Abstract
Background:
Particulated juvenile articular cartilage (PJAC) implantation has demonstrated promising early results in the treatment of symptomatic articular cartilage defects of the patella. However, some uncertainty exists regarding the stability of this cell-based technique in lesions that are not well contained or shouldered.
Purpose:
To compare clinical and magnetic resonance imaging (MRI) outcomes of PJAC treatment in shouldered versus unshouldered full-thickness cartilage defects of the patella.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A retrospective review of prospectively collected data from an institutional knee registry was conducted to identify patients treated with PJAC for full-thickness symptomatic patellar cartilage lesion between January 2009 and August 2017. Cartilage defects were graded arthroscopically according to the Outerbridge classification and characterized as shouldered or unshouldered. For the primary outcome, postoperative MRI studies were read by a musculoskeletal fellowship-trained radiologist who characterized the percentage of fill based on both coronal and sagittal images. Patient-reported outcome measures (PROMs) were obtained at baseline and 2-year follow-up. MRI and PROM results were compared between the shouldered and unshouldered cohorts.
Results:
A total of 64 knees in 60 patients (mean age, 26.3 ± 7.6 years) were identified, of which 62 (97%) knees underwent a concomitant patellar stabilization or offloading procedure. There were 32 (50%) shouldered and 32 (50%) unshouldered defects. On postoperative MRI, 68.8% of shouldered lesions demonstrated 67% to 100% fill, compared with 59.4% of unshouldered lesions; 12.5% of shouldered lesions and 15.6% of unshouldered lesions demonstrated 34% to 66% fill; and 18.8% of shouldered lesions and 25.0% of unshouldered lesions demonstrated 0% to 33% fill (P = .604). At 2 years, significant improvements were observed in Knee Injury and Osteoarthritis Outcome Score (KOOS) Quality of Life (QOL) (23.1 to 65.3; P < .001), International Knee Documentation Committee (IKDC) (41.2 to 76.0; P < .001), KOOS Physical Function Shortform (PS) (34.8 to 13.9; P < .001), Kujala (52.0 to 87.5; P < .001), 12-Item Short Form Survey (SF-12) Physical Health (39.5 to 51.5; P < .001), Veterans Rand 12 Item Health Survey (VR-12) Physical Health (40.7 to 52.8; P < .001), Veterans Rand 6D (0.66 to 0.79; P < .001), SF-12 Mental Health (48.9 to 53.4; P = .015), and VR-12 Mental Health (49.4 to 54.0; P = .019) values. No significant change was observed in Pediatric Functional Activity Brief Scale score (9.1 to 10.3; P = .384). All PROMs were equivalent between shouldered and unshouldered defects at the 2-year follow-up (P = .318-.980). There was a greater improvement in both KOOS PS (27.2 vs 10.7; P = .015) and Kujala (44.3 vs 26.9; P = .039) values from baseline to the 2-year follow-up in the shouldered group.
Conclusion:
PJAC implantation with concomitant patellar stabilization led to significantly improved PROMs for both shouldered and unshouldered patellar cartilage lesions over time. Additionally, minimal differences were observed between shouldered and unshouldered defects treated with PJAC at the 2-year follow-up.
Keywords
Cartilage restoration in the patellofemoral joint can present unique challenges, such as the contour of the articular surface, the unique thickness of the cartilage, and the high dynamic shear and compressive forces at the trochlear articulation. 13 Particulated juvenile articular cartilage (PJAC), minced hyaline cartilage from juvenile donors, has demonstrated promising clinical results in treating symptomatic articular defects of the patella and other surfaces.6,8,12,15 Additionally, PJAC is more cost-effective than both nonoperative management and matrix-induced autologous chondrocyte implantation (MACI). 15
A critical factor historically thought to optimize the repair of full-thickness cartilage defects is whether a stable rim of healthy cartilage fully shoulders the lesion. In microfracture procedures, this contributes to the success of achieving and containing a stable clot.3,22,23 Similarly, with MACI procedures, cartilage defect edges are debrided to create contained, fully-shouldered lesions to ensure a watertight seal with the implant using fibrin glue.11,31 Previous studies have demonstrated significant improvement clinically and radiographically with PJAC implantation for the treatment of contained lesions. 10 The described technique for PJAC recommends a minimum of 50% to 60% containment of native cartilage when preparing the defect for grafting, suggesting that the properties and characteristics of PJAC grafting may be well suited for tolerating unshouldered lesions; however, it has yet to be formally studied.30,35
This study aimed to compare the clinical outcomes of utilizing PJAC for shouldered and unshouldered full-thickness cartilage defects of the patella. By utilizing magnetic resonance imaging (MRI) findings, this study correlates patient outcomes to the morphology of the lesions with respect to postoperative lesion fill. We hypothesized that PJAC implantation for shouldered cartilage defects would have similar clinical outcomes and lesion fill on MRI compared with PJAC implantation for unshouldered cartilage defects.
