Abstract
Background:
Cartilage restoration procedures for patellar cartilage defects have produced inconsistent results, and optimal management remains controversial. Particulated juvenile articular cartilage (PJAC) allograft tissue is an increasingly utilized treatment option for chondral defects, with previous studies demonstrating favorable short-term outcomes for patellar chondral defects.
Purpose:
To identify whether there is an association between defect fill on magnetic resonance imaging (MRI) with functional outcomes in patients with full-thickness patellar cartilage lesions treated with PJAC.
Study Design:
Case series; Level of evidence, 4.
Methods:
A retrospective review of prospectively collected data was conducted on patients treated with PJAC for a full-thickness symptomatic patellar cartilage lesion between March 2014 and August 2019. MRI was performed for all patients at 6, 12, and 24 months postoperatively. Patient-reported outcome measures (PROMs) were obtained preoperatively and at 1, 2, and >2 years postoperatively. Clinical outcome scores—including the International Knee Documentation Committee (IKDC) score, the Kujala, the Knee injury and Osteoarthritis Outcome Score–Physical Function Short Form (KOOS-PS), the Knee Injury and Osteoarthritis Outcome Score–Quality of Life (KOOS-QoL), and the Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS)—were analyzed and evaluated for a relationship with tissue fill on MRI.
Results:
A total of 70 knees in 65 patients (mean age, 26.6 ± 8.1 years) were identified, of which 68 knees (97%) underwent a concomitant patellar stabilization or offloading procedure. Significant improvements were observed on all postoperative PROM scores at the 1-, 2-, and >2-year follow-up except for the Pedi-FABS, which showed no significant difference from baseline. From baseline to the 2-year follow-up, the KOOS-QoL improved from 24.7 to 62.1, the IKDC improved from 41.1 to 73.5, the KOOS-PS improved from 35.6 to 15, and the Kujala improved from 52 to 86.3. Imaging demonstrated no difference in the rate of cartilage defect fill between the 3-month (66%), 6-month (72%), 1-year (74%), and ≥2-year (69%) follow-ups. No association was observed between PROM scores and the percent fill of cartilage defect on MRI at the 1- and 2-year follow-up.
Conclusion:
PROM scores were significantly improved at the 2-year follow-up in patients who underwent PJAC for full-thickness patellar cartilage defects. On MRI, a cartilage defect fill of >66% was achieved by 3 months in most patients. In our sample, PROM scores were not significantly associated with the defect fill percentage at the short-term follow-up.
Keywords
Patellar chondral defects are a significant source of knee morbidity that can pose unique challenges during surgical intervention. Cartilage lesions of the patellofemoral joint can be associated with reentry trauma after a patellar dislocation or excessive load due to malalignment or maltracking.7,15,31 Recurrent patellar instability is associated with high rates of patellar cartilage injury, with some studies noting >50% of patients with high-grade lesions.17,30 Defects of the patellar cartilage are inherently difficult to treat because of the relatively thick and contoured patellar cartilage surface, high-density subcortical bone, and high shear and compressive forces. 27 Several surgical cartilage restoration techniques have been developed—including marrow stimulation, autograft and allograft osteochondral transfer, and cell-based treatments. 11
Particulated juvenile articular cartilage (PJAC) (DeNovo Natural Tissue; Zimmer Biomet) is a cell-based allograft restoration approach for contained cartilage defects of the patella with healthy associated subchondral bone. The graft substrate consists of minced live cartilage from juvenile donors. Unlike cell-based methods—such as matrix-induced autologous chondrocyte implantation (MACI)—PJAC is a single-stage technique, potentially adding the benefit of decreased morbidity and cost. 24 Several studies have reported promising results in short-term patient outcomes.5,37,38 High fill rates and evidence of matured grafts with hyaline-like cartilage have also been reported.14,18,38 A previous study from this institution looking at postoperative magnetic resonance imaging (MRI) after PJAC implantation found moderate to good cartilage fill at 6, 12, and 24 months postoperatively. To date, limited data are available on the association between radiographic and functional outcomes after treatment of patellar cartilage defects with PJAC.
This study aimed to assess the association between patellar cartilage defect fill and patient-reported outcome measures (PROMs) in patients treated with PJAC for symptomatic full-thickness patellar cartilage defects. We hypothesized that patients with greater defect fill would report improved subjective outcomes than those with suboptimal fill.
Methods
Patient Identification and Data Collection
After obtaining approval from the institutional review board, a review of prospectively collected data was conducted from an institutional knee registry to identify consecutive patients who were treated with PJAC for full-thickness, symptomatic patellar cartilage lesions and who subsequently underwent postoperative MRI of their knee. Patient characteristics, patellar cartilage characteristics, and allograft tissue specifications were collected. Patient characteristics included age, sex, body mass index, duration of knee symptoms, previous knee surgery, and concomitant procedures. Lesion-specific factors included size, location, cause of the cartilage defect (contact vs noncontact), and concomitant Outerbridge grade IV cartilage defects obtained intraoperatively. Graft-specific factors collected for analysis included donor age and the number of allograft packets utilized for reconstruction. Upon inclusion into the institutional registry, patients completed baseline clinical PROMs—including the Knee injury and Osteoarthritis Outcome Score–Physical Function Short Form (KOOS-PS) and –Quality of Life (KOOS-QoL) subscales, the International Knee Documentation Committee (IKDC) questionnaire, the Pediatric Functional Activity Brief Scale (Pedi-FABS), and the Kujala. Postoperative PROM questionnaires were sent to patients at 1- and 2-year follow-up time points to complete via email or phone.
