Abstract
Background:
Hamstring autograft (HA) is a popular graft for anterior cruciate ligament reconstruction (ACLR). Although multiple studies have demonstrated that increasing the graft diameter decreases the risk of failure or revision surgery, the exact minimum graft diameter remains controversial, with the unofficial standard being 8 mm.
Purpose:
To evaluate the risk of aseptic revision after HA-ACLR in patients aged ≤25 years, comparing graft diameters of (1) <8 versus ≥8 mm and (2) ≤8 versus >8 mm. A secondary purpose was to determine whether there was a threshold for graft diameter that best identifies patients at a higher risk of aseptic revision.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A total of 5972 primary HA-ACLRs were identified using data from the ACLR registry of Kaiser Permanente. Propensity score–weighted Cox regression was used to evaluate revision risk for HAs with <8 versus ≥8 mm diameter and with ≤8 versus >8 mm diameter. An effect plot and receiver operating characteristic curve and area under the curve (AUC) analysis were also created to model the probability of revision based on the HA diameter to determine whether there was a specific diameter threshold to minimize revision risk.
Results:
In the propensity score–weighted Cox model, <8 mm autografts had a higher risk of aseptic revision compared with ≥8 mm autografts (hazard ratio [HR], 1.32 [95% CI, 1.01-1.71]; P = .039); a higher risk was also observed when comparing ≤8 to >8 mm grafts (HR, 1.32 [95% CI, 1.06-1.66]; P = .015). In receiver operating characteristic analysis, an HA diameter that best identified patients at a higher risk for revision after ACLR could not be identified (AUC, 0.56). However, the adjusted effect plot showed a direct decrease in revision risk with each increasing increment in graft size.
Conclusion:
A 32% higher risk of revision was observed in smaller graft diameter groups (<8 or ≤8 mm) compared with larger graft sizes (≥8 or >8 mm), regardless of specific diameter cutoff. In a cohort of almost 6000 HA-ACLR, the authors were unable to determine a definitive minimum graft diameter threshold that should be used.
Keywords
The anterior cruciate ligament is the main stabilizer of the knee joint and requires reconstruction in young and high-demand patients to stabilize the knee, prevent meniscal and cartilage injury, and allow for a return to cutting and pivoting sports. 10 Several graft choices exist, with the bone-patellar tendon-bone autograft being the gold standard, while the hamstring autograft (HA) has gained popularity because of improved soft tissue fixation techniques and lower donor site morbidity.1,16,32
Several factors have been identified as predictors of failure and revision after anterior cruciate ligament reconstruction (ACLR) using the HA, with one of them being graft diameter.2,20,34,37 Biomechanical studies have shown increased tensile strength with increasing graft diameter,5,30 while clinical studies 34 have reported that for each 0.5-mm increase in the HA diameter, the risk of revision is lowered by 18% (between 7 and 9 mm) to 33 14% (between 7 and 10 mm). A meta-analysis of 19,333 ACLRs by Itoh et al 14 also confirmed a significant reduction in the failure rate for each 0.5-mm increase in the HA diameter when going from <7 (failure rate, 3.6%) to >9 mm (failure rate, 1.8%).
Despite studies finding a lower risk of revision surgery with increasing HA graft diameter, the minimum acceptable diameter remains controversial. The unofficial minimum graft diameter is 8 mm.8,20 However, some studies have reported a diameter >8 mm as the threshold,20,21 while others have stated that ≥8 mm8,25,32 or ≥7 mm2,15 are also acceptable. The inconsistency in reporting is likely due to variation across studies in terms of study cohorts and sample sizes, lack of standardization in outcome definitions, and different statistical analyses, thus making it difficult to interpret findings across studies and allow clinicians to determine a specific minimum HA diameter for primary ACLR.
