Abstract
Background:
Lateral elbow pain affects 1% to 3% of the general population. Neuropathy involving the posterior interosseous nerve (PIN) is a potential cause of lateral elbow pain but shares overlapping symptoms with other pathologies, particularly lateral epicondylitis, which may be difficult to distinguish based solely on patient history, physical examination, or electrodiagnostic testing (EDX). Conventional magnetic resonance imaging may reveal denervation edema or atrophy in PIN-innervated muscles, but abnormal signal intensity within the nerve may not be apparent.
Hypothesis:
By incorporating magnetic resonance neurography (MRN) as a diagnostic tool, physicians may be able to identify peripheral nerve pathologies and differentiate PIN neuropathy from other conditions.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
The authors conducted an institutional review board–approved retrospective study to evaluate MRN findings of patients with clinically suspected PIN neuropathy. Patients were initially identified via a radiology report database. Patients with previous elbow surgery or trauma and those with EDX-confirmed Parsonage-Turner syndrome were excluded. The cohort was age-matched and compared with a control group of patients with clinically suspected cubital tunnel syndrome. Two radiologists, blinded to patient symptoms, graded the MRN examinations focusing on peripheral nerves, muscles, and extensor tendon pathology. Independent-samples t tests and chi-square tests were used to compare means and proportions, respectively, between the 2 cohorts.
Results:
The final cohort consisted of 47 patients with suspected PIN neuropathy and 47 control patients. No significant demographic differences were found between the groups. Statistically significant findings in the study cohort compared with the control group included abnormal T2-weighted hyperintensity of the PIN-innervated extensor muscles (P = .0052), and the PIN (P = .0002), as well as enlargement of both the PIN (P < .0001), and the radial nerve proper (P = .0410).
Conclusion:
This study found that MRN can identify imaging characteristics indicative of PIN involvement compared with the control group. Future studies should focus on determining the prognostic value of these MRN findings for nonsurgical versus surgical intervention.
Keywords
Lateral elbow pain affects 1% to 3% of the general population, with most cases being diagnosed as lateral epicondylitis. Although uncommon, with an annual incidence of 0.03%, involvement of the posterior interosseous nerve (PIN) sometimes presents with symptoms that overlap those of lateral epicondylitis and may therefore be underdiagnosed.4,5 Individuals engaged in repetitive pronation and supination movements, such as racquet sports or woodworking, are most susceptible to both lateral epicondylitis and PIN entrapment. It is essential to distinguish between the 2 pathologies to provide the appropriate treatment to the patient.
The PIN is the deep motor branch of the radial nerve and theoretically susceptible to entrapment along its course in the elbow and forearm regions. 16 PIN neuropathy is often characterized by a sensation of a deep ache in the posterior forearm that can be accompanied by weakness of the forearm extensors and/or sensory alterations. 17 The potential compression sites are as follows 3 :
Fibrous bands at the proximal edge of the extensor carpi radialis brevis
Thickened fascial tissue superficial to the radiocapitellar joint between the brachialis and brachioradialis
Leash of Henry—arterial branches that arise from the recurrent radial artery and cross over the PIN
Arcade of Frohse—proximal superior portion of the supinator
Distal edge of the supinator
Accurate diagnosis of PIN neuropathy is challenging because of overlapping symptoms and its coexistence with lateral epicondylitis. These include variations in the nature of the pain, inconsistent locations of maximal tenderness, and a history of episodic pain that precedes the onset of weakness. Conversely, lateral epicondylitis typically presents with pain and burning around the lateral epicondyle of the humerus, which may radiate to the forearm or upper arm. 12
In addition to the difficulty in diagnosing these pathologies using physical examination alone, electrodiagnostic testing (EDX) may not always reveal abnormalities in the radial nerve; in fact, electromyography (EMG) and nerve conduction velocity studies in patients with these conditions are typically negative. 15 EMG may detect signs of denervation in the extensor muscles in PIN neuropathy; however, these findings lack specificity as similar EMG changes may also occur in Parsonage-Turner syndrome, an idiopathic spontaneous neuropathy. 3 For these reasons, providers are increasingly reliant on imaging modalities to substantiate their clinical suspicion of PIN pathology.
