Abstract
Background:
Heterotopic ossification (HO) may occur after multiligament knee injuries (MLKIs), with the potential to cause significant functional deficits. However, risk factors for HO after MLKI are not well understood.
Purpose:
To identify the incidence and risk factors for HO after MLKI.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
Patients were included if they had undergone treatment for an MLKI by a single surgeon at a level 1 trauma center between 2001 and 2023, with radiographs at the time of injury and at a minimum 6-month follow-up. All data were obtained from electronic medical records. Two senior musculoskeletal radiologists reviewed all cases for the presence of HO in the most recent radiographs, using the initial radiographs obtained at the time of injury for comparison. Patients were grouped by whether they had developed HO after MLKI. Univariable analyses were performed to compare patient, injury, and treatment characteristics between groups. Odds ratios (ORs) for HO were assessed by multivariable logistic regression, adjusted for ligament injury classification, mechanism of injury, documented dislocation, central nervous system (CNS) trauma, and knee-spanning external fixation (KSEF).
Results:
The studied cohort included 128 patients, of whom 28 were excluded due to insufficient follow-up. Of the remainder, 35 (35%) developed HO, with a mean follow-up of 35.17 months. In the univariable analysis, the HO group had significantly higher rates of documented dislocation, CNS trauma, and KSEF. In the multivariable analysis, KSEF (OR, 8.51; 95% CI, 21.81-31.717; P = .001) and CNS trauma (OR, 6.63; 95% CI, 1.507-29.209; P = .012) remained independent predictors of HO after MLKI.
Conclusion:
The incidence of HO after MLKI was 35%. KSEF and CNS trauma were independently associated with HO. Recognition of these risk factors may help identify at-risk patients for closer monitoring and consideration of HO prophylaxis and should therefore be included in the decision-making strategy for KSEF versus early surgical treatment after MLKI.
Keywords
Heterotopic ossification (HO) can develop directly in response to skeletal or soft tissue trauma or indirectly after neurologic damage.29,47 Given the high burden of trauma associated with knee dislocations (KDs) and multiligament knee injuries (MLKIs), along with prolonged immobilization, vascular stasis, and edema, patients with these injuries are particularly susceptible to HO, with an incidence of 13% to 43% in the literature.22,26,27,31,38,43,47 While the precise underlying physiology of trauma-induced HO is complex, it generally involves hypoxia-driven chondrogenic differentiation and endochondral ossification of local mesenchymal progenitor cells secondary to the inflammatory and osteogenic milieu induced by musculoskeletal trauma.1,20,23,32 HO can itself lead to pain, functional limitations, and decreased range of motion, thereby impairing the patient's rehabilitation and recovery after MLKI.27,41,47 As such, it is important to understand the nonmodifiable and potentially modifiable risk factors for HO after MLKI to identify at-risk patients. This may minimize the impact of HO after MLKIs, which are frequently associated with poor functional outcomes at 2 years postoperatively, particularly for posterior cruciate ligament (PCL)-based injuries. 19
A few studies have investigated the risk factors for HO after KD or MLKI with contradictory results.27,38,43,47,48 This lack of consensus is in part due to the rarity of MLKIs and insufficient patient follow-up, leading to few large studies on this topic. 35 A case series by Scarcella et al 38 is the largest cohort to date, with 119 patients with KDs and 14 (13%) developing HO. Prior studies have had varying results on whether the surgical approach, Injury Severity Score, and presence of closed head injury were significant risk factors for HO.27,38,43,47 Only 2 studies have assessed independent explanatory factors for HO after MLKI, both of which identified PCL reconstruction as the only independent risk factor.47,48 There are individual reports of HO after PCL reconstruction and after open reduction and internal fixation of a PCL avulsion fracture as part of an MLKI, as well as a case of symptomatic isolated PCL ossification.17,21,22,28,33 The patellar tendon and anterior cruciate ligament (ACL) have also been implicated in reactive HO of the knee.6,7,11,24,44,46 In contrast, Mills and Tejwani 27 found that the type of surgery had no meaningful impact on HO in their cohort of 35 patients, of whom 15 (42.9%) developed HO. However, like the remaining studies in the literature that assessed risk factors for HO after MLKI, this finding was based on univariable analysis.27,38,43
The purpose of this study was to define independent risk factors for HO in a large cohort of patients with MLKI. Understanding the risk factors for HO is useful in identifying patients who may be at high risk, adjusting treatment options and surgical strategy, or considering HO prophylaxis.13,36 Additionally, some potential risk factors are generally modifiable, such as the application of a knee-spanning external fixator (KSEF). It was hypothesized that HO would be associated with mechanism of injury (MOI), ligamentous injury combinations, associated injuries, and documented KDs.
