruptures were confirmed with 3.0-T knee MRI using proton density–weighted, fat-suppressed sequences in sagittal, coronal, and axial planes, interpreted by a musculoskeletal radiologist blinded to graft type. Additional variables recorded included age, sex (assigned at birth), graft thickness, and graft type. PROMs (KOOS, IKDC, Lysholm) were collected through standardized in-office clinical visits at baseline, 6 months, 1 year, and 2 years. No telephone surveys were used. Data entry and compilation were performed by 2 independent research coordinators, each with >5 years of experience in prospective orthopaedic outcomes research and certified in clinical research data management. Both coordinators were blinded to the graft allocation during data collection and entry.
Statistical Analysis
Descriptive data were presented as mean ± standard deviation for continuous variables and frequencies (n, %) for categorical variables. Continuous variables were compared using the Mann-Whitney U test. Categorical variables were analyzed using Fisher exact test. Kaplan-Meier survival analysis was used to illustrate rerupture-free survival. Given the limited number of events (n = 8), log-rank comparisons and multivariable logistic regression analyses (including graft preparation method, age, sex as recommended by the Sex and Gender Equity in Research [SAGER] guidelines, and graft diameter) were performed as exploratory only and are not interpreted as confirmatory. A post hoc power analysis was conducted using graft rerupture as the primary endpoint. With an alpha of .05 and the observed event rates (2/78 vs 6/76), the achieved statistical power was <80%, indicating that the study was likely underpowered to detect small to moderate between-group differences.
Results
A total of 154 patients were included in the study, with 78 in the braided group (73 male and 5 female) and 76 in the nonbraided group (69 male and 7 female). The mean age was 29.92 ± 7.72 years for the braided group and 29.97 ± 7.45 years for the nonbraided group. Table 1 provides a summary of baseline demographic characteristics, including age, sex distribution, and preoperative functional scores (IKDC, KOOS, Lysholm).
Baseline Demographic Characteristics and Preoperative Scores a

Values are expressed as mean ± SD unless otherwise noted. Boldface indicates statistical significance. IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score.
Fisher exact test (2-sided) for sex. All other P values from Mann-Whitney U test (2-sided).
Mean graft thickness was significantly greater in the braided group, with a mean of 9.39 ± 0.63 mm compared with 8.87 ± 0.74 mm in the nonbraided group (P < .001). All braided grafts achieved a final diameter of at least 8.0 mm; none remained below this threshold. Rerupture rates also differed numerically between the groups, with 2 of 78 (2.6%) reruptures in the braided group and 6 of 76 (7.9%) in the nonbraided group; however, this difference was not statistically significant (2-sided Fisher exact test, P = .16) and was statistically fragile (fragility index = 1). Figure 3A illustrates graft thickness stratified by sex and graft type: Men with braided grafts had the greatest mean diameter, whereas women with nonbraided grafts had the smallest mean diameter, but the overlapping 95% CIs indicated that sex-specific differences should be interpreted cautiously. Figure 3B shows rerupture rates for braided and nonbraided grafts in men and women. Rerupture was numerically lower in men with braided grafts (2/73; 2.7%) than in men with nonbraided grafts (5/69; 7.2%), and no reruptures occurred among women with braided grafts (0/5) compared with 1 in 7 (14.3%) in women with nonbraided grafts.

Graft thickness (mean and 95% CI) and rerupture rate stratified by sex and graft type (braided vs nonbraided). Error bars represent the 95% CI calculated from subgroup-specific standard deviations. Raw rerupture counts (events/total) are displayed above each bar.
Kaplan-Meier survival curves illustrated numerically higher rerupture-free survival in the braided group over the 2-year follow-up (24-month rerupture-free survival, 97.44% vs 92.11%). Given the low number of events (n = 8), these survival analyses are presented descriptively, and log-rank comparisons were considered exploratory and are not interpreted as confirmatory (Figure 4).

Kaplan-Meier survival curves, with rerupture-free survival rates at 24 months of 97.44% for the braided group (blue) and 92.11% for the nonbraided group (yellow).
