Abstract
Background:
Tendon graft soaking in vancomycin solution minimizes surgical infection risk and has no effect on graft strength, but the intermittent vancomycin shortages, presence of vancomycin-resistant organisms, and treatment needs of patients with vancomycin hypersensitivity merit investigation of alternative antibiotics. Tobramycin prevents infection at low concentrations and is a cost-effective alternative, but it is unknown if tobramycin alters graft mechanical properties.
Purpose/Hypothesis:
The purpose of this study was to investigate the effects of tobramycin soaking on tendon graft mechanical properties. It was hypothesized that tobramycin soaking will not affect tendon graft mechanical properties.
Study Design:
Controlled laboratory study.
Methods:
A total of 30 tibialis tendon grafts were wrapped in saturated gauze swabs containing saline (control; n = 10), vancomycin (n = 10; 5 mg/mL), or tobramycin (n = 10; 1 mg/mL) for 10 minutes. Grafts were then removed from the treated gauze swab wraps and mechanically tested under uniaxial tension loading conditions. Grafts were pulled at a rate of 10 mm/min to failure. Force and displacement data from each test were used to calculate the Young modulus (MPa), ultimate tensile stress (MPa), and elasticity limit (MPa).
Results:
There were no significant differences in Young modulus (552 ± 108, 583 ± 98, and 660 ± 237 MPa; P = .62), ultimate tensile stress (91.5 ± 20.8, 96.6 ± 17.8, and 99.7 ± 33.3 MPa; P = .85), or elasticity limit (51.7 ± 16.4, 53.2 ± 13.8, and 52.3 ± 15.3 MPa; P = .98) between the control, vancomycin, and tobramycin groups, respectively.
Conclusion:
Soaking of tibialis tendon grafts with tobramycin does not appear to alter mechanical properties of the tendon graft under uniaxial load conditions.
Clinical Relevance:
If vancomycin use is not possible or contraindicated for certain patients, surgeons can soak grafts in tobramycin to achieve similarly effective infection mitigation without weakening the graft.
Keywords
Anterior cruciate ligament reconstruction (ACLR) is a routine, safe, and effective procedure. One rare complication associated with this procedure is septic arthritis, in which the primary infecting organisms are Staphylococcus aureus or coagulase-negative staphylococci.20,24,28 While infection risk is small, the consequences of this complication can be significant and include additional medical costs, extended antibiotic treatment, and increased risk of arthritis and knee dysfunction.7,23,41 Perioperative intravenous antibiotic administration using first- or second-generation cephalosporins, vancomycin, or cloxacillin is common practice for protecting against infection after ACLR.28,42,44 In cases involving only intravenous antibiotic use, the reported infection risk ranges from roughly 0.35% to 2.5%.8,20,24,28,30,41,42,44
Compelling evidence indicates that utilizing intravenous antibiotics, as well as graft soaking with vancomycin, further reduces the infection risk compared with intravenous antibiotic use alone.8,27,28,33,34 Graft soaking consists of either wrapping the graft in gauze saturated with vancomycin or direct placement of the graft in a vancomycin solution.8,28,33,34 Graft soaking with vancomycin is cost-effective, has no negative effects on graft mechanical properties, and has no significant effects on the graft at a molecular level.3,17,21,28
Despite positive outcomes of anterior cruciate ligament graft soaking with vancomycin, questions remain regarding its exclusive use in this setting. The literature suggests that vancomycin-resistant or intermediately vancomycin-sensitive strains of S. aureus and vancomycin-resistant strains of coagulase-negative staphylococci have been found.15,16,36 While rare, infection with these organisms can lead to persistent infection, which typically requires additional antibiotic therapy. 10 Additionally, antibiotic shortages are increasingly common worldwide.11,25,31 Finally, there are patients who have hypersensitivity or adverse reactions to vancomycin.22,32 These considerations justify the search for vancomycin alternatives for graft soaking during ACLR. One potential alternative is tobramycin. Data from a preliminary study suggest that soaking grafts in a 1-mg/mL tobramycin solution nearly eliminates the presence of bacteria in comparison with saline control. 5 This same study found that a vancomycin soaking concentration of 2.5 mg/mL was required to have effects similar to the 0.1-mg/mL tobramycin solution. 5 Additionally, in a study evaluating the cost-effectiveness of intrawound vancomycin and tobramycin use in orthopaedic trauma, both drugs were found to be cost-effective when administered alone. 19 Similar to vancomycin, availability and antibiotic resistance are also concerns with tobramycin use.9,12,25,37 Regardless, the above advantages of tobramycin justify further investigation.
