Abstract
Background:
It has been demonstrated that exogenous testosterone can lead to higher rates of tendinous rupture and that injectable testosterone replacement therapy (TRT) is associated with more systemic complications. While this is the case, there are few studies exploring associations between injectable TRT and complications after anterior cruciate ligament reconstruction (ACLR) surgery.
Purpose/Hypothesis:
The purpose of this study is to evaluate the association between preoperative injectable TRT and ACLR revision surgery. It was hypothesize that ACLR revision surgery rates will be higher in patients undergoing TRT preoperatively compared with a matched cohort.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A retrospective cohort of patients who underwent ACLR between 2015 and 2022 was obtained through the PearlDiver Database (Mariner). Patients with a history of ACLR, 2 years of follow-up, and injectable TRT use before ACLR were identified using International Classification of Diseases, 10th Revision, and Current Procedural Terminology codes. Three TRT usage intervals were analyzed to evaluate ACLR revision rates in those with usage of injectable TRT compared with those with propensity score–matched demographics and Charlson Comorbidity Index with no history of injectable TRT use who underwent ACLR. Cohorts were via univariate analysis, chi-square tests, Student t tests, and log-rank tests.
Results:
TRT use ≤1 year before primary ACLR surgery demonstrated a 17.05% (66/387; mean age, 40.3) ACLR revision rate compared with 5.17% (20/387; mean age, 40.8) in the matched cohort control group (P < .001), yielding a relative risk of revision surgery of 3.30 (95% CI, 2.04-5.33). Statistical analysis for distant TRT use >1 year before surgery (n = 160) was underpowered but supported trends toward increased revision risk with TRT exposure with a 14.38% revision rate (n = 23). TRT users with any exposure before or after surgery (n = 2614) had similar revision rates (n = 168; 6.36%) compared with the control group (n = 164; 6.21%); the difference was not significant (P = .82).
Conclusion:
Our study demonstrated that injectable TRT use within 1 year of primary ACLR is associated with a >3-fold increase in ACLR revision rates with 2 years of follow-up. Physicians should consider incorporating this increased risk into patient counseling during shared decision-making discussions, as this correlation may influence clinical and surgical decision making for patients with previous ACLR.
Anterior cruciate ligament (ACL) injuries are one of the most common knee injuries in young active patients, accounting for approximately 200,000 injuries in the United States per year.9,14 ACL reconstruction (ACLR) is a vital procedure for those patients who wish to return to high-level pivoting and cutting sports. Traditionally, ACLR is a successful procedure with an overall revision rate of <5% at 2 years of follow-up. Of those who undergo revision ACLR surgery, over half are because of graft rupture.16,20 Therefore, perioperative optimization is essential for patients who undergo ACLR to provide excellent outcomes and try to prevent postoperative graft rupture and the need for revision surgery.8,23,32,34
Testosterone replacement therapy (TRT) has increased dramatically over the past several decades, specifically in the active 18- to 45-year-old male age group.6,31 In addition to its traditional clinical role in treating symptomatic hypogonadism, testosterone has seen a recent increase in use in “gender-affirming” hormone therapy and as an illicit substance used by athletes over the past decade.5,13,21,26
TRT has been associated with various systemic complications, with some studies suggesting that oral and injectable formulations may carry a higher risk of adverse cardiovascular events than some other formulations.1,15 This may be due to the varying basal serum concentrations, washout periods, and supratherapeutic concentration intervals. 7 However, these findings have been inconsistently replicated and remain underdocumented in current literature. In addition to systemic effects, exogenous testosterone and anabolic steroid use have been well-documented to increase the risk of tendinous rupture in both athletes and the general population.2,25,28 On a molecular level, testosterone has been shown to elevate collagen turnover and dysplasia, leading to increased stiffness and reduced stress adaptability of native muscles and tendons.12,29 While many studies have analyzed TRT in relation to musculotendinous health, only 1 other study 24 has specifically investigated the effects of TRT concerning ACLR revision rates. No studies have investigated the effects of specific TRT formulations on ACLR revision rates, let alone the effects of different TRT formulations on musculotendinous structures as a whole.
