Abstract
Background:
Pediatric meniscal injury rates and their arthroscopic repair are on the rise. With the growing popularity of all-inside devices used in this potentially higher-risk population, studies examining the proximity of these devices to neurovascular structures are needed.
Purpose/Hypothesis:
The purpose of this study was to provide distances to neurovascular structures from all-inside devices placed arthroscopically at both high-risk approach angles and zones of the lateral menisci of pediatric cadaveric specimens. It was hypothesized that the neurovascular structures would be situated closely to arthroscopically placed all-inside lateral meniscal devices, particularly the popliteal artery (PA), popliteal vein (PV), and tibial nerve (TN) to devices placed at the posterior root when placed via a lateral portal, as well as the peroneal nerve (PN) to devices placed just medial to the popliteal hiatus via a medial portal.
Study Design:
Descriptive laboratory study.
Methods:
The study involved 12 fresh-frozen cadaveric knee specimens (6 matched pairs) between the ages of 6 and 10. Two all-inside meniscal repair implants were placed in the lateral meniscus: 1 via a medial port just medial to the popliteal hiatus and 1 via a lateral port at the visible edge of the posterior root. Specimens were then dissected posteriorly to expose the posterior knee capsule and meniscal implants. The distance measurements between the implants and the PA, PV, TN, and PN were recorded.
Results:
The distances from the anchors to the PA, PV, TN, and PN ranged from 2.3 to 59.8 mm, 3.5 to 58.0 mm, 5.3 to 63.0 mm, and 6.3 to 40.1 mm, respectively. Implants at the meniscus root were closer to the PA, PV, and TN than implants at the popliteal hiatus (P≤ .001 for all). There was no statistically significant difference in distances to the PN between the 2 implant positions (P = .06).
Conclusion:
This study illustrates the proximity of neurovascular structures to all-inside devices in pediatric knee specimens, particularly those directed at the posterior horn using the anterolateral portal. Under the conditions of this study, all-inside devices appeared to be safe.
Clinical Relevance:
This study provides much-needed data to the growing body of knowledge regarding meniscal injuries and repair in the pediatric population.
Keywords
Meniscal injury rates in skeletally immature patients are increasing rapidly.2,6,19,24,27 In the pediatric population, arthroscopic treatment of meniscal tears, particularly those of the lateral meniscus, is becoming increasingly common for both isolated tears and those occurring in the context of a discoid meniscus or anterior cruciate ligament injury.8,12,13,15,22,26,30
The menisci play a crucial role in shock absorption, reduction of femorotibial contact forces, and as secondary stabilizers within the knee joint. 29 Partial or total meniscectomies can lead to early joint degeneration and subsequent osteoarthritis.16,20,28 Moreover, there is evidence that suggests meniscal repair in the pediatric population is more efficacious than in the adult population.14,18,24,27 For these reasons, arthroscopic meniscal treatment should aim to preserve the functioning meniscus, particularly in pediatric patients.
Inside-out and outside-in meniscal repair have been considered the gold standard for meniscal fixation.17,31 However, all-inside fixation has gained prominence in the arthroscopic field because of its relative ease of implantation, reduced surgical time, and decreased risk of complications.11,24 The latest generation of all-inside devices seeks to combine the strength of inside-out repair with the advantages of the all-inside technique. 9 Many all-inside devices rely on hollow needle penetration of the meniscus and joint capsule that may penetrate well beyond the posterior limits of the meniscus during repair. While inside-out meniscal repair allows for placement of retractors that may protect neurovascular structures posterior to the knee joint, 11 all-inside repair does not utilize this technique, which may put neurovascular structures at risk.