Methods
Patient Identification and Data Collection
After institutional review board approval (IRB 2018-0734), a retrospective evaluation of prospectively collected data from an institutional registry was conducted to identify consecutive patients treated with PJAC for full-thickness symptomatic patellar cartilage lesions from January 2009 to August 2017. An electronic medical record review was performed to identify those with at least 2 years of clinical follow-up and postoperative MRI of their knee. Patients lacking intraoperative arthroscopic images of the lesion were excluded (Figure 1). Collected data included patient characteristics, lesion-specific characteristics, and features of the implanted allograft. Patient-specific factors included age, sex, duration of knee symptoms, and any concurrent knee pathology, including patellar instability. Lesion-specific factors included size, location, and origin of the cartilage defect. Graft-specific factors included donor age and number of allograft packets utilized.

Flow diagram of the study using CONSORT (Consolidated Standards of Reporting Trials) guidelines.
Surgical Technique
The decision to perform a cartilage repair procedure with PJAC was made based on the presence of a full-thickness cartilage lesion on the articular surface of the patella, as demonstrated by preoperative MRI and confirmed during diagnostic arthroscopy.
Patellar cartilage repair with PJAC was then performed via a medial or lateral parapatellar arthrotomy. The recipient bed was gently debrided with a combination of ring curette and scalpel, ensuring that all affected cartilage was removed with care to not violate the subchondral plate. Whenever possible, an attempt was made to create a well-shouldered lesion with vertical walls at the site of the patellar cartilage defect. For unshouldered lesions, a portion of the circumferential cartilage remained uncontained. The PJAC tissue utilized in this study was DeNovo NT (Zimmer Biomet). The allograft cartilage was implanted using a previously described surgical technique. 7 This defect reconstruction method involves templating the lesion’s geometry and size with sterile foil, which helps to determine the amount of allograft that is needed to fill the defect, as 1 package of DeNovo NT can cover approximately 2.5 cm2 of defect. 33 These measurements were obtained with a ruler and documented in the operative report. Once the template was created, allograft tissue was placed inside the template to create a mold and secured with fibrin glue. This technique allows for complete lesion fill.
Concomitant procedures for patellar stabilization or additional cartilage procedures were performed to address patients’ underlying patellofemoral pathology during the index PJAC implantation. These concomitant procedures were based on clinical and radiographic indications. All bipolar lesions were managed with osteochondral allograft (OCA) or chondroplasty of the trochlear or femoral condyle lesion depending on the characteristics and location of the lesions.
Postoperatively, all patients were placed in a hinged knee brace for 6 weeks until adequate quadriceps strength returned. Passive range of motion using a continuous passive motion device was initiated on postoperative day 2. Initially, passive motion was restricted to 0° to 60° for the first 2 weeks; at this point, patients were allowed to progress to 90° of flexion. Patients were initially allowed toe-touch weightbearing in the brace, progressing to partial weightbearing with the brace locked in full extension. The rehabilitation protocol was modified as indicated by other associated procedures (tibial tubercle osteotomy [TTO] or medial patellofemoral ligament [MPFL] reconstruction), and the length of protected weightbearing was contingent on the addition of a TTO. Continuous passive motion was used for 4 to 6 hours per day for the first 6 weeks with the goal of obtaining 0° to 90° by the end of 6 weeks. Eccentric quadriceps exercises were initiated within the first week of surgery. After the initial healing phase, range of motion was unrestricted, strengthening exercises were started, and the patients began sport-specific training at the 3- to 4-month time point.