Surgical Technique
PJAC transplantation was performed by a single surgeon (B.E.S.S.) using a consistent surgical technique. 12 All patients underwent a diagnostic knee arthroscopy to document the cartilage status of all knee compartments. The Outerbridge classification repair of the patellar cartilage defect was performed after the arthroscopic portion of the procedure. There were several different causes of patellar cartilage lesions in this study—including sequela of instability, maltracking, or a combination of both—thus, most patients underwent concomitant procedures to address these underlying pathologies. Secondary procedures depended on the pathoanatomy of the patient at the time of surgery and included medial patellofemoral ligament (MPFL) reconstruction, tibial tubercle osteotomy, loose body removal, trochlear cartilage reconstruction, and lateral retinacular release or lengthening.
Patellar cartilage reconstruction with PJAC was performed via medial parapatellar arthrotomy or lateral parapatellar arthrotomy if a lateral lengthening was concomitantly performed. The decision to perform a cartilage reconstruction procedure with this graft type was made based on preoperative MRI, demonstrating the presence of a full-thickness cartilage lesion on the undersurface of the patella with healthy subchondral bone. After arthrotomy, the recipient bed was gently debrided to ensure that the damaged cartilage was removed, the subchondral plate was not violated, and no bleeding bone was exposed. Care was taken to create a well-shouldered recipient site with vertical walls whenever possible. The geometry of the lesion was then templated with sterile foil. Once the template was created, allograft tissue was placed inside the template and secured with fibrin glue, effectively making a mold with the allograft implant to be placed into the defect and sealed with fibrin glue. This technique allowed for complete lesion fill with the implant material.
Postoperatively, all patients were placed in a hinged knee brace locked in extension for ambulation for 6 weeks. Patients were made weightbearing as tolerated unless they underwent concomitant tibial tubercule osteotomy (TTO), in which case they were made nonweighting for 4 weeks. A passive range of motion using a continuous passive motion device was initiated on postoperative day 2. Initially, passive motion was 0° to 30° and progressed as tolerated. Continuous passive motion was used for 6 hours per day (2-hour sessions 3 times per day) for the first 6 weeks. Eccentric quadriceps exercises were initiated within the first week of surgery. After the initial healing phase, strengthening exercises were started, and the patients began sport-specific training at the 3- to 4-month time point. The rehabilitation protocol was modified as indicated by other associated procedures (tibial tubercle transfer and/or MPFL reconstruction).
MRI Acquisition and Grading
A 1.5-T (450 model) or a 3-T (750 model) imaging system (General Electric Healthcare) was utilized for MRI purposes. Fast spin echo images were obtained in 3 planes to allow assessment of articular cartilage using a previously validated cartilage-sensitive pulse sequence. 35 The moderate echo time (TE) used to acquire images allowed for high-contrast resolution between articular cartilage, subchondral bone, and joint fluid while avoiding the susceptibility artifacts of the postoperative knee seen in gradient echo imaging techniques. A repetition time (TR) of 3500 to 6000 ms, a TE of 34 ms (effective), a field of view of 13 to 16 cm2, and a matrix of 512 × 256 to 416 provided a minimum in-plane resolution of 254 μm in the frequency direction by 312 μm in the phase direction by slice resolution of 3 to 3.5 mm with no gap. A wider receiver bandwidth of 31.2 to 62.5 kHz was used over the entire frequency range to minimize chemical shift misregistration. A fat-suppressed pulse sequence in the sagittal plane was used to evaluate the presence of subchondral bone marrow edema.
T2 relaxation time mapping was performed using a multislice, multiecho-modified CPMG (Carr-Purcell-Meiboom-Gill) pulse sequence, which uses interleaved slices and tailored refocusing pulses to minimize the contribution from simulated echoes. 34 Standard T2 mapping pulse sequence parameters used were a TR of 800 ms, 8 echoes sampled using sequential multiples of the first TE (9-10 ms), a field of view of 16 cm2, and a matrix of 256 to 384 × 256, providing a minimum in-plane resolution of 254 μm in the frequency direction by 312 μm in the phase direction by slice resolution of 2 to 3 mm with no gap, and a receiver bandwidth of 62.5 kHz. After image acquisition, data sets were analyzed pixel by pixel with a 2-parameter weighted least-squares fit algorithm, assuming a monoexponential fit (Functool 3.1; General Electric Healthcare). Quantitative T2 values were calculated by taking the natural logarithm of the signal decay curve in a selected region of interest. Regions of interest were obtained in a standardized fashion from the treated area of articular cartilage at the interface with adjacent uninvolved tissue, as well as of the adjacent and opposite articular cartilage surfaces. All T2 relaxation times are reported in milliseconds.