The purpose of this study was to investigate the relationship between preoperative injectable TRT usage and the rate of ACLR revision using a series of data analyses stratified by exposure intervals to gather the overall influence of TRT over time. We hypothesized that patients with any preoperative injectable TRT exposure would have higher revision surgery rates than a matched cohort without TRT exposure.
Methods
Patient Selection
This was a retrospective cohort study carried out through the PearlDiver Database (MARINER). The data were queried utilizing International Classification of Diseases, 10th Revision (ICD-10) diagnosis and procedure codes as well as Current Procedural Terminology (CPT) codes. This study was exempt from institutional review board approval because all data were made available to researchers on a deidentified basis.
The 2015 to 2022 data were queried in April of 2024 using ICD-10 procedure codes to identify patients who underwent primary ACLR from October 2015 to December 2022. Patients who had not reached 2 years of postprocedural follow-up time by the data extraction date were excluded from the analysis. The laterality of the procedure was determined by assessing the associated ICD-10 diagnosis in conjunction with the CPT code and then assessed for revision CPT code with the same laterality. TRT was evaluated with CPT codes for injections completed within a specific time frame from their primary ACLR surgery.
We delegated those with a history of TRT use >1 year prior to surgery as “distant use” and those that had used TRT ≤1 year prior as “recent use.” Our data analyses were stratified accordingly to help assess the temporal relationship between TRT and ACLR with the granularity of the data made available to us.
Patients were included if they had ≥2 years of follow-up. Patients were excluded if they had any coded soft tissue diseases or rheumatologic diseases such as Marfan syndrome, Ehlers-Danlos syndrome, systemic lupus erythematosus, rheumatoid arthritis, Sjogren syndrome, polymyositis, dermatomyositis, and any form of mitochondrial disease. A complete list of relevant codes used to retrieve patient records, along with inclusion and exclusion criteria, can be found in Appendix Tables A1 and A2.
Statistical Analysis
Experimental and control groups were propensity score matched with nearest matching by age, sex, and the Charlson Comorbidity Index (CCI). The “any use” cohort was successfully matched for age, sex, and CCI with standardized mean differences of .015, 0, and .021, respectively, and the “recent use” cohort was also matched with standardized mean differences of .011, 0, and .064, respectively. The risk for ACLR revision was stratified by TRT use interval as determined by the last documented injection of TRT. Distant TRT use was analyzed by isolating all patients who had their last documented dose of TRT >1 year before the index procedure. The second interval included TRT users who had a last-documented injection ≤1 year of the surgery. We also analyzed the effect of TRT by including patients with any perioperative TRT exposure.
Statistical significance was set a priori at.05. Cohorts were compared using univariate analysis, chi-square tests, and Student t tests to compare demographics and outcomes between groups. A log-rank test was used to compare overall survival rates. All statistical analyses were performed using the R Statistical Package within the PearlDiver user interface.
Results
The calculated sample sizes for each TRT use interval were n = 160 patients for distant TRT use >1 year before the index procedure, n = 387 patients for recent use ≤1 year before, and n = 2641 patients for any TRT use before or after the index procedure. Populations for each TRT usage interval were matched in terms of age, sex, and CCI (Tables 1 and 2).
Any Testosterone Replacement Therapy Exposure Versus Control Demographics and Comorbidities a

Data are reported as n (%) unless otherwise indicated. P values reported in bold indicate statistical significance. CCI, Charlson Comorbidity Index; TRT, testosterone replacement therapy.
PearlDiver does not publish populations with <11 members.
Testosterone Replacement Therapy Exposure Within 1 Year Versus Control Demographics and Comorbidities a

Data are reported as n (%) unless otherwise indicated. P values reported in bold indicate statistical significance. ACLR, anterior cruciate ligament reconstruction; DVT, deep venous thrombosis; PE, pulmonary embolism; SSI, surgical-site infection; TRT, testosterone replacement therapy.
PearlDiver does not publish populations with <11 members.
