Abstract
Background:
Despite the promising clinical outcomes of artificial polyethylene terephthalate (PET) ligaments in isolated anterior cruciate ligament reconstruction (ACLR), their biomechanical performance after combined anterolateral ligament reconstruction (ALLR)/ACLR in anterolateral structure (ALS)/anterior cruciate ligament (ACL)–deficient knees has not been investigated.
Purpose/Hypothesis:
The purpose of this study was to compare biomechanical performance in cadaveric knees between combined artificial ALLR/ACLR and isolated artificial ACLR using PET ligaments. It was hypothesized that combined artificial ALLR/ACLR would restore native knee stability and outperform isolated artificial ACLR in ALS/ACL-deficient knees.
Study Design:
Controlled laboratory study.
Methods:
Eight fresh-frozen cadaveric knees were tested using a robotic manipulator. Each knee was tested in 4 states: (1) ALS/ACL intact, (2) ALS/ACL deficient, (3) ACLR, and (4) ALLR/ACLR. The anterior tibial translation (ATT) and tibial internal rotation (IR) in each knee condition were measured under 3 loads: (1) 89 N of anterior tibial loading, (2) 5 N·m of IR torque, and (3) simulated pivot shift (combined 5 N·m of IR torque and 7 N·m of valgus load).
Results:
During 89 N of anterior tibial loading, there were no significant differences in ATT between the isolated ACLR and ALLR/ACLR knees. During 5 N·m of IR torque, the mean tibial IR at 45° of flexion was significantly higher in the ACLR knees (32.49°± 7.96°) than in the ALLR/ACLR knees (21.78°± 3.03°) (P < .05). During the simulated pivot shift, the mean ATT and tibial IR at 30° and 45° of flexion were significantly higher in the ACLR knees (ATT: 5.09 ± 2.74 mm at 30°, 5.43 ± 2.79 mm at 45°; IR: 30.08°± 7.31° at 30°, 32.55°± 6.48° at 45°) than in the ALLR/ACLR knees (ATT: 1.93 ± 2.71 mm at 30°, 1.17 ± 2.26 mm at 45°; IR: 22.12°± 4.05° at 30°, 22.18°± 3.37° at 45°) (P < .05).
Conclusion:
Combined artificial ALLR/ACLR restored native knee stability across multiple flexion angles and outperformed isolated artificial ACLR in ALS/ACL-deficient knees, particularly with respect to ATT and tibial IR during the pivot-shift test.
Clinical Relevance:
The indications of the artificial PET ligament may be expanded to include combined ALLR/ACLR to restore knee stability better than isolated artificial ACLR in ALS/ACL-deficient knees.
Anterior cruciate ligament (ACL) rupture is one of the most common sports injuries, affecting 68.6 per 100,000 individuals annually in the United States. 28 Despite significant advancements in ACL reconstruction (ACLR), current evidence suggests that up to 30% of patients experience persistent instability after surgery,10,19 with only about 33% to 41% reporting normal knee function. 3 Furthermore, the rate of graft failure after ACLR is unacceptably high, ranging from 12.9% to 28.3%, 38 especially in young females.17,21 Combined anterolateral ligament (ALL) reconstruction (ALLR) and ACLR is gaining popularity, with ALL considered part of the anterolateral structure (ALS), as it has been shown to be effective in reducing graft rupture rates, controlling rotatory instability (pivot shift), and improving functional outcomes compared with isolated ACLR. 1
Autografts (hamstring tendon, quadriceps tendon, or bone–patellar tendon–bone [BPTB]) are the gold standard in ACLR with excellent clinical outcomes. 22 Allografts are less commonly used, whereas artificial grafts have fallen out of favor because of the high failure rates, knee synovitis, and early development of knee osteoarthritis with the first- and second-generation synthetic ligaments in the early 1980s and 1990s. 37 Despite their excellent clinical outcomes, autograft harvest may lead to donor-site morbidity, such as anterior knee pain and knee extensor strength deficits for BPTB autografts and deficits in knee flexor and internal tibial rotation strength for hamstring autografts.2,9 Furthermore, in multiligament reconstructions or revision ACLR, their supply may be limited. Allografts reduce operation time, eliminate donor-site morbidity, and as a result allow for faster rehabilitation compared with autografts 29 ; however, they may result in disease transmission and immunological rejection response. The sterilization process and irradiation may weaken the allograft. 