All MRI studies were read by a single musculoskeletal radiologist (A.J.B.) without knowledge of the patient or treating surgeon. 35 The images were scored according to a previously described cartilage repair criteria: signal intensity of the repaired area relative to the surrounding cartilage (hypointense, isointense, or hyperintense), subchondral edema (none, mild, moderate, or severe), geometry, bony overgrowth (absence or presence), interface with adjacent cartilage (absence, presence, or size of fissure), percentage of fill, based primarily on axial images and with sagittal imaging used as an adjunct for craniocaudal distribution fill (0%-33%, 34%-66%, or 67%-100%), integrity of adjacent cartilage (Outerbridge grading) (Figure 1), fat pad scarring (mild, moderate, or severe), synovial reaction, graft hypertrophy, and graft displacement.4,25

Evaluation of patellar cartilage fill on magnetic resonance imaging. PJAC, particulated juvenile articular cartilage.
Statistical Analysis
Descriptive statistics were calculated for patient and clinical characteristics for the study cohort. For continuous data, means with standard deviations and ranges are reported, while frequencies and percentages are reported for discrete variables. To account for the missingness of data, longitudinal assessments of PROMs were analyzed using linear mixed models and parameter estimates summarized using means and standard errors. Changes across qualitative MRI findings against study time points were analyzed using chi-square tests. Linear mixed modeling was used to analyze the change in quantitative MRI measures across time. Kruskal-Wallis tests were used to compare the outcome survey scores between cartilage fill levels at each follow-up time in the study. Critical threshold for statistical significance was set to an alpha level of 0.05. All reported p-values were adjusted for any potential type I errors from multiple comparisons. P-values of 0.05 or below were considered statistically significant. All analyses were performed using SPSS Version 22.0 (IBM Corp).
Results
Patient Characteristics
From March 2014 to August 2019, a total of 70 lesions in 65 patients were treated with PJAC for patellar cartilage lesions (Table 1). The mean patient age was 26.6 years (range, 14-51 years), and 76% (n = 53) were women. A total of 25 (36%) knees had undergone previous surgery, and the mean symptom duration was 7.5 years. The mean patellar lesion size was 277 mm2 (range, 77-800 mm2), the mean number of allografts packets used was 2.4 (range, 1-5), and the mean donor age was 46.1 months (range, 1-120 months). Of the 70 knees, 68 (97%) underwent a concomitant patellar stabilization or offloading procedure: 35 (50%) a combined MPFL and TTO, 11 (16%) an isolated MPFL, and 22 (31%) an isolated TTO. A total of 33 (47%) knees had a lateral release, 2 of which were in isolation. A concomitant nonpatellar grade IV lesion was found in 27% of knees, with 17 (24%) on the trochlea. Twelve trochlear lesions were treated with osteochondral allograft (OCA), and 1 was treated with microfracture. The remaining were managed nonoperatively.
Patient Demographic and Clinical Characteristics a

BMAC, bone marrow aspirate concentrate; BMI, body mass index; MPFL, medial patellofemoral ligament; OCA, osteochondral allograft; ROH, removal of hardware; TTO, tibial tubercle osteotomy.
Clinical Outcomes
All patients reached the 2-year postoperative time point, with 58 (83%) of knees completing at least 2-year PROMs. The mean time to the last follow-up was 2.8 ± 1.6 years. A total of 23 knees (33%) had >2 years of follow-up (range, 3-7 years). There was a significant improvement in all postoperative patient-reported outcome (PRO) scores at the 1-, 2-, and >2-year follow-up, except for the Pedi-FABS, which showed no significant difference from baseline (Table 2, Figure 2). From the mean baseline to 2-year scores, the KOOS-QoL improved from 24.7 to 62.1, the IKDC score improved from 41.1 to 73.5, the KOOS-PS improved from 35.6 to 15, and the Kujala improved from 52 to 86.3. Six patients did not complete the baseline PROMs, and 7 patients did not complete follow-up PROMs.
PROMs at Baseline and Postoperatively a

Values are presented as mean (standard error of the mean). Bold P values indicate significance. IKDC, International Knee Documentation Committee questionnaire; KOOS, Knee injury and Osteoarthritis Outcome Score; Pedi-FABS, Pediatric Functional Activity Brief Scale; PROM, patient-reported outcome measures; PS, Physical Function Short Form; QoL, Quality of Life.