Methods
Record Review Approval was secured at the Yale University institutional review board (2000028912). No funding was received for the completion of this study. Patients who had undergone treatment for a KD or MLKI by the senior author (M.J.M.), a sports medicine fellowship-trained orthopaedic surgeon, at a single level 1 trauma center between January 2001 and May 2023 were identified by specific Current Procedural Terminology and International Classification of Diseases codes related to MLKI treatment. Nonoperative treatment cases were identified through a comprehensive analysis of an established MLKI database including patients treated by the senior author.
Patient records were carefully assessed to ensure that cases met the criteria for MLKI, defined as complete injury to at least 2 of the 4 major knee ligaments: ACL, PCL, superficial medial collateral ligament (MCL), and lateral collateral ligament (LCL). Central nervous system (CNS) trauma was defined as traumatic brain injury or spinal cord injury and was based on physician documentation.
Descriptive information, injury details, neurovascular status, laterality, MOI, KSEF, type of surgery (arthroscopic vs open), time to first surgical intervention, time from injury to earliest HO, and radiographs were obtained from electronic medical records. MOI was stratified by the classification described by Ockuly et al, 30 which included high energy (motor vehicle collision, pedestrian versus automobile, falls from heights >10 ft, polytrauma), low energy (athletes in sports falls, height of 5-10 ft), and ultra-low velocity (activities of daily living such as stepping off a curb in patients with morbid obesity).
Management of MLKIs
Importantly, the heterogeneity of MLKI patterns led to considerable variation in surgical techniques, influenced by specific injury and surgeon preference. Ligament and other soft tissue repairs were frequently performed, including the LCL, PCL, MCL, and biceps femoris, as well as those involving fixation of avulsion fractures. Allografts and autografts were utilized in ligament reconstructions, with graft selection varying by patient factors. Postoperative care followed standard institutional practices for MLKIs. Patients were placed in a hinged knee brace after surgery, with early controlled range of motion exercises initiated as soft tissue healing permitted. Weightbearing progression was introduced gradually over the early postoperative period and individualized by the specific ligaments repaired or reconstructed. Rehabilitation was tailored to each patient's injury pattern and clinical recovery.
For all KDs and MLKIs, ankle brachial indices were initially assessed to screen for vascular injuries. If the indices were asymmetric or below the normal threshold of 0.9, a computed tomography angiography was conducted, and vascular surgery was consulted. Any patient who required operative intervention by vascular surgery was defined as experiencing a vascular injury. A peroneal nerve injury was defined as any loss of sensation in peroneal nerve distribution or loss of strength in superficial or deep peroneal nerve distribution and was separated into partial or complete motor and sensory components. A modified Schenck classification of KDs and an anatomic classification of MLKIs were used to stratify injuries by severity depending on whether the patient experienced a radiographically or clinically documented tibiofemoral dislocation and which ligaments were injured (Table 1).12,34,39,42 The modification of the Schneck KD classification system included adding the Stannard et al. subclassification for KDV injuries, 42 as well as the Green et al. subclassification for KDI injuries. 12 Injuries involving a fracture were classified as KDV if they met the consensus definition outlined by Medvecky et al. 25
ACL, anterior cruciate ligament; KD, knee dislocation; LCL, lateral cruciate ligament; MCL, medial collateral ligament; MLKI, multiligament knee injury; PCL, posterior cruciate ligament; PLC, posterolateral corner; sMCL, superficial medial collateral ligament.
Radiographic Review of MLKIs
All MLKIs were evaluated by magnetic resonance imaging (MRI) on a Siemens Verio machine with a field strength of 1.5 or 3 T, utilizing a 15-channel knee coil and employing fat suppression and proton density sequences. Those patients with KSEF in place could not use a knee coil and were placed into the MRI gantry either without a coil or with a body (wrap) coil that could fit under the KSEF. A board-certified, fellowship-trained musculoskeletal radiologist generated every official radiology report, which was corroborated by assessment of the senior author. The surgeon's final postoperative diagnosis was considered the definitive diagnosis of the patient's ligament injury if there was any inconsistency between the findings. Patients treated nonoperatively because of head injury or polytrauma were included within the study cohort, but the final diagnosis was made per a consensus agreement of the radiologist and the senior author.