Conventional magnetic resonance imaging (MRI) may reveal denervation edema or atrophy in muscles along the distribution of the PIN. 8 However, abnormal T2-weighted hyperintensity within the nerve may not always be apparent. Recent technical advancements have refined magnetic resonance neurography (MRN), a technique that utilizes high-resolution, heavily T2-weighted imaging to effectively identify pathological changes of peripheral nerves, 18 but its use in diagnosing PIN neuropathy remains largely unreported.
In this study, we aimed to investigate the imaging features of the PIN and associated muscle changes on MRN in patients with clinically suspected PIN neuropathy. Findings were compared with those from a control group of patients with medial elbow pain suggestive of cubital tunnel syndrome, a completely different clinical entity, using age-matched comparisons.
Methods
Study Participants
Institutional review board approval was received from the Hospital for Special Surgery, and informed consent was waived for this retrospective study. To compare patients with and without PIN neuropathy, we divided participants into 2 groups: those with PIN pathology and the control group. Patients included in the PIN pathology group were identified through a search of the radiology report database using the keywords “posterior interosseous nerve” and “PIN” for MRN examinations of the elbow and forearm performed between January 1, 2016, and July 7, 2023. Patient age, sex, and body mass index were recorded. Clinical notes, including EDX reports when available, were reviewed to refine patient selection based on the clinical suspicion of PIN compression, manifested by symptoms such as lateral elbow pain, paresthesia/numbness, and/or motor weakness. We found a total of 74 patients fitting the original criteria. However, we excluded any patients who developed symptoms of PIN injury due to trauma and those with confirmed Parsonage-Turner syndrome.
The control group was identified by searching the radiology report database using the keyword “cubital tunnel syndrome,” also spanning the same date range as the PIN group. The control group consisted of an equal number of patients with clinically suspected ulnar neuropathy and without symptoms such as lateral elbow pain or weakness to suggest possible concomitant PIN involvement.
Image Acquisition
MRN examinations were performed on a 1.5-T unit (Optima MR450w or Optima MR450; GE Healthcare) in 6 cases and on a 3.0-T unit (Discovery MR750 or Signa Premier; GE Healthcare) in 88 cases using a flexible coil (16-21 channels) wrapped around the elbow region. The MRN protocol included axial 2-dimensional (2D) intermediate-weighted sequences (parameters at 3.0-T: field of view [FOV], 10-15 cm; echo time [TE], 29 milliseconds; repetition time [TR], 5475 milliseconds; matrix, 512 × 352; receiver bandwidth [RBW], 244 Hz/pixel; slice thickness, 3 mm) and axial and coronal 2D T2-weighted Dixon sequences (parameters at 3.0-T: FOV, 10-16 cm; TE, 87 milliseconds; TR, 4324 milliseconds; matrix, 320 × 224; RBW, 244 Hz/pixel; slice thickness, 2.5 mm). Occasionally, an axial inversion recovery sequence was obtained (parameters at 1.5-T: FOV, 10-16 cm; TE, 18 milliseconds; TR, 5206 milliseconds; acquired matrix, 320 × 192; RBW, 122 Hz/pixel). Three-dimensional (3D) sequences, including time-reversed FISP (PSIF) and double-echo steady-state (DESS), were also added during the scan, at the radiologist's discretion. PSIF was performed in 25 patients (parameters at 3.0-T: FOV, 11-15 cm; TE, 5 milliseconds; TR, 9.8 milliseconds; matrix, 320 × 224; slice thickness, 2 mm). DESS was performed in 5 patients (parameters at 3.0-T: FOV, 11-15 cm; TE, 5.3 and 9.5 milliseconds; TR, 14.7 milliseconds; matrix, 320 × 320; slice thickness, 1.6 mm).