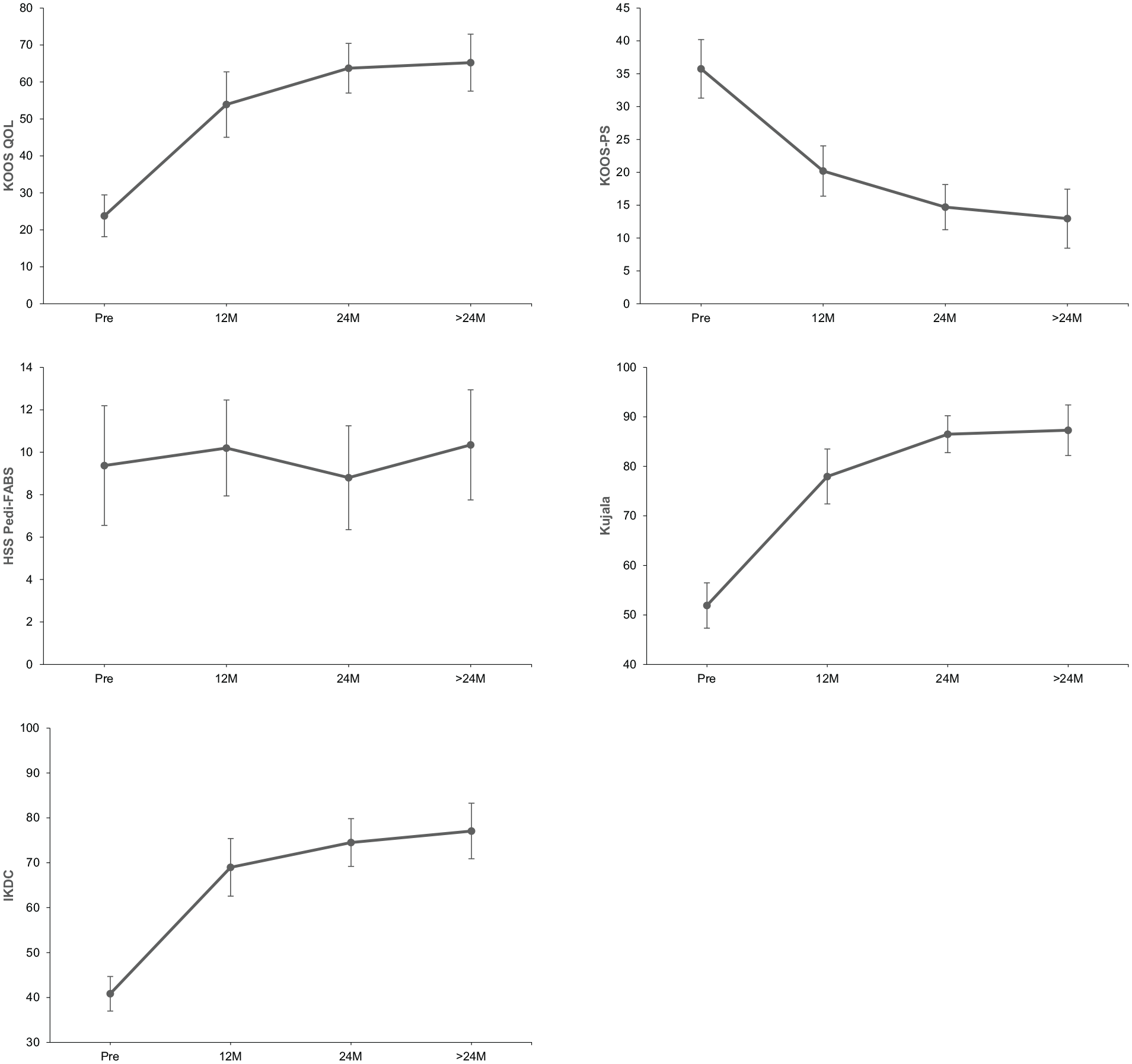
PROMs demonstrate significant improvements from baseline in most measures. IKDC, International Knee Documentation Committee questionnaire; KOOS, Knee injury and Osteoarthritis Outcome Score; Pedi-FABS, Pediatric Functional Activity Brief Scale; PROM, patient-reported outcome measure; PS, Physical Function Short Form; QoL, Quality of Life.
MRI Findings
Postoperative MRI demonstrated high rates of good defect fill (67%-100%) and low rates of graft failure, displacement, and hypertrophy (Table 3, Figure 3). Regarding graft fill, 39 (66%) defects had good fill at 3 months. Similar proportions of good fill were found at 6 months (72%), 1 year (74%), and ≥2 years (69%). Graft failure (no fill) was found in 10% of MRIs at 3 months and ranged from 7% to 13% across postoperative MRIs. None of the patients with graft failure demonstrated further fill on subsequent MRIs at 3 months. There were no documented cases of graft displacement at any time point. A small percentage of grafts had evidence of hypertrophy, ranging from 2% to 4%. There was a higher proportion of isointense grafts relative to native cartilage (31%; P < .001) and bony overgrowth (13%; P = .004) at the ≥2-year follow-up compared with earlier time points. The mean T2 relaxation time of the deep graft showed a significant decrease from 3 months to 1 year (55.3 [3.8] vs 43.9 [2.9]; P < .001) (Table 4). No other differences were observed in T2 relaxation times in the deep or superficial graft, cartilage, or integration zone.
