Abstract
Background:
Anterior cruciate ligament reconstruction (ACLR) requires optimal graft healing for successful outcomes. Magnetic resonance imaging (MRI) is widely used to monitor graft maturation, with lower graft signal intensity suggesting better tissue organization and advanced healing. Hyperbaric oxygen therapy (HBOT) has shown promise in enhancing healing, but its effect on anterior cruciate ligament graft maturation in humans remains unclear.
Purpose:
To evaluate the impact of adjuvant HBOT on graft maturation and integration in patients undergoing ACLR, using 4-month postoperative MRI.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
All patients who underwent primary anatomic ACLR with hamstring autografts and adjuvant HBOT were identified and matched 1:1 by sex and age (<25, 25-39, 40-54, and >55 years) with controls who did not receive HBOT. MRI was performed at 4 months postoperatively. Graft signal-to-noise quotient (SNQ) was measured to quantify graft maturation and analyzed using the Mann-Whitney U test; bone marrow edema at the graft-bone tunnel interface (graded as absent, mild, or accentuated) was used to infer graft integration and analyzed using the Fisher exact test.
Results:
In total, 52 patients (26 HBOT, 26 controls; mean age 37.0 ± 13.5 years; 61.5% male) were included. Patients receiving HBOT exhibited significantly lower graft SNQ compared to controls (median difference, 83.9; P = .031), indicating improved graft maturation. Bone marrow edema at the graft-bone tunnel interface was also lower in the HBOT group (P = .029), suggesting enhanced graft integration.
Conclusion:
In this pilot study, adjuvant HBOT following ACLR was associated with significantly improved signs of graft maturation and integration, as evidenced by lower graft signal intensity and interface signal on MRI at 4 months, suggesting accelerated early healing. These findings may help refine strategies to enhance early graft incorporation and support the development of rehabilitation protocols that optimize postoperative outcomes for patients undergoing ACLR.
Anterior cruciate ligament (ACL) tears are common knee injuries that frequently require surgical reconstruction to restore joint stability and function, particularly in active individuals. 27 While ACL reconstruction (ACLR) usually yields positive results, the risk of rerupture and other complications during recovery poses significant challenges.26,31 The limited spontaneous healing environment within the joint and the lengthy osteointegration and ligamentization process underline the need for improved strategies to enhance graft healing and long-term success. 7 Efforts continue to focus on optimizing surgical techniques, rehabilitation, and biological augmentation to support graft incorporation and functional recovery. 13
Hyperbaric oxygen therapy (HBOT) has emerged as a safe and promising adjunctive treatment to enhance tissue healing. HBOT involves administering 100% oxygen at increased atmospheric pressure, typically around 2.0 to 2.5 atmospheres absolute (ATA), for specified durations. 49 This approach enhances healing through several mechanisms, including higher oxygenation, neovascularization, and extracellular matrix restoration.6,33 In this regard, the elevated pressure and oxygen concentration significantly increase the amount of dissolved oxygen in the plasma, which boosts oxygen delivery to tissues with limited blood supply. Additionally, HBOT stimulates the formation of new blood vessels and promotes fibroblast activity and collagen production, steps that are crucial for graft maturation and remodeling after ACLR. 23
Although preclinical studies have shown that HBOT effectively enhances ACLR, with evidence of improved graft maturation and osteointegration, as well as superior biomechanical properties,22,36,56 no research has yet explored the effects of HBOT after ACLR in humans. Advanced imaging techniques, such as magnetic resonance imaging (MRI), are commonly used to monitor graft maturation and reveal significant changes occurring as early as the initial postoperative period.28,55,57 Therefore, the purpose of this study focuses on assessing early graft changes using postoperative MRI at 4 months following ACLR. Lower graft signal intensity on MRI is generally interpreted as a marker of more advanced maturation, reflecting improved tissue organization and decreased water content. We hypothesize that patients undergoing adjuvant HBOT exhibit lower graft signal intensity, indicating a more advanced maturation process compared to control patients who did not receive HBOT.