No research exists examining the effects of tobramycin soaking on graft mechanical properties. The purpose of this study was to investigate the role of tobramycin soaking on the mechanical properties of a tendon graft. We hypothesized that tobramycin soaking would not affect tendon graft mechanical properties.
Methods
Tibialis Graft Preparation
Human tibialis tendons (anterior or posterior; n = 30) were acquired from MTF Biologics and stored in a freezer (−20°C) and removed the day before testing to reach ambient room temperature. On the day of testing, tendon grafts were submerged in 100 mL of sterile water (McKesson) and carefully trimmed to remove any residual muscle. After cleaning and trimming, whole grafts were marked with a 6-cm segment to represent the graft’s working length. Photos of each graft were taken with a ruler in the same image to generate a millimeter per pixel ratio for each image. Images were uploaded to NIH ImageJ software (https://imagej.net/ij/), and width measurements were performed at 6 locations along the length of each graft. The mean graft width was used to approximate the cross-sectional area (CSA), assuming an elliptical cross section. A sample calculation of CSA is provided in Table 1. After CSA measurements, grafts were then randomly separated into 3 different soak groups: normal saline (control; n = 10), vancomycin (n = 10; 5 mg/mL), or tobramycin (n = 10; 1 mg/mL).
Sample CSA Calculation a
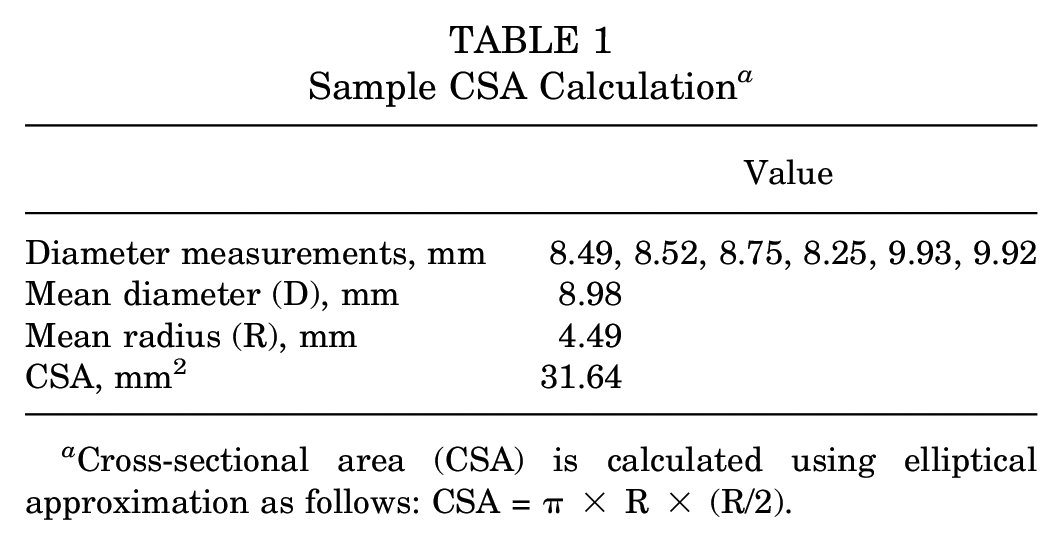
Cross-sectional area (CSA) is calculated using elliptical approximation as follows: CSA = π× R × (R/2).