Prior pediatric imaging studies have demonstrated that neurovascular structures and menisci, particularly the popliteal and posterior areas of the lateral meniscus, are in close proximity.3,23,25 Moreover, certain arthroscopic approach angles may place all-inside meniscal devices even closer to neurovascular structures. 25 While there are both imaging10,21,33 and anatomic 1,5,7 studies in the adult population that address vascular safety of all-inside devices, there remains a paucity of comparable anatomic studies in the pediatric population. 32
The purpose of this study was to provide distances to neurovascular structures from all-inside devices placed arthroscopically at both high-risk approach angles and zones of the lateral menisci of pediatric cadaveric specimens. We hypothesized that the neurovascular structures would be situated closely to arthroscopically placed all-inside lateral meniscal devices, particularly the popliteal artery (PA), popliteal vein (PV), and tibial nerve (TN), to devices placed at the posterior root when placed via a lateral portal; and the peroneal nerve (PN) to devices placed just medial to the popliteal hiatus via a medial portal.
Methods
This cadaveric study was considered exempt from institutional review board oversight and approval because of the following criteria: (1) cadaveric tissue was utilized, (2) donor families gave consent to use tissue for research purposes, (3) no genetic information was obtained, and (4) there was no additional contact with the donor families.
Specimen Preparation and Device Placement
This cadaveric study involved 12 fresh-frozen cadaveric knee specimens (6 matched pairs) of 3 male and 3 female children between the ages of 6 and 10 years, encompassing the entire knee joint from the distal half of the femur to the proximal half of the tibia, along with all surrounding tissues except the skin and subcutaneous tissue. All specimens were sourced from an allograft harvest facility.
Prior to the study, the knees were appropriately thawed to enable unrestricted joint movement. Knee arthroscopy was then performed, and all-inside meniscal repair devices were placed using a depth limiter with polyetheretherketone anchors and sutures designed to capture the peripheral aspect of the meniscus, the meniscotibial ligament complex, and the meniscal capsule. The knee was maintained at 90° of flexion in all cases with varus stress applied to ensure adequate visualization of the lateral meniscus during device placement.
Stryker All Inside Repair (AIR) (second generation) (Stryker Corp) devices were placed in 2 previously identified high-risk zones and arthroscopic approach angles of the lateral meniscus (device A: just medial to the popliteal hiatus via the anteromedial portal; device B: at the visible edge of the posterior root via the anterolateral portal) (Figure 1). 25 All device anchors were deployed with a depth limiter set at 10 to 12 mm of penetration and traversed the meniscus with the intention of engaging the posterior joint capsule. Surgeons (P.W. and H.B.E.) were instructed to simulate a meniscal repair of the red/white zone and to penetrate the capsule with the repair device. Each meniscal repair device had 2 implant anchors that attached the meniscal repair suture to the posterior aspect of the meniscal/joint capsular region. For each meniscal repair device, a measurement was taken from each of the 2 implant anchors to the relevant neurovascular structures. Sutures were placed in a vertical mattress configuration. Implant placements were conducted by 2 fellowship-trained orthopaedic sports medicine surgeons in a consistent manner (P.W. and H.B.E.).

High-risk zones and approach angles of repair for the medial and lateral menisci. Red arrows indicate high-risk zones and approach angles, orange arrow indicates intermediate risk zone and approach angle, and green arrows indicate low-risk zones and approach angles as determined by Shea et al. 25 High-risk zones denoted with “A” and “B” represent the 2 high-risk zones where all-inside devices A and B were placed. AVN, artery, vein, nerve; m, muscle; n, nerve.
Device A: Medial Side of the Popliteal Hiatus
The all-inside device was placed into the joint through an anteromedial portal and inserted at the most medial corner of the popliteal hiatus with the curved tip of the needle aimed laterally toward the PN. Arthroscopic images of device placement at the popliteal hiatus can be seen in Figure 2.

Arthroscopic images of all-inside device A placement at the medial side of the popliteal hiatus via the anteromedial portal. Devices were placed into the left knee of a 10-year-old female specimen. The arthroscopic camera was inserted into the joint space via the anterolateral portal while the all-inside device was placed into the joint through an anteromedial portal and inserted at the most medial corner of the popliteal hiatus with the curved tip of the needle aimed laterally toward the peroneal nerve. Sutures were placed in a vertical mattress configuration. The knee was maintained at 90° of flexion in all cases with varus stress applied to ensure adequate visualization of the lateral meniscus during device placement.