Therefore, using data from a United States (US)–based health care system, we sought to evaluate using 8 mm as a minimum HA diameter and aseptic revision risk in a cohort of patients with a minimum age of 25 years. Two comparisons were made: (1) <8 versus ≥8 mm and (2) ≤8 versus >8 mm. A secondary purpose was to determine whether there was a threshold for graft diameter that best identifies patients who are at a higher risk of aseptic revision after ACLR.
Methods
Study Design, Setting, and Data Source
We conducted a retrospective cohort study using data from the Kaiser Permanente ACLR registry. This integrated health care system covers over 12 million people throughout 8 geographical US regions—including Colorado, Georgia, Hawaii, the mid-Atlantic, Northern California, the Northwest, Southern California, and Washington. Health care plan membership has previously been shown to be demographically representative of the geographical areas that it covers. 9 The protocol for this study was approved by our organization’s institutional review board.
A summary of details on the data collection procedures, coverage, and participation rates for the Kaiser Permanente ACLR registry has been published previously.26,27 Briefly, patient, procedure, implant, surgeon, and hospital information for all ACLR procedures performed within our health care system is collected into this surveillance tool using electronic intraoperative forms that are completed at the point-of-care by the operating surgeon. Information is then supplemented using data from the electronic health record (EHR), administrative claims data, membership data, and mortality records. Outcomes, such as revisions, are prospectively monitored using electronic screening algorithms and validated by trained clinical content experts using the EHR.
Study Sample
The study population included patients who underwent a primary isolated ACLR between January 1, 2005, and December 31, 2020. The population was restricted to patients aged ≤25 years and the use of HA at the index ACLR. Patients were excluded if they had a previous procedure in the same knee, underwent a double-bundle procedure, had a reported infection, or were missing information on graft diameter size. The final study sample included 5972 ACLRs performed by 172 surgeons at 41 health care centers (Figure 1).

Flowchart of patient inclusion in the study. ACLR, anterior cruciate ligament reconstruction; HS, hamstring.
Graft Diameter Measurements
Two board-certified orthopaedic surgeons with subspecialty certification in sports medicine (R.M. and G.B.M.) retrospectively reviewed all operative reports for included ACLRs to record graft diameters to the nearest 0.5 mm. Information on the number of strands was not comprehensively collected by the registry across all study years; for ACLR where this information was reported, >80% were 4-strand grafts. Each surgeon made the graft diameter measurements (in mm) using commercially available sizing tools. There were variabilities in the graft measurement recordings. Therefore, the following steps were followed by the 2 authors for consistency in recordings. Where a single graft diameter was stated in the operative report, that diameter was recorded and used for the analysis. If the graft diameter varied between the femoral and tibial sides, the smaller of the 2 diameters was used. For example, if the femoral side was 8 mm and the tibial side was 8.5 mm, then 8 mm was the diameter used for analysis. This was done to ensure that the minimum graft diameter was used for analysis, as it was felt that the graft would be most likely to fail at its smallest diameter. When the graft diameter was not directly stated in the operative report, the femoral tunnel diameter was used. Patients were excluded if the graft diameter or reamer diameter was not reported.
Exposure of Interest
The exposure of interest was the HA diameter. Table 1 summarizes the number of ACLRs for each graft diameter by 0.5-mm increment. Most graft diameters (90.6%) were between 7 and 9 mm, with the most common being 8 mm (n = 2231). For analysis, 2 different graft diameter comparisons were made (Figure 1): (1) <8 versus ≥8 mm and (2) ≤8 versus >8 mm.
Summary of Primary HA-ACLR Included in the Final Study Sample and Revisions Observed by Primary ACLR Graft Diameter in 0.5-mm increments a

ACLR, anterior cruciate ligament reconstruction; HA, hamstring autograft.
Crude incidence was the cumulative revision probability, calculated as follows: 1 – (Kaplan-Meier estimator at 2-year follow-up).