Qualitative Postoperative Cartilage MRI Characteristics a
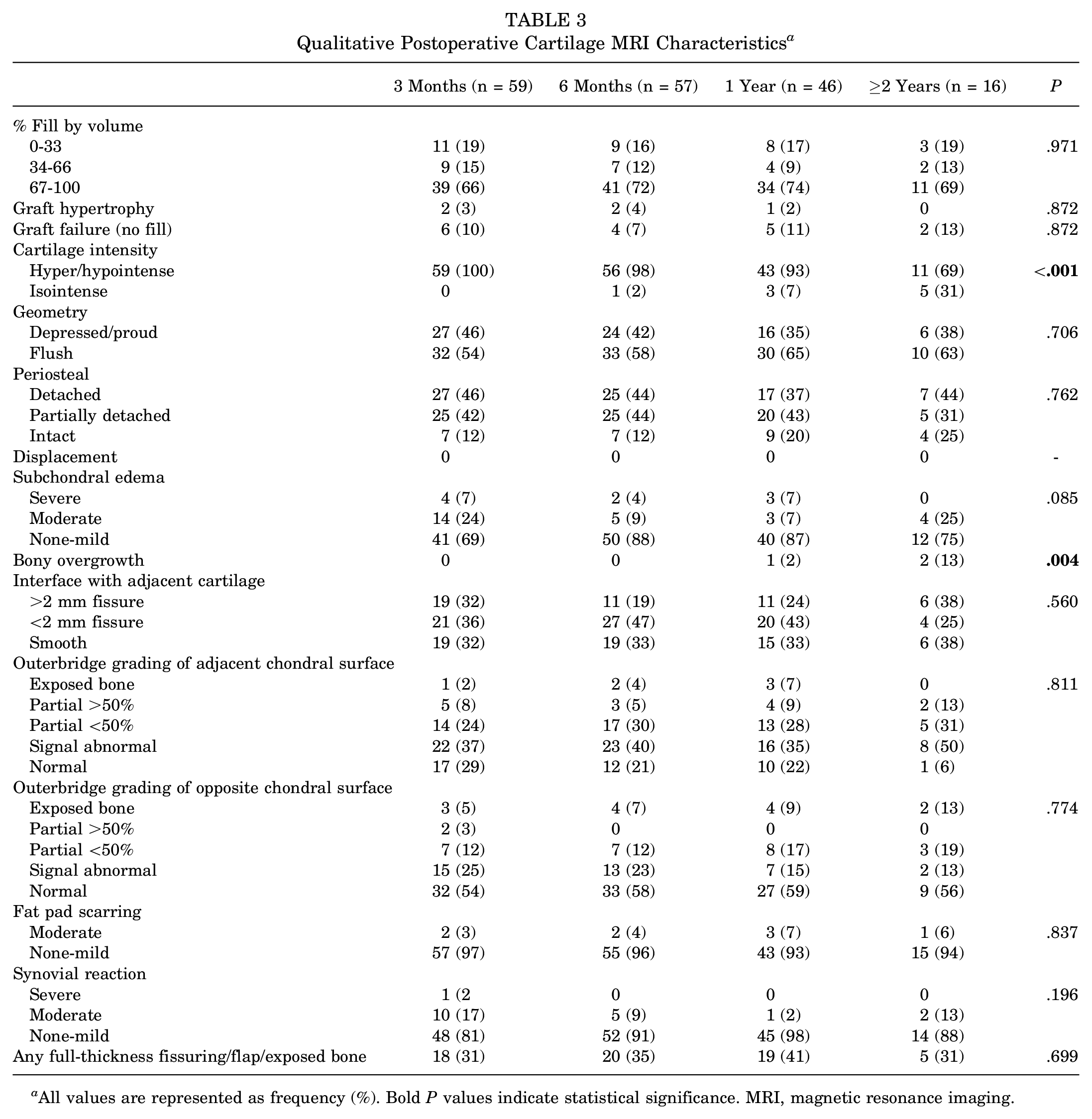
All values are represented as frequency (%). Bold P values indicate statistical significance. MRI, magnetic resonance imaging.

Postoperative patellar cartilage defect fill demonstrated that most patients had excellent fill (67%-100%). No significant differences were found in fill rates between postoperative time points.
Quantitative Postoperative Cartilage MRI Characteristics a
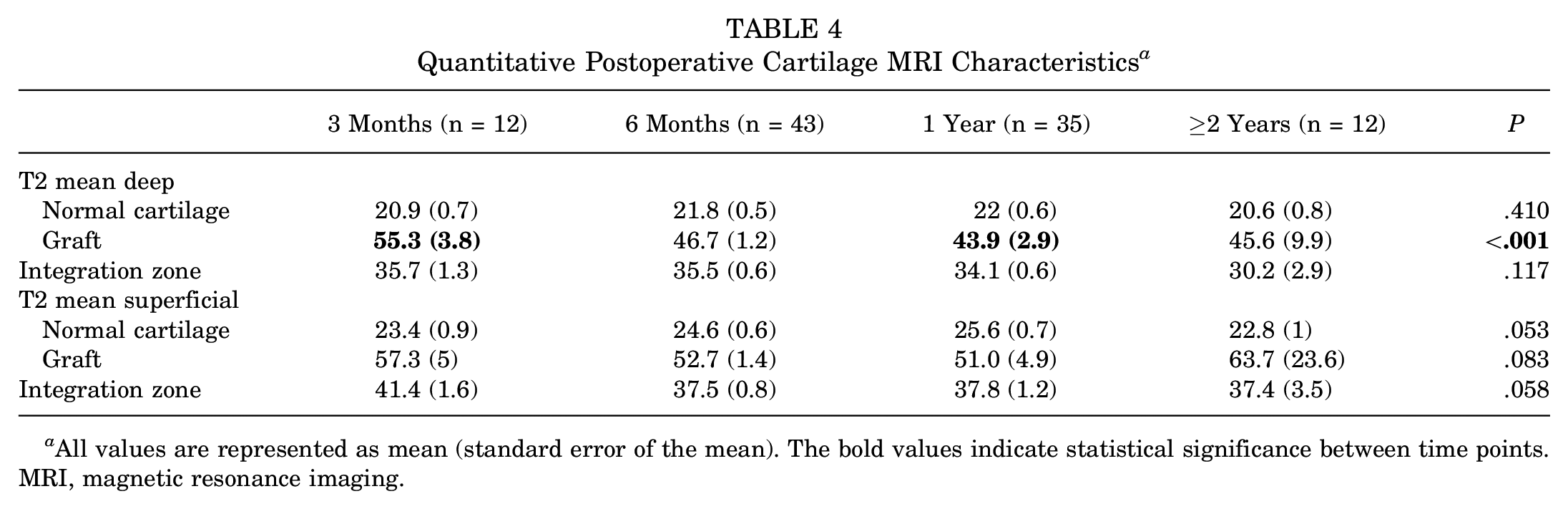
All values are represented as mean (standard error of the mean). The bold values indicate statistical significance between time points. MRI, magnetic resonance imaging.