Device B: Posterior Horn of the Lateral Meniscus
The all-inside device was placed into the joint through an anterolateral arthroscopy portal and inserted at the visible edge (within 1-2 mm) of the posterior root of the lateral meniscus with the curved tip of the needle aimed medially toward the neurovascular bundle (NVB). Arthroscopic images of device placement at the posterior horn can be seen in Figure 3.

Arthroscopic images of all-inside device B placement at the visible edge of the posterior root via the anterolateral portal. Devices were placed into the right knee of a 10-year-old female specimen. The arthroscopic camera was inserted into the joint space via the anteromedial portal, while the all-inside device was placed into the joint through an anterolateral portal and inserted at the visible edge (within 1-2 mm) of the posterior root of the lateral meniscus with the curved tip of the needle aimed medially toward the neurovascular bundle. Sutures were placed in a vertical mattress configuration. The knee was maintained at 90° of flexion in all cases with varus stress applied to ensure adequate visualization of the lateral meniscus during device placement.
Measurements
Following implantation, specimens were dissected by the same surgeons performing the repairs through a direct posterior approach to expose the posterior knee capsule and identify the implants. Dissection was performed in a systematic way with care taken not to disturb the native anatomy of the PA, PV, TN, and PN during dissection. Upon exposure of the devices, the distance measurements between the anchors and the PA, PV, TN, and PN were systematically performed by the same surgeons using digital calipers (Mitutoyo) with a nominal precision of 0.02 mm. Four caliper measurements (distance to PA, PV, TN, and PN) were taken on each specimen at each of the 4 locations of the all-inside device anchors (2 anchors per device, recorded as “first anchor” and “second anchor”) for a total of 16 possible measurements per specimen and 192 possible measurements overall. The measurements for all specimens were performed within a 4-hour period immediately after devices were placed in the specimens.
Data Collection and Statistical Analysis
Raw data were collected by hand in the cadaver labs at the allograft harvest facility by the study coordinator (H.B.G.B). Raw data were then collated and coded using Microsoft Excel. With our small sample size, determining the distribution of the distance measurements was important for choosing an appropriate statistical method. A Shapiro-Wilk test was performed and showed that the distribution of distance measurements departed significantly from normality (W = 0.915; P < .001). Based on this outcome, continuous data (ie, distance measurements from device anchors to neurovascular structures) are presented as median with IQR. Nonparametric methods were used to assess potential differences in anchor distance distributions: Mann-Whitney U test was used to assess differences by neurovascular structure (PA, PV, TN, and PN), sex (male and female), and age group (<10 and ≥10); while the Kruskal-Wallis test was used to assess differences by age- and sex-matched subgroups (<10 and female vs ≥10 and female, <10 and male vs ≥10 and male, <10 and female vs <10 and male, and ≥10 and female vs ≥10 and male). Post hoc power analyses were conducted to assess the study’s ability to detect differences in distance to neurovascular structures from implants for 3 key comparisons. First, for differences in distance to the PA, PV, TN, NVB, and PN between device A (popliteal hiatus) and device B (meniscus root), with a sample size of 12 cadaveric knee specimens and the significance level (α) corrected for multiple comparisons using the Bonferroni method (corrected α = .05/5 = .01). For the PA, the calculated effect size was 1.40 with a power of 0.725. For the PV, the calculated effect size was 2.52 with a power of 0.999. For the TN, the calculated effect size was 2.59 with a power of 0.999. For the NVB, the calculated effect size was 2.76 with a power of 0.999. For the PN, the calculated effect size was 0.189 with a power of 0.017. Overall, the analyses for PA, PV, TN, and NVB showed high power values, suggesting the study was well-powered to detect differences in these distances, whereas the analysis for PN was underpowered and should be interpreted with caution. Second, for differences in distance to all neurovascular structures from implants between <10-year and ≥10-year age cohorts, with equal sample sizes (6 for <10 years old and 6 for ≥10 years old) and α of .05, the effect size was −0.208 with a power of 0.062 for males, and the effect size was 0.810 with a power of 0.246 for females, indicating insufficient power to detect age-related differences. Third, for differences in distance to all neurovascular structures from implants between males and females, with equal sample sizes (6 males and 6 females) and α of .05, the effect size was −1.03 with a power of 0.365 for <10 years old, and the effect size was −0.005 with a power of 0.050 for ≥10 years old, suggesting the study is underpowered for detecting sex differences. A P < .05 was considered statistically significant, with Dunn-Bonferroni correction for multiple-hypothesis testing. All statistical analyses were performed using SPSS, Version 28.0.1.0 (IBM Corp).