Tears of the cruciate and collateral ligaments were stratified into partial, complete, midsubstance, proximal, and distal avulsions, similar to methods outlined by Kahan et al. 18 Partial tears observed on MRI were characterized by intact structural continuity accompanied by edema or abnormal signal within the surrounding ligamentous and myotendinous structures. Midsubstance tears were identified as complete disruptions occurring through the middle portion of the structures. Avulsions were delineated as the separation of the soft tissue or bony attachment from the individual structure and were classified by the proximal and distal attachment site. Fractures of the patella, medial and lateral femoral condyles, medial and lateral tibial plateau, and fibular head were recorded.
Radiograph Review of HO
Only cases with (1) initial anteroposterior and lateral radiographs from the time of injury and (2) follow-up radiographs at a minimum 6-month follow-up were included, similar to a study by Whelan et al. 47 Two senior musculoskeletal radiologists reviewed all patients for the presence of HO in the most recent radiograph as well as the radiograph performed at the time of injury, which was used as the baseline comparison film. Because of the retrospective nature of the study, the precise time interval between the traumatic event and acquisition of the baseline radiograph was not consistently available. However, among patients who developed HO, the baseline radiograph was assessed for the presence of HO to assess the within-patient development of new HO. A patient was deemed to have HO if at least 1 musculoskeletal radiologist read the radiograph as having any new extraskeletal bone formation in the knee.
Statistical Analysis
Patient characteristics were summarized by descriptive statistics, including mean and standard deviation for continuous variables and frequency and percentage for categorical variables. Student t test or chi-square, Fisher exact, or Kruskal-Wallis test were used for group comparisons. Univariable and multivariable logistic regression analyses were performed to explore the association between risk factors for HO, with odds ratios and 95% confidence intervals reported for all regressions. Consistent with methodology by Whelan et al, 47 covariates were selected for inclusion based on a combination of their statistical significance in the univariable analysis (P < .05) and their clinical relevance as established or theoretical risk factors for HO after MLKI. These covariates included ligament injury classification, MOI, KSEF, documented KD, and CNS trauma.
To account for potential overlap between predictors, sensitivity analyses were conducted. First, soft tissue injuries that were significantly associated with HO in the univariable analysis were substituted into the model as a covariate in place of ligament injury classification to evaluate whether individual ligamentous injuries provided additional predictive value beyond overall dislocation/ligamentous injury severity. Similarly, fracture was substituted into the model instead of ligament injury classification to assess whether fracture status alone independently predicted HO.
Model fit was evaluated through several approaches. The omnibus test of model coefficients was used to confirm that the final model provided a significant improvement in prediction as compared with the null model. The proportion of variance explained was summarized with Cox and Snell R2 and Nagelkerke R2 statistics. Model calibration was assessed with the Hosmer-Lemeshow goodness-of-fit test. Correlation matrices among covariates were examined to assess for multicollinearity.
SAS 9.4 (SAS Institute) and SPSS Grad Pack Premium 31.0 (IBM) were used to perform and validate all analyses. Statistical significance was defined as P < .05.
Results
Study Cohort
The studied cohort screened 128 patients, of whom 28 were excluded owing to insufficient follow-up. Of the final cohort of 100 patients, 40 had a documented KD and 60 had an MLKI with no documented dislocation. The mean follow-up time for the included patients was 35.17 ± 34.7 months. Of the final cohort of 100 patients, 35 (35%) developed HO, with 100% agreement between the radiologists in assessing for the presence or absence of HO. The mean time from injury to the earliest HO was 12.7 ± 27.6 months.