Methods
The study was approved by the local institutional review board (IOT protocol approval number: 1671). This is a retrospective analysis of prospectively collected data evaluating patients who underwent anatomic ACLR with or without adjuvant HBOT. Initially, skeletally mature patients who underwent primary ACLR using hamstring tendon autografts followed by HBOT from 2023 to 2024 were evaluated (HBOT group). During this period, all patients undergoing ACLR by the senior author (M.K.D.) were offered HBOT as part of the standard postoperative recovery protocol. HBOT was administered to those who willingly accepted the treatment after receiving detailed information regarding its potential benefits and risks. Prior to initiating HBOT, all patients underwent a thorough medical evaluation, including a comprehensive medical history review and physical examination. Patients with contraindications, such as active upper respiratory tract infections or claustrophobia, were excluded from receiving HBOT. Postoperative MRIs were routinely performed at 4 months on those patients as part of a research protocol. Concomitant meniscal procedures, including repair or meniscectomy, were not considered exclusion criteria. Patients were excluded if they did not undergo MRI at 4 months post-ACLR or if they had other ligament injuries requiring repair or reconstruction. Following the identification of the HBOT group, a matched control group was established. This group consisted of a randomly selected cohort of patients who underwent primary anatomic ACLR with hamstring tendon autografts using a similar surgical technique and standard postoperative recovery protocol without adjuvant HBOT and had a postoperative MRI performed at 4 months. Patients were matched 1:1 based on sex and age categories (<25, 25-39, 40-54, and >55 years). Demographic data were collected for all included patients. The presence and frequency of adverse events following HBOT were also documented.
ACL Reconstruction
Arthroscopic single-bundle anatomic ACLR using a hamstring tendon autograft was performed. Briefly, the semitendinosus and gracilis tendons were harvested and prepared as a quadruple-stranded autograft. Anatomic ACLR was then carried out, with femoral fixation achieved using a suspension fixation with a button, and tibial fixation was secured using an interference screw. Postoperatively, all patients followed a standardized rehabilitation protocol that included immediate weightbearing as tolerated with the use of a brace, with early emphasis on achieving full extension and quadriceps activation. Progressive range of motion and closed-chain strengthening exercises were initiated in the first few weeks. This was followed by neuromuscular training and proprioceptive exercises.
Hyperbaric Oxygen Therapy Protocol
HBOT was administered in a monoplace hyperbaric chamber equipped with a vital sign monitoring device (Figure 1). In this study, HBOT was administered once daily at 2.5 ATA for 90 minutes per session. Each session included 60 minutes at isobaric pressure (2.5 ATA), with approximately 15 minutes allocated for compression and 15 minutes for decompression. Compression and decompression were performed at controlled rates of 0.03 to 0.06 ATA/min and 0.03 ATA/min, respectively. Each patient underwent a total of 5 HBOT sessions within the first 10 days following ACLR, beginning the day after surgery. This treatment protocol was empirically adapted from previously published studies.22,44

Monoplace hyperbaric chamber used for hyperbaric oxygen therapy.
Imaging Evaluation
MRI conducted at 4 months after ACLR was used for imaging evaluation. Scans were performed on 1.5- or 3.0-T MRI scanners with the knee in the standard position—slightly flexed, in neutral rotation, and with the patient in the supine position. MRI sequences included T2-weighted fat-saturated images in the coronal, sagittal, and axial planes; proton density images in the sagittal plane; and T1-weighted images in the coronal plane. Images were acquired with a slice thickness of 3 to 4 mm. For graft evaluation, the sagittal T2-weighted image that displayed the full length of the ACL graft was selected. Coronal and axial images were used to assess overall knee joint morphology, while sagittal images were used to measure the signal-to-noise quotient (SNQ) of the graft and evaluate the signal intensity of bone marrow edema adjacent to the graft-bone tunnel interface.