Results
A total of 36% (69/192) anchor-to-anatomic structure measurements could not be obtained for one of the following reasons: the anchors could not be isolated, the anchors were accidentally displaced during dissection, or the native anatomic structure of the PA, PV, TN, or PN was significantly disrupted during dissection (Table 1). The distances from the anchors to the PA, PV, TN, and PN ranged from 2.3 to 59.8 mm, 3.5 to 58.0 mm, 5.3 to 63.0 mm, and 6.3 to 40.1 mm, respectively (Appendix Table A1). The median (IQR) distance of all anchors to all neurovascular structures, the NVB (mean of measurements to PA, PV, and TN), PA, PV, TN, and PN were 21.0 (12.0-44.0) mm, 28.4 (11.1-45.0) mm, 26.0 (10.3-45.0) mm, 28.8 (10.1-44.8) mm, 31.7 (12.8-48.7) mm, and 16.4 (12.1-28.0) mm, respectively (Appendix Table A1). There were no statistically significant differences in anchor distance between the groups (P > .05 for all) (Appendix Table A1).
Age, Sex, and Laterality of Specimens With Individual Distances From Suture Anchors to Neurovascular Structures (mm) a
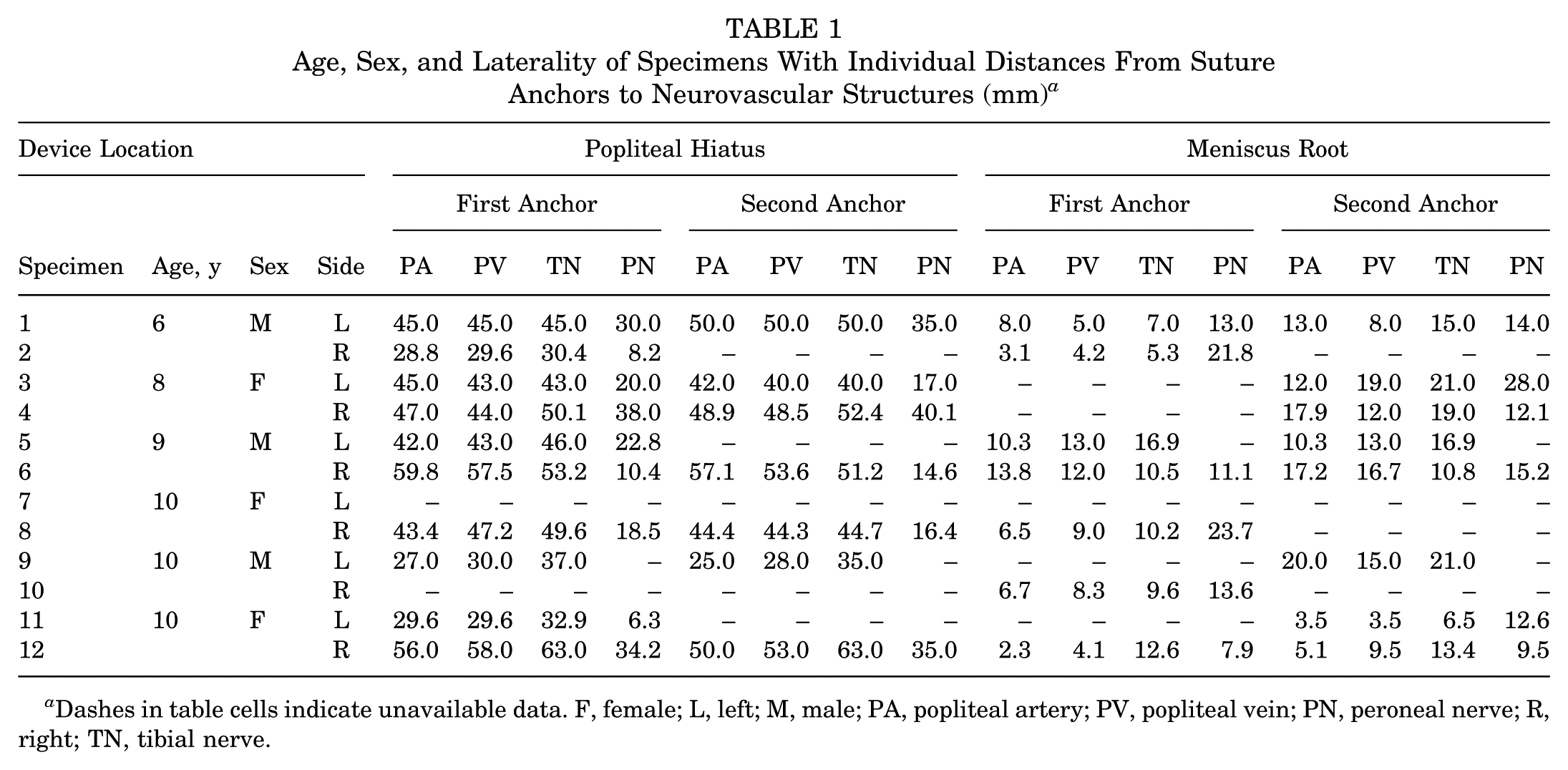
Dashes in table cells indicate unavailable data. F, female; L, left; M, male; PA, popliteal artery; PV, popliteal vein; PN, peroneal nerve; R, right; TN, tibial nerve.
Device A: Medial Side of the Popliteal Hiatus
The median (IQR) distances from both anchors to all neurovascular structures, NVB, PA, PV, TN, and PN were 41.1 (26.5-48.6) mm, 44.3 (30.4-50.1) mm, 45.0 (35.8-50.0) mm, 44.3 (35.0-51.5) mm, 46.0 (38.5-51.8) mm, and 20.0 (14.6-35.0) mm, respectively (Table 2).
Comparative Analysis of Distances (mm) of Anchors to Neurovascular Structures by Device Location a