Patients with documented TRT use ≤1 year before the index procedure had a significantly increased risk for ACLR revision, with n = 66 (17.05%) patients undergoing ACLR revision in comparison with only n = 20 (5.17%) in the control group, yielding a relative risk of 3.30 for ACLR revision (P < .001; 95% CI, 2.04-5.33) (Table 3). A log-rank survival curve demonstrates a significant difference in group outcome rates that is apparent approximately 400 days after index ACLR (P < .001) (Figure 1).
Testosterone Replacement Therapy Exposure Within 1 Year Versus Control Revision Anterior Cruciate Ligament Reconstruction and Complications a

Data are reported as n (%) unless otherwise indicated. P values reported in bold indicate statistical significance. ACLR, anterior cruciate ligament reconstruction; DVT, deep venous thrombosis; PE, pulmonary embolism; SSI, surgical-site infection; TRT, testosterone replacement therapy.
PearlDiver does not publish populations with <11 members.

Kaplan-Meier survival analysis comparing anterior cruciate ligament reconstruction (ACLR) revision-free survival between patients who received testosterone replacement therapy (TRT) within 1 year before surgery (red lines) and a matched control group with no TRT exposure (blue lines). Solid lines represent estimated survival probabilities; dashed lines indicate 95% CIs. Treat, treatment.
The cohort with distant TRT use >1 year before the index procedure experienced n = 23 revisions (14.38%) but the control group had too few occurrences of ACLR revision to qualify for a sufficiently powered analysis. However, assuming the highest possible number of patients for the control group given PearlDiver's data disclosure policy (n = 11) would demonstrate a >2-fold increased rate for ACLR revision. Groups consisting of <11 patients are not disclosed by PearlDiver for preservation of patient anonymity.
When accounting for any TRT use before or after the index procedure, there were similar incidences of ACLR revision in the TRT cohort (n = 168; 6.36%) and the control group (n = 164; 6.21%); these values were not significantly different (P = .82; 95% CI, 0.83-1.26) (Table 4). To investigate the isolated effect of postoperative TRT, we conducted a subgroup analysis only including those who had started TRT ≤6 months after their index procedure. The analysis yielded only 110 patients in each group and was underpowered due to the PearlDiver data disclosure policy.
Any Testosterone Replacement Therapy Exposure Versus Control Revision Anterior Cruciate Ligament Reconstruction and Complications a
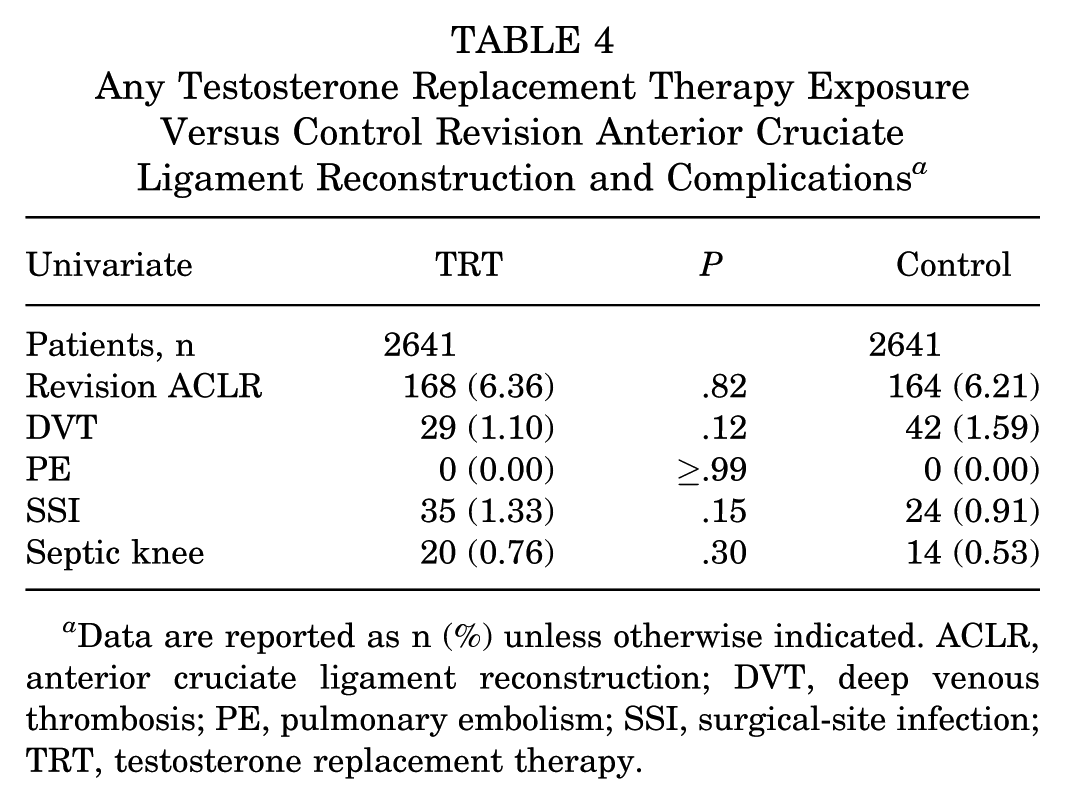
Data are reported as n (%) unless otherwise indicated. ACLR, anterior cruciate ligament reconstruction; DVT, deep venous thrombosis; PE, pulmonary embolism; SSI, surgical-site infection; TRT, testosterone replacement therapy.