Lower graft signal indicates reduced water content, which theoretically corresponds to greater graft maturity and improved healing. To quantitatively assess graft signal intensity and compare graft maturity between the 2 groups, SNQ was measured. The sagittal section that best demonstrated the full intra-articular length of the ACL graft was selected for analysis. An area encompassing the intra-articular portion of the graft was manually outlined, and the mean signal intensity was calculated. This value was then normalized using the mean signal intensity of the quadriceps tendon and background noise, measured approximately 1 cm anterior to the patellar tendon. Both regions were assessed using circular regions of interest with a diameter of 10 mm.25,42,55 The SNQ for each graft was calculated using the following formula:
The signal intensity of the bone marrow edema adjacent to the graft-bone tunnel interface was visually assessed and graded using a system adapted from previous studies1,9,48 as follows: (1) absent, when no hyperintense signal was detected between the bone tunnel and the surrounding bone; (2) mild, when a hyperintense signal was present along the interface but not markedly elevated; and (3) accentuated, when a pronounced and intense signal was observed (Figure 2). Both the femoral and tibial tunnels were evaluated, and the most severe score between the 2 was considered for analysis. Signal intensity was determined by comparison with the adjacent bone, with careful attention to distinguishing true bone marrow signal change localized to the tunnel regions.

Sagittal magnetic resonance imaging T2-weighted fat-suppressed images following anterior cruciate ligament reconstruction, illustrating the bone marrow edema adjacent to the graft-bone tunnel interface (yellow arrows). (A) Absent bone marrow edema: no hyperintensity observed around the tunnel. (B) Mild bone marrow edema: subtle hyperintensity around the tunnel. (C) Accentuated bone marrow edema: pronounced hyperintensity surrounding the tunnel.
All measurements were performed by 2 independent radiologists (L.R.M., A.G.O.F.) specialized in musculoskeletal imaging, both of whom were blinded to the study groups. Blinding was ensured by providing anonymized MRI scans labeled with coded identifiers only. The average of their measurements was used for analysis. Any discrepancies were resolved by consensus. To assess intraobserver reliability, 1 investigator reevaluated 20 patients in a second review conducted at least 1 month later. Interobserver and intraobserver reproducibility were assessed using intraclass correlation coefficients (ICCs) for all measured parameters. ICC values >0.9 were considered excellent; values between 0.8 and 0.9, good; values between 0.7 and 0.8, moderate; and values below 0.7, poor.
Statistical Analysis
Numerical variables are described as mean ± standard deviation when normally distributed and as median [interquartile range] when nonnormally distributed, based on the Shapiro-Wilk test. Categorical variables are reported as absolute values and percentages. Comparisons between 2 continuous variables were conducted using either the ttest or the Mann-Whitney U test, depending on data distribution. Categorical variables were compared using either the Fisher exact test or the chi-square test, as appropriate. For the patients who underwent HBOT, correlation analysis between adverse events following HBOT and sexor age was performed using the phi coefficient for categorical variables and Spearman rank correlation for continuous variables. Statistical analyses were performed using GraphPad Prism version 10 (GraphPad Software), with statistical significance set at P < .05.
Sample size was calculated using G*Power (version 3.1). Given the novelty of this study as the first to investigate the effects of HBOT after ACLR in humans, we based our analysis on early MRI-based SNQ values of ACL grafts that have shown correlation with clinical parameters. Specifically, graft laxity within the first 3 months post-ACLR, measured by remanent anterior tibial translation, was considered. Based on previously reported data indicating a moderate correlation (r≈ 0.51), a sample size of 25 participants per group was estimated to provide 80% power at a significance level of α = .05. 24
Results
A total of 52 patients were included: 26 patients (26 knees) in the HBOT group and 26 patients (26 knees) in the control group.The overall mean age was 37.0 ± 13.5 years, ranging from 13 to 62 years. In total, 32 patients (61.5%) were male.
Regarding HBOT-related adverse events, 4 patients (15.4%) in the HBOT group reported mild ear discomfort during treatment sessions. Notably, this did not interfere with the completion of therapy, and no other adverse events were reported. There was no significant correlation between the occurrence of HBOT-related adverse events and sex (P = .867) or age (P = .653).