Data are presented as median (IQR). Bold P values are statistically significant. NVB, neurovascular bundle; PA, popliteal artery; PN, peroneal nerve; PV, popliteal vein; TN, tibial nerve.
Mean measurements of the distances from the implant to the PA, PV, and TN.
Device B: Posterior Horn of the Lateral Meniscus
The median (IQR) distances from both anchors to all neurovascular structures, NVB, PA, PV, TN, and PN were 12.1 (8.2-18.5) mm, 12.0 (6.6-17.1) mm, 10.3 (5.1-13.8) mm, 9.5 (5.0-13.0) mm, 12.6 (9.6-16.9) mm, and 13.3 (11.4-20.2) mm, respectively (Table 2).
Comparative Analysis of Anchor Distances by Device, Sex, and Age
The distance of both anchors of device B at the meniscus root of the posterior horn to neurovascular structures (PA, PV, TN, and PN) was statistically significantly smaller than the distance of both anchors of device A at the popliteal hiatus (P < .001) (Table 2). The same trend was seen for the NVB (mean of measurements to PA, PV, and TN) and the PA, PV, and TN individually (P≤ 0.001 for all) (Table 2). There was no statistically significant difference in distances to the PN between the 2 devices (P = .06) (Table 2).
Although female and <10-year-old specimens tended to have larger distances than male and ≥10-year-old specimens, respectively, there were no statistically significant differences (P > .05 for all) (Appendix Figures A1 and A2). Similar trends were seen when specimens were matched by age and sex with a notable exception that male specimens ≥10 years old tended to have larger distances than male specimens <10 years old (Appendix Figures A1 and A2, respectively); however, there were no statistically significant differences (P > .05 for all).