Outcomes of Interest
The primary outcome was aseptic revision surgery, defined as any reoperation performed after the index ACLR where removal and replacement of the original graft for aseptic reasons was required. Within our health care system, patients are continuously monitored for revisions after the index ACLR through surgeon reports and EHR surveillance until either health care plan membership termination or death. The identified revisions are manually validated by trained research associates. During the study period, the median time to revision surgery for those who experienced one was 1.7 years (interquartile range [IQR], 1.1-3.2 years). In the study sample, 1808 (30.3%) patients were lost to follow-up through health care plan membership termination at a median time of 3.7 years (IQR, 1.6-6.4 years). The number of revisions observed for each graft diameter group is presented in Table 1.
Covariates
Covariates included patient age in years (continuous), sex (men vs women), body mass index (BMI, continuous), race/ethnicity (Asian, Black, Hispanic, other, White), activity at the time of injury (basketball, football, soccer, other sports, other activity), femoral fixation device type (combination, crosspin, interference, suspensory), tibial fixation device type (combination, interference, suspensory), and femoral tunnel drilling method (lateral approach, medial portal, tibial tunnel). Combination fixation was defined as >1 fixation device used on the same side.
Statistical Analysis
Propensity score weighting was used to balance covariates between comparison groups before outcome assessment. After applying propensity score weighting, the standardized mean difference (SMD) for each covariate was measured. An SMD of <0.2 indicates a balance achieved for the covariate between comparison groups. Propensity scores were calculated using a multivariable logistic regression model, which included all specified covariates as predictors of treatment assignment. Rosenbaum approach was used to address missing covariate values; we created separate levels for nominal variables with missing values and a missing indicator variable for continuous variables with missing data while also imputing the mean.
Revisions were modeled as time-to-events from the index ACLR until the revision surgery date for those who experienced an event or until the last follow-up date for those who did not have a revision. The last follow-up date was defined as the date of health care membership termination, death, or study end date (December 31, 2020)—whichever came first. Crude cumulative aseptic revision probabilities were calculated as 1 minus the Kaplan-Meier estimate and cumulative incidence curves were reported up to the time point at which at least 50 ACLRs remained at risk in the smallest group to ensure a reliable estimate. The mean treatment effect propensity score–weighted Cox proportional hazards regression was used to evaluate the risk of aseptic revision by graft diameter comparison group. Robust standard errors were used to account for the correlation from ACLR performed by the same operating surgeon. The proportional hazards assumption for the exposure variable was checked by testing time interaction terms and met, implying that the factors investigated have a constant impact on the hazard over time. Hazard ratios (HRs) with 95% CIs are reported.
To determine whether there is a graft diameter threshold that best identifies patients who are at a higher risk of aseptic revision, we assessed individual intervals of the HA diameter using receiver operating characteristic (ROC) curve and area under the curve (AUC) analyses. ROC curves show the tradeoff between sensitivity and specificity of increasing graft diameter (in mm) in differentiating the risk of aseptic revision. The AUC, which ranges from 0.5 to 1, is a measure of accuracy; an AUC of <0.7 is considered a poor predictor, with an AUC of 0.5 interpreted the same as random chance. 24 An effect plot was also created to model the probability of revision based on the HA diameter. The effect plot displays a fitted logistic regression model for increasing intervals of the HA diameter, adjusting for patient age, sex, and BMI. These covariates were selected for the model, as they changed the estimates by >10% or were determined to be clinically relevant. Analyses were performed using R (R Version 3.6.2; R Foundation for Statistical Computing). P < .05 was the statistical significance threshold used for this study, and all tests were 2-sided.
Results
Comparison 1: HA Diameter <8 vs ≥8 mm
Cohort characteristics for the first comparison are presented in Table 2. The median follow-up time for the entire cohort was 4.4 years (IQR, 2-7.5 years). After propensity score weighting, the balance was observed with an SMD of <0.1 for all covariates. Figure 2A presents the cumulative incidence of revision during the follow-up according to the study group. At the 10-year follow-up after primary ACLR, the cumulative aseptic revision probability was 10.9% for <8 mm and 9.3% for ≥8 mm (Table 3). In the propensity score–weighted Cox model, <8-mm autografts had a higher risk of aseptic revision compared with ≥8-mm autografts (HR, 1.32 [95% CI, 1.01-1.71]; P = .039).