40 Moreover, graft rerupture rates after ACLR range from 6% to 25% in younger patients. 5 Furthermore, their usage may be limited by the shortage of available tissue banks in China and other Asian countries. 11
Several advances in biomaterial technology have driven the development of contemporary artificial ligaments used in ACLR. The Ligament Advanced Reinforcement System (LARS; Movmedix) is the most popular among the artificial ligaments and has demonstrated surprisingly impressive clinical outcomes. 16 LARS is made of polyethylene terephthalate (PET) fibers, which allow fibroblast adherence in vitro 36 and induction of autologous collagen growth with neoligament formation in vivo. 30 A recent study demonstrated no significant differences among autograft (hamstring tendon), allograft, and LARS ligament in terms of the clinical outcomes at 5 years after ACLR. 34 Despite the promising clinical outcomes of artificial PET ligaments in isolated ACLR, their biomechanical performance in combined ALLR/ACLR has not yet been investigated.
The aim of the present study was to compare the biomechanical performance in cadaveric knees between combined artificial ALLR/ACLR and isolated artificial ACLR using PET ligaments. Our hypothesis was that a combined artificial ALLR/ACLR with an artificial PET ligament would restore native knee stability and outperform isolated artificial ACLR in ALS/ACL-deficient knees.
Methods
Preparation of Knee Specimens
After ethics committee approval of the study protocol, 8 unpaired fresh-frozen human cadaveric knees (3 left and 5 right knees; age range, 45-76 years) were obtained from our department of anatomy. The specimens were preserved at −20°C and thawed at room temperature for 24 hours before testing. The tibia and femur were cut 20 cm from the knee joint line. Skin and subcutaneous fat were removed, and the ligaments and popliteal tendon were carefully preserved. All soft tissue was then removed from the distal 10 cm of the tibia and fibula and the proximal 10 cm of the femur for potting. The femur and the tibia were potted in cylindrical molds with plaster compound for fixation in custom clamps. The femur was secured to a fixed base, and the tibia was fixed to the end effector of the robotic arm.
In this study, each specimen was tested under 4 knee states, in the following order: (1) ALS/ACL intact, (2) ALS/ACL deficient, (3) ACLR, and (4) ALLR/ACLR.
Robotic Testing System
A robotic testing system equipped with a 6 degrees of freedom industrial robot (KUKA KR 120 R2500 pro; KUKA Robotics) was used for kinematic testing of all knee specimens as described previously4,25 (Figure 1). The measurement accuracy of the robot was ±0.06 mm of position repeatability. Attached to the robot’s end effector was a universal force/moment sensor (Delta; ATI Industrial Automation) with a force and moment accuracy of ±0.25 N and ±0.008 N·m, respectively. The ACL-intact knees were subjected to a robotically determined passive path, ranging from full extension to 90° of knee flexion, at incremental steps of 1°. The passive path of the cadaveric knee was determined by minimizing forces (<0.5 N) and moments (<0.5 N·m) in all remaining degrees of freedom. 20 Data acquisition was performed using a custom MATLAB program (The MathWorks) to control translations and rotations and measure force/moments across all 6 degrees of freedom.

A cadaveric knee was mounted in a 6 degrees of freedom robotic testing system. This allowed knee testing from full extension to 90° of knee flexion.
