Abstract
Background:
Previous research has shown that tendon graft soaking in vancomycin or tobramycin solution has no negative effects on graft mechanical properties, but there are no studies that have investigated graft mechanical properties after soaking grafts in gentamicin. Additionally, nearly all published biomechanical studies are based on data collected from a mechanical load frame or strain gauge, which does not provide insight on local graft strains compared with 3-dimensional digital image correlation (3D-DIC).
Purpose/Hypothesis:
The purpose of this study was to use 3D-DIC to investigate the effects of vancomycin, tobramycin, and gentamicin soaking on tendon graft mechanical properties. It was hypothesized that (1) no significant difference in mechanical properties exists between the saline control, vancomycin, tobramycin, and gentamicin groups and (2) local graft strain at the graft failure location will be greater than global strain spanning the entire graft length.
Study Design:
Controlled laboratory study.
Methods:
Human tibialis anterior, peroneus longus, and tibialis posterior tendon grafts were prepared and evenly separated into 4 groups: control, vancomycin (5.0 mg/mL), tobramycin (1.0 mg/mL), and gentamicin (0.8 mg/mL). Grafts were soaked in antibiotic solution for 10 minutes, then removed and painted via airbrush with water-based black paint. Uniaxial tension testing was then completed at a strain rate of 10 mm/min. Data collected were used to calculate Young modulus (YM), elasticity limit (EL), ultimate tensile strength (UTS), and failure strain (FS).
Results:
There were no significant differences in YM (P = .49), EL (P = .62), UTS (P = .98), and FS (P = .14) between control, vancomycin, tobramycin, and gentamicin, respectively. Additionally, local strain at graft failure location was larger than global strain across the length of the graft.
Conclusion:
Soaking tendon grafts in vancomycin, tobramycin, or gentamicin does not alter the mechanical properties of grafts under uniaxial loading.
Clinical Relevance:
If vancomycin use is not possible or is contraindicated for certain patients, surgeons can soak grafts in tobramycin or gentamicin to achieve similarly effective infection mitigation without weakening the graft.
Keywords
Anterior cruciate ligament reconstruction (ACLR) is a routine, safe, and effective procedure. One rare complication associated with this procedure is septic arthritis.15,19,23 While infection risk is small, consequences of this complication can be significant and include additional medical costs, extended antibiotic treatment, possible removal of graft, and increased risk of arthritis and knee dysfunction.6,18,28 The use of perioperative intravenous antibiotics, typically first- or second-generation cephalosporins, clindamycin, or vancomycin, is one common method of decreasing infection risk.23,29,32
Evidence has also shown that, in addition to the use of perioperative intravenous antibiotics, soaking tendon grafts in an antibiotic solution can further reduce infection risk compared with perioperative intravenous antibiotics alone.7,23,26 Most research investigating this comparison studied vancomycin as the antibiotic used in graft soaking. In addition to vancomycin, recent evidence has also shown that tobramycin and gentamicin solutions, which are typically used as alternatives when vancomycin is unavailable or contraindicated, can reduce infection risk to a similar extent. One preliminary study investigating tobramycin found that a vancomycin soaking concentration of 2.5 mg/mL was required to have effects similar to the 0.1-mg/mL tobramycin solution and that soaking grafts in a 1-mg/mL tobramycin solution nearly eliminated the presence of bacteria in comparison with saline control. 5 Research on gentamicin found that a concentration of 0.8 mg/mL significantly reduced infection risk when compared with saline control and that a concentration of 0.08 mg/mL had similar infection-reducing ability compared with 1-mg/mL vancomycin solution.4,20 Given these antimicrobial effects, many surgeons who utilize intraoperative graft soaking use antibiotic solutions with the above concentrations.
Despite the investigation of these antibiotics’ effects on decreasing infection risk, less research has been done to study the effects of antibiotic soaking on graft mechanical properties. Several studies have shown that vancomycin has no significant effect on multiple mechanical properties of tendon grafts.3,13,16,23 A recent study found that graft mechanical properties are not significantly altered by graft soaking in either vancomycin or tobramycin solution. 8 However, there are no studies that have investigated the effects of gentamicin soaking on tendon graft mechanical properties.