PROs and Cartilage Fill
At the 1- and 2-year follow-up, patients with higher fill (67%-100%) reported improved scores in all PROMs compared with those with lower percent cartilage fill (0%-66%). However, those differences in the outcome scores were not statistically significant (Table 5). All PROMs trended toward improvement in lesions with good fill relative to poor to moderate fill. Similar findings were observed at the 2-year and final follow-up. At the 2-year follow-up, patients with good fill had a KOOS-QoL score improvement of 18.3 compared with patients with poor or moderate fill (P = .105). Improvements in all other PROM scores were also observed; however, these differences were not statistically significant. At the final follow-up, most PROMs were higher in the good fill group for the KOOS-QoL, Pedi-FABS, KOOS-PS, and Kujala scores; however, these findings were not significant.
PROM Stratified by Postoperative Cartilage Defect Fill a
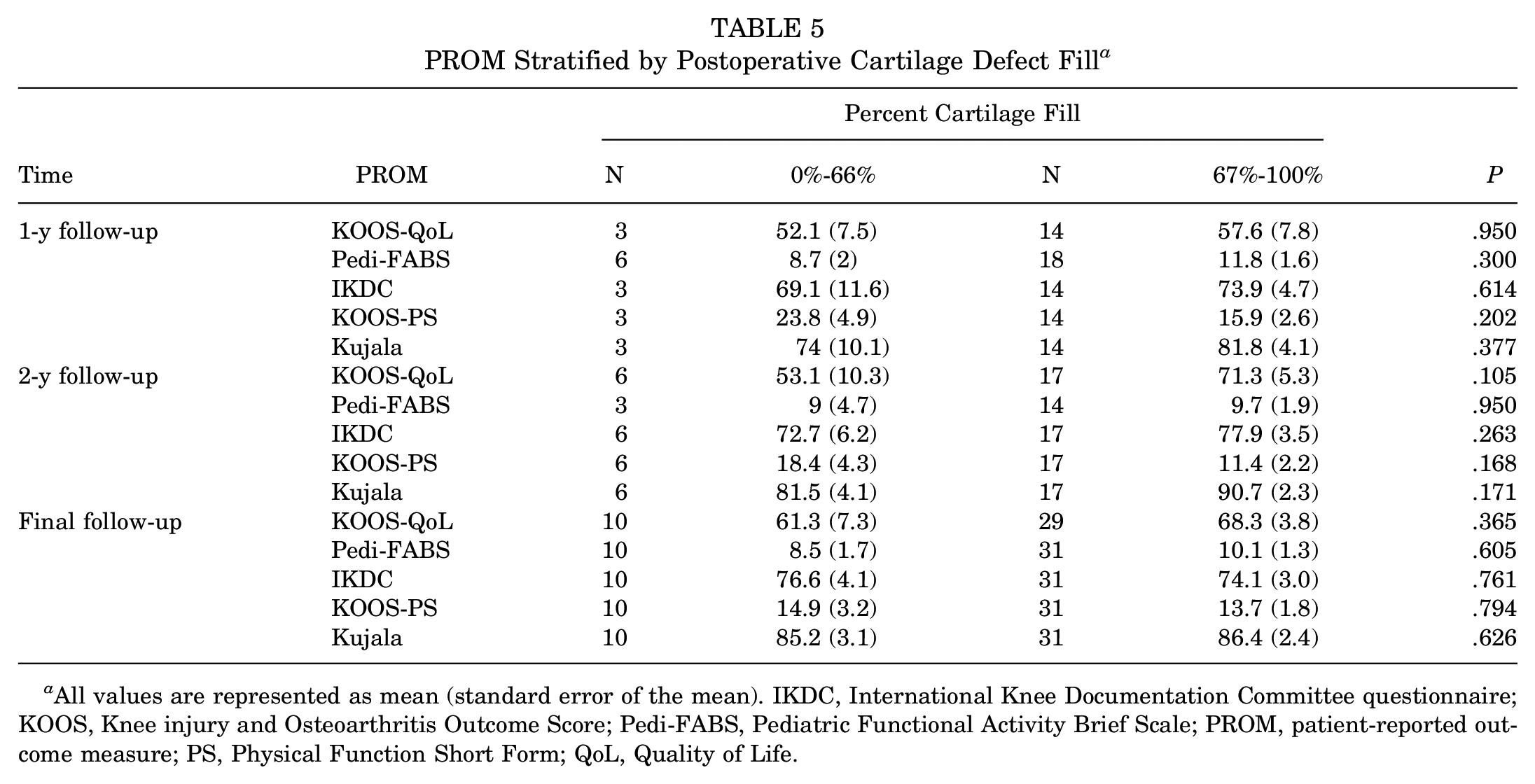
All values are represented as mean (standard error of the mean). IKDC, International Knee Documentation Committee questionnaire; KOOS, Knee injury and Osteoarthritis Outcome Score; Pedi-FABS, Pediatric Functional Activity Brief Scale; PROM, patient-reported outcome measure; PS, Physical Function Short Form; QoL, Quality of Life.