A complete case representation is provided in Table 2. Overall, 17 (17%) patients had a KDI or MLKI-1; 3 (3%), KDII or MLKI-2; 40 (40.0%), KDIII or MLKI-3; 14 (14.0%), KDIV or MLKI-4; and 26 (26.0%), KDV. Among the KDIII/MLKI-3 subgroup (n = 40), 10 (25.0%) were medial (KDIIIM/MLKI-3M) and 30 (75.0%) were lateral (KDIIIL/MLKI-3L). Among the KDI or MLKI-1 subgroup (n = 17), 2 (11.8%) had a KDI-DAM or MLKI-1-AM; 4 (23.5%), KDI-DAL or MLKI-1-AL; 1 (5.9%), KDI-DALM or MLKI-1-AML; 3 (17.6%), KDI-DPM or MLKI-1-PM; 6 (35.3%), KDI-DPL or MLKI-1-PL; and 1 (5.9%), KDI-DPLM or MLKI-1-PML. 12 Of the patients who had a KDV (n = 26), 4 (15.4%) had a KDV.1; 13 (50.0%), KDV.3L; 5 (19.2%), KDV.3M; and 4 (15.4%), KDV.4. 42
Univariable Analysis of Risk Factors for Heterotopic Ossification a
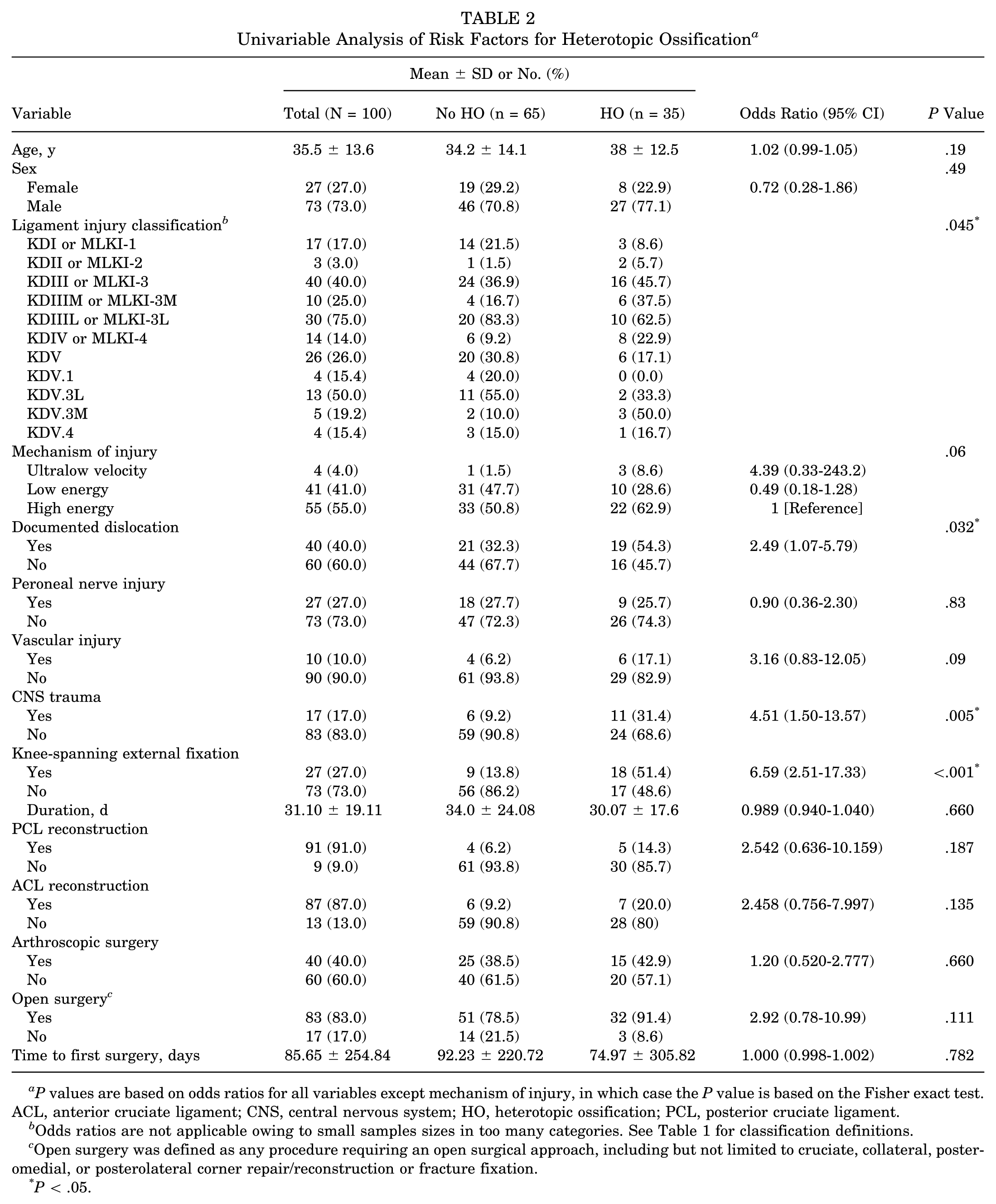
P values are based on odds ratios for all variables except mechanism of injury, in which case the P value is based on the Fisher exact test. ACL, anterior cruciate ligament; CNS, central nervous system; HO, heterotopic ossification; PCL, posterior cruciate ligament.