Lesion Characterization
Arthroscopic photographs taken intraoperatively were stored in patients’ medical records. Images of the cartilage defects were collected, blinded, and then reviewed by 1 sports medicine fellowship-trained orthopaedic surgeon (K.K.) as well as 1 orthopaedic surgery resident (B.A.). Cartilage defects were individually characterized as either shouldered (Figure 2) or unshouldered (Figure 3), depending on the presence or absence of a stable rim of native cartilage surrounding the defect. The determination of whether a lesion was shouldered between the 2 raters was evaluated for interrater reliability.
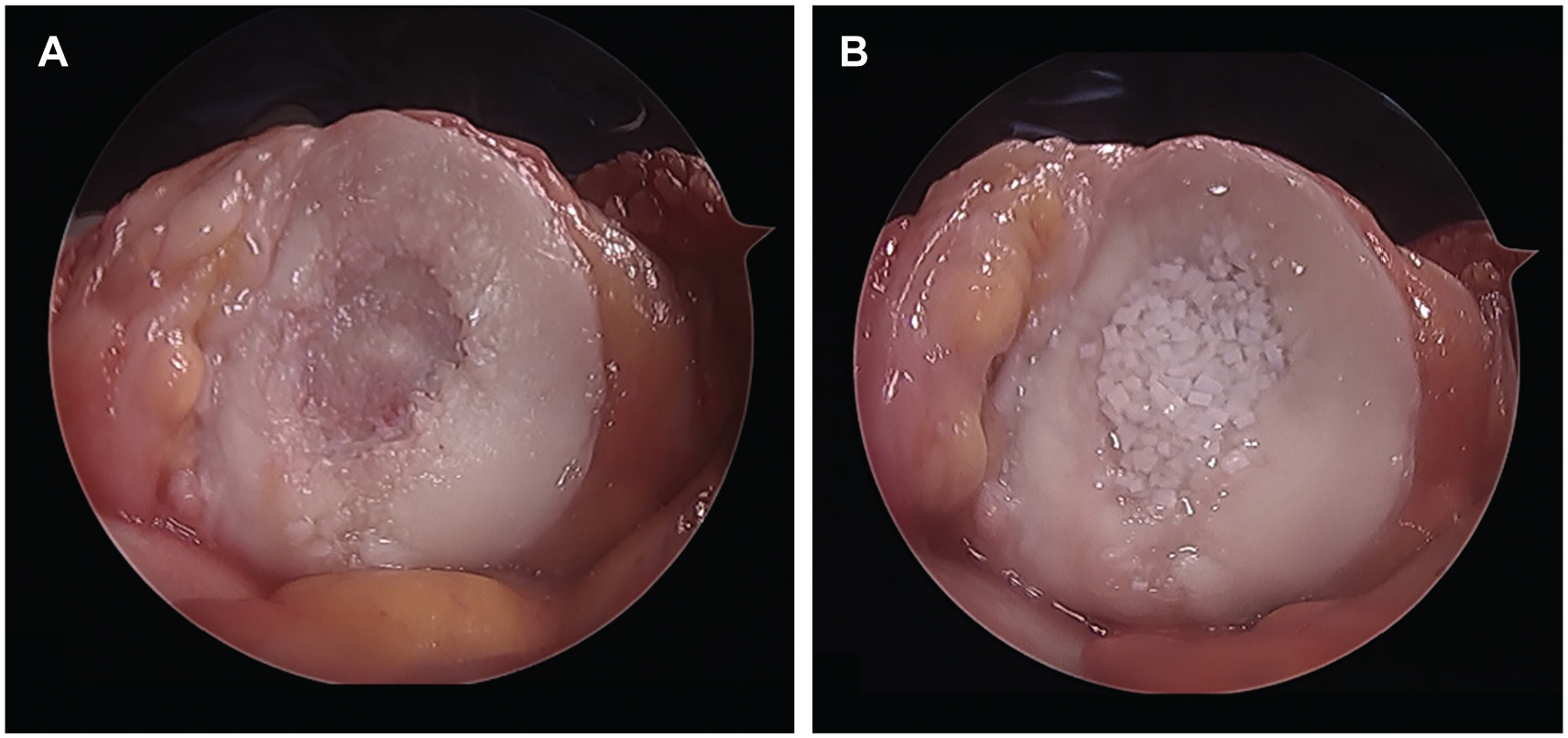
(A) Shouldered patellar cartilage defect. (B) Shouldered patellar cartilage defect after treatment with particulated juvenile allograft cartilage at time point 0.