Discussion
Treatment of symptomatic patellar cartilage defects with PJAC demonstrated significant improvements in clinical outcomes with evidence of successful defect fill on MRI. Our study did not identify an association between cartilage fill and PROs over time. Functional knee outcome and quality of life scores significantly improved at the 1- and 2-year follow-up compared with baseline. Findings on MRI demonstrated high rates of good cartilage fill, ranging from 66% to 74%. Evidence for deep graft maturation was also observed with a significant decrease in T2 relaxation time from 3 months to 1 year.
Numerous strategies are currently employed to treat focal cartilage defects; however, optimal management is not yet clear. 11 PJAC, MACI, marrow stimulation, and osteochondral autograft transfer system (OATS) are some of the most commonly utilized techniques. PJAC may offer a desirable benefit profile for patellar chondral lesions compared with other treatments. Unlike MACI, only a single procedure is necessary for PJAC, decreasing the theoretical risk of knee morbidity and overall cost. 24 Moreover, in cases where the subchondral bone of the patella is not also compromised, PJAC provides a less invasive technique that can be molded to the patient-specific anatomy compared with alternative options that inherently include the bone, such as OCA and can be challenging to match the topography of the patella. To date, no alternative cartilage restoration technique has demonstrated superior clinical outcomes. A review by Noyes and Barber-Westin 32 compared the results of autologous chondrocyte implantation (ACI) and OCA for patellar lesions and could not identify a preferred treatment. Marrow stimulation techniques, such as microfracture, have been shown to produce fibrocartilage, which is structurally weaker than native hyaline cartilage. 28 Clinical outcomes after marrow stimulation have not been consistent; a systematic review of long-term outcomes after marrow stimulation showed varying degrees of success with up to 32% failure rates at 10 years. 33
This study found that functional knee outcomes and QoL scores significantly improved at the 1- and 2-year follow-up compared with baseline. No differences in Pedi-FABS scores were observed, suggesting similar patient activity pre- and postoperatively. Recent studies have demonstrated that PJAC is a safe and effective cartilage restoration technique for lesions of the knee.3,5,8,10,14,18,36-38 Buckwalter et al 5 reported short-term outcomes at 8 months in 13 patients with patellar chondral lesions treated with PJAC, of which 6 were treated with concomitant TTO. They found significant improvements in overall KOOS scores postoperatively; however, no differences were found in individual KOOS subscales. A study by Tompkins et al 37 reported clinical and MRI outcomes in 15 patellar lesions treated with PJAC at a mean follow-up of 29 months. Postoperative clinical outcomes showed high postoperative KOOS, IKDC, and Kujala scores. This study lacked preoperative subjective outcomes, and therefore, improvements from baseline could not be determined. Farr et al 14 conducted a prospective study of 25 patients with lesions of the trochlea or femoral condyles treated with PJAC and assessed clinical, MRI, and histological outcomes at 2 years and found significant improvements in IKDC and KOOS scores. A study by Wang et al 38 observed 27 patients with a minimum of 2-year follow-up and found significant improvements from baseline in the mean IKDC and Knee Outcome Survey Activities of Daily Living scores but no improvements in the Marx Activity score. 38 Also, 16 of their patients underwent concomitant MPFL (6), MPFL + TTO (4), or isolated TTO (6). The presence of a concomitant TTO during surgery did not influence outcomes.
In the present study, postoperative MRI demonstrated high rates of good cartilage fill, ranging from 66% to 74%. Evidence for deep graft maturation was also observed with a significant decrease in T2 relaxation time from 3 months to 1 year. Grawe et al 18 found moderate to good cartilage defect fill in up to 85% of knees and significant graft maturation on MRI up to 2 years postoperatively. Wang et al 38 found good cartilage fill (>66%) in 69% of their patients on the 2-year follow-up MRI. 38 Dawkins et al 10 assessed MRI outcomes in 36 patellofemoral lesions in patients <21 years old, which showed that 78% of patients had >50% defect fill. Tompkins et al 37 demonstrated overall improved joint cartilage, with a mean defect fill of 89% ± 19.6%. Farr et al 14 found graft maturation with characteristics similar to native cartilage and good lesion fill. The mean lesion fill increased from 44% at 3 months to >100% at 24 months. Similar findings have been reported with PJAC in different lesion locations, such as the talus, suggesting that PJAC may provide a robust and versatile option for cartilage lesions.1,6,9,16,19,23,39
The association of MRI and subjective outcomes in patients treated with PJAC has not been well defined in the literature. Wang et al 38 found no relationship between defect fill and subjective outcomes in patients treated with PJAC for patellofemoral lesions. However, unlike this study, which only included lesions of the patella, Wang et al included 22 lesions from the patella and 8 from the trochlea. The lack of association between defect fill and clinical outcomes in the present study may be the result of the overall clinical improvement because of concomitant patellar stabilization or unloading procedures, making it difficult to observe a more subtle association to be observed between patient outcomes and lesion fill. In our cohort, 68 (97%) patients were concomitantly treated with a combined MPFL and TTO, isolated MPFL, or isolated TTO. These procedures are likely strong contributors to improved subjective outcomes, making the association of outcomes with defect fill more difficult to identify.