Odds ratios are not applicable owing to small samples sizes in too many categories. See Table 1 for classification definitions.
Open surgery was defined as any procedure requiring an open surgical approach, including but not limited to cruciate, collateral, posteromedial, or posterolateral corner repair/reconstruction or fracture fixation.
P < .05.
Risk Factor Analysis
In the univariable analysis, the distribution of ligamentous injuries between the HO and no-HO cohorts differed significantly (P = .045). Overall, higher-grade ligamentous injuries were more common in the HO group as compared with the no-HO group. Additionally, patients who developed HO had significantly higher rates of documented dislocation (P = .032), CNS trauma (P = .005), and KSEF use (P < .001). Higher-energy MOI approached statistical significance (P = .06). Other variables that did not reach statistical significance included age (P = .19), sex (P = .49), peroneal nerve injury (P = .77), vascular injury (P = .09), duration of KSEF (P = .660), PCL reconstruction (P = .187), ACL reconstruction (0.135), arthroscopic surgery (P = .660), open surgery (P = .111), and time to first surgery (P = .782).
Multivariable logistic regression was performed, adjusting for covariates that were statistically significant in univariable testing and/or clinically relevant to HO risk, including ligament injury classification, MOI, documented KD, CNS trauma, and KSEF (Table 3). The analysis demonstrated that KSEF was independently associated with HO (odds ratio, 8.51; 95% CI, 2.28-31.72; P = .001). Similarly, CNS trauma was an independent predictor of HO (odds ratio, 6.63; 95% CI, 1.51-29.21; P = .012). In contrast, documented dislocation (P = .927), ligament injury classification (P = .513), and MOI (P = .580) were not significantly associated with HO after adjustment.
Multivariable Analysis of Risk Factors for Heterotopic Ossification a

Covariates included ligament injury classification, mechanism of injury, knee-spanning external fixation use, documented knee dislocation, and central nervous system trauma. OR, odds ratio.
P < .05.
Model diagnostics demonstrated that overfitting and multicollinearity were not concerns. The omnibus test of model coefficients confirmed overall model significance (P < .001), and the Hosmer-Lemeshow test indicated good calibration (P = .607). Cox and Snell R2 (0.31) and Nagelkerke R2 (0.42) suggested moderate explanatory power. Examination of correlation matrices showed no concerning multicollinearity among predictors.
Association Between Specific Structural Injuries and HO
Of all the specific ligament injury patterns, only posterior oblique ligament (POL) injuries (any, not isolated) were associated with an increased risk of HO (Table 4). Of the 13 patients with a POL tear, 9 developed HO (69.2%) yielding a statistically significant difference between groups (P = .01). However, owing to its overlap with overall dislocation severity as captured by the ligament injury classification, as well as to limit model overfitting, POL injury was not included in the primary multivariable model. In a sensitivity analysis substituting POL injury for ligament injury classification, the direction of association between HO and KSEF, as well as between HO and CNS trauma, remained consistent. Tears of the MCL (P = .07), LCL (P = .06), PFL (P = .08), and MPFL (P = .08) approached but did not reach statistical significance.
Association Between Ligaments and Tendons Injured and Heterotopic Ossification a

Data are presented as No. (%). P values are based on chi-square test or Fisher exact test. ACL, anterior cruciate ligament; HO, heterotopic ossification; LCL, lateral collateral ligament; MCL, medial collateral ligament; MPFL, medial patellofemoral ligament; PCL, posterior cruciate ligament; PFL, popliteofibular ligament; POL, posterior oblique ligament.
P < .05.