Surgical Technique
Each specimen was rigorously inspected by a single experienced surgeon (H.Y.) via an anteromedial parapatellar arthrotomy incision to confirm the absence of any prior surgical interventions, injuries, or anatomic abnormalities. 23 All reconstruction procedures were performed through the same incision of the knee by the same surgeon. After closing this initial incision, the stability of the ALS/ACL-intact knee was assessed. The intact ACL was cut through the anteromedial parapatellar arthrotomy incision of the knee (Figure 2A), and the ALS-deficient state was simulated by cutting all femoral attachments between the lateral collateral ligament and the patellar tendon, including the capsule, ALL, and distal deep insertions of the iliotibial band (ITB). 15

Shown are all surgical procedures on the same right knee: (A) ACL- and ALS-deficient states; (B) polyethylene terephthalate ligament used for ACLR/ALLR, with the ACL and ALL portions indicated; (C) after ACLR; (D) after ALLR/ACLR. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; ALL, anterolateral ligament; ALLR, anterolateral ligament reconstruction; ALS, anterolateral structure.
ACL Reconstruction
An artificial PET ligament (Ligatech Bioscience Company) with identical properties as the LARS was used in the present study (Figure 2B). The aim was to standardize the properties and eliminate the variability of allografts in the specimens. 12 All ACLRs were conducted in accordance with the technique developed by Laboureau, as described by Dericks. 8
The diameters of the tibial and femoral drilling tunnel were 7.5 mm, identical to the diameter of the graft. To drill the tibial tunnel, the drill guide was inserted into the joint through an anteromedial approach. The tibial tunnel was created by inserting a guide pin at the centrum of the ACL tibial posterolateral (PL) bundle footprint using fluoroscopy, at the junction of approximately 50% of the width of the tibial plateau and approximately 50% of the distance between the most anterior border of the tibial plateau and the posterior border of the medial tibial plateau. 13 Correspondingly, the intra-articular entry of the femoral tunnel was located at an isometric position according to the technique developed by Laboureau, as described by Dericks. 8 A line parallel to the Blumensaat line was drawn forward through the most prominent point of the posterior condylar cartilage of the femur, with the isometric point located at 60% of the line from front to back using fluoroscopy. After drilling the Kirschner wire through the femur, the femoral tunnel was established by drilling over the Kirschner wire with a cannulated drill from the anterolateral thigh into the knee joint. Subsequently, a wire loop was passed sequentially through the femoral tunnel, across the joint, and then through the tibial tunnel. The artificial PET ligament was threaded into this loop and pulled through the joint, and it was inserted into the osseous tunnels with the longitudinal free fibers of the graft entering the joint. The ligament was fixed at the lateral femoral cortex using titanium interference screws with blunt thread edges, with a diameter of 8 mm on the femoral side and 9 mm on the tibial side. Once the femoral end of the graft was secured, the knee was moved from 0° to 90° to test the isometry. If the graft had good isometric properties, the knee was flexed to approximately 90°, and the tibial end of the graft was fixed with a screw (Figure 2C). Otherwise, the tibial end of the graft was fixed at the flexed angle that the longest intra-articular length of the graft allowed.
ALL Reconstruction
The ALLR procedure was performed following the guidelines outlined in a prior technical note. 24 Briefly, the tibial tunnel was positioned 10 mm below the joint line, halfway between the center of the Gerdy tubercle and the center of the fibular head. 31 The initial location of the femoral attachment of ALL was determined with the knee fully extended, placing it 8 mm proximal and 8 mm posterior to the lateral epicondyle, based on the reported optimal isometric region of its femoral attachment. 41 Then, an isometry assessment was made: 2 wires were drilled into the femoral and tibial sites, respectively; a suture was placed around the femoral and tibial wires; and the knee was taken through a full range of motion to see if the suture became too tight in flexion. If not satisfied, the initial femoral site was adjusted. A 15 × 20-mm spiked ligament staple (Ligatech Bioscience Company) was used to secure the femoral attachment of the ligament. A guide pin was drilled transversely through the tibia, and the suture attached to the tibial end of the graft was threaded to the medial side of the tibia. To secure the tibial end of the graft when the knee is taken into full extension and the tibia is in neutral rotation, a 7 × 25-mm polyether ether ketone interference screw (Ligatech Bioscience Company) was used, with the graft being tensioned to 20 N 14 (Figure 2D).