Functional outcomes (KOOS, IKDC, and Lysholm scores) were assessed at baseline, 6 months, 1 year, and 2 years. Both groups showed improvement over time, with the braided group achieving slightly higher scores, although these differences were not statistically significant between groups (P > .05). At the final 24-month assessment, KOOS values were 85.3 ± 5.9 in the braided group and 83.1 ± 6.2 in the nonbraided group; IKDC scores were 79.5 ± 8.4 (braided) vs 77.2 ± 8.7 (nonbraided); and Lysholm scores were 84.6 ± 6.1 (braided) vs 82.4 ± 6.5 (nonbraided) (Figure 5).
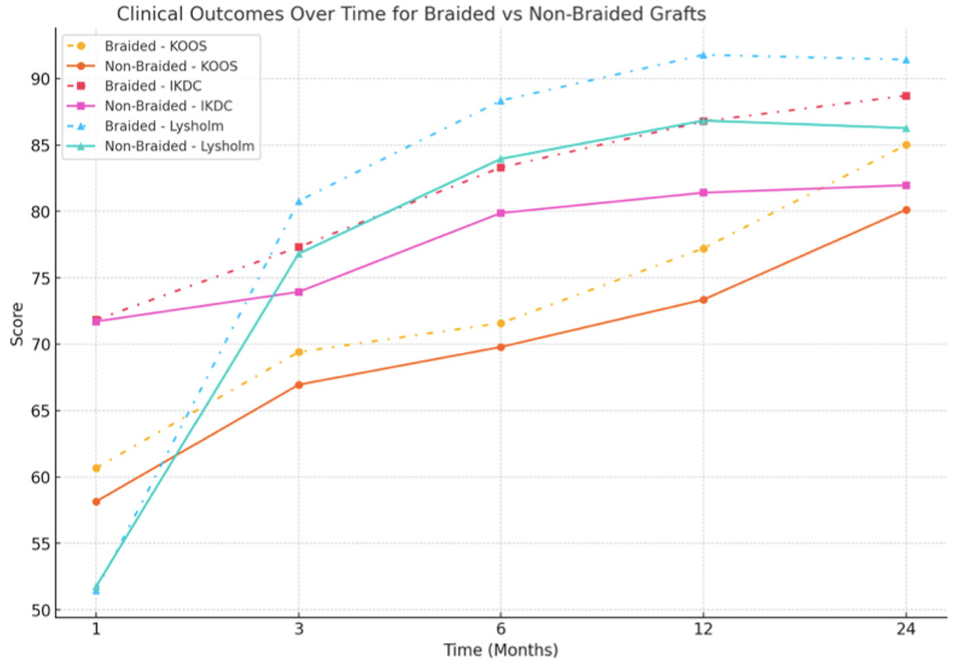
Comparison of clinical outcomes over time for patients in braided and nonbraided graft groups. IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score.
Logistic regression analysis indicated that graft thickness was a significant predictor of rerupture risk, with thicker grafts associated with a lower risk of failure (coefficient = −1.93; P = .009). Kaplan-Meier curves stratified by graft thickness are presented in Figure 6 and are shown descriptively given the low number of rerupture events. However, graft type (braided vs nonbraided) alone was not a significant predictor when controlling for age, sex, and graft diameter (P = .816). Sex and age were also not significantly associated with rerupture risk (P > .05).