Patients who received adjuvant HBOT demonstrated significantly improved graft maturation, as indicated by a lower graft SNQ compared to the control group (median difference, 83.9; P = .031; Figure 3). Interobserver and intraobserver reliability for SNQ measurements were excellent, with ICCs of 0.909 (95% CI, 0.846-0.946) and 0.998 (95% CI, 0.995-0.999), respectively.
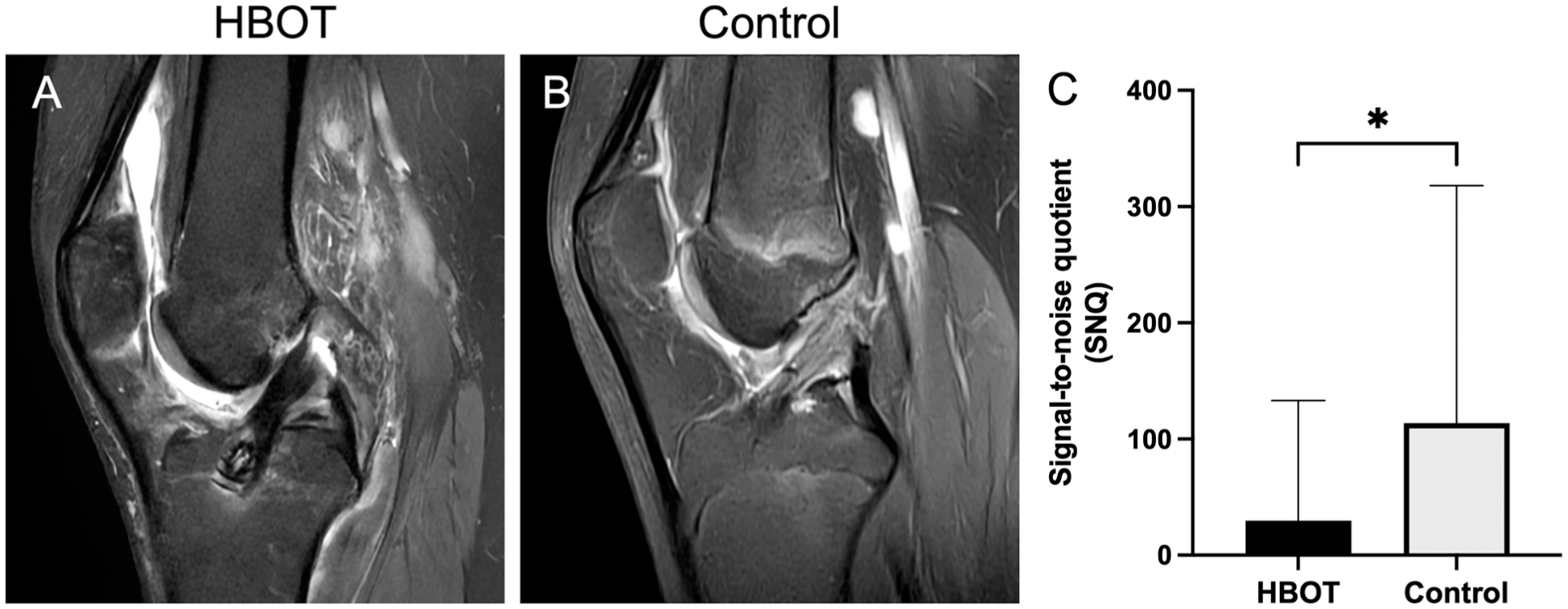
Sagittal T2-weighted magnetic resonance imaging of the knee following anterior cruciate ligament reconstruction. The hyperbaric oxygen therapy (HBOT) group (A) exhibited decreased graft signal intensity compared to the control group (B). (C) Quantitative analysis of the signal-to-noise quotient (SNQ). Data are presented as median + interquartile range. *P < .05.
