Abstract
Background:
Bridge-enhanced anterior cruciate ligament (ACL) restoration (BEAR) surgery has demonstrated, through clinical outcomes and magnetic resonance imaging evaluation, the ability to restore the injured native ACL. However, the integrity of the quality of the tissue restored has yet to be studied histologically in the setting of retear.
Purpose/Hypothesis:
The purpose of this study was to evaluate the histologic composition of BEAR procedure retear tissue in human patients. It was hypothesized that tissue biopsy specimens taken at the location of retear after BEAR procedures will resemble torn native ACL tissue.
Study Design:
Case Series; Level of evidence, 4.
Methods:
Patients undergoing revision ACL surgery after a failed BEAR procedure were consented for tissue biopsy at their treating institution. Tissue biopsy specimens were taken at several locations across the site of the retear, with cluster biopsy specimens taken at the retear site. Hematoxylin and eosin–stained slides were prepared and examined by a single pathologist. The intraoperative location of retear was recorded to be cross-referenced with the original site of tear based on the Sherman classification. Histologic parameters (0-4) included presence of acute inflammation, lymphocytes, eosinophils, macrophages, giant cells, organisms/evidence of infection, evidence of neovascularization, and fatty infiltrate. Foreign material, malignancy, ligamentous/fibrous tissue, and synovial tissue were assessed. The appearance of the tissue in relation to normal-appearing ACL was also evaluated.
Results:
Five patients were identified for inclusion. The retear of the BEAR and analysis occurred at a mean of 482.4 days (16 months) (range, 366-623 days [12-20.7 months]) after implantation. No cases demonstrated evidence of acute inflammation (neutrophilic infiltrate), while lymphocytic infiltrate was noted in all cases, ranging from rare (80%) to heavy infiltrate (20%). Foreign materials from previous procedures were present in 2 cases (40%) with associated histiocytic infiltrate and giant cells. Ligamentous tissue and synovial tissue were noted in all cases (100%) with evidence of neovascularization. Fatty tissue was identified in 3 cases (60%), while no cases showed evidence of organisms/infection or malignancy. Absence of grossly fibrotic tissue was noted at the biopsy sites.
Conclusion:
Histological evaluation of BEAR retears in human patients supports the hypothesis that the tissue restored by the implant is similar to native ACL collagen.
Anterior cruciate ligament (ACL) tears represent a prevalent condition frequently encountered by orthopaedic surgeons, with an annual incidence of 400,000 cases. 5 Despite its common occurrence, there remains ongoing debate regarding the optimal treatment approach. For the past 3 decades, ACL reconstruction (ACLR) has stood as the gold standard for addressing these injuries. 4 However, in part based on growing research and technological advances, renewed interest has occurred in examining the ability to repair or restore the native ACL.
The bridge-enhanced ACL restoration (BEAR) procedure provides an alternative to reconstruction of the torn ACL. The BEAR implant is a decellularized, bovine-derived, type I collagen implant that resorbs within 8 weeks of implantation. After surgical implantation, the proprietary composition of the implant allows for a fibrin clot to be maintained at the site of healing when activated with the patient’s own blood. The function of the implant allows for a “bridge” between the torn ACL and the femoral attachment site, allowing for cellular migration to complete the healing process between the torn ends of the ligament (Figure 1). 9 To date, clinical trials performed by Murray et al8,9 have established non-inferiority in patients who received the BEAR procedure compared to those who received either a semitendinous-gracilis or bone-tendon-bone ACLR autograft reconstruction. Furthermore, Proffen et al 12 noted psychological readiness to return to sport was higher in BEAR patients than in reconstruction patients.

The bridge-enhanced anterior cruciate ligament (BEAR) implant. The BEAR implant is a proprietary protein-based scaffold implant between the 2 torn ends of the ligament. It is rehydrated with the patient’s own whole blood which allows it to become gelatinous for implantation within the body.