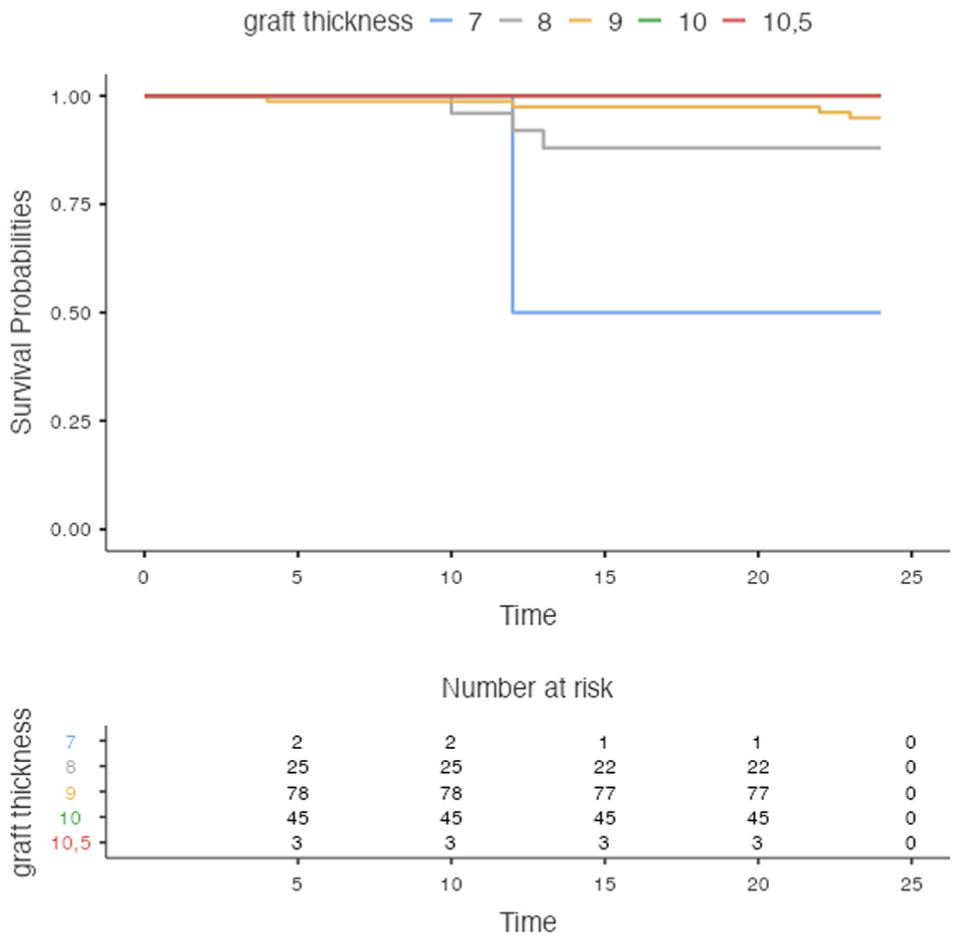
Survival curves, with rerupture-free survival rates at 24 months of different graft thicknesses.
The mean operative time was 67.5 ± 11.2 minutes in the braided group and 64.9 ± 10.7 minutes in the nonbraided group (P = .184), indicating no statistically significant difference.
Discussion
The most consistent finding of this study was the larger graft diameter obtained with braided constructs. Although rerupture rates were numerically lower, the difference was not statistically significant and demonstrated high fragility. This increase in graft thickness may be clinically relevant for surgeons when confronted with small-diameter hamstring tendons; however, any effect on graft strength, stability, or rerupture risk remains speculative. The rerupture difference was small in absolute terms, was statistically nonsignificant (P = .16), and had a fragility index of 1, indicating that the comparison is unstable.
Previous studies have underscored graft thickness as a key determinant of biomechanical strength and long-term stability. For example, studies by Spragg et al 29 and Figueroa et al 13 established that ACL grafts with diameters <8 mm carried a significantly higher failure risk, whereas even minor increases in thickness, such as 0.5 mm, could enhance graft durability and longevity. In our study, braided grafts consistently achieved diameters exceeding this threshold, providing additional support for the hypothesis that thicker grafts offer added durability.
Braiding increases the apparent diameter of the graft but does not increase collagen cross-sectional area. Human hamstring studies by Kim et al 17 , and Tis et al 32 showed that twisting and braiding reduced linear tensile strength and stiffness, likely due to fiber reorientation and increased inter-fascicular spacing. These biomechanical limitations highlight that a larger braided diameter does not imply mechanical superiority. Our clinical findings therefore should not be interpreted as evidence of a biomechanical advantage but rather as preliminary observations that braided constructs did not demonstrate short-term clinical harm in this cohort.