Likewise, the bone marrow edema adjacent to the graft-bone tunnel interface was significantly lower in the HBOT group compared to controls (P = .029), suggesting enhanced graft integration. Reliability for interface signal measurements was also excellent, with an interobserver ICC of 0.953 (95% CI, 0.922-0.971) and an intraobserver ICC of 0.960 (95% CI, 0.904-0.984).
Patient demographics and imaging outcomes are summarized in Table 1.
Patient Demographics and Imaging Outcomes a

Data are presented as mean ± standard deviation (range), median [interquartile range], or number (percentage). P values <.05 are shown in bold to indicate statistical significance. HBOT, hyperbaric oxygen therapy; SNQ, signal-to-noise quotient.
Discussion
The main finding of this study is that patients who received adjuvant HBOT after ACLR exhibited grafts with lower signal intensity on MRI compared to those who did not receive HBOT. Additionally, HBOT-treated patients showed lower bone marrow edema adjacent to the graft-bone tunnel interface. These observations suggest that HBOT may support graft maturation and integration during the early stages of healing.
HBOT is a medical treatment modality that involves exposing patients to 100% oxygen at elevated pressures, typically between 2 and 2.5 ATA. 49 Physiologically, HBOT is based on 2 mutually dependent fundamental principles: increased environmental pressure and hyperoxia. The elevated pressure in a hyperoxic environment enhances the diffusion of oxygen into the plasma, significantly increasing oxygen delivery to hypoxic tissues. 30 Additionally, the hyperoxic environment not only influences the amount of oxygen dissolved in the plasma but also triggers several therapeutic effects, including modulation of reactive oxygen species production, regulation of inflammation, promotion of neovascularization, and modulation of extracellular matrix metabolism.6,23,33,49 Together, these mechanisms help explain how HBOT may support and accelerate tissue healing. HBOT is a safe treatment modality typically administered in specialized chambers, which can be either monoplace (for individual patients) or multiplace (for multiple patients). Although no significant clinical differences in outcomes have been reported between the two, monoplace chambers, despite being potentially more claustrophobic, offer advantages such as lower cost, increased privacy, and the ability to deliver more personalized care. 20 In this study, all patients in the HBOT group received treatment in monoplace chambers. The sessions were overall well tolerated by all patients. Mild ear discomfort was reported in a few cases, but it did not prevent any patient from completing the treatment. This finding aligns with previous literature, which identifies ear barotrauma, particularly ear discomfort, as the most commonly reported adverse effect of HBOT. 58 Noteworthy, while HBOT is generally considered a safe intervention, other potential complications have been described, including sinus barotrauma, transient myopia, and pulmonary oxygen toxicity. Rare cases of oxygen-induced seizures have also been reported. No such complications were observed in our cohort, but these risks highlight the importance of proper patient screening and monitoring during HBOT administration.17,40,58
Understanding the actual role of HBOT in tendon and ligament injuries remains uncertain, as most studies have been conducted in preclinical settings.2,14,16,18,19,29,46 In tendinopathy models, HBOT at 2.5 ATA for 2 hours a day over 30 days has been shown to improve collagen alignment and structure, reduce inflammation, increase vascularity, and enhance tendon load capacity in rabbits. 16 Additionally, HBOT promoted patellar tendon healing by increasing type I procollagen levels following injury in rats exposed to 2 ATA for 1 hour a day, for 10 days. 18 In models of ligament injury, HBOT has been found to enhance scar tissue formation, promote collagen type I synthesis, and strengthen biomechanical properties of the transected medial collateral ligament (MCL) in rats exposed to 2.5 ATA, 2 hours a day, for 5 to 10 days.2,14,29,46 Notably, in rats with an MCL transection, exposure to 1.5 ATA for 30 minutes a day over 10 days resulted in signs of improved ligament tissue repair. Increasing the exposure to 2.0 ATA for 30 minutes further enhanced healing, and extending the duration to 1 hour per session yielded even better results, suggesting a positive relationship between pressure, duration, and the healing process. 19 The limited clinical trials available have shown that, in patients with acute MCL injuries treated conservatively, HBOT at 2 ATA for 1 hour over 5 or 10 days improved pain, swelling, and range of motion, as well as accelerated recovery, including a faster return to play in rugby players.44,54