Qualitative Image Evaluation
MRN examinations were evaluated independently by 2 musculoskeletal radiologists in the research team, one with 9 years of dedicated experience in MRN (Y.E.) and 1 with 3 years of experience (Y.L.), blinded to the patients’ clinical symptoms. Before independent grading, the 2 radiologists attended a consensus session to review the grading scheme and scales on separate image datasets not analyzed in this study. The following intrinsic nerve features were evaluated in the PIN, radial nerve proper, and superficial sensory branch of the radial nerve (SSBRN): signal hyperintensity (on T2-weighted Dixon sequences) (normal, mild, or moderate to marked, based on the signal intensity of the other nerves and muscles for reference), size (normal or enlarged), and perineural fat planes (normal or loss). Additionally, the number of affected slices (<2 or ≥3), along with the following locations of these abnormalities, was recorded: (1) proximal to the supinator tunnel, (2) at the entrance of the supinator tunnel, and (3) midway within the supinator tunnel. The PIN-innervated extensor muscles typically included within the field of view of elbow MRN—namely, the supinator, extensor carpi radialis brevis, extensor digitorum, extensor digiti minimi, extensor carpi ulnaris—were evaluated for the presence of T2-weighted hyperintensity (none, mild, or moderate to severe), muscle bulk (normal, mild atrophy, or moderate to severe atrophy), and presence of disproportionate fatty infiltration (none or present). The arcade of Frohse was assessed as normal in thickness or thickened if it was more than just a thin sliver of tissue. As symptoms of PIN neuropathy are known to overlap with those of lateral epicondylitis, the common extensor tendon was graded for tendinosis (none, mild, moderate, or severe) and tear (low-grade partial, moderate to high-grade partial, or full-thickness).
Statistical Analysis
Statistical analyses were performed by a biostatistician (J.T.N.). Continuous variables were analyzed using t tests to compare means between the PIN group and the control group. Categorical variables were evaluated using chi-square tests and Fisher exact tests to identify differences in proportions. Interreader agreement was assessed using the Cohen kappa (κ). The strength of agreement was categorized as follows: <0 = poor, 0.00 to 0.2 = slight, 0.21 to 0.40 = fair, 0.41 to 0.60 = moderate, 0.61 to 0.80 = substantial, and 0.81 to 1 = almost perfect. 14
A P value <.05 was considered statistically significant.
Results
From the initial search, 74 patients were initially identified. Four patients were excluded due to the absence of PIN symptoms documented in the clinical records. An additional 18 patients were excluded due to symptom on set following trauma or surgery, including direct injury to the elbow or proximal forearm, or recent surgical intervention in the lateral elbow region. Three patients with EDX-confirmed Parsonage-Turner syndrome were excluded. The final cohort comprised 47 patients after excluding 2 additional patients to meet the age-matching criteria with the control group (±3 years). An equal number of age-matched patients with clinically suspected ulnar neuropathy, but without signs of PIN involvement, served as the control group for comparison. Both groups underwent MRN using similar techniques.
In terms of demographic characteristics, there were no significant differences in mean age between the PIN group (50.7 ± 13.5 years) and the control group (50.6 ± 13.7 years) (P = .952). For age, the standardized difference was significant (<0.10), indicating that the age match was successful.
Table 1 shows the reliability of MRN interpretations performed by 2 radiologists for the PIN group. The analysis of interrater reliability revealed substantial to almost perfect agreement between the 2 radiologists for all measurements.