(A) Unshouldered patellar cartilage defect. (B) Unshouldered patellar cartilage defect after treatment with particulated juvenile allograft cartilage at time point 0.
MRI Acquisition and Grading
A 1.5-T (450 model) or 3.0-T (750 model) imaging system (GE HealthCare) was utilized for MRI. Qualitative (fast spin echo) MRI scans obtained for all patients preoperatively and postoperatively at 12 months. Fast spin echo images were obtained in 3 planes to allow assessment of articular cartilage using a previously validated cartilage-sensitive pulse sequence.17,25 The moderate echo time used to acquire images allowed for high-contrast resolution between articular cartilage, subchondral bone, and joint fluid while avoiding the susceptibility artifacts of the postoperative knee seen in gradient echo imaging techniques. Regions of interest were evaluated from the treated area of articular cartilage at the interface with adjacent uninvolved tissue and the adjacent and opposite articular cartilage surfaces. All MRI studies were read by a single musculoskeletal fellowship-trained radiologist (A.B.) without knowledge of the patient or treating surgeon. Images were scored according to previously described cartilage repair criteria, characterizing the percentage of fill assessed by determining volume filling of the defect based on both coronal and sagittal images: 0% to 33%, 34% to 66%, or 67% to 100% (Figure 4).4,17,18,25

Postoperative magnetic resonance imaging grading, characterizing percentage of fill based on coronal and sagittal images. Each pair of images illustrates an example of percentage fill, shown from top to bottom as follows: 0-33%, 34-66%, and 67-100%.
Patient-Reported Outcome Measures
Patient-reported outcome measures (PROMs) in the form of International Knee Documentation Committee (IKDC), Kujala, Knee Injury and Osteoarthritis Outcome Score (KOOS) Physical Function Shortform (PS), KOOS Quality of Life (QOL), 12-Item Short Form Survey (SF-12), Veterans Rand 12 Item Health Survey (VR-12), Veterans Rand 6D (VR-6D), and Pediatric Functional Activity Brief Scale (Pedi-FABS) values were collected at preoperatively (baseline) and postoperatively at 2-year time points.
Statistical Analysis
Patient characteristics and percent fill were assessed in individuals with shouldered versus unshouldered full-thickness cartilage defects of the patella. Categorical variables were compared using the chi-square or Fisher exact test, as appropriate. Continuous variables were evaluated through independent 2-sample t tests or Wilcoxon Mann-Whitney tests, as appropriate. Patient-reported outcome scores are reported descriptively. Scores are summarized using means and standard deviations. Across the entire cohort, irrespective of cartilage defect type, scores were compared between baseline and 2-year follow-up. This comparison was conducted using paired t tests or Wilcoxon signed-rank tests, as appropriate. Subsequently, deltas of patient-reported outcome scores between time points were calculated and compared between the shouldered versus unshouldered groups. These comparisons were also performed using independent 2-sample t tests or Wilcoxon Mann-Whitney tests.
Results
Patient Characteristics
The cohort consisted of 64 patellar cartilage defects in 60 patients treated with PJAC. Patients were predominantly female (n = 47; 73.4%) with a mean patient age of 26.3 years (range, 14 – 44 years) and mean follow-up of 2.8 ± 1.7 years. The mean symptom duration was 8 years (Table 1). A total of 24 (37.5%) knees had undergone previous surgery. The mean patellar cartilage lesion size was 277 mm2 (range, 77-800 mm2), the mean number of allograft packets utilized was 2.4 (range, 1-5), and the mean donor age was 48.6 months (range, 1-120 months). There was no significant difference in laterality of lesion, sex, age at the time of intervention, or body mass index (BMI) between individuals with shouldered versus unshouldered chondral lesions. Individuals with unshouldered lesions had a significantly longer mean symptom duration of 10.1 ± 9.9 years compared with 5.3 ± 7.1 years (P = .023). More knees in the unshouldered group also had a history of previous knee surgery: 53.1% (n = 17) compared with 21.9% (n = 7) (P = .019).