Antibiotic Solution Preparation
Individual solutions were prepared in concurrence with each graft tested. The saline solution was prepared by placing 100 mL of sterile saline (McKesson) in a sterile 8-oz bowl (Medline). Vancomycin solution was prepared by diluting 500 mg of vancomycin hydrochloride powder (Slate Run Pharmaceuticals) with 100 mL of sterile saline. The concentration of the vancomycin solution was based on previous literature.28,33,34,43 Tobramycin solution was prepared by diluting 100 mg of tobramycin powder (Mylan Pharmaceuticals) with 100 mL of sterile saline. The concentration of the tobramycin solution was based on previous data. 5
Graft Soaking Procedure
The graft soaking procedure followed the protocol from previous literature. 43 For brevity, a 4 × 4-inch (10.16 × 10.16-cm) Medline Accu-Sorb Raytec gauze sponge was soaked in the desired solution for 1 minute. After 1 minute, the gauze was removed from the solution and wrapped around the tendon for 10 minutes. 13
Mechanical Testing and Data Collection
After soaking, grafts underwent uniaxial tension mechanical testing on an MTS Systems Bionix servohydraulic mechanical testing frame. Tendon specimens were gripped using custom fabricated, corrugated cryo-clamps reinforced with dry ice to improve adhesion. To further minimize tendon slipping, grips were reinforced using 2 C-clamps. Specimens were gripped on each side, leaving 6 cm of length between the grips. To begin each test, samples were preconditioned with 10 cycles of 1-mm extension. After a 5-second rest, the graft was pulled in tension to failure at a rate of 10 mm/min. 17
Force and displacement data during each test were collected using MTS Multipurpose Elite software. These data were exported and used to calculate stress-strain curves for each test. Stress-strain curves were then used to find the Young modulus (MPa), elasticity limit (MPa), and ultimate tensile stress (UTS) (MPa). The Young modulus was found by creating a first-degree polynomial curve fit for the elastic region of each test (Figure 1). The elastic region was conservatively approximated with a strain range of 3% to limit use of nonlinear data. The elasticity limit was found by taking the stress value at the transition point between elastic and plastic regions. UTS is the maximum stress value in the data set for each respective test. Calculations were performed using MATLAB (MathWorks).

Mechanical testing setup.
Statistical Analysis
Statistical analysis was performed using SYSTAT (Inpixon) analysis software. To assess the normality of data, a Shapiro-Wilk test was performed. Differences in the Young modulus and UTS between treatment groups were analyzed using Kruskal-Wallis nonparametric tests. The difference in mean elasticity limit between treatment groups was analyzed using an analysis of variance. A P value ≤.05 was used to determine the significance of all tests.
Results
Of the tests performed, only 2 samples showed failure within the cryo-clamps. One sample was part of the control group, and the other was part of the tobramycin group. Data from both tests were included in the results. All other samples showed failure at a location between the cryo-clamps in the 6-cm working length of the graft.
The Young modulus, elasticity limit, and ultimate tensile strength were not significantly different between groups (Table 2).
Biomechanical Data for Treatment Groups Reported a

Data are presented as mean ± SD. UTS, ultimate tensile stress.
Stress-strain curves for these tests show that tendon grafts produce 3 distinct regions: a nonlinear initial loading region, a linear elastic region representing elastic deformation, and a yield region representing plastic deformation and graft failure. A representative curve from one of the tests can be seen in Figure 2.

Representative stress versus strain curve. Red lines separate different curve regions.
Discussion
The primary finding in this study was that tendon graft mechanical properties were not significantly different among grafts soaked with tobramycin, vancomycin, and a saline control. Tobramycin is an attractive alternative to reduce the incidence of septic arthritis after ACLR. 5 Additionally, as demonstrated here, tobramycin is not detrimental to graft mechanical properties in vitro.
Septic arthritis is a rare complication associated with ACLR, but one that often results in arthroscopic debridement, extended antibiotic treatment, and graft dysfunction after reconstruction.1,7,23,35,41 Thus, establishing effective antibiotic regimens that minimize infection risk without altering mechanical properties on the graft is desirable.