While no clinical studies to date have reported the use of HBOT for ACL tears, preclinical research suggests potential benefits.22,46,56 In a rat model of ACL transection, daily HBOT sessions at 2.5 ATA for 2 hours over 5 consecutive days did not result in macroscopic signs of spontaneous healing. However, increased levels of tissue inhibitor of metalloproteinases and type I collagen were observed in the ligament, suggesting that HBOT may modulate the molecular environment during ACL healing. 46 Additionally, in a rabbit model of ACLR, HBOT administered at 2.5 ATA for 2 hours daily over 5 days significantly improved graft integration and mechanical strength compared to animals maintained under ambient air. 56 A more recent study assessing imaging outcomes in rabbits undergoing ACLR and adjuvant HBOT at the same pressure and duration demonstrated enhanced biomechanical properties of the graft, reduced tunnel widening, and decreased graft signal intensity on MRI, findings indicative of improved graft integration and maturation. 22 In line with these preclinical results, our current study, using a comparable HBOT protocol (2.5 ATA for 90 minutes daily over 5 days), found that patients who received adjuvant HBOT exhibited lower graft signal intensity on MRI compared to controls.
MRI is a commonly used tool to assess graft healing following ACLR, with previous studies supporting its utility in monitoring graft maturation through postoperative signal intensity changes.* During the ligamentization process, graft signal intensity on MRI typically increases within the first month, peaks around 6 months, and gradually declines between 12 and 48 months, reflecting the biological progression of graft maturation.15,35 Mechanistically, the initial hyperintense signal is primarily attributed to revascularization and elevated water content, which gradually decreases over time as the graft matures, indicating the transition from granulation tissue to collagen fibers. 34 Elevated graft signal intensity in the early postoperative period has been associated with inferior graft mechanical properties, increased residual knee laxity, and a higher risk of graft rupture.5,53 Recent studies further support this association and have demonstrated that higher graft signal intensity correlates with an increased incidence of rupture at longer follow-ups,35,55,57 with 1 study reporting that a hyperintense graft on MRI performed during the second postoperative year was associated with a 12-fold greater likelihood of rupture. 35 However, it is important to note that this evidence is not entirely uniform. Some studies have shown limited correlation between MRI signal and functional or clinical outcomes, including inconsistencies between histologic parameters and SNQ-based assessments. This may indicate that graft appearance on MRI should be interpreted as supportive evidence of graft remodeling rather than a definitive predictor of function.24,52 In our study, MRI assessments were focused on the 4-month postoperative period, which represents a transitional phase between the early reparative stage at 3 months and the peak graft signal intensity typically seen around 6 months after ACLR. Research has suggested that graft signal intensity within the first 6 months postsurgery may provide a more reliable prediction of future residual knee laxity compared to later follow-ups.5,37,38,53 Furthermore, studies indicate that MRIs conducted at 3 months might be less influenced by the peak hyperintensity observed at 6 months, making early assessments potentially more precise in identifying initial graft deterioration and residual laxity. 5 By examining the 4-month time frame, this study aimed to capture a pivotal stage in graft maturation while avoiding the confounding effects of later-phase hyperintensity.