Testing Protocols
Each of the 4 knee states was tested under 3 loads 25 : (1) 89 N of anterior tibial loading to test anterior tibial translation (ATT), (2) 5 N·m of internal rotation (IR) tibial torque to test tibial IR, and (3) simulated pivot-shift test consisting of a combined 5 N·m of IR tibial torque and 7 N·m of valgus load to test ATT and IR. The ATT during the anterior tibial loading was measured at full extension and 30°, 60°, and 90° of knee flexion. The IR during the IR tibial torque was measured at full extension and 15°, 30° and 45° of knee flexion. The ATT and IR during the pivot-shift test were measured at full extension and at 15°, 30°, and 45° of knee flexion.
Statistical Analysis
Descriptive statistics were used, with kinematic data (ATT and IR) reported as means with standard deviations. All parameters underwent Shapiro-Wilk testing for normality. One-way repeated-measures analyses of variance were conducted to compare the differences in ATT and IR at all measured flexion angles between the ALS/ACL-intact, ALS/ACL-deficient, isolated ACLR, and combined ALLR/ACLR states. To adjust for multiple comparisons, post hoc Bonferroni corrections were calculated using GraphPad Prism Version 7.0 (GraphPad Software). The level of statistical significance was set at P < .05.
According to the past studies on knee laxity,6,39 a sample size of 8 was found to be sufficient to identify changes in translation and rotation with 80% power based on the means and standard deviations.
Results
ATT During 89 N of Anterior Tibial Loading
The ATT values for each knee state are shown in Table 1 and Figure 3. The ALS/ACL-deficient knees demonstrated significantly higher ATT compared with ALS/ACL-intact knees at all tested knee flexion angles (P < .05). The ATT was significantly higher in ACLR compared with ALS/ACL-intact knees at 60° of flexion (P < .05) but not at the other tested flexion angles. No significant differences in ATT were observed at any flexion angle in ALLR/ACLR compared with ALS/ACL-intact knees.
ATT During 89 N of Anterior Tibial Loading and Tibial IR During 5 N·m of IR Torque According to Knee State and Flexion Angle a

Data are presented as mean ± SD. Statistically significant difference (P < .05) between +ALS/ACL-intact versus ALS/ACL-deficient states, #ALS/ACL-intact versus ACLR states, and *ACLR versus ALLR/ACLR states. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; ALLR, anterolateral ligament reconstruction; ALS, anterolateral structure; ATT, anterior tibial translation; IR, internal rotation.
Mean anterior tibial translation (ATT) under 89 N of anterior tibial loading at different flexion angles. Error bars indicate standard deviations. Statistically significant difference (P < .05) between +ALS/ACL-intact versus ALS/ACL-deficient states and #ALS/ACL-intact versus ACLR states. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; ALLR, anterolateral ligament reconstruction; ALS, anterolateral structure.
Tibial IR During 5 N·m of IR Torque
The tibial IR values for each knee state are shown in Table 1 and Figure 4. The ALS/ACL-deficient knees demonstrated significantly higher tibial IR compared with the ALS/ACL-intact knees at all tested knee flexion angles (P < .05). After ACLR, the tibial IR was significantly higher than in the ALS/ACL-intact knees at 45° of knee flexion (P < .05), but no difference was found at the other flexion angles. After combined ALLR/ACLR, no significant differences in tibial IR at any flexion angle were observed compared with the ALS/ACL-intact knees. The tibial IR at 30° and 45° of knee flexion was significantly higher after isolated ACLR than combined ALLR/ACLR (P < .05).