Clinical Characteristics Between Shouldered and Unshouldered Lesions a

Values are presented as mean ± SD or n (%) unless otherwise indicated. Bold P values indicate statistical significance. BMI, body mass index.
Lesion Characterization
Based on the intraoperative photographs, 32 lesions were classified as shouldered with a continuous stable rim, and 32 were classified as unshouldered without a stable rim. The determination of whether a lesion was shouldered between the 2 raters was evaluated for interrater reliability using the Cohen kappa, which revealed a perfect level of agreement (κ = 1.00).
Patellar chondral lesion characteristics are outlined in Table 2. Notably, there were no statistically significant differences in patellar lesion dimensions, location, or number of allograft packets required to address the lesions between unshouldered and shouldered defects.
Patellar Chondral Lesion Characteristics and Treatment a

Values are presented as mean ± SD or n (%) unless otherwise indicated. PJAC, particulated juvenile allograft cartilage.
A concomitant nonpatellar grade 3 lesion was found in 27% of knees, with most nonpatellar grade 3 lesions (67.2%) located on the trochlea (Table 3). Twelve of the trochlear lesions were addressed with concomitant OCA transfers, with the remainder undergoing chondroplasty. Regarding additional concomitant procedures, 62 (97%) knees underwent a patellar stabilization or offloading procedure. A total of 31 (48%) knees underwent a combined MPFL and TTO, 11 (17%) an isolated MPFL, and 20 (31%) an isolated TTO. In total, 29 (45.3%) had a lateral release, only 1 (1.6%) of which was in isolation. Additionally, 1 lateral lengthening was performed as an isolated procedure (Table 4). Of note, the isolated lateral release and isolated lengthening were utilized to help with maltracking, as neither patient exhibited patellar instability.
Concomitant Chondral Lesion Location and Characteristics a

Values are presented as n (%) unless otherwise indicated.
Concomitant Procedures at Time of Particulated Juvenile Articular Cartilage Implantation a
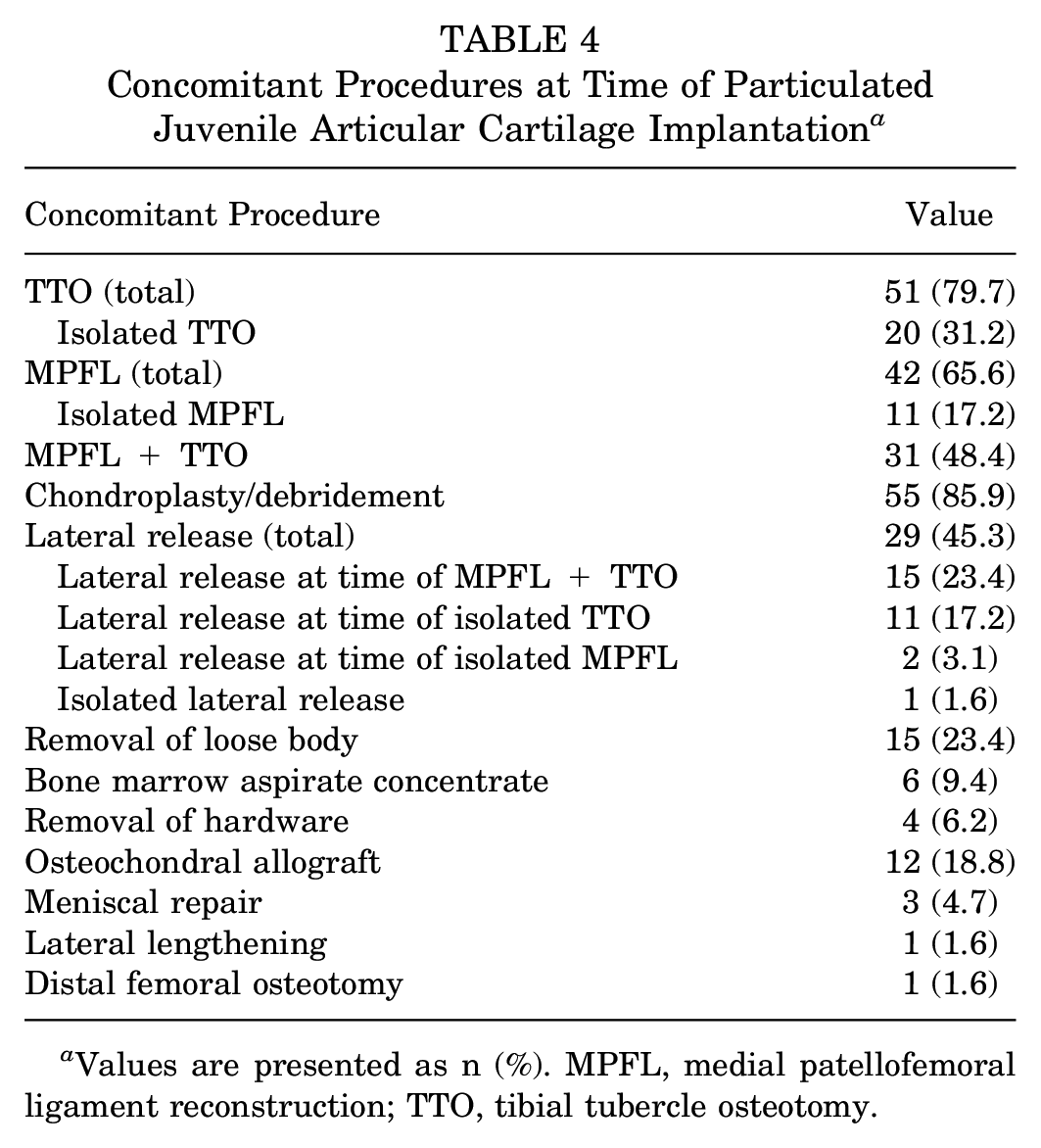
Values are presented as n (%). MPFL, medial patellofemoral ligament reconstruction; TTO, tibial tubercle osteotomy.
MRI Findings