Reliability Measurements Between Independent Radiology Raters a
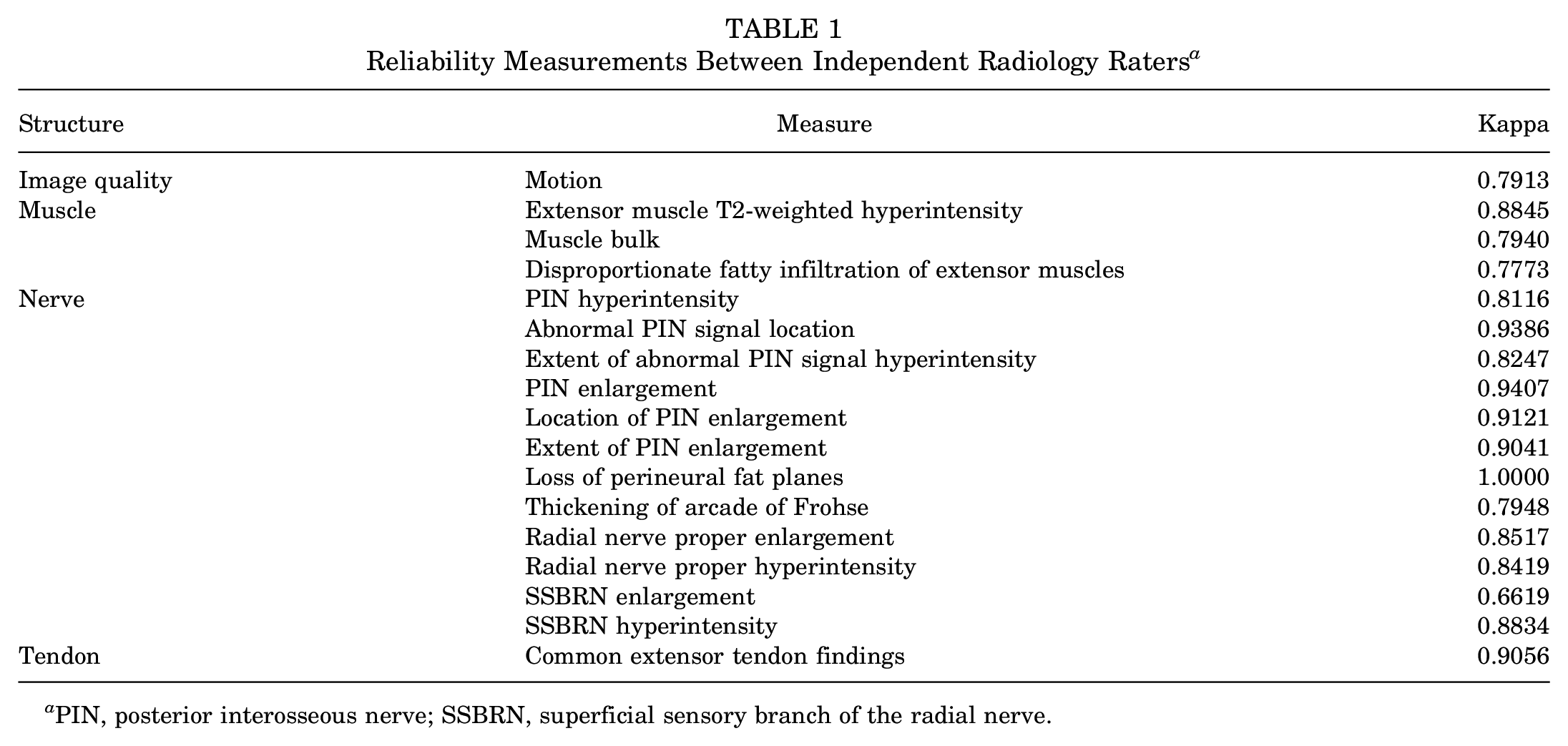
PIN, posterior interosseous nerve; SSBRN, superficial sensory branch of the radial nerve.
Considering the high interrater reliability demonstrated between both radiologists’ measurements, the measurements of 1 radiologist (Y.E.) were used for comparative analysis of the differences between the PIN group and the control group (Table 2).
Differences in Radiographic Measurements Between PIN and Control Groups a

Comparative analysis was done using 1 radiologist's measurements with chi-square tests and Fisher exact tests. Bold P values indicate statistical significance. NA, not applicable; PIN, posterior interosseous nerve; SSBRN, superficial sensory branch of the radial nerve.
In the evaluation of nerves, our study shows a statistically significant difference (P = .0002) in abnormal T2-weighted hyperintensity of the PIN. Specifically, 25 patients (53%) exhibited mild signal hyperintensity, and 10 patients (21%) exhibited marked signal hyperintensity, compared with 19 patients (40%) and 0 patients, respectively, in the control group (Table 2, Figures 1 and 2).