Despite these clinical results, skepticism remains over the quality of the tissue that has been restored using the BEAR. Ligamentization is the process by which an ACL graft undergoes healing and remodeling after ACLR.13,15 This process has been studied extensively and described well in the literature. Histologically, as early as 3 weeks after patellar tendon autograft ACLR, original fibroblasts and new cells arising from the neovascularity have been observed. 14 Biomechanically, Marumo et al 7 found ACLR had remodeled sufficiently at 1 year to have similar characteristics to native ACL tissue.
While histological remodeling of ACL graft reconstructions has allowed for a better understanding of the healing process, little is known histologically of the ACL after primary repair via the BEAR procedure. In animal models, the BEAR ACL was noted to gradually change from a relatively disorganized fibrovascular scar to a more highly aligned collagenous structure over the first year after surgery. 9 However, in human patients, most studies to date examining BEAR ACL restoration have focused on magnetic resonance imaging (MRI) findings, clinical examination, healing potential, and risk of retear and revision surgery.2,6,9,10
The purpose of this study is to evaluate the histologic composition of BEAR procedure retear tissue in human patients. It is hypothesized that tissue biopsy specimens taken at the location of retear after BEAR procedures will resemble torn native ACL tissue.
Methods
Patients undergoing revision ACL surgery after a failed BEAR implant procedure were consented for tissue biopsy at their treating institution. All included patients were revised to a traditional autograft ACLR. Tissue biopsy specimens were taken at several locations across the site of the retorn ligament, with cluster biopsy specimens taken at the retear site. Biopsy specimens were taken using an arthroscopic punch and grasper. After multiple punch biopsy specimens were taken, the remainder of the ACL stump was excised using arthroscopic scissors and a grasper when possible. Histology slides of the biopsy specimens and remaining specimen were prepared and examined by a single pathologist (J.P.). The location of retear was recorded based on intraoperative evaluation to be cross-referenced with the original site of the tear (Table 1). Classification of the tear site was uniform based on the Sherman classification and was recorded by the surgeon per protocol in the BEAR III clinical trial (Table 2). 17
Location of the Index ACL Tear of the Native Ligament Recorded and Cross-Referenced With the Location of Retear a
Tissue biopsy specimens were taken for histological analysis clustered at the site of the retear. ACL, anterior cruciate ligament; MRI, magnetic resonance imaging.
Sherman Classification Describing Location of the ACL Disruption and Quantifying Remaining ACL Tissue a

The location of the injury is named from the femoral side of the attachment of the ACL.
Pathology
Tissues submitted by the surgeons were entirely for histologic evaluation. As noted in Table 1, the site of the most recent tear was identified intraoperatively and labeled for evaluation. The remaining ACL tissue was evaluated histologically along a vertical plane from proximal to distal. The approximate location of the original tear location had been noted from the original operative report for cross-referencing. Similarly, the extent and location of the retear was also noted for comparison. For each case, hematoxylin and eosin–stained slides were reviewed by a central pathologist (J.P.). Histologic parameters included acute inflammation (0-4), presence of lymphocytes (0-4), presence of eosinophils (0-4), presence of macrophages (0-4), presence of giant cells (0-4), presence of organisms/evidence of infection (0-4), evidence of neovascularization (0-4), and fatty infiltrate (0-4) (Table 3). The presence of foreign material, malignancy, ligamentous/fibrous tissue, and synovial tissue was assessed. The appearance of the tissue in relation to normal-appearing ACL was also evaluated.
Scoring System for Each Histologic Parameter Used by a Single Pathologist on All 5 Patient Specimens

Results
Five patients across 2 institutions were identified with retears after a BEAR procedure and were consented for histological analysis over a 5-year period. The retear of the BEAR and analysis occurred at a mean of 482.4 days (16 months) (range, 366-623 days [12-20.7 months]) after implantation (Table 4). Four of the 5 patients had passed functional testing and were cleared to return to sport. One patient had arthrofibrosis that was documented 3 months after procedure. This patient (patient 5) underwent an arthroscopic lysis of adhesions and manipulation at 5 months after the procedure. At the time of the lysis, the ACL was noted to be intact (Figure 2). The mean time from retear to tissue biopsy was 191.6 days (range, 77-440 days) (Table 5). The mechanism of injury was described for each retear. In 3 of the patients, the mechanism of action was described as a pivoting maneuver, while the other 2 patients noted jumping injuries. MRI was performed on each patient after reinjury. MRI identified partial tears in each 5 patients. In each instance, heterogeneous signal intensity consistent with high-grade partial retear of the ACL was noted.