Evaluation of percent fill at the latest postoperative MRI time point was performed. The mean MRI time points were 14.8 and 12.4 months postoperatively for shouldered and unshouldered lesions, respectively (P = .240). Within the shouldered group, 6 (18.8%) patients reached 0% to 33% fill, 4 (12.5%) reached 34% to 66% fill, and 22 (68.8%) reached 67% to 100% fill. With the unshouldered group, 8 (25.0%) patients achieved 0% to 33% fill, 5 (15.6%) reached 34% to 66% fill, and 19 (59.4%) reached 67% to 100% fill of their defects after PJAC. Comparatively, there was no statistically significant difference in the percent fill achieved between the shouldered and unshouldered lesions (P = .604) (Table 5).
Differences in MRI Cartilage Fill Achieved Between Shouldered and Unshouldered Lesions a

Values are presented as mean ± SD or n (%) unless otherwise indicated. MRI, magnetic resonance imaging.
Clinical Outcomes
The entire cohort showed a statistically significant improvement from baseline to 2 years in KOOS QOL, IKDC, KOOS PS, Kujala, SF-12 Mental Health, SF-12 Physical Health, VR-12 Mental Health, VR-12 Physical Health, and VR-6D values (all P < .05), with the exception of Pedi-FABS, which showed no significant difference (P = .384) (Table 6).
Patient-Reported Outcome Measurements From Baseline to 2-Year Follow-up a

Values are presented as mean ± SD unless otherwise indicated. Bold P values indicate statistical significance. IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; Pedi-FABS, Pediatric Functional Activity Brief Scale; PS, Physical Function Shortform; QOL, Quality of Life; SF-12, 12-Item Short Form Survey; VR-12, Veterans Rand 12-Item Health Survey; VR-6D, Veterans Rand 6D.
A subanalysis of the PROMs was performed to compare shouldered and unshouldered defects. Overall, in addition to improvement seen from baseline to 2 years in both groups, there were no significant differences in baseline and 2-year PROMs between shouldered and unshouldered groups (Table 7). However, the shouldered group had a larger improvement from baseline to 2 years in KOOS PS (27.2 vs 10.7; P = .015) and Kujala (44.3 vs 26.9; P = .039) values (Table 8). While the other outcome measures did not show significant differences between groups, the shouldered group broadly had larger mean improvements compared with the unshouldered group, except for Pedi-FABS.