Association Between Fracture Patterns and HO
Of the 100 patients in the study, 45 sustained a fracture with their MLKI or KD (Table 5). Of these 45 patients, 9 developed HO (20.0%), making fracture a statistically significant risk factor for HO development in the univariable analysis (P = .004). However, owing to the primary analysis adjusting for fracture-dislocations via the ligament injury classification variable, fracture was not included in the primary multivariable model to avoid overfitting. In a sensitivity analysis substituting fracture for KD classification, the direction of association between HO and KSEF, as well as between HO and CNS trauma, remained consistent. Of these 45 patients with fractures, 6 (13.3%) had patellar fractures, 18 (40.0%) fibular head fractures, 19 (42.2%) medial tibial plateau fractures, 15 (33.3%) lateral tibial plateau fractures, 9 (20.0%) medial femoral condyle fractures, 5 (11.1%) lateral femoral condyle fractures, and 5 (11.1%) tibial spine fractures. No specific fracture type was associated with HO on its own. Of these patients, 26 (57.8%) had fractures that met criteria for a KDV as defined by Medvecky et al. 25 Fibular head fractures and lateral tibial plateau fractures approached significance, as 3 patients (16.7%) with fibular head fractures developed HO (P = .07) and 2 (13.3%) with lateral tibial plateau fractures developed HO (P = .06).
Association Between Fractures and Heterotopic Ossification a
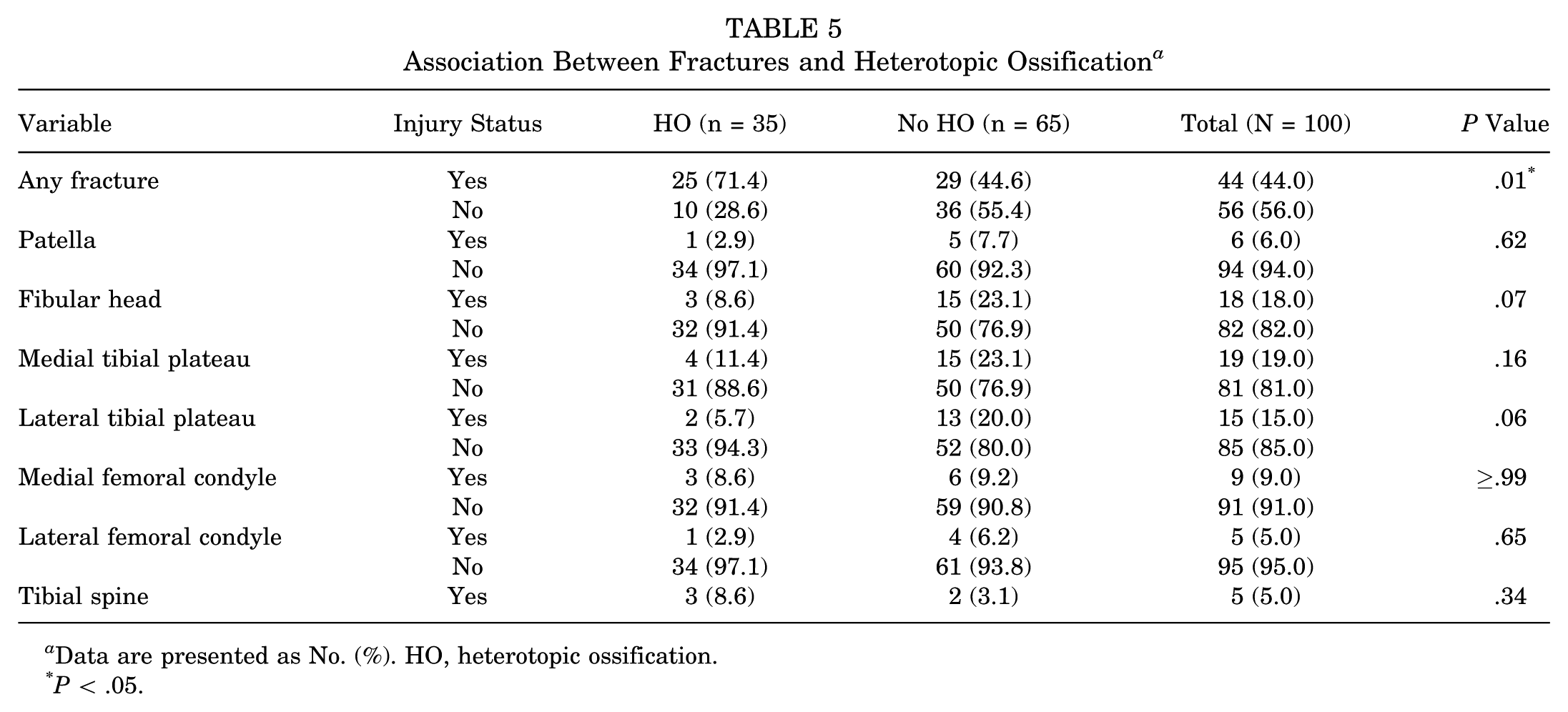
Data are presented as No. (%). HO, heterotopic ossification.