Most published research studying the mechanical properties of tendon grafts is based on force and displacement data collected via a mechanical load frame or strain gauge. These methods of measurement provide force data, but they may not provide strain data throughout the entirety of the graft because human tendons are anisotropic and can deform in multiple planes of motion.11,17 One way to address this potential issue is to use digital image correlation (DIC) when measuring graft displacement. DIC is a 2-dimensional (2D-DIC) or 3-dimensional (3D-DIC) noncontact imaging technique that measures displacement of an object by comparing images of the object before and during load application. These comparison images can provide detailed insight of strain throughout an entire area of interest and can identify the strain state at the eventual failure location.
The use of DIC in tendon graft testing has been shown to be accurate. 17 However, its application in evaluating tendon graft mechanical properties is limited.
The purpose of this study was to investigate the effects of vancomycin, tobramycin, and gentamicin soaking on mechanical properties of tibialis and fibularis longus tendon grafts by using 3D-DIC. We hypothesize that no significant difference in mechanical properties will exist between the 4 groups. We further hypothesize that local strain at the graft failure location will be greater than global strain spanning the entire graft length.
Methods
Graft Preparation
Human tibialis anterior (TA), peroneus longus (PL), and tibialis posterior (TP) tendons (N = 40) were acquired from MTF Biologics and stored in a freezer (–20° C) until the day before testing when they were removed to reach ambient room temperature. Graft type was selected based on supply at the facility where testing was completed. These 3 graft types have similar mechanical properties. 2 On the day of testing, tendon grafts were submerged in 100 mL of sterile water (Medline) and carefully trimmed to remove any residual muscle. Following cleaning and trimming, grafts were separated into 4 different soaking groups: normal saline (control, n = 10: TA, 3; PL, 5; TP, 2), vancomycin (5.0 mg/mL, n = 10: TA, 5; PL, 5), tobramycin (1.0 mg/mL, n = 10: TA, 5; PL, 5), or gentamicin (0.8 mg/mL, n = 10: TA, 5; PL, 5). Whole grafts were then marked with a 6-cm segment to represent the graft's working length. Photos of each graft were taken with a ruler in the same image to generate a mm/pixel ratio for each image. Images were uploaded to National Institutes of Health ImageJ software (https://imagej.nih.gov/ij/) and width measurements were performed at 6 locations along the length of each graft. The mean graft width was used to approximate the cross-sectional area (CSA) assuming an elliptical cross-section. CSA for each graft was used when calculating graft stress. The equation below shows elliptical approximation, pi times semi-major radius time semi-minor radius. In this case, the semi-major radius was measured, and the semi-minor radius was estimated to be half of the semi-major radius.
Antibiotic Solution Preparation
Individual solutions were prepared in concurrence with each graft being tested. Solution concentrations were based on data from previous literature standards at concentrations known to decrease bacterial bioburden.4,5,23,26,31 The saline solution was prepared by placing 100 mL of sterile saline (McKesson) in a sterile 8-oz bowl (Medline). Vancomycin solution was prepared by diluting 500 mg of vancomycin hydrochloride powder (Fisher Scientific) with 100 mL of sterile saline. Tobramycin solution was prepared by diluting 100 mg of tobramycin powder (Fisher Scientific) with 100 mL of sterile saline. Gentamicin solution was prepared by diluting 80 mg of gentamicin powder (Fisher Scientific) in 100 mL of sterile saline.
Graft Soaking and Painting Procedure
The graft-soaking procedure followed the protocol from previous literature. 31 For brevity, a 10.16 × 10.16–cm Medline Accu-Sorb Raytec gauze sponge was soaked in the desired solution for 1 minute. After 1 minute, the gauze was removed from the solution and wrapped around the tendon for 10 minutes. 12 Grafts were then placed on a ConMed graft preparation table (ConMed), pulled in slight tension, and painted using an airbrush containing water-based black paint. This paint provided a color that contrasted with the native graft color, allowing the DIC cameras and software to accurately detect certain locations along the graft's length. The ability to detect and track these locations during each test was essential to calculate strain. An image of a painted graft just before testing is presented in Figure 1.

Image of painted graft.