Patient-Reported Outcome Measurements Compared Across Unshouldered and Shouldered Cartilage Defects Treated With PJAC Implantation a

Values are presented as mean ± SD unless otherwise indicated. IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; Pedi-FABS, Pediatric Functional Activity Brief Scale; PJAC, particulated juvenile articular cartilage; PS, Physical Function Shortform; QOL, Quality of Life; SF-12, 12-Item Short Form Survey; VR-12, Veterans Rand 12-Item Health Survey; VR-6D, Veterans Rand 6D.
Change in Patient-Reported Outcome Measures From Baseline to 2 Years a

Values are presented as mean ± SD unless otherwise indicated. IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; Pedi-FABS, Pediatric Functional Activity Brief Scale; PS, Physical Function Shortform; QOL, Quality of Life; SF-12, 12-Item Short Form Survey; VR-12, Veterans Rand 12-Item Health Survey; VR-6D, Veterans Rand 6D.
Discussion
In this patient cohort undergoing PJAC patellar cartilage repair for patellar defects, PJAC implantation with concomitant patellar stabilization resulted in significantly improved PROMs for both shouldered and unshouldered patellar cartilage lesions over time. Shouldered and unshouldered defects demonstrated similar rates of cartilage fill on postoperative MRI at 1 year (P = .604). Overall, postoperative PROMs were equivalent between shouldered and unshouldered patellar cartilage defects at baseline and at 2 years. However, shouldered lesions showed greater improvements in KOOS PS (27.2 vs 10.7; P = .015) and Kujala (44.3 vs 26.9; P = .039) values from baseline to the 2-year follow-up.
The management of patellar cartilage lesions has evolved over time with the development of novel cartilage restoration techniques.21,28 Current indications for the treatment of patellar chondral lesions with PJAC include well-shouldered lesions, >2 cm, with intact subchondral bone. 21 However, uncontained lesions, which are not uncommon on the patella, present challenges when attempting to create vertical stable cartilage walls. Despite this challenge, our study found that most unshouldered lesions (59.4%) and shouldered lesions (68.8%) achieved two-thirds fill on 1-year postoperative MRI. Similar results were reported by Wang et al, 32 with 69.2% of patients showing majority fill after PJAC treatment for patellar lesions. Interestingly, there was no association between depth of lesion fill and patient-reported outcome scores. This was later corroborated by Marmor et al, 19 who found significant improvement in all PROM scores after implantation at 1, 2, and > 2 years postoperatively, regardless of percent fill. Additional studies analyzing autologous chondrocyte repair similarly demonstrated no correlation between MRI findings and clinical outcomes.24,27 Likewise, in the current study, both shouldered and unshouldered patellar cartilage lesions showed significant PROM improvements, independent of MRI fill.
Earlier investigations have demonstrated improved short-term functional outcomes for patellar cartilage lesions treated with PJAC.5,26,32 The current study found significant improvements in all PROMs from baseline to 2 years, except for Pedi-FABS, which assesses activity and function in sport or leisure. 9 No differences were found between shouldered and unshouldered lesions in PROMs at 2 years. However, while all patients regardless of shouldered or unshouldered status had improvement in PROMs from baseline to 2 years, the shouldered group showed greater improvement in KOOS PS and Kujala values. It is important to note that the unshouldered group had a longer symptom duration (10.1 vs 5.3; P = .023) and higher rate of previous knee surgery (53.1% vs 21.9%; P = .019), which may influence patient perception and therefore reported outcomes. These similar PROMs at 2 years indicate that PJAC stability may not actually rely on an intact cartilage rim. However, to definitively determine if PJAC can withstand the biomechanical demands of the patellofemoral joint in treating shouldered and unshouldered defects, larger and longer-term studies are necessary.