Images obtained in a 48-year-old man with persistent right lateral elbow pain despite injections around the lateral epicondyle. (A) Axial intermediate-weighted image demonstrates a thickened edge of the arcade of Frohse (arrowheads) and enlargement of the posterior interosseous nerve (PIN) (arrow). (B and C) Sequential axial double-echo steady-state images, with (D) a curved multiplanar reformation, illustrate hyperintensity and enlargement of the PIN throughout the supinator tunnel (arrows). Note the normal size and signal intensity of the ulnar nerve (dashed arrows in B and C).

Images obtained in a 69-year-old man with left forearm paresthesias and weakness. (A) Axial T2-weighted Dixon image shows a diffuse edema pattern of the extensor musculature (dashed ellipse), compatible with active denervation. (B) Axial gradient echo image depicts enlargement and hyperintensity of the posterior interosseous nerve (PIN) at the entrance to the supinator tunnel (arrow). (C) Curved multiplanar reformatted gradient echo image further demonstrates enlargement and hyperintensity of the PIN throughout the supinator tunnel (arrows).
PIN enlargement was also significantly more common in the PIN group (43%) compared with the control group (4%) (P < .01). Radial nerve proper enlargement also differed significantly between the 2 groups (P = .04), while its T2-weighted hyperintensity did not (P = .18). In contrast, no statistical differences between the MRN features of SSBRN were present between the PIN and control groups. While the location and intensity of abnormal PIN signal, PIN enlargement, and abnormal thickening of the arcade of Frohse were not statistically different, the PIN group tended to show abnormal MRN findings to a greater degree compared with the control group, as shown in Table 2.
For the evaluation of muscles, a statistically significant difference was observed in the T2-weighted signal of the extensor muscle, in which 16 patients (34%) of the PIN group displayed an abnormal hyperintensity compared with 4 patients (9%) in the control group (P = .0052) (Table 2). Disproportionate fatty infiltration of extensor muscles was also not statistically different between the groups (P = .2163), although this finding was present in 8 PIN patients (17%) compared with 4 control patients (9%).
Regarding tendon abnormalities, our study showed no significant differences in the presence of tendinosis or the degree of tears of the extensor tendon between groups (P = .2837) (Table 2). The PIN group had a higher frequency of low-grade partial tears (12 patients [26%] vs 6 patients [13%] in the control group). In fact, 27 of the PIN patients (57%) had some degree of tendinosis or tendon tear. On the other hand, the control group had a higher frequency of moderate to high-grade partial tears (4 patients [9%] vs 3 patients [6%] in the PIN group) and complete tears (1 patient [2%] vs 0 patients in the PIN group).
Among the 47 patients in the PIN group, 19 had available EMG results. Of these, 5 demonstrated findings consistent with PIN neuropathy, 2 with radial nerve proper neuropathy, and 2 with cervical radiculopathy. An additional 5 patients had other neuropathies, including median neuropathy and multifocal acquired demyelinating sensory and motor neuropathy. Notably, 5 patients had normal EMG findings. Although all 19 patients had clinical symptoms suggestive of PIN neuropathy, 9 also exhibited findings of radial nerve proper involvement, which, when correlated with MRN and clinical findings, were consistent with a diagnosis of PIN neuropathy. Additionally, the diagnosis was further supported in 12 patients in the PIN group by the response to various interventions, including PIN release/decompression (n = 4), ultrasound-guided anesthetic injection (n = 6), or tenotomy of the supinator/extensor carpi radialis brevis (n = 2).
Discussion
This study evaluated MRN findings in patients with clinically suspected PIN neuropathy compared with an age-matched control group. Our findings revealed a significantly higher percentage of abnormal PIN T2-weighted hyperintensity, PIN enlargement, and abnormal T2-weighted hyperintensity of the extensor muscles in the PIN group compared with the control group.
PIN neuropathy may result from entrapment, trauma, or compression by adjacent lesions—many located within the radial tunnel. PIN neuropathy is a rare condition with an estimated annual incidence rate of 0.03%.7,20 For reference, the most prevalent peripheral neuropathy is carpal tunnel syndrome, with an estimated annual incidence of 0.1% to 0.35%.1,20 Like many peripheral neuropathies, the diagnosis of PIN neuropathy has traditionally relied on a combination of clinical history, physical examination, and electrodiagnostic studies, although these clinical assessments may not always accurately localize the site of pathology.