Patient, Implant, and Surgery Characteristics of 5972 Primary Isolated HA-ACLR by Autograft Diameter Size (2005-2020) a

Data are presented as mean ± SD or n (%). ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; BSD, balanced standardized difference; HA, hamstring autograft; USD, unbalanced standardized difference.

Cumulative probability of aseptic revision after primary isolated ACLR by HA diameter. (A) Comparison 1 (<8 vs ≥8 mm) and (B) comparison 2 (≤8 vs >8 mm). Shaded areas around the solid lines represent 95% CIs. The plot is based on incidence after propensity score weighting on the following covariates: patient age, BMI, sex, race/ethnicity, femoral fixation, tibial fixation, femoral tunnel technique, and activity at injury. ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; HA, hamstring autograft.
Crude Incidence and Adjusted Risk for Revision After ACLR by HA Diameter a
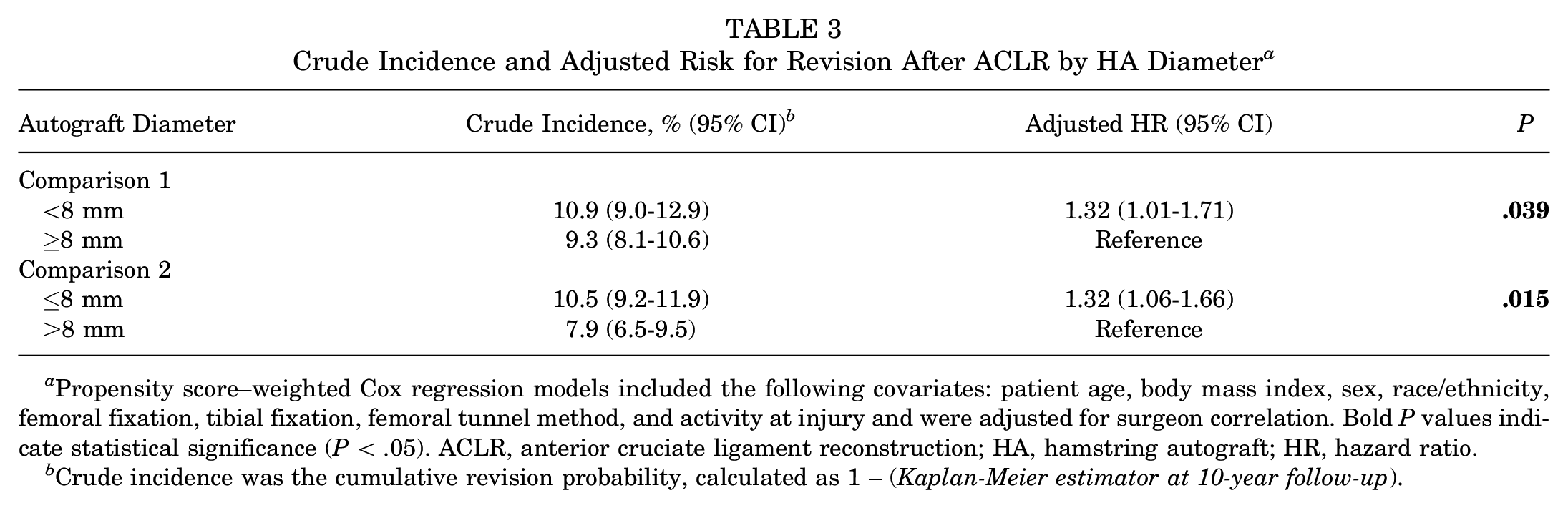
Propensity score–weighted Cox regression models included the following covariates: patient age, body mass index, sex, race/ethnicity, femoral fixation, tibial fixation, femoral tunnel method, and activity at injury and were adjusted for surgeon correlation. Bold P values indicate statistical significance (P < .05). ACLR, anterior cruciate ligament reconstruction; HA, hamstring autograft; HR, hazard ratio.