A key advantage of braided grafts is the reduction in additional tendon harvesting, especially in patients with smaller tendons, such as young athletes and women. Traditional solutions to insufficient graft thickness, such as additional harvesting or allograft use, can lead to increased donor site morbidity, extended recovery time, and potential immunologic complications.8,12,18,30,33 Our findings suggest that braiding offers a practical alternative, achieving adequate graft thickness without additional harvesting. Park et al 24 found similar benefits with braiding techniques, observing an enhanced graft diameter despite minor reductions in length. This capability to optimize graft thickness without extra harvesting holds significant promise for minimizing surgical impact.
Despite the larger apparent diameter of braided constructs, our study found no statistically significant differences between braided and nonbraided groups in functional outcomes (KOOS, IKDC, Lysholm scores) over the 2-year follow-up period. These findings indicate that our data do not demonstrate clinical biomechanical superiority of braided grafts, but they also do not suggest short-term functional harm associated with their use. Our findings are consistent with previous research suggesting that although graft diameter contributes to reconstruction success, a combination of factors such as surgical technique, rehabilitation adherence, and graft integration also influence outcomes. In a related study, Duerr et al 11 reported that a 1-mm increase in graft diameter was associated with significantly improved KOOS results at 2 years. However, our comparison of KOOS, Lysholm, and IKDC scores revealed no significant differences between braided and nonbraided groups despite improvements in graft thickness, suggesting that factors beyond thickness alone contribute to clinical success. An additional observation was that the proportion of women in the braided group was lower than expected, despite women typically having smaller hamstring tendons. This discrepancy is explained by the very small number of female patients in the overall cohort and the natural variability in tendon dimensions; several female patients had 4-strand diameters ≥8.5 mm and therefore did not meet the intraoperative threshold for braiding. Accordingly, this does not reflect a biological pattern but rather the demographic composition and diameter variability within our sample.
Limitations
This study has several limitations. First, its retrospective, single-center design introduces inherent risks of selection and information bias. Moreover, prebraiding diameters were not systematically recorded, which prevented us from quantifying the exact increase in graft diameter attributable to braiding in individual cases. Second, graft allocation was nonrandomized, as braiding was preferentially performed in cases with smaller intraoperative diameters; therefore, confounding by indication cannot be excluded. Third, the number of rerupture events was low (n = 8), yielding a fragility index of 1, which indicates that the findings regarding rerupture are statistically fragile. Consequently, the Kaplan-Meier analyses and multivariable logistic regression should be interpreted as exploratory rather than confirmatory, as the small event count limits model stability. Fourth, detailed data on concomitant meniscal or chondral injuries, return-to-sport exposure, and the specific mode of reinjury were not systematically collected, which prevents adjustment for these potentially relevant confounders. Baseline height, body mass index, and activity level (eg, Tegner scores) were not systematically collected for all patients and therefore could not be included in the comparative analyses. Fifth, the cohort was predominantly male (approximately 92%), limiting the generalizability of the results, particularly for female patients.
Conclusion
Braided hamstring tendon grafts used in ACL reconstruction achieved a significantly greater mean diameter compared with nonbraided grafts (9.39 ± 0.63 vs 8.87 ± 0.74 mm; P < .001). Rerupture rates were numerically lower in the braided group (2.6% vs 7.9%), but this difference was not statistically significant (P = .16) and was statistically fragile. Functional outcomes, as measured by KOOS, IKDC, and Lysholm scores, improved similarly in both groups over the 2-year follow-up, with no statistically significant differences. Taken together, these findings suggest that braided grafts are a practical intraoperative option for increasing hamstring graft diameter without compromising short-term clinical outcomes, particularly in patients with small tendons; however, any potential benefit regarding graft survival remains speculative and should be confirmed in prospective, adequately powered randomized studies.
Footnotes
Final revision submitted December 29, 2025; accepted January 4, 2026.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Ethics Committee of Kütahya Health Sciences University (Decision No. 2025/01-22; January 13, 2025).