Conventional MRI has previously been reported to be a useful tool for identifying signs of PIN neuropathy by showing denervation edema or atrophy within the supinator and/or extensor muscles, 8 findings that were also demonstrated in this study. However, conventional MRI might fail to detect abnormal signal intensity in the PIN 8 due to (1) inadequate spatial resolution to effectively visualize the PIN, (2) insufficient T2-weighted contrast, and (3) inadequate vascular suppression due to the PIN's frequent proximity to neighboring blood vessels. 9 With advancements including greater access to high magnet field strengths (3.0-T) and gradient performance, MRN offers enhanced visualization of peripheral nerves and associated pathologies through a combination of 2D and 3D sequences, 10 and its role is becoming increasingly significant in the management of diverse peripheral nerve disorders. 6
Using MRN, our study identified a significantly higher percentage of T2-weighted signal hyperintensity of the PIN in the PIN group. This hyperintensity is thought to reflect intraneural edema secondary to inflammation and/or ischemia of the PIN. 13 The location of abnormal signal hyperintensity was further analyzed as the PIN is anecdotally observed as being hyperintense at the entrance of the supinator tunnel (arcade of Frohse) even in asymptomatic patients. 2 While most abnormalities were located at the entrance of the supinator tunnel for both groups, in the PIN group, the abnormal signal intensity often extended proximally beyond the entrance and continued distally through the tunnel. This contrasts with a previous report suggesting that most lesions occurred at the fascicular level of the more proximal radial nerve. 2 The differences may stem from cohort variations, as Parsonage-Turner syndrome, typically involving the fascicular level above the elbow, was not excluded in that study. 19
In addition to hyperintensity of the PIN, enlargement of the PIN could be a valuable indicator, which has not been specifically reported before. Our findings suggest that these nerve features are indicative of PIN involvement and may serve as supportive diagnostic criteria when assessed using MRN. However, the analysis of the location and extent of abnormal T2-weighted signal or enlargement of the PIN could be impacted by small sample sizes. Lastly, the higher percentage of cases exhibiting hyperintensity and enlargement of the radial nerve proper and SSBRN warrants that radiologists extend their evaluation beyond the PIN in patients evaluated with lateral elbow pain or clinically suspected PIN neuropathy.
Regarding overlap with extensor tendon abnormalities, our study shows that 27 patients (57%) in the PIN group had some degree of tendinosis or tendon tear. Notably, the control group had a higher frequency of moderate to high-grade partial and complete extensor tendon tears. As these control patients were not experiencing lateral elbow pain, this could imply that tendon abnormalities are common and may not correlate with clinical symptoms.
Limitations
There were several limitations to our study. The retrospective nature and relatively small sample size may limit the generalizability of our findings. However, we age-matched the PIN group with the control group to improve the comparability of results. Notably, this study represents one of the largest cohorts to date for evaluating PIN neuropathy using MRN. It is also important to acknowledge that most patients did not undergo electrodiagnostic testing, which limited correlation with MRN findings beyond clinical evaluation. Additionally, most patients in the PIN group did not have surgical confirmation of compression, primarily due to the nonsurgical approach to initial treatment of PIN neuropathy, with surgical intervention reserved for refractory cases. 11 Future research is needed to examine the correlations between MRN findings and surgical findings in patients with PIN compression.
Conclusion
PIN neuropathy is a potential cause of lateral elbow pain but shares overlapping symptoms with lateral epicondylitis. MRN may complement clinical evaluation of suspected PIN neuropathy by identifying imaging characteristics such as PIN T2-weighted hyperintensity, PIN enlargement, and associated T2-weighted hyperintensity of the PIN-innervated extensor muscles. Further studies are warranted to correlate MRN findings with surgical outcomes.
Footnotes
Final revision submitted February 7, 2025; accepted April 14, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: J.T.N. was supported in part by funds from the Clinical Translational Science Center (CTSC), National Center for Advancing Translational Sciences (NCATS) grant No. UL1-RR024996. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding source, NCATS, based in Rockville, Maryland, USA. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Hospital for Special Surgery.