Crude incidence was the cumulative revision probability, calculated as 1 – (Kaplan-Meier estimator at 10-year follow-up).
Comparison 2: HA Diameter ≤8 vs >8 mm
Table 2 presents characteristics of the study sample by an HA diameter of ≤8 and >8 mm; balance improved after applying propensity score weighting with an SMD of <0.1 for all covariates. At 10 years after primary ACLR, the cumulative revision probability was 10.5% for ≤8 mm and 7.9% for >8 mm (Figure 2B and Table 3). In the propensity score–weighted Cox model, ≤8-mm autografts were observed to have a higher risk of revision compared with >8-mm autografts (HR, 1.32 [95% CI, 1.06-1.66]; P = .015).
HA Threshold
In ROC analysis, an HA diameter that best identifies patients at a higher risk for revision after ACLR could not be identified (Figure 3). The AUC indicated diameter was not adequate in differentiating a cutoff for revision risk (AUC, 0.56). However, the adjusted effect plot showed a direct decrease in revision risk with each increasing increment in graft size (Figure 4).

A ROC curve for increasing increments of the HA diameter for patients who underwent ACLR. The reference line (white line) is a guideline of a test unable to identify patients at risk for aseptic revision better than chance, or where the AUC = 0.50. ACLR, anterior cruciate ligament reconstruction; AUC, area under the curve; HA, hamstring autograft; ROC, receiver operating characteristic.

Adjusted effect size plot for increasing increments of the HA diameter (x-axis) and revision risk (y-axis) for patients who underwent ACLR. The logistic regression model included age, sex, and BMI; the shaded area represents the 95% CI. ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; HA, hamstring autograft.
Discussion
The HA diameter is a well-recognized risk factor that may potentially be modified by the operating surgeon.2,20,34,37 Graft harvest can sometimes lead to inconsistent graft diameters due to patient anthropometric differences, inadequate harvest, or truncation of the tendon during surgical harvest, 7 which has led surgeons and investigators to try to determine the minimum acceptable diameter. In our cohort of patients ≤25 years old who underwent primary HA-ACLR in a health care organization, we observed a 32% higher risk (HR, 1.32) of revision for graft diameter comparisons (<8 vs ≥8 mm and ≤8 vs >8 mm)—the minimum cut-point suggested by previous literature. Further, we were unable to determine a definitive minimum graft diameter threshold that should be used.
Biomechanical studies have shown a clear increase in load to failure with increasing graft diameter. Boniello et al 5 reported mean failure loads of the HA and found that the mechanical strength of grafts was related to their size: the smaller the size, the weaker the grafts. However, they reported that the benefit of graft strength increased most between 6 and 7 mm (38%) but sequentially decreased with the lowest increase in strength being from 8 to 9 mm (12%). Schimoler et al 30 also tested 20 quadrupled looped HAs ranging from 6 to 9.5 mm in 0.5-mm increments and found that load to failure increased linearly with increasing the graft diameter and the cross-sectional area.