Besides ligamentization, the graft must undergo progressive osteointegration to ensure long-term stability. This complex and gradual process involves the development of a fibrous interface between the graft and bone tunnel, where collagen fibers resembling Sharpey-like fibers anchor the graft to the surrounding bone.12,39,41 These fibers, first detected as early as 3 to 4 weeks postsurgery, are considered the earliest histologic sign of osseous integration and correlate with pullout strength.11,39,41 However, only a dense concentration of Sharpey fibers, initially composed of type III collagen that matures into type I, constitutes a solid indirect tendon insertion.11,45,50 Their presence is not uniform until late healing stages, and solid integration is characterized by low cellularity, minimal vascularity, and a collagen-rich matrix, appearing as low signal intensity on MRI. 43 Histologic analysis of human ACL hamstring autografts collected 12 to 15 weeks postsurgery demonstrated early tendon-to-bone integration with fibroblast proliferation, woven bone formation, and fiber continuity across the interface. 39 Failure in this osteointegration process can lead to inadequate graft fixation and failure, and HBOT has shown promise in enhancing osteointegration.22,56 Indeed, preclinical studies revealed that, compared to air ambient controls, HBOT-treated grafts demonstrated more organized fibrovascular tissue, greater fibroblast proliferation, and denser, well-structured Sharpey-like fibers bridging the graft and bone. 56 Additionally, HBOT has been found to reduce bone tunnel diameters to below the initial drill size, increase bone mineral density, and improve graft tensile strength, findings that suggest a beneficial role in promoting early and effective graft osteointegration. 22 Consistent with these preclinical findings, our study demonstrated that HBOT-treated patients exhibited lower bone marrow edema adjacent to the graft-bone tunnel interface, further supporting the potential of HBOT to enhance graft osteointegration.
This study has limitations. First, it is a retrospective study, which is subject to inherent biases, such as the inability to control for confounding factors. Additionally, while 2 groups were analyzed, the lack of randomization could introduce selection bias. We attempted to mitigate this by matching patients from both groups, although this does not fully replicate the benefits of randomization. Another limitation is that the control group was not exposed to sham HBOT, which would involve placing patients inside the chamber with slightly increased pressure without any increase in oxygen concentration. This could introduce bias, particularly in clinical assessments, as it does not account for the placebo effect that HBOT may generate. However, since our study focused primarily on imaging parameters, which are objective measurements, the placebo effect is less likely to influence the results. Furthermore, this study evaluated the effects of HBOT following ACLR using hamstring grafts only. Other graft types may respond differently and warrant further investigation. Moreover, variability in SNQ measurements was higher than what has been reported in some previous studies.8,32 This variation may reflect differences in MRI acquisition parameters, region of interest definition, graft type and tensioning technique, or rehabilitation protocols. Although absolute SNQ values differ across studies due to methodological heterogeneity, the relative differences observed between the HBOT and control groups in our sample were consistent and formed the basis for the comparative analysis. MRI scans were acquired on both 1.5-T and 3-T systems, which could introduce some variability, although standardized imaging protocols were used, and prior studies have shown that relative graft signal measurements are comparable across field strengths.4,51 On the other hand, to our knowledge, this pilot study is the first to evaluate the use of HBOT in patients undergoing ACLR, and its promising results provide a foundation for future protocol development. By enhancing graft healing and osteointegration, HBOT may shorten the time required for patients to return to sports and normal activities and reduce the risk of failure. Moreover, its anti-inflammatory effects may also reduce postoperative pain and improve patient comfort. Further studies evaluating the clinical outcomes after HBOT are ongoing. Furthermore, HBOT does not require any changes to the surgical technique and is compatible with other mechanical or biological augmentation strategies, if indicated. While these results suggest a potential benefit of adjuvant HBOT on ACL graft healing, definitive conclusions require larger randomized controlled trials incorporating imaging, patient-reported outcomes, and biomechanical assessments.
Conclusion
In this pilot study, adjuvant HBOT following ACLR was associated with significantly improved signs of graft maturation and integration, as evidenced by lower graft signal intensity and interface signal on MRI at 4 months, suggesting accelerated early healing. These findings may help refine strategies to enhance early graft incorporation and support the development of rehabilitation protocols that optimize postoperative outcomes for patients undergoing ACLR.
Footnotes
Final revision submitted November 30, 2025; accepted December 6, 2025.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Instituto de Ortopedia e Traumatologia, Hospital Das Clinicas, Faculdade de Medicina, Universidade de São Paulo (reference number 1671).