Mean internal rotation (IR) under 5 N·m of IR tibial torque at different flexion angles. Error bars indicate standard deviations. Statistically significant difference (P < .05) between +ALS/ACL-intact versus ALS/ACL-deficient states and #ALS/ACL-intact versus ACLR states, *ACLR vs ALLR/ACLR states. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; ALLR, anterolateral ligament reconstruction; ALS, anterolateral structure.
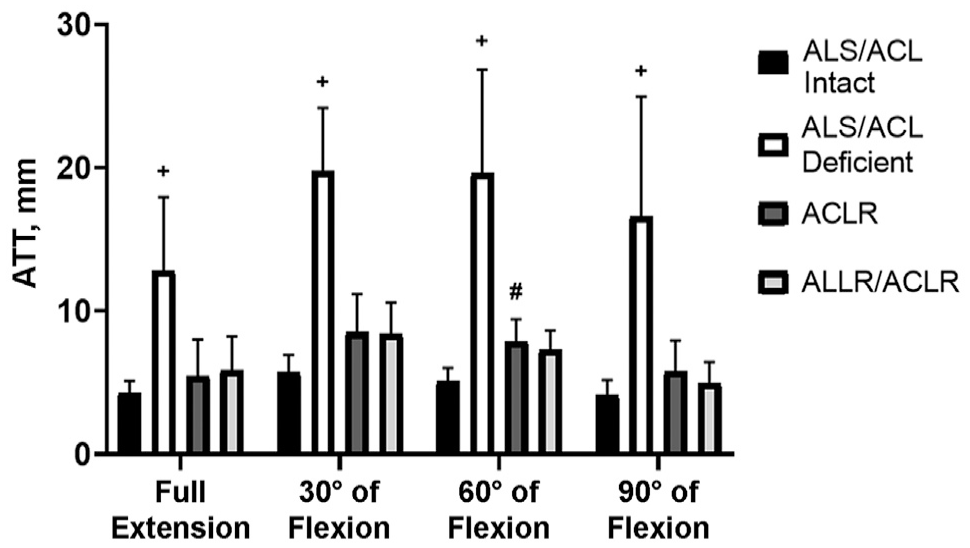
Simulated Pivot-Shift Test
ATT and tibial IR values during the simulated pivot-shift test (5 N·m of IR tibial torque and 7 N·m of valgus load) are shown in Table 2 and Figure 5.
ATT and IR During the Simulated Pivot-Shift Test According to Knee State and Flexion Angle a
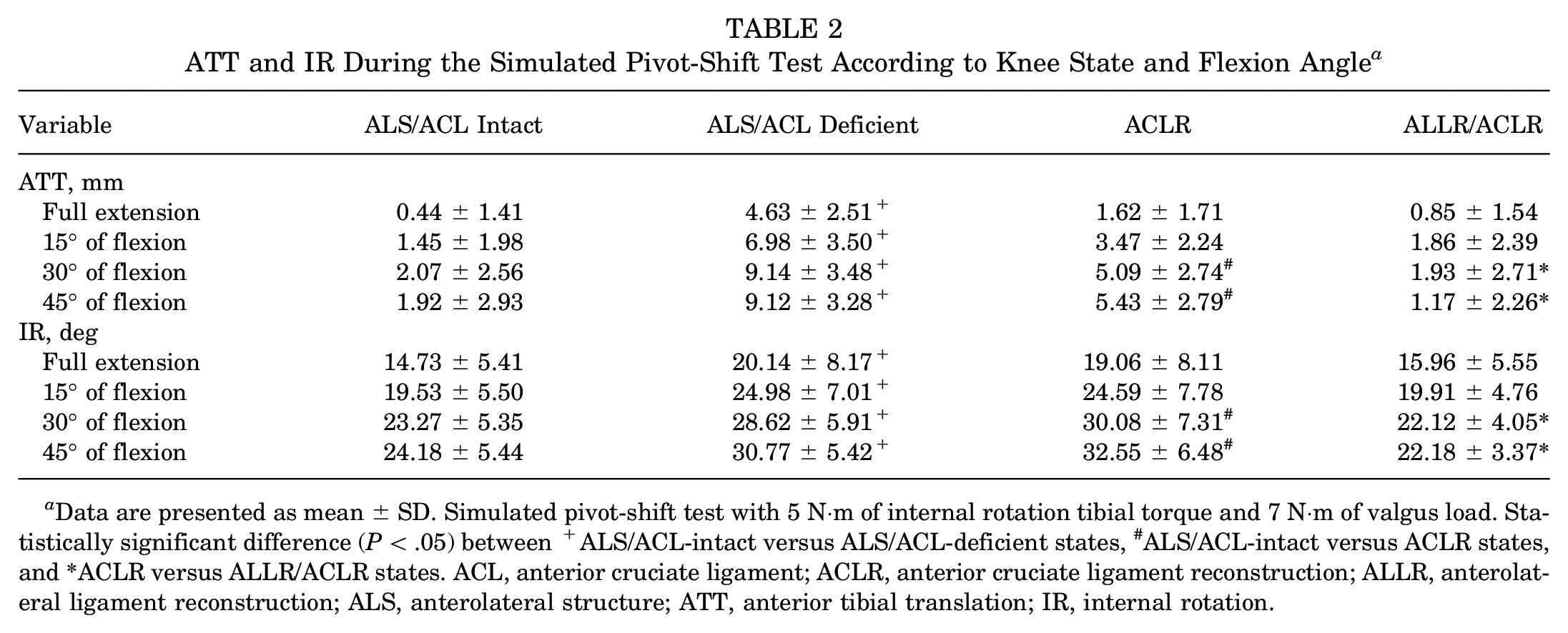
Data are presented as mean ± SD. Simulated pivot-shift test with 5 N·m of internal rotation tibial torque and 7 N·m of valgus load. Statistically significant difference (P < .05) between +ALS/ACL-intact versus ALS/ACL-deficient states, #ALS/ACL-intact versus ACLR states, and *ACLR versus ALLR/ACLR states. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; ALLR, anterolateral ligament reconstruction; ALS, anterolateral structure; ATT, anterior tibial translation; IR, internal rotation.
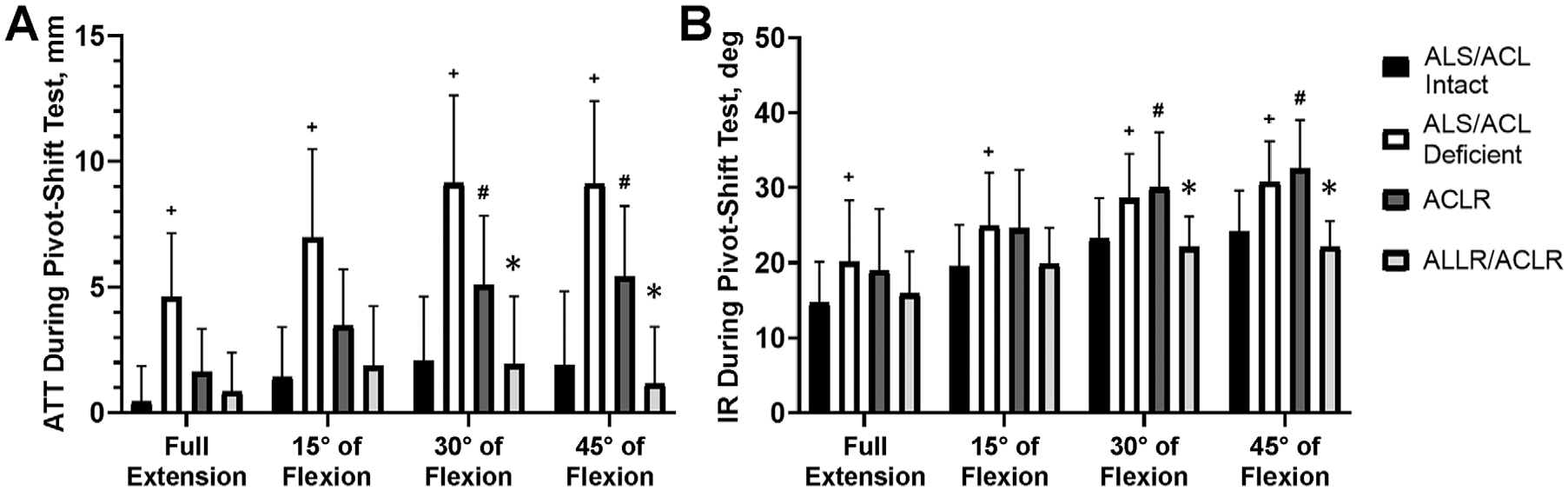