One method of further mitigating infection rates is soaking the graft used during ACLR with vancomycin.27,28,33,34 Although grafts used in previous studies (ie, hamstring, bone-tendon-bone, or quadriceps tendons) differ from those herein, antibiotic soaking parameters were similar and at concentrations previously found to decrease the infection rate after ACLR.27,33,34 Given this similarity, the results herein support current literature stating that vancomycin does not significantly alter tendon graft mechanical properties.17,21,27,33,34
Tobramycin has been found to be an effective alternative to vancomycin in preventing infection. 5 Compared with saline control solution, soaking in 0.1 mg/mL tobramycin solution for both 10 minutes and 20 minutes led to a significant decrease in bacteria recovered. Compared with the 0.1-mg/mL tobramycin solution, soaking in 2.5 mg/mL vancomycin solution was required to cause a similar decrease in bacteria recovered. 5 Taken collectively with the results presented herein, data suggest that tobramycin is an effective alternative to minimize infection without significantly altering mechanical properties of a tendon.
A novelty of the present study was the use of cryo-clamps to grip the tendon graft. This is a supported gripping method, but reports indicate a wide range of tendon graft mechanical properties with the use of cryo-clamps.2,38,39 Furthermore, while previous studies have used tibialis tendon grafts and the cryo-gripping technique, graft treatment and mechanical testing parameters differ from those here, making it difficult to compare the present findings with previous ones. However, graft treatment and mechanical testing properties in this study are comparable to those of Jacquet and colleagues. 17 Despite similarities, discrepancies in the type of graft and gripping method are potential factors contributing to contrasting results between Jacquet et al, and those in the present study. 2 Based on the available evidence, data clearly underscore the importance of intrinsic factors (eg, age and type of graft)6,18 and testing procedures (eg, soaking parameters and testing strain rate)14,17,21,29,40,45 when investigating and interpreting tendon graft mechanical properties. Additional research examining the role of antibiotics on graft mechanical properties is warranted.
Limitations
There are some limitations in this study worth discussion. First, the use of displacement control in the preloading phase of mechanical testing was implemented in this study. This control mode contributed to variability in the initial loading region between samples. Displacement control eliminated the ability to analyze an additional mechanical property: ultimate tensile elongation, which is the strain achieved at the graft’s UTS. While all results for this study are reliable and sufficiently contribute to the study’s conclusion, ultimate tensile elongation would provide further insight on graft deformability in loading conditions beyond the elasticity. Second, tibialis tendon grafts were used to conduct this study, and mechanical properties vary between graft types.2,6 Therefore, results from this study may not apply to other graft types commonly used in ACLR. Third, the sample size of this study was relatively small. The data from this investigation will be used to establish appropriately powered studies for future research. Lastly, the controlled 10-minute graft soaking time in this study may not always be applicable to operating room practices.
Additional research is needed to determine the optimal use of tobramycin to prevent infection without deleterious consequences on the graft mechanical properties. Differences in antibiotic concentration, soaking time, and time between soaking and mechanical testing are examples of independent variables to investigate. This study only examines the immediate effects of graft soaking on mechanical properties. Using time between graft soaking and mechanical testing as an independent variable could be a good way to study long-term effects. In addition to tobramycin, gentamycin is another aminoglycoside that has been shown to reduce infection rates when used in tendon graft soaking solution.4,26 Its effects on graft mechanical properties are also undescribed in the literature. The inclusion of a gentamycin test group in future studies should be considered.
Conclusion
Soaking of tibialis tendon grafts with tobramycin does not appear to alter mechanical properties of the tendon graft under uniaxial load conditions.
Footnotes
Final revision submitted September 16, 2024; accepted November 6, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.M. has received a grant from DJO. D.F. has received consulting fees from Linvatec, Medical Device Business Services, Smith & Nephew, Vericel, DePuy Synthes Products, Bioventus, and Zimmer Biomet Holdings; nonconsulting fees from Linvatec, Smith & Nephew, Vericel, KARL STORZ Endoscopy-America, Pacira Pharmaceuticals, and Trice Medical; and honoraria from Vericel. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