Discussion
The major findings of our study demonstrate a 330% increase in risk for ACLR revision surgery for patients who have received ≥1 injection of TRT ≤1 year of their primary ACLR; the revision rates between TRT exposure and control groups are 17.05% and 5.17%, respectively. While not statistically assessable because of data disclosure limitations from PearlDiver, our results for distant TRT use >1 year before the procedure suggest that TRT injection may be associated with long-term risks. To our knowledge, this is the first study to demonstrate a direct association between ACLR revision rates and TRT use before primary ACLR. Additionally, it is one of the few studies to investigate the effects of testosterone on nonnative musculotendinous structures.
TRT is frequently prescribed to patients for its propensity to increase energy levels and improve libido, which significantly improves the quality of life for symptomatic hypogonadal men. 3 Other benefits include increased fat catabolism and increased muscular anabolism which serve to improve physical performance, cosmetic appearance, and self-esteem. 3 However, TRT use also comes with its own set of drawbacks ranging from mild inconveniences to increased risks of life-threatening conditions. Common adverse effects include increased or worsening acne vulgaris, new-onset mental health conditions, and reductions in fertility.3,22 Exposure has been associated with increases in several major health risks including stroke, acute coronary syndrome, and all-cause mortality.17,22
Given there are several existing formulations of TRT with varying pharmacokinetic profiles and routes of administration, differences in effect duration and toxicity are important to consider when evaluating health risks. There are several forms of both short-acting and long-acting testosterone; most long-acting forms are either administered subcutaneously or intramuscularly while most short-acting forms are administered in transdermal, oral, buccal, or intranasal fashions.11,15 Topical gels and patches have been reported to provide the most consistent basal testosterone levels, while injections exhibit higher peak-to-trough ratios, resulting in less time within the therapeutic range. 7 Likely for this reason, transdermal applications have been found to have fewer adverse systemic effects such as cardiac events and mortality in comparison with injected formulations.7,15 Some injectable formulations can remain detectable in the body for several months and can cause even longer-lasting endocrine effects (ie, azoospermia), lasting up to 15 months after the previous dose. 11 This prolonged presence may contribute to increased rates of musculoskeletal morbidity and complications as suggested by the data in our study. Existing literature analyzing rates of musculoskeletal morbidity between formulations or routes of administration is sparse, although injections have been associated with more dramatic increases in muscle mass. 4
Several other studies have analyzed the effects of TRT on musculotendinous structures of the knee, but none has attempted to analyze the effects of individual TRT formulations. A 2024 study by Quinn et al 24 performed a retrospective cohort study to quantify the association between TRT use with both primary ACL rupture and reinjury after ACLR. The study demonstrated an increase in primary ACL rupture rates with TRT use but determined there was no difference in ACLR revision rates. 24 Our analysis of the cohort with any perioperative TRT exposure supports their findings. However, their study only focuses on postoperative TRT use and does not specifically include any cohort of patients using TRT before ACLR. Our study found it vital to include this cohort to assess patients who underwent surgery with elevated basal serum TRT levels, where the risk of adverse musculotendinous events is the highest. Additionally, Quinn et al did not attempt to stratify for TRT formulation. Given that different forms of TRT have widely variable half-lives, peak serum concentrations, and washout periods, the deleterious effects of more potent or long-acting formulations (ie, injectable) may have been diluted in their statistical analysis that included short-acting formulations.4,7,11,15 Additionally, our study has documented verification that each patient has received his TRT dose, optimizing internal validity that may have contributed to the statistical significance of our results. Briefly, the findings of the current study build upon theirs likely given our methodological differences that account for the stratification of injectable TRT formulations, preoperative TRT exposure, and verified in-clinic TRT use.