Time From Implantation of BEAR to Histologic Biopsy a

All 5 patients were biopsied 1 year following the BEAR procedure. BEAR, bridge-enhanced anterior cruciate ligament restoration.

Right knee of a patient who underwent bridge-enhanced anterior cruciate ligament (ACL) restoration procedure and developed arthrofibrosis. The patient was taken back to the operating room at 5 months after implantation for a lysis. At this time, the ACL was found to be intact (arrow). The patient subsequently went on to tear the ACL after return to sport at month 10 after implantation.
Time From Retear to Histologic Biopsy

Restoration Cases
Using the scoring system identified in Table 2, the histological classification of each biopsy was identified. No infection or malignancy was identified. All 5 samples demonstrated ligamentous tissue and synovial tissue.
Histologic information is summarized in Table 6. None of the cases demonstrated evidence of acute inflammation (neutrophilic infiltrate), while lymphocytic infiltrate was noted in all cases (100%), ranging from rare (4 of 5; 80%) to heavy infiltrate (1 of 5; 20%) (Figure 3). Foreign materials from previous procedures were present in 2 cases (40%) with associated histiocytic infiltrate and giant cells. Ligamentous tissue and synovial tissue were noted in all cases (100%) with evidence of neovascularization (Figures 4 and 5). Fatty tissue was identified in 3 cases (60%), while no cases showed evidence of organisms/infection or malignancy (Figures 3-5). Type I collagen was noted predominantly at the biopsy sites. Absence of grossly fibrotic tissue was noted at the biopsy sites. There was no evidence of remnant collagen implant identified in any of the biopsy specimens.
Histologic Findings of Failed Bridge-Enhanced Anterior Cruciate Ligament (ACL) a

Using the scoring system identified in Table 2, the histological classification of each biopsy was identified. No infection or malignancy was identified. All five samples demonstrated ligamentous tissue and synovial tissue.

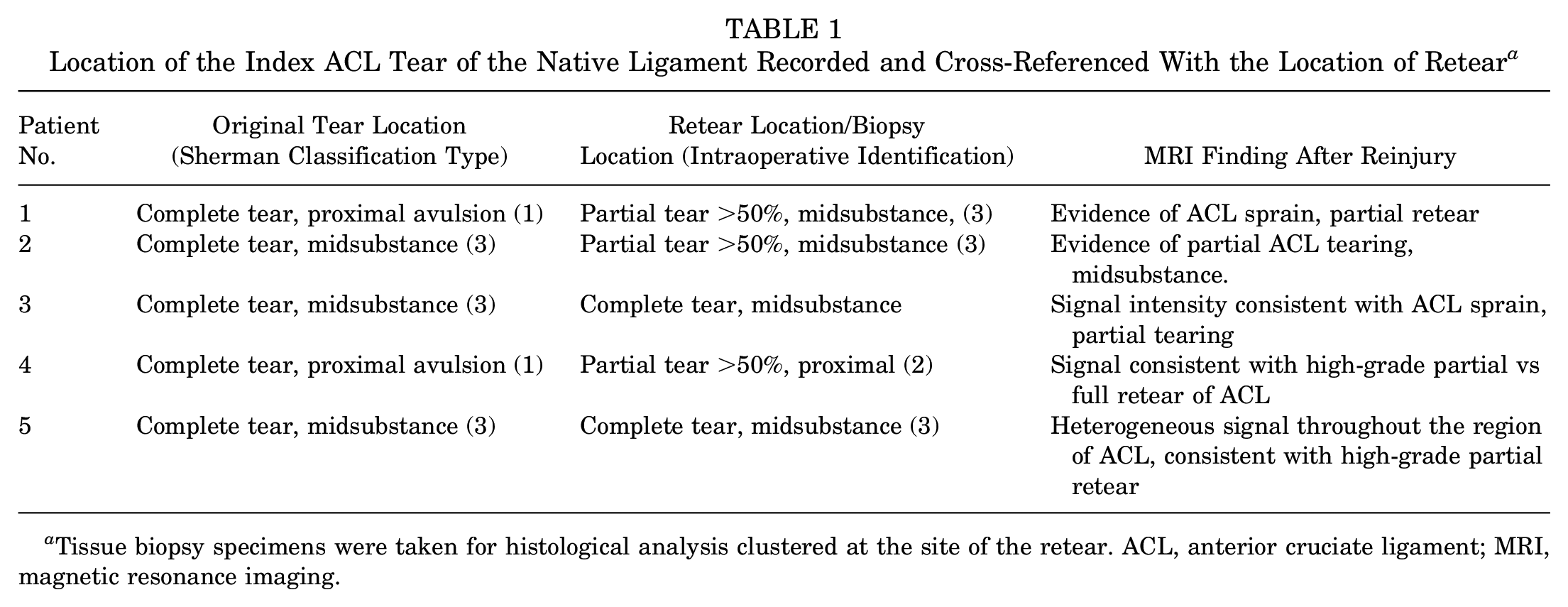
Synovial tissue from patient 3 shows (A) multinucleated giant cells (arrow) with associated foreign materials and (B) perivascular lymphocytic inflammation (arrowheads). Hematoxylin and eosin 10X.

Tissue from patient 4 demonstrates (A) dense fibrous tissue with (B) focal area of neovascularization characterized by capillary proliferation (bordered by arrowheads). Hematoxylin and eosin 10X.
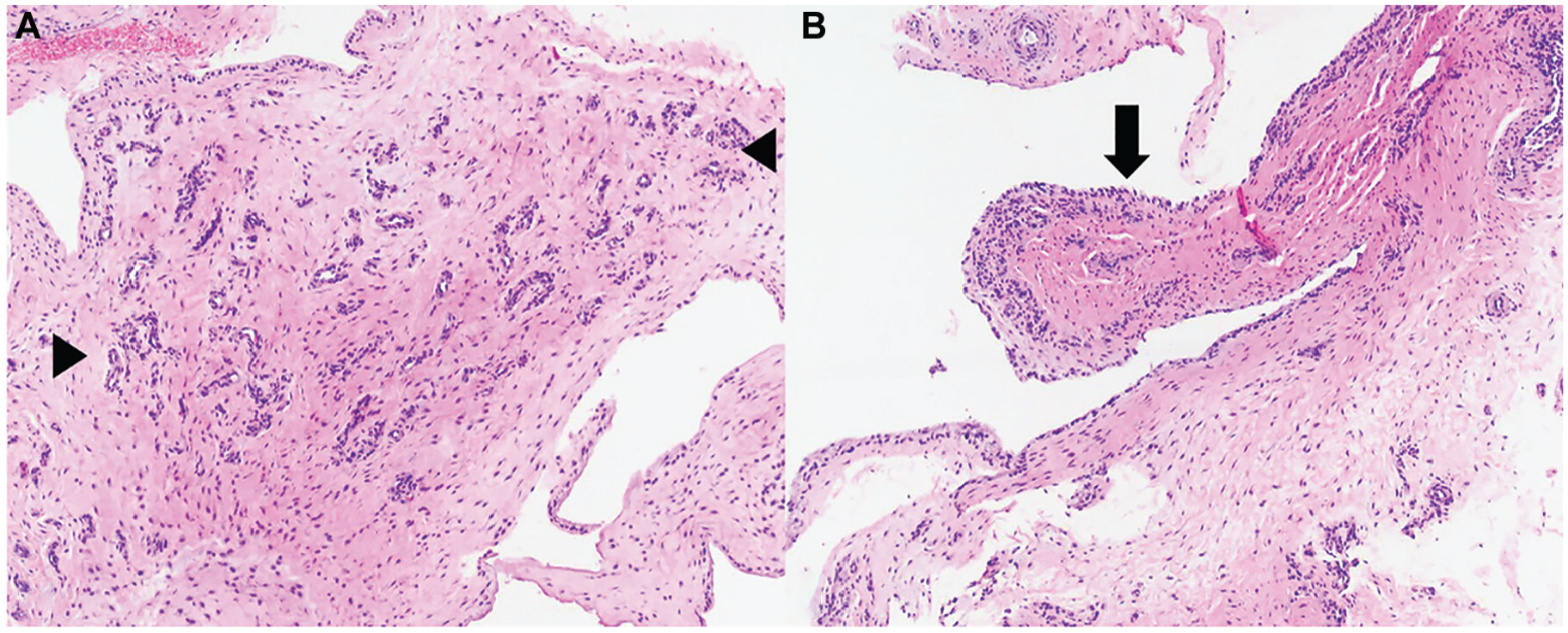
Tissue from patient 5 shows (A) neovascularization with capillary proliferation (the area between arrowheads) and (B) reactive synovial lining (arrow). Hematoxylin and eosin 10X.