P < .05.
Discussion
This study found that in a cohort of 100 patients with KDs and MLKIs, the incidence of HO was 35%. Risk factors for HO identified on univariable analysis included documented KD, CNS trauma, KSEF, POL tear, and fracture. Conversely, the following were not statistically significant risk factors: patient sex, age, MOI, vascular injury, peroneal nerve injury, PCL reconstruction, ACL reconstruction, and time to first surgery, as well as other fractures, ligament injuries, and tendon injuries. On multivariable analysis, only CNS trauma and KSEF remained independent risk factors for HO after MLKI. These findings provide a reference for further investigations of larger cohorts to help predict and explain risk factors for HO that could alter treatment timing, prognosis, and overall functional recovery. This may enable orthopaedic surgeons to better anticipate the potential for HO and incorporate this into their operative and nonoperative treatment decision-making related to traumatic knee injuries.
In contrast to knee HO after total knee arthroplasty, the prevalence and risk factors for HO after MLKI have been rarely studied and are poorly understood, with wide variation in relative risk factors. One systematic review including 2988 patients with total knee arthroplasty found a prevalence of HO in 14% of patients. 10 There are a number of studies on the development of knee HO after other knee injuries or surgery, including PCL and ACL reconstruction, patellar tendon injuries, high tibial osteotomy, and arthroscopy. ¶ As MLKIs are rare injuries, few studies have assessed the development of HO after MLKI, with incidences between 26% and 43%.27,43,47 In a study by Whelan et al, 47 30 of 88 (34%) patients with KDs developed HO. Stannard et al 43 reported an incidence of 25% in 55 patients with MLKIs, Mills and Tejwani 27 an incidence of 43% in 35 patients with KDs, and Scarcella et al 38 an incidence of 13% in 119 patients with KDs. Thus, the incidence of 35% in the current study is consistent with findings in the literature.
A couple of studies have assessed independent risk factors for HO after MLKI.47,48 In both, PCL reconstruction was the only independent explanatory factor for HO after MLKI.47,48 Unlike the present study, Whelan et al 47 demonstrated that KSEF was not an independent predictor of HO. In their study, the authors included age, KSEF, time from injury to surgical reconstruction, and posterolateral corner/PCL reconstruction as covariates after a preliminary univariable analysis. With the same statistical methodology, the current findings demonstrated that, of these preliminary variables, only KSEF differed significantly between groups and was independently associated with HO, whereas in the Whelan et al 47 study, it failed to achieve significance after adjusting for covariates. While the duration of KSEF may be expected to affect the incidence of HO owing to differences in immobilization time, KSEF duration did not differ significantly between the HO and no-HO groups in the current study. In a separate univariable analysis of 57 knees, Stannard et al 43 found that closed head injury, MOI, periarticular fracture, posteromedial corner injury, and posteromedial surgical approach were not risk factors for developing HO. In a univariable analysis of 35 patients, Mills and Tejwani 27 reported that Injury Severity Score and traumatic brain injury increased the risk of HO, but bicruciate reconstruction, surgery within 3 weeks of injury, open surgery, and medial surgery did not. Finally, in an analysis of outcomes after KD that included 119 patients, Scarcella et al 38 noted that HO was more likely in patients with a higher Injury Severity Score, arterial injury, wound infections, and open injury. However this study did not adjust for confounding variables. While Injury Severity Score, open injury, and wound infection were not assessed in the current study, vascular injury was and did not demonstrate any difference between groups.