The acceptable minimum graft diameter remains controversial. While most reports agree that an 8-mm graft is the minimum acceptable diameter,14,20,31 some studies have stated >8 mm, 21 while others have reported it to be ≥8 mm.8,25,32 In our cohort, higher revision risks were observed when considering ≤8 mm compared with >8 mm and <8 mm compared with ≥8 mm. Magnussen et al 20 reported that an HA diameter of >8 mm was a minimum acceptable graft diameter. In a retrospective analysis of 256 patients with a mean follow-up of 14 months, the authors found that 17 of 18 revisions occurred in patients with an HA of ≤8 mm. However, all revisions in grafts <7 mm occurred in skeletally immature patients. Along with the short mean follow-up time, the authors may not have captured adult patients who may not have returned to full sporting activities. 20 Mariscalco et al 21 examined 263 patients and showed that smaller HA size was a predictor of poorer Knee injury and Osteoarthritis Outcome Scores at 2 years after primary ACLR. Of the 14 revisions, all patients had autografts with diameters <8 mm, but analysis accounting for confounders such as age was not performed. Another retrospective study by Park et al 25 examined 296 patients with quadrupled HA and found superior results in patients with a graft diameter of >8 mm. All 12 of the graft failures (defined as International Knee Documentation Committee grade ≥C or revision surgery) had a graft diameter of <8 mm. However, an adjusted analysis for potential confounders was not performed. Conte et al, 8 in a systematic review of 4 studies with evidence levels 3 or 4 (N = 913 patients), with a minimum 1-year follow-up, found that failure rate was associated with a 6.8 times greater risk if the graft diameter was ≤8 mm. Evaluating 18,245 patients with a minimum 2-year follow-up from within the Swedish and Norwegian National Knee Ligament registries, Snaebjörnsson et al 32 reported a higher revision risk among patients treated with HA with a diameter of <8 mm compared with those ≥8 mm. Murgier et al, 23 using the New Zealand ACLR Registry, found a similar failure rate of 6% for those with graft diameters of <8 mm and ≥8 mm.
Some have also reported a graft diameter as low as 7 mm to be acceptable and found no difference in revision rates. Kang et al 15 in a meta-analysis of 9 studies (N = 2243 knees) found no significant difference in the risk of failure for grafts ≤7 mm compared with those >7 mm. However, a significant difference was found in the failure rate for grafts >6.5 mm compared with those ≤6.5 mm, suggesting that a cutoff value of 7 mm rather than 8 mm is appropriate. In a recent meta-analysis of pooled data of 19,799 ACLRs, Alomar et al 2 also reported a graft diameter of ≥7 mm to be associated with significantly lower failure rates than a graft diameter of <7 mm. Unfortunately, the event rate was too low in our cohort to analyze a graft diameter of ≤7 mm. Still, with higher revision risks observed for grafts <8 mm, a graft diameter of <8 mm may not be recommended.
As higher revision risks were observed regardless of the 8-mm threshold, we sought to determine whether there was a specific cut point could be identified in our cohort. Unfortunately, an “optimal” threshold when performing HA-ACLR could not be identified. As a continual decrease in revision risk was observed in the adjusted effect plot for each increasing increment in graft diameter, a larger graft is preferred to optimize revision risk. This is consistent with a previously matched case-control study using data from the same registry. 34 Spragg et al 34 included 132 revisions after HA-ACLR and 396 matched HA-ACLR controls without revision. They reported that for every 0.5-mm decrease in the HA diameter from 9 to 7 mm, the likelihood of needing revision surgery increased by 22% and concluded that an increase in diameter >7 mm, even as small as 0.5 mm, could benefit patients. The present study goes beyond the matched case-control study by Spragg et al, 34 as the full cohort of HA-ACLR in the registry was included in the present study while only patients with a revision and a matched sample of nonrevised counterparts were included in the previous study. This allowed for an evaluation of the HA diameter as a predictor of revision. Snaebjörnsson et al, 33 using data from the Swedish National Knee Ligament Registry, compared 560 cases with 1680 matched controls. Similar to the study by Spragg et al, 34 Snaebjörnsson et al also found that the likelihood of revision surgery for every 0.5-mm decrease in diameter between 10 and 7 mm was 1.16.