The association between MRI and clinical outcomes has been better studied in several other restoration techniques, such as MACI, microfracture, and OATS. Niemeyer et al 29 conducted a study on clinical and MRI outcomes after MACI of the patellofemoral joint. Lesion locations included the patella, trochlea, and femoral condyles. While patients experienced improvements in functional outcomes, no association between MRI and clinical outcomes was observed. Blackman et al 2 conducted a meta-analysis in which MRI and clinical outcomes were evaluated after ACI, OATS, or microfracture. Graft hypertrophy, repair tissue signal, and subchondral edema had the strongest association with MRI outcomes in ACI and microfracture. Several studies included in the analysis found defect filling to be associated with clinical outcomes for both ACI and microfracture.20-22,26 De Windt et al 13 conducted a similar meta-analysis and found that for most MRI parameters, there was limited or no association with subjective outcomes. However, 9 of the 32 included studies found an association with defect fill.
In the present study, of the 70 patients, 6 (8.6%) experienced treatment failure at 3 months, evidenced by no lesion fill on MRI. Two patients had bilateral knees and underwent surgery at different time points; one patient had good fill on their contralateral knee and the other patient had no follow-up MRI. All patients had concomitant cartilage lesions in the trochlea, femoral condyles, or tibial plateau. The patellar lesion size ranged from 120 to 528 mm, and the number of PJAC packets used ranged from 2 to 5. One of the patients underwent concomitant MPFL + TTO, 5 underwent a TTO without an MPFL reconstruction, and 4 had a lateral release. Despite fill failure, patients, on average, showed improvements in PROM scores. Grawe et al 18 found that of the patients who had follow-up MRIs at 12 and 24 months postoperatively, 15% and 18% had poor graft fill (0-33%), respectively. Wang et al 38 reported that 31% of their patients at the 2-year follow-up had <67% lesion fill; however, they did not report graft failure rates. Dawkins et al 10 found that 22% of their patients had <50% fill, with 2 patients experiencing graft failure. In a study by Tompkins et al, 37 of the 16 patients observed, 3 patients required reoperation due to symptomatic grafts, 1 of which was due to poor defect fill. Farr et al 14 found that of their 25 patients, 2 (8%) experienced partial graft failure, and 1 experienced complete graft failure.
There are several limitations and strengths of this study. First, our cohort included patients with different degrees of PROM completion. Six of our patients had no baseline scores, and 7 patients had no 1- or 2-year follow-up scores. There were also incomplete rates of postoperative MRI follow-up, which may partially be explained by limitations in access to nonessential imaging during the COVID-19 pandemic. Our analysis of defect fill and subjective outcomes was limited by our sample size. The heterogeneous mix of concomitant surgical procedures also provides a limitation to our study, as it is difficult to determine the weight of each procedure on improvement in subjective outcomes. This study has several strengths. To the author's knowledge, no previous studies have assessed the association of cartilage fill after PJAC treatment and subjective outcomes. In addition, the prospective nature of our study design avoids the challenges associated with retrospective studies. Relative to the present literature, our sample size of 70 patients is large and includes both objective outcomes, such as MRI data, and subjective clinical outcomes.
Conclusion
This study found significant improvements in subjective clinical outcomes in patients undergoing PJAC for full-thickness patellar cartilage defects at the 1- and 2-year follow-up. MRI findings demonstrated that the cartilage defect fill of > 66% is achieved by 3 months. Moreover, PROMs were not significantly associated with the percent defect fill at the short-term follow-up in our sample of patients who underwent concomitant patellar instability procedures. At the short-term follow-up, clinical outcomes are difficult to associate with cartilage fill, likely because of the beneficial intervention of concomitant procedures. Future studies with long-term follow-up and larger cohorts are needed to determine outcomes at mid- and long-term time points and detect the potential association between MRI fill and subjective outcomes.
Footnotes
Final revision submitted November 5, 2023; accepted November 16, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: E.R.D. has received education payments from Arthrex, Gotham Surgical Solutions & Devices, and Smith & Nephew. S.S.B. has received education payments from Smith & Nephew and hospitality payments from Stryker. B.E.S. has received nonconsulting fees from Arthrex; consulting fees from Arthrex; education fees from Gotham Surgical Solutions & Devices; and her spouse is a consultant for Arthrex and receives royalties.
Ethical approval for this study was obtained from the Hospital for Special Surgery (reference No. 2018-0734).