Consistent with findings from Mills and Tejwani 27 but in contrast to those from Stannard et al 43 and Whelan et al, 47 the current study found that CNS trauma was an independent risk factor for the development of HO. This is consistent with findings that patients with neurologic injury have a 20% chance of forming HO anywhere in the body, with most occurring in a large joint, and the combination of traumatic brain injury and femur fractures together increases the risk of HO to >50%. 14 Since neurologic injury alone has been found to be associated with HO development in large joints, including the knee, there may a synergistic effect of HO development when neurologic injury and traumatic musculoskeletal injury occur.8,14,15
Given that trauma and surgery induce an inflammatory state, HO may develop secondary to the initial injury or subsequent surgical treatments, or from a synergistic effect of both. Ogilvie-Harris and Sekyi-Otu 31 described 4 cases of HO on the lateral distal femur after arthroscopic ACL reconstruction at the area of the femoral tunnel, postulating that bone deposits or hematoma from tunnel drilling could have induced osteogenesis. Similarly, Patton and Tew 33 described 3 cases where reaming of graft tunnels in PCL reconstruction contributed to the development of HO, and Jones et al 16 reported that HO development was independently associated with reamed tibial nailing. In the current study, neither PCL nor ACL reconstruction was associated with HO on univariable analysis and therefore was not included in the multivariable analysis. However, KSEF did independently increase the risk of HO. While the pathophysiology of HO in the knee joint remains unclear, particularly in relation to KSEF, a plausible explanation based on the available literature is from prolonged immobilization and inflammation from transcutaneous access to the bone.4,37,40,45 Yet, it remains plausible that KSEF is more likely an indication of a severe or highly unstable knee injury with extensive soft tissue disruption, or it was applied to facilitate nursing care in patients with multiple associated injuries. Thus, it would be informative to assess whether the relationship between HO and KSEF persists with the use of hinged KSEF constructs, as these enable early controlled range of motion and are often used as a postoperative adjunct rather than as a temporizing measure.2,3 Future studies will need to use large-enough sample sizes to reliably investigate the structures injured as well as the surgical approaches used as risk factors for HO spatially and temporally.
Orthopaedic surgeons should at least be aware of the high incidence of HO after KD and MLKI when establishing treatment decisions and prognostic considerations. Given that nearly half of patients with CNS trauma, documented KD, or KSEF developed HO and that CNS trauma and KSEF emerged as independent predictors of HO, clinicians should recognize these as important risk factors. HO prophylaxis should be strongly considered in such high-risk groups, although in patients with concomitant CNS trauma, nonsteroidal anti-inflammatory drug (NSAID)-based prophylaxis would represent a relative contraindication owing to concern for cerebrovascular bleeding. This highlights the need for future studies assessing the safety and efficacy of traditional and alternative prophylactic strategies in various MLKI cohorts.
Limitations
There are several limitations to this retrospective cohort study. First, while this is one of the largest investigations of HO after MLKI to date, the sample size (N = 100) still limits statistical power and constrains multivariable adjustment. Similarly, generalizability is limited by the single-center, single-surgeon design and the level 1 trauma setting, where indications for KSEF and management of concomitant CNS trauma may differ from other institutions. Temporal changes across 2001 to 2023 may also influence applicability to contemporary practice. Nevertheless, this study identified statistically significant associations without introducing multicollinearity and performed sensitivity analyses where appropriate, supporting the robustness and practical relevance of the findings. Second, the reliance on retrospective data constrained the number of soft tissue structures that could be investigated, as the current study was limited to the documentation found in operative notes and MRI reports. Third, given the low numbers of KDs classified as ultra-low velocity, generalizations in this injury pattern are not possible. Fourth, the exact time stamps for baseline radiographs were not uniformly available. However, the use of each patient's own initial radiograph as the reference limits misclassification bias, as HO had to be radiographically new to be classified as such. Fifth, HO prophylaxis was not systematically ascertained because a retrospective review of NSAID use was difficult to verify, as many patients were taking formally prescribed and over-the-counter NSAIDs for pain from injury and surgery. Thus, unmeasured variation in anti-inflammatory exposure may have contributed to HO incidence and residual confounding of estimates. Last, a grading of HO severity was not incorporated because of the absence of a validated knee HO severity classification system. This limited the extent to which radiographic findings could be correlated with clinical outcomes, as HO is expected to differentially affect range of motion, pain, and functional outcomes based on severity.
Conclusion
In 100 patients sustaining MLKIs, the overall incidence of HO was 35%. KSEF and CNS trauma were independently associated with HO, whereas ligament injury classification, MOI, and documented dislocation were not. Future studies with larger cohorts are needed to better understand the unique contributions of various patient characteristics, injury patterns, and surgical interventions on the development of HO after MLKI.
Footnotes
Final revision submitted December 11, 2025; accepted December 15, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: M.J.M. has received speaking fees from DePuy Mitek and Smith & Nephew, nonconsulting fees from Synthes GmbH, and consulting fees from Smith & Nephew.
Ethical approval for this study was waived by the Yale University institutional review board (2000028912).