Mechanical Testing and Data Collection
Quickly following painting, grafts underwent uniaxial tension mechanical testing on an MTS Systems Bionix servohydraulic mechanical testing frame. Tendon specimens were gripped using custom fabricated, corrugated cryoclamps reinforced with dry ice to improve adhesion (Figure 2). To further minimize tendon slipping, grips were reinforced using 2 C-clamps. Specimens were gripped on each side, leaving 6 cm of length between the grips, and a tensile load of roughly 10N was applied to ensure the grafts were taut before testing. At this point, an initial image was recorded to provide a reference image for DIC.

Mechanical testing setup.
Grafts then went through a preconditioning cycle to reduce graft viscoelasticity. This process varies greatly according to literature. 14 For this study, the preconditioning was based on Nurmi and colleagues’ 22 protocol where grafts were manually displaced to a tensile load of 80N and held at this load for several seconds. The graft was then cyclically loaded from 80N to 0N for 25 cycles over a period of 100 seconds. An 80N load was then reapplied for several seconds before the grafts were pulled to failure in uniaxial tension at a rate of 10 mm/min. 13
Force data were collected using a myDAQ data acquisition device (National Instruments). DIC images were collected using 2 Teledyne Point Grey cameras (Teledyne Vision Solutions). The myDAQ was synchronized with the DIC images to ensure measured forces corresponded to the correct point in each test. To ensure accuracy, force and displacement data during each test were also collected using MTS Multipurpose Elite (MPE) software. Cameras recorded images at a rate of 4 frames per second. These images were uploaded to 3D-DIC software, VIC-3D (Correlated Solutions Inc), and VIC-3D was used to create an area of interest for each graft, seen in Figure 3. Settings were then adjusted to create a grid of squares ranging between 31 × 31 pixels and 51 × 51 pixels. Strain within each of these squares represented local strains in the graft throughout the entire area of interest. Local strains and failure location were compared qualitatively as seen in Supplementary Videos 1 and 2 (available separately). Also within this area of interest, a 6-cm digital extensometer was placed. The extensometer was used to calculate global strain across the entire length of the graft.

VIC-3D black and white image of graft (left). Image with overlying area of interest and extensometer (right). VIC-3D, 3D-DIC software.
Following strain calculations, strain data from VIC-3D and load data from MPE software were uploaded to MATLAB (MathWorks). MATLAB was used to calculate stress, read in strain data from VIC-3D, and generate stress-strain curves for each test. It was also used to calculate Young modulus (YM) (MPa), elasticity limit (MPa), ultimate tensile stress (UTS) (MPa), and failure strain percentage. YM was found by creating a first-degree polynomial curve fit for the elastic region of each test. The elastic region was conservatively approximated with a strain range of 3% to limit use of nonlinear data and can be seen in Figure 4. Elasticity limit was found by taking the stress value at the transition point between elastic and plastic regions. UTS was the maximal stress value in the data set for each respective test.

Stress-strain representative curve. DIC, digital image correlation; UTS, ultimate tensile strength.
Statistical Analysis
Statistical Analysis was performed using IBM SPSS (IBM; Version 30) analysis software. To assess normality of data, a Shapiro-Wilk test was performed. Difference in mean YM, elasticity limit, and UTS between treatment groups was analyzed using an analysis of variance. Difference in failure strain between treatment groups was analyzed using Kruskal-Wallis nonparametric tests. A P value of ≤.05 was used to determine significance of all tests. A power analysis was performed to ensure the study was appropriately powered to detect a 30% change in mechanical strength with antibiotic soaking with an alpha of .05 and beta of 0.2 (80% power). An effect size of 1.0 was estimated based on preliminary standard deviation data.
Results
There was no statistically significant difference in YM, UTS, elasticity limit, or failure strain between the control, vancomycin, tobramycin, and gentamicin groups (Table 1).