Mean (A) anterior tibial translation (ATT) and (B) internal rotation (IR) under simulated pivot-shift loading (5 N·m of IR tibial torque and 7 N·m of valgus load) at different knee flexion angles. Error bars indicate standard deviations. Statistically significant difference (P < .05) between +ALS/ACL-intact versus ALS/ACL-deficient states, #ALS/ACL-intact versus ACLR states, and *ACLR versus ALLR/ACLR states. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; ALLR, anterolateral ligament reconstruction; ALS, anterolateral structure.
Anterior Tibial Translation
The ALS/ACL-deficient knees demonstrated significantly higher ATT compared with ALS/ACL-intact knees at all tested flexion angles (P < .05). After isolated ACLR, the ATT was significantly higher than in the ALS/ACL-intact knees at 30° and 45° of knee flexion (P < .05), but no difference was found at the other flexion angles. After combined ALLR/ACLR, no significant differences in ATT were observed at any flexion angle compared with ALS/ACL-intact knees. The ATT at 30° and 45° of knee flexion was significantly higher after isolated ACLR than combined ALLR/ACLR (P < .05).
Tibial IR
The ALS/ACL-deficient knees demonstrated significantly higher tibial IR than the ALS/ACL-intact knees at all tested flexion angles (P < .05). After isolated ACLR, the tibial IR was significantly higher compared with the ALS/ACL-intact knees at 30° and 45° of flexion (P < .05), but no difference was found at the other flexion angles. After combined ALLR/ACLR, no significant differences in tibial IR were observed at any flexion angle compared with ALS/ACL-intact knees. The tibial IR at 30° and 45° of knee flexion was significantly higher after isolated ACLR than combined ALLR/ACLR (P < .05).
Discussion
The most important finding of the present study was that combined ALLR/ACLR demonstrated similar knee stability, in terms of ATT and tibial IR, to ALS/ACL-intact knees and surpassed the stability provided by isolated ACLR in ALS/ACL-deficient knees, which demonstrated both higher ATT and tibial IR, especially during the simulated pivot-shift test at midflexion (30° and 45°). Therefore, the hypothesis of this study was confirmed, as combined ALLR/ACLR with an artificial PET ligament restored native knee stability at different tested knee flexion angles and outperformed the knees that underwent isolated artificial ACLR.
Combined ALLR/ACLR is associated with excellent outcomes in graft rupture rates, knee stability, and return to sports.26,27 Considering the advantages of artificial ligaments over auto- and allografts, several studies have investigated their clinical performance in isolated ACLR, with promising outcomes.16,34 However, the biomechanical performance of artificial PET ligaments in combined ALLR/ACLR had not been investigated in ALS/ACL-deficient knees. To the best of our knowledge, this is the first cadaveric study that has compared the biomechanical performance of combined artificial ALLR/ACLR with isolated artificial ACLR.