The results of our study, along with the established effects of testosterone and its metabolites on bone, ligament, and tendon health, prompt us to explore plausible mechanisms behind the observed increase in ACLR revision risk. From a simplified surgical perspective, an ACLR typically stems from failure in ≥1 of 3 major locations: the osseous tunnel, the bone-graft interface, or the graft itself. Testosterone and its metabolites (including estrogen) have a direct positive effect on bone health by modulating stress-induced bone remodeling, leading to the creation of stronger and denser bones that are more efficacious at distributing mechanical force exerted upon musculotendinous implants. 19 The bone-graft interface is also positively affected by testosterone on a molecular level, demonstrating increased extracellular matrix production and higher bone-graft contact surface areas in vivo.10,30 However, as previously mentioned, the effects of testosterone and related metabolic products on musculotendinous integrity have been well-described as deleterious. TRT has been shown to increase collagen turnover and dysplasia, leading to higher stiffness and lower stress adaptability of native muscles and tendons.12,29 Several other studies have also described other negative biophysical consequences of musculotendinous remodeling including dysregulated matrix metalloproteinase activity, disproportionate muscle and tendon growth rates, attenuated muscle response to relaxin, and altered cell metabolism.10,12,18,29 These deleterious effects have widely been accepted as the major mechanism for increased rates of native muscle injury. It must also be noted in this context that a 2017 study by Wu et al 33 demonstrated that perioperative TRT supplementation is associated with increased quadriceps muscle mass and preservation of strength. However, these studies only demonstrated significant differences in muscular biomechanical properties and not patient-oriented outcomes while placing patients at an increased risk for testosterone-associated complications. Preserved muscle strength theoretically aids postoperative recovery but its role in clinical outcomes has yet to be proven as positive.
Because TRT has been documented to enhance the biomechanical properties of bone and facilitate stronger bone-graft interface formation, its well-documented deleterious effects on musculotendinous structures, either preoperatively on the native muscle in vivo or postoperatively on the graft, cannot be overlooked as a potential explanation for the increased ACLR revision risks in our study. However, the strength of this conclusion must be weighed against the presence of potential confounders that were not accounted for in our matching process, including differences in graft type, surgical technique, and patient activity level between groups.
Currently, because only preoperative exposure to TRT has been associated with increased ACLR revision risk, physicians should consider incorporating the increased risk into patient counseling. This information should be discussed during the shared decision-making process when determining the most appropriate and patient-centered management for ACL injury for TRT users. Additionally, physicians may consider TRT cessation for up to 1 year in the elective ACLR setting. Delaying ACLR in the hopes of allowing a full injectable TRT washout period, if medically and logistically feasible, may be recommended, as long-term outcomes between early and delayed ACLR are negligible. 27 More studies must be conducted to translate the significance of these data into actionable items that will improve patient outcomes. First, the pathophysiology behind the increased revision risk should be firmly established so appropriate interventions or preventative measures can be recommended. Additionally, it should be determined which graft types for ACLR are most susceptible to revision after long-term TRT exposure (allograft vs autograft, patellar tendon vs quadriceps tendon, etc). Finally, because our data granularity precluded the quantification of TRT dose exposure, further research should focus on establishing a dose- and formulation-dependent relationship between TRT use and the risk of poor outcomes in ACLR to work toward establishing causality and defining the bounds of safe TRT usage in a surgical context. 15
Limitations
This study has several limitations. The study design additionally relies on consistent and accurate practitioner CPT and ICD-10 coding for cohort generation, accuracy of prescriptions, and appropriate surgical categorization. Additionally, the indications for both testosterone therapy and ACLR revision surgery were not specified in CPT coding, which potentially dilutes the strength of the results given that causes of revision may not specifically indicate graft failure. Further, the nature of a US-based claims database limits the external validity of our results given that TRT prescription practices and ACLR techniques may differ between other patient populations. The most limiting factor in our study design was the lack of power for some of the temporal subgroup analyses. Given that CPT coding does not account for TRT self-administration out of office, some injection TRT users were missed by our database query, which potentially led to insufficient power in our distant use TRT cohort. Finally, the study design cannot account for potential confounding from the hypothesis that patients seeking TRT may be generally more physically active and therefore more susceptible to graft injury, a sentiment echoed by similar studies.24,28
Conclusion
Our study demonstrated that injectable TRT use within 1 year of primary ACLR is associated with a >3-fold increase in ACLR revision rates with 2 years of follow-up. Physicians should consider incorporating this increased risk into patient counseling during shared decision-making discussions, as this correlation may influence clinical and surgical decision making for patients with previous ACLR.