The 2 means by which authors have tried to predict the HA diameter are anthropometric data and the use of preoperative imaging studies. Anthropometric factors—such as thigh circumference3,29 and weight12,29,36—have been shown to play a role in predicting diameter, with a patient’s height being the strongest predictor.3,11,13,19,28,29,35 BMI has been shown to have a weak positive correlation 29 or none at all. 35 One study 6 suggested an equation to calculate the HA diameter based on patient height: graft diameter in mm = (height in cm × 0.052) − 1.07. Some authors have used preoperative magnetic resonance imaging (MRI) measurements—including the cross-sectional area of the gracilis and semitendinosus—to predict graft diameter.4,13,22,36 Vivekanantha et al, 36 in a recent systematic review including 46 studies comprising 4140 patients, found that 12 of 19 (63.2%) studies reporting on the semitendinosus and gracilis cross-sectional area had a moderate to very high correlation with intraoperative graft diameter. Leiter et al 18 found the strongest predictor to be the cross-sectional area of the semitendinosus and gracilis on MRI plus the patient’s weight. One study found that a smaller cross-sectional area of the semitendinosus and gracilis tendon on MRI, shorter stature, lower weight, smaller thigh circumference, and female sex are associated with a smaller 4-stranded HT autograft diameter. 12 A systematic review and meta-analysis of studies using ultrasound to predict graft diameter found preoperative ultrasound measurements of the target tendons were moderately correlated with the intraoperative HA size and that there were no significant differences between ultrasound and MRI measurements in predicting the sizes of the gracilis and semitendinosus tendon autografts. 17
Limitations
Our study has several limitations, one of which is that it was observational, and only observed associations were reported. While the study inclusion and exclusion criteria and the analysis used were to address confounding, there is potential for residual confounding due to unmeasured factors. For example, our ACLR registry does not collect information on the posterior tibial slope, return to sports, and postoperative rehabilitation. These factors are left to the discretion of the surgeon, patient, or physical therapist and are not standardized. However, both return to sports and postoperative rehabilitation occur after the primary ACLR and therefore do not meet the definition of a confounder. Further, we do not anticipate these factors to differ depending on graft diameter size. There was no standardization of HA diameter measurement before the study but was instead determined by each operating surgeon. Further, almost 3000 patients were excluded due to missing information on the HA diameter. This was largely due to the availability of the HA diameter information in the EHR for retrospective collection; characteristics of patients excluded due to missing diameter information were not different from those included.
The Kaiser Permanente ACLR registry only collects revision surgery as the primary endpoint, which likely underestimates the actual graft failure rate, as patients with graft failure detected through clinical examination or imaging studies who do not undergo revision surgery cannot be captured through the registry. However, the decision to undergo a revision surgery was determined by both the surgeon and the patient and not solely determined by the graft diameter size that was used during the primary ACLR. The registry does not collect patient-reported outcome scores and therefore those were not evaluated. The study was limited to younger patients because they are more likely to show differences in revision rates; therefore, the results may not be generalizable to older patients. Finally, we are only able to identify revisions performed within the health care system and there is potential for patients to seek outside care. As outside care, including revisions, is not covered by the health care system’s insurance plan, we do expect outside care to be minimal. Furthermore, loss to follow-up was 30.3%, with a median time of 3.7 years. However, the survival analysis used was able to incorporate this loss to follow-up in the regression model. Patient follow-up was included in the effect estimation and then censored at the last follow-up date if the membership was terminated without experiencing a revision.
Conclusion
A 32% higher risk of revision was observed for smaller graft diameters compared with larger graft sizes regardless of the graft diameter comparison considered. In a cohort of almost 6000 HA-ACLR, we were unable to determine a definitive minimum graft diameter threshold that should be used, a larger graft diameter was associated with a lower revision risk. While others have suggested there may be a minimum acceptable graft diameter, findings from the present cohort suggest that there may not be a minimum acceptable graft diameter for clinicians to use for all patients.
Footnotes
Acknowledgements
The authors acknowledge the orthopaedic surgeons who contribute to the Kaiser Permanente Anterior Cruciate Ligament Reconstruction Registry as well as the staff in the Department of Surgical Outcomes and Analysis at Kaiser Permanente, which coordinates registry operations.
Presented as a poster at the AOSSM Annual Meeting, July 13-17, 2022, Colorado Springs, Colorado
Final revision submitted June 27, 2024; accepted July 2, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.M. has received nonconsulting fees from Arthrex and Empire Medical and consulting fees from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Kaiser Permanente Southern California/Hawaii (reference No. 5691).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