Several biomechanical studies have investigated knee stability after ACLR in a combined ALS/ACL injury and reported that isolated ACLR failed to restore intact knee kinematics. Inderhaug et al 15 compared ATT during the anterior drawer test and tibial IR during 5 N·m of IR torque between ALS/ACL-intact knees, ALS/ACL-deficient knees, and ACL-reconstructed knees (10 mm of autologous BPTB) and reported that although ACLR improved both ATT (about 7 mm) and tibial IR (about 4°), it failed to restore intact knee kinematics (about 2 mm increased ATT and 2° increased tibial IR at all knee flexion angles [0°-90°]). Delaloye et al, 7 using a similar experimental setting, reported that in knees with a combined ALS/ACL deficiency, ATT and tibial IR remained significantly increased after isolated ACLR (autologous semitendinosus graft) compared with the ALS/ACL-intact knee (2.33 ± 1.44 mm and 1.98°± 1.06°, respectively; P < .01) throughout all the tested flexion angles (0°-90°). In the present study, knees that underwent isolated ACLR with an artificial PET ligament demonstrated residual increased ATT only at 60° and increased tibial IR only at 45° of flexion, whereas during the pivot-shift test, the residual increased ATT and tibial IR were between 30° and 45° of flexion. Contrary to the studies by Inderhaug et al and Delaloye et al, isolated artificial ACLR was able to restore native knee stability during and near full extension (0°-15°) in all tested conditions. These differences in outcomes might be attributed to different experimental designs, weakening of the autograft during preparation of the cadaver, or different graft fixation techniques (suspensory vs interference screw, fixation angle, tension, etc), rather than isolated graft type. Nevertheless, the artificial PET ligament demonstrated excellent performance in improving knee stability after ACLR, especially at lower flexion angles.
The biomechanical effects of combined ALLR/ACLR in combined ALS/ACL injury have been scarcely investigated in the literature. Yet, the available body of evidence suggests that combined ALLR/ACLR is able to restore native knee stability and is superior to isolated ACLR in terms of ATT and tibial IR. Inderhaug et al 15 investigated the effect of several anterolateral procedures (modified Lemaire, MacIntosh, and ALLR) in addition to ACLR in ALS/ACL-deficient cadaveric knees and reported that only the deep Lemaire and MacIntosh procedures with 20 N of tension restored rotational kinematics to the intact state, whereas the ALLR underconstrained and the superficial Lemaire overconstrained the tibial IR. Delaloye et al 7 also investigated the effect of modified Lemaire and ALLR in addition to ACLR in knee stability and reported that both procedures restored native knee stability in ATT and tibial IR (contrary to isolated ACLR), and neither overconstrained the knee. Similarly, in the present study, a combined ALLR/ACLR with an artificial PET ligament restored knee stability (both ATT and tibial IR) in all tested conditions and at all knee flexion angles. Increased knee stability during the initial postoperative period can have a protective effect on the operated knee and may explain the lower ACL graft 33 and medial meniscal repair 32 failures observed clinically after combined ALLR/ACLR, as opposed to isolated ACLR.
Limitations
The present study should be interpreted in light of its limitations, mostly inherent to the experimental study design. The results represent the initial postoperative period (time zero) as they do not take into account possible changes due to healing or in response to cyclic loading and rehabilitation. Second, the age of the human cadaveric knees did not represent the typical age for patients experiencing an ACL rupture. Unfortunately, it was virtually impossible to test young human knees in sufficient numbers given the scarcity of human donors. Furthermore, the ALS injury inflicted on the knees represents the worst-case scenario because all femoral attachments between the lateral collateral ligament and the patellar tendon, including the capsule, ALL, and distal deep insertions of the ITB band, were transected. Several ALSs play an important role in resisting tibial IR. Specifically, Kittl et al, 18 in a cadaveric study performing sequential dissection of ALSs, reported that the deep and capsulo-osseous layers of the ITB were the most important restraints of tibial IR, whereas the ALL only made a minor contribution. Although Terry et al 35 reported that a majority of patients with a concomitant ACL and anterolateral injury had a lesion to the deep and capsulo-osseous layers