Discussion
The present paper is the first to histologically evaluate the composition of tissue associated with the BEAR implant after a retear in humans. Histological analysis can prove valuable in appraising the quality and maturation of the healing tissue and substantiate clinical outcomes associated with biologic implants.16,18 Arnoczky et al 1 examined tissue biopsy specimens of the rotator cuff during second-look arthroscopies in 7 patients who underwent treatment for rotator cuff disease using a type I bovine collagen implant. Their findings demonstrated highly organized collagen at 6 months, which suggests functional loading of the newly generated tissue, which had the histologic appearance of tendon. While a small subset of patients, their findings nonetheless provided an early look and confidence for the tissue produced from the use of a bioinductive implant.
Before the BEAR procedure, successful ACL repair across the spectrum of ACL tear patterns was often not feasible based on tear location and the caustic intra-articular environment created after a tear.11,17 Murray et al 11 previously examined the histological changes in human ACL after rupture. Their findings noted a layer of synovial tissue over the ruptured surface, which potentially impeded repair of the ligament. Moreover, a large number of cells were observed in this synovial layer and in the epiligamentous tissue that expressed the gene for a contractile actin isoform, alpha-smooth muscle actin, thus differentiating into myofibroblasts. They believed that these factors contributed to the retraction and inhibition of the ACL to heal appropriately after repair.
The process of ligament healing starts with an early inflammatory response characterized by the infiltration of immune cells and the release of cytokines. 15 Fibroblasts, which are responsible for producing collagen, begin to migrate into the damaged area and lay down new connective tissue. Blood vessels also start to form, aiding in the delivery of oxygen and nutrients to support healing. Over time, there is the presence of collagen fibers bridging the gap between the torn ends of the ACL, indicating the progress of healing. In addition, histological analysis can reveal the biomechanical characteristics, organization, and alignment of collagen fibers, which is crucial for restoring the structural integrity of the ACL. The presence of mature collagen bundles with a similar orientation to the original ligament suggests a successful restoration (Figure 4). The histological examination can also provide insight into the degree of neovascularization, which refers to the formation of new blood vessels (Figure 5). A well-vascularized healing tissue is essential for adequate nutrient supply and can contribute to better outcomes.
In the present study, the histological findings confirmed that the tissue submitted during the procedure is in keeping with native type I collagen tissue (ligamentous tissue/synovial lining), showing reactive changes (lymphocytic infiltrate and neovascularization), which are part of the normal healing and maturation process. Notably, chronic fibrotic tissue was not observed in the samples; rather, the tissue appeared consistent with native ruptured ACL fibers. In addition, there were no findings to suggest additional pathology/complications (eg, acute inflammation, infection). Two patients demonstrated foreign material consistent with suture. The importance of the location of the biopsy at the time of revision for comparison with the site of initial injury is substantial. Through intraoperative classification in the index procedure, targeted biopsies at the site of injury during the revision procedure provide the best chance of examining the tissue to histological consistency. We recognize that this is an inexact science; however, the ability to cross-reference these locations across time points provides the best chance of study.