Biomechanical Data for Treatment Groups Reported a
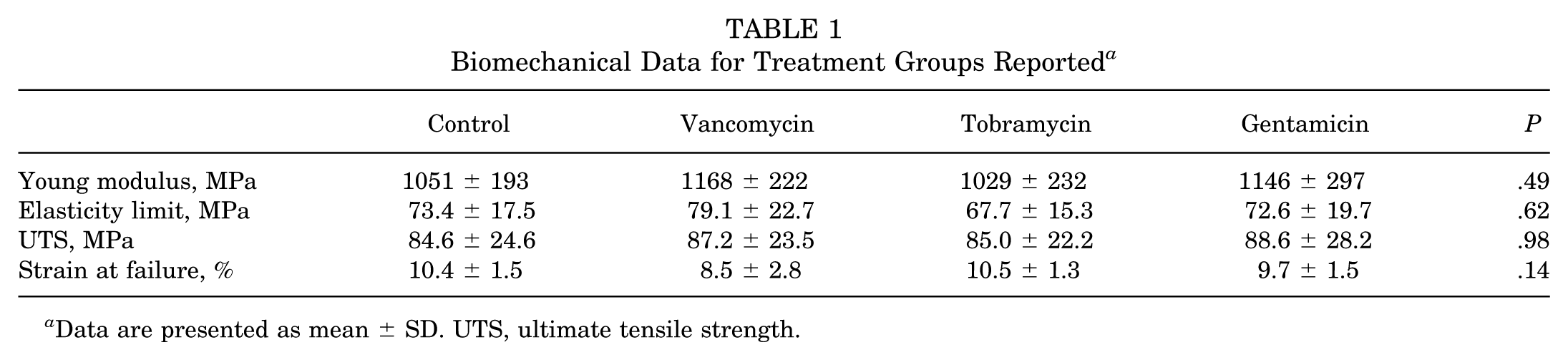
Data are presented as mean ± SD. UTS, ultimate tensile strength.
All grafts tested demonstrated failure within the 6-cm distance between the cryoclamps. A nonlinear toe region, linear elastic region, and a final plastic deformation region were distinctly identified in all stress-strain curves and are representatively shown in Figure 4.
In addition to global strain data collected above, for all tests in all groups, local strain at the graft failure location was greater than global graft strain at the point of failure.
Discussion
The most important finding in this study is that tendon graft mechanical properties are not significantly different among grafts soaked with gentamicin, tobramycin, vancomycin, and saline control. This supports the use of gentamicin, tobramycin, and vancomycin to reduce risk of septic arthritis following ACLR without altering mechanics of the graft tissue.
When comparing results of this study with a similar study comparing vancomycin and tobramycin, this study shows a larger YM and elasticity limit. The increased elasticity limit is likely explained by the increased YM. Increased YM can potentially be explained by several things. First is the graft preconditioning cycle in this study, which was not present in the other study. The addition of a preconditioning cycle was based on its effect on graft mechanics. Previous studies have shown that an increased load during preconditioning can increase the stiffness of the material being tested. 10 While stiffness does not directly correlate with YM, it is conceivable that the preconditioning could have caused the increase. In addition to preconditioning increasing stiffness, it also increased the duration of time the graft was sitting in the cryoclamps. Increased time in cryoclamps likely decreased graft temperature relative to our previous study, making decreased graft temperature a potential contributing factor to the increased YM. Also, graft properties have been shown to vary depending on characteristics of the graft donor. 1 The use of peroneus longus tendons was also a potential contributor to the increase in YM, as this study and others have shown that, although not statistically significant, peroneus longus grafts exhibit higher YM than tibialis anterior and tibialis posterior grafts. 2 Last, the similar study used load frame displacement to approximate strain. Using load frame displacement may overestimate strain, which in turn would underestimate YM. Despite these differences, it is important to keep in mind that, within this study, there was no significant difference between antibiotic groups, which still supports the viability of gentamicin use in tendon graft soaking.
DIC proved to be an effective method of measuring graft strain both locally within small portions of each graft and globally across the entire graft length. The combination of the speckle pattern used and DIC processing parameters used in this study allowed us to measure failure strains up to approximately 14%. A study recently published by Ashton and colleagues 2 indicates that failure strain for most lower extremity tendons is approximately ≤17%, with the Achilles tendon being the most at roughly 23.5%. Comparing strains measured in this study with strains measured by Ashton et al, 2 it is reasonable to suggest that DIC can be used to accurately collect biomechanical data for nearly all lower extremity tendon grafts.