Discussion
This study illustrates the proximity of neurovascular structures to all-inside meniscal repair device anchors in pediatric knee specimens. Prior pediatric anatomic research has suggested that the higher risk zones are the posterior meniscal region extending from the root to the popliteal hiatus, and these locations were the focus of this study.3,23,25 The distances between these devices and critical neurovascular structures can be as close as 2.3 to 6.3 mm, with devices placed at the meniscus root via a lateral portal being the closest. In adult studies, <5 mm between the repair device and the neurovascular structures is considered to pose a high risk.1,7 With this definition, lateral meniscal repair at both device sites and approach angles utilized in our study should be regarded as higher risk, as the PA and PV were within 5 mm of the device anchors in multiple instances. The TN and PN may also be at higher risk when repairing the meniscus at both sites or just medial to the popliteal hiatus, respectively, as anchors came within 7 mm.
While the incidence of neurovascular injury during all-inside meniscal repair in adults is considered low, 4 there are several adult cadaveric studies that illustrate the proximity of neurovascular structures to lateral meniscal implants. For instance, Cohen et al 5 found that the tip of an all-inside device without the penetration limiter was ≤3 mm from the artery. Abouheif et al 1 reported damage to the PA, PV, and PN in 33% and 10% of their 31 knees, respectively. Cuellar et al 7 demonstrated that lateral meniscal repair was safer at 90° of knee flexion with increased risk in extension. More recently, an magnetic resonance imaging–based study revealed significant risks to the neurovascular structures with lateral meniscal repair. 10 However, the incidence and risks of such complications in children undergoing meniscal repair are unknown, despite the increasing incidence of these cases in the pediatric population.6,2,19,24,27
Risk for neurovascular injury using a straight all-single device through a lateral portal has been described in adult cadaveric specimens. 1 Based on these results, we hypothesized that we would identify the same relationship. Our study yielded similar results in pediatric cadaveric specimens and supported our hypothesis, as devices were closest to the NVB and PA, PV, and TN individually when placed at the posterior meniscus root via a lateral portal. Interestingly, we did not see a statistical difference in proximity to the PN of devices placed just medial to the popliteal hiatus via a medial portal when compared with devices placed at the posterior meniscus root via a lateral portal. We hypothesized that this difference would exist, as the PN is located posterior to the biceps femoris tendon and adjacent to the popliteal hiatus. Our failure to illustrate this difference in our results may be attributable to our limited sample size.
Our results also align with Yen et al’s 32 pediatric anatomic study; however, devices in that study were not implanted. Instead, device needle points were positioned after extensive exposure of the lateral meniscus via dissection of the knee extensor mechanism, medial collateral ligament, and lateral collateral ligament. After this “open” approach, measurements to neurovascular structures were made with needle points left in situ. 32 Our study sought to implant devices under simulated arthroscopic conditions and thus provide more representative and clinically relevant distances to neurovascular structures in high-risk meniscal zones when these implants are placed in live patients.
Prior pediatric imaging studies examining the vascular safety of all-inside devices have shown that the distance between neurovascular structures and the posterior lateral meniscus increases linearly with age and is greater in male anatomy.3,23,25 Interestingly, the trends identified in this study, except for male specimens >10 years old, appear to contradict these previous findings. However, as none of these trends were statistically significant, this is likely due to our limited sample size.
Ultimately, this study sought to determine the proximity of neurovascular structures in a "worst-case" scenario by intentionally directing an all-inside meniscal repair device toward the neurovascular structures and engaging the posterior joint capsule at high-risk zones of the meniscus. It is crucial to mention that if the all-inside device were used as recommended, with appropriate precautions and depth penetration limitations, the device would be positioned further away from the neurovascular structures, potentially reducing the risk of injury in most cases.