We believe these histological evaluations help support the premise that ACL tears treated with the BEAR implant contain ligamentous tissue that matures and heals into native-like tissue resembling the ACL. The biopsy results reported in this review are the first to histologically analyze the composition of the reruptured ligament following the BEAR procedure.
Of note, 4 of the 5 BEAR implant retears occurred at the same location as the index tear per chart review. Pathology review of the retear sites in these 4 patients did not reveal chronic fibrosis of the tissue per the pathology review. MRI was performed on each of the patients after reinjury. Interestingly, in each of the 5 MRIs, the radiologist noted a pivot-shift contusion, consistent with instability, as well as high-grade partial retears based on abnormal ACL signal within the ligament. While this sample size is too small to draw meaningful conclusions, we recognize that further evaluation of the retear locations of future BEAR implant patients should be cross-referenced with the index tear location to determine if incomplete healing may have occurred.
Retears associated with ACL surgery are an unfortunate inevitability in all methods of treatment. Multiple factors, such as failed biology, limb malalignment, tibial slope, technical error, and trauma, have been cited as reasons for failed index procedures. 3 Within the BEAR surgical cohort of patients, histological confirmation of native-like tissue has yet to be examined in human patients to date. Concerns over the integrity of the tissue have rightfully existed, as nonnative fibrotic scarring of the ACL could potentially lead to delayed failures long term. The results of this paper suggest that, in spite of retears in patients who underwent BEAR procedures, the tissue that had been restored at the index procedure was consistent with viable ACL-like tissue that would otherwise be found with a native ACL rupture. Further biopsy collection of intact and disrupted BEAR procedures at various time points are warranted to make a broader statement in regard to these findings and longer-term implications.
Limitations
The authors recognize limitations exist in this writing. The sample size of 5 patients is notably small. However, as reported in the Arnoczky et al 1 manuscript analyzing rotator cuff repair biopsy, a small sample size can nevertheless yield valuable information. Samples of tissue are limited by the fact that they are only taken during second-look procedures due to retear of the restored ligament. Additionally, tear location and restoration at the index procedure may vary. Based on operative records, the biopsy specimens were taken at multiple locations of the ACL retear with a focus placed on the site of the actual visualized tear. Further collection of biopsy specimens taken at the time of revision surgery for retears and correlation with the location of index tear and retear location should be considered. Additionally, en bloc resection of the retorn ACL, while attempted in this current review, would be of benefit, although may be technically challenging across the board.
Conclusion
Histological evaluation of BEAR retears in human patients supports the hypothesis that the tissue restored by the implant is similar to native ACL collagen.
Footnotes
Final revision submitted October 6, 2024; accepted December 2, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.M. is a consultant for Depuy Synthes, Con Med, and BD; has been a consultant for Miach orthopedics; and has received consulting fees from Zimmer Biomet Holdings, Ethicon, Maruho Medical, Anika Therapeutics, Biorez, C.R. Bard Inc & Subsidiaries, Davol, Linvatec, Medical Device Business Services, Miach Orthopaedics, Pacira Pharmaceuticals Incorporated, Trice Medical, and DePuy Synthes Products; nonconsulting fees from Biorez, Paladin Technology Solutions, Smith & Nephew, Davol, and Liberty Surgical; honoraria from Davol; hospitality payments from Eclipse Technology Solutions; acquisitions from Linvatec; and education payments from Prodigy Surgical Distribution and Arthrex. D.E.K. has been a consultant for Miach Orthopaedics and has received consulting fees from DePuy Synthes Products and Smith & Nephew; education payments from Kairos Surgical and Gotham Surgical Solutions & Devices; and hospitality payments from Smith & Nephew. S.A. has been a consultant for Miach Orthopaedics. E.A.F. has received education payments from Liberty Surgical, Arthrex, and Prodigy Surgical Distribution and hospitality payments from Eclipse Technology Solutions, Liberty Surgical, and Medical Device Business Services. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility.
Ethical approval for this study was obtained from WCG (No. 20203658) and Boston's Children's Hospital (IRB No. P00026162).