Footnotes
Appendix
Exclusion Criteria via ICD-10 Codes a

| Exclusion Criteria Diagnosis | ICD-10 Code |
|---|---|
| Marfan syndrome | ICD-10-D-Q8740, ICD-10-D-Q87410, ICD-10-D-Q87418, ICD-10-D-Q8742, ICD-10-D-Q8743 |
| Ehlers-Danlos syndrome | ICD-10-D-Q796, ICD-10-D-Q7960, ICD-10-D-Q7961, ICD-10-D-Q7962, ICD-10-D-Q7963, ICD-10-D-Q7969 |
| Rheumatoid arthritis | ICD-10-D-M069, ICD-10-D-M0579, ICD-10-D-M0609, ICD-10-D-M059, ICD-10-D-M0600, ICD-10-D-M0689, ICD-10-D-M0589, ICD-10-D-M0540, ICD-10-D-M0800, ICD-10-D-M0570, ICD-10-D-M0680, ICD-10-D-M0580, ICD-10-D-M083, ICD-10-D-M0569, ICD-10-D-M0550, ICD-10-D-M0560, ICD-10-D-M06861, ICD-10-D-M05761, ICD-10-D-M06862, ICD-10-D-M06061, ICD-10-D-M0820, ICD-10-D-M05762, ICD-10-D-M06062, ICD-10-D-M0809, ICD-10-D-M0559, ICD-10-D-M05712 |
| Systemic lupus erythematosus (SLE) | ICD-10-D-M329, ICD-10-D-M3210, ICD-10-D-M3219, ICD-10-D-M328, ICD-10-D-M3214, ICD-10-D-M320, ICD-10-D-M3213, ICD-10-D-M3212, ICD-10-D-M3215, ICD-10-D-M3211 |
| Sjogren syndrome | ICD-10-D-M3500, ICD-10-D-M3501, ICD-10-D-M3502, ICD-10-D-M3503, ICD-10-D-M3504, ICD-10-D-M3509 |
| Dermatomyositis | ICD-10-D-M3310, ICD-10-D-M3313, ICD-10-D-M3312, ICD-10-D-M3300, ICD-10-D-M3319, ICD-10-D-M3311, ICD-10-D-M3302, ICD-10-D-M3309, ICD-10-D-M3301, ICD-10-D-M3303 |
| Polymyositis | ICD-10-D-M3320, ICD-10-D-M3321, ICD-10-D-M3322, ICD-10-D-M3329, ICD-10-D-M3390, ICD-10-D-M3391, ICD-10-D-M3392, ICD-10-D-M3393, ICD-10-D-M3399 |
| Mitochondrial disease | ICD-9-D-27787, ICD-10-D-E8840, ICD-10-D-E8849, ICD-10-D-G713 |
ICD-10, International Classification of Diseases, 10th Revision.
Final revision submitted July 28, 2025; accepted September 12, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.S. is a board or committee member of AOSSM and American Shoulder and Elbow Surgeons, is a paid consultant for Arthrex Inc, and receives royalties from Medacta. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