Limitations
This study has some limitations inherent to cadaveric studies, including lack of physiological function of the tissue, tissue quality, and a limited sample size. Access to pediatric and adolescent tissue for research purposes is severely limited, making sample size a consistent issue with any pediatric tissue-based study. Figure-of-4 positioning, commonly used in the surgical setting, is difficult to achieve with cadaveric specimens and may have limited our ability to fully replicate operative conditions during device placement. Furthermore, dissection posteriorly after device insertion may have allowed for tissue movement, potentially altering the true relationship of the meniscus with the neurovascular structures. Despite meticulous dissection, the small implants could not be detected or were excluded in 36% of the specimens, potentially affecting study power. While we excluded anchor measurements in cases where dissection altered the native anatomy to the extent that obtaining accurate measurements became challenging, it should be noted that anchors that could not be isolated were likely intra-articular or embedded in the meniscotibial ligament as opposed to the capsule. Last, it is important to note that only the AIR II) all-inside device system was utilized in this study; however, several other systems with varying needle size, angles/radius of curvature, and depth limits exist.
Conclusion
This study illustrates the proximity of neurovascular structures to all-inside devices in pediatric knee specimens, particularly those directed at the posterior horn using the anterolateral portal. Under the conditions of this study, all-inside devices appeared to be safe.
Footnotes
Appendix
Distances (mm) of All Anchors to Neurovascular Structures a

| Neurovasculature | All | NVB | PA | PV | TN | PN |
|---|---|---|---|---|---|---|
| Range | 2.3-63.0 | 2.3-63.0 | 2.3-59.8 | 3.5-58.0 | 5.3-63.0 | 6.3-40.1 |
| Median (IQR) | 21.0 (12.0-44.0) |
28.4 (11.1-45.0) |
26.0 (10.3-45.0) |
28.8 (10.1-44.8) |
31.7 (12.8-48.7) |
16.4 (12.1-28.0) |
| P | PA vs PV | PA vs TN | PA vs PN | PV vs TN | PV vs PN | TN vs PN |
| ≥.99 | ≥.99 | ≥.99 | ≥.99 | ≥.99 | .23 |
NVB, neurovascular bundle; PA, popliteal artery; PN, peroneal nerve; PV, popliteal vein; TN, tibial nerve.
Acknowledgements
The authors extend their gratitude to Todd Huft, Lisa Houck, and Tom Cycota, the CEO of Allosource located in Centennial, Colorado, for their generous contribution of specimens and the use of their facilities, which enabled this study. Additionally, they appreciate Stryker for donating the all-inside devices utilized in this cadaveric investigation. They also wish to acknowledge the medical artists of Texas Scottish Rite for their assistance in preparing ![]() and to recognize and honor the families whose donations to the study group were instrumental. Their generosity has not only made this research feasible but will also affect the lives of other families and their children.
and to recognize and honor the families whose donations to the study group were instrumental. Their generosity has not only made this research feasible but will also affect the lives of other families and their children.
Final revision submitted April 27, 2025; accepted June 16, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: Y.-M.Y. is a consultant or advisor for Smith & Nephew Inc, serves on the editorial board of The American Journal of Sports Medicine, and serves on the executive board of the American Academy of Pediatrics Section on Orthopedics. M.T. is a paid consultant for Abys Medical; receives other financial or material support from Allosource-ROCK group and Vericel-ROCK group; is an editorial or governing board member of The American Journal of Sports Medicine and Knee Surgery, Sports Traumatology, Arthroscopy, and Sports Health; and is a board or committee member of AOSSM, Pediatric Research in Sports Medicine Society, and ROCK. M.Schmitz is a board member of the International Orthopaedic Diversity Alliance and is a consultant or advisor for Wishbone Medical. T.G. is a board or committee member of the American Academy of Orthopaedic Surgeons, American Academy of Pediatrics Section on Orthopaedics, International Pediatric Orthopaedic Symposium, Pediatric Orthopaedic Society of North America, Pediatric Research in Sports Medicine, and Research on Osteochondritis Dissecans of the Knee; receives research support from Allosource and Vericel; and is an editorial or governing board member and receives publishing royalties and financial or material support from The American Journal of Sports Medicine. H.B.E. receives speaking and lecture fees from OrthoPediatrics; has received grants from Pediatric Orthopaedic Society of North America, Department of Defense, Washington University in St Louis, and AOSSM; is a board member of the American Academy of Orthopaedic Surgeons and Pediatric Research in Sports Medicine; and receives nonfinancial support from Arthrex Inc. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