The clinical improvements demonstrated in patients treated with PJAC in this study may be attributed to any combination of the following features of PJAC. One notable benefit lies in the facilitation of a single-stage operation, which enables faster recovery, quicker return to activity, and is more cost-effective than alternative treatments. 15 PJAC also avoids donor site morbidity, reducing complications and symptoms. However, PJAC has drawbacks, such as its cost—approximately $4950 for a 2.5-cm area, with increased cost for larger defects—and inability to reuse partially used packets.2,20 Additionally, it has a short shelf life of up to 49 days, which can contribute to challenges with respect to the timing and coordination of surgery. 15 Although concerns regarding the potential for disease transmission and immune host rejection exist, these risks are mitigated by extensive serological testing and viral screening during processing. 29 Isolated juvenile chondrocytes are immune privileged, with no reports of immunogenic responses. 1 Additionally, recent studies have shown that elevated CD24 expression in juvenile chondrocytes offers protection against inflammatory responses. 16
Patellofemoral cartilage restoration has historically had poor outcomes and high failure rates.14,34 However, with improved risk factor identification, new surgical techniques, and patient-tailored approaches, outcomes have improved with lower rates of complications. A 2014 review by Yanke et al 34 found better and more predictable results for patients >30 years of age, with low BMI and lesions <4 cm. The current study highlights the variability in lesion size (277 ± 140 mm2), which can possibly affect outcomes. A meta-analysis by Hinckel et al 14 showed that modern techniques lead to improved outcomes with low rates of complications, although OCA still demonstrated a relatively higher failure rate (22%) compared with other restoration techniques.
There are several limitations in this study. First, the retrospective nature of this study makes it subject to selection bias. Another limitation is the potential lack of power to detect differences between groups. However, we hypothesize that outcomes will not differ between groups, and given the sample size constraints, we did not expect a large effect size. Additionally, the study cohort includes a heterogeneous group of concurrent procedures at the time of PJAC. This poses notable challenges to understanding the interaction between the morphology of the lesion and cartilage intervention with PJAC in isolation. However, it is important to acknowledge that cartilage restoration procedures are rarely performed alone, as it is crucial to address the underlying origin by offloading the patellofemoral compartment or changing the biomechanics of the patient’s knee. Therefore, we view our cohort as a more generalizable and clinically applicable analysis of the real-world challenges presented by patients who undergo patellar cartilage treatment. The use of both 1.5-T (450 model) and 3.0-T (750 model) MRI systems may introduce bias in percent fill interpretation, but this is thought to be minimal due to the musculoskeletal radiologist’s experience and training. The study focused on short-term outcomes, specifically 1-year MRI follow-up and approximately 2-year clinical follow-up, and it did not include comparator cartilage restoration treatments or patients undergoing just a patellar stabilizing or offloading procedure, making it difficult to assess the relative efficacy of PJAC. Therefore, this study was unable to determine if the improvement in PROMs was a result of the offloading procedures or what potential additional affects PJAC implantation may have contributed. Future studies will need longer follow-up, rigorous controls, and larger samples powered to assess equivalence or noninferiority to draw more definitive conclusions.
Conclusion
In this cohort, PJAC implantation with concomitant patellar stabilization resulted in significantly improved PROMs for both shouldered and unshouldered patellar cartilage lesions over short-term follow-up. Shouldered and unshouldered defects demonstrated similar rates of cartilage fill on postoperative MRI. Clinically, PROMs were comparable between shouldered and unshouldered patellar cartilage defects. These results strongly support the use of PJAC with concomitant patellar stabilization for treating both shouldered and unshouldered patellar cartilage lesions. However, long-term follow-up is essential to confirm the durability and sustainability of these outcomes.
Footnotes
Final revision submitted April 21, 2025; accepted May 14, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: E.D. has received education payments from Arthrex, Gotham Surgical Solutions & Devices, and Smith & Nephew and is a consultant for ConMed. Her spouse is a consultant for Medtronic. B.E.S.S. has received nonconsulting fees from Arthrex, royalties from Arthrex, consulting fees from Arthrex and ConMed, education fees from Gotham Surgical Solutions & Devices, is on the editorial board for Orthopedics Today, and is on the publishing board for The American Journal of Sports Medicine. Her spouse is a consultant for Arthrex and receives royalties. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Hospital for Special Surgery (IRB 2018-0734).