While failure strains were relatively similar from graft to graft, local strains were more variable. For one particular test, shown in Supplemental Video 1, it can be seen that local strain is greatest in the top, middle right, and bottom left portions of the graft being tested. In another test, shown in Supplemental Video 2, local strain is greatest in the top right and bottom right of the graft. These differences in local strain and failure location provide objective evidence that tendon grafts are anisotropic and are consistent with findings in published literature.11,17 With this anisotropy in mind, the utility of 3D-DIC in biomechanical studies becomes increasingly important, as it may be helpful in showing strain patterns, locations of maximal/minimal strain, and location of graft failure.
While not directly pertaining to this study's primary objective, there were several lessons learned during this study that may assist in further optimizing the use of 3D-DIC in biomechanics research. First, the use of black, water-based spray paint is likely the best way to create a speckle pattern on tendon grafts. Water-based paint has been used to successfully generate high-resolution images in other studies that incorporated DIC, and there are no known direct side effects of its use.17,30 One alternative to the painting method used herein is saturation of tendon grafts in methylene blue and using white water-based paint to create contrast. While this has generated quality DIC data in previous studies, we found that at a nontoxic concentration of methylene blue at 0.5 mg/mL suggested by Peng et al 24 did not create the amount of contrast necessary for quality DIC images. Second, moisture of each graft after saturation in antibiotic solution initially created a glare that was detected by the cameras used for DIC data collection. Two effective methods to reduce glare in this study were closing the camera aperture and adjusting lighting accordingly for each test. Last, regarding DIC data processing, the use of a relatively large subset size was helpful in capturing DIC data at high strains. When increasing subset, this increases the number of pixels that make up each unique element within the tendon graft area. In this study, a subset range of 31 to 51 pixels was found to generate the most quality data for the duration of a test.
In addition to its application in studies like this, there are additional ways to utilize 3D-DIC in biomechanics research. One use would be to investigate the changes in local strains during a variable or cyclic loading cycle. As described by Elias and colleagues, 10 graft stiffness has been seen to change when under different magnitudes of force and when force is applied for longer times. This suggests that local strains within grafts may be dependent on force magnitude or time, and 3D-DIC would be an effective way to investigate this. It was also noted in several tests that the location of maximal strain within a graft was not always the location at which the graft failed, so studying the rate with which this occurs is potentially warranted. Finally, using 3D-DIC to study graft behavior under loading patterns other than uniaxial tension, like compression or torsion, may be a worthwhile investigation.
Limitations
There are some limitations to this study. First, as stated above in the discussion, the use of cryoclamps may have contributed to an increased YM. While cryoclamps may have affected graft mechanical properties, they were necessary in completing testing. Several noncryo alternatives were attempted when developing our test protocol. Despite efforts, for all alternatives, grafts either slipped in grips before failure or failed outside of the working gauge section. Second, although initially planning to use a nontoxic concentration of methylene blue solution, it did not provide adequate contrast for DIC. Black water-based aerosol paint was utilized instead for the speckled pattern. While there is a risk of graft toxicity, previous literature using water-based paint does not report any graft weakening. 17 Additionally, antibiotic soaking parameters for this study may vary from the practices of some surgeons. Grafts in this study were soaked for 10 minutes whereas common ACLR graft-soaking measures can range from 5 to 30 minutes.21,27,33 Both tibialis and peroneus longus grafts were utilized in testing, which although biomechanically similar to each other, do not encompass all ACLR graft options.9,25 Finally, this study is limited to a small sample size.
Conclusion
Soaking tendon grafts in vancomycin, tobramycin, or gentamicin does not alter the mechanical properties of the grafts under uniaxial loading. Additionally, local graft strain at the failure location is larger than global graft strain at the point of failure. The anisotropy, variable max strain points, and variable failure location of tendon grafts merits further use of DIC in biomechanics research. This study supports multiple antibiotic alternatives that can safely be used for decontaminating grafts at the time of surgery without altering tissue biomechanics.
Footnotes
Final revision submitted November 21, 2025; accepted December 1, 2025.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study
Supplemental Material
A Video Supplement for this article is available at http://journals.sagepub.com/doi/suppl/![]()
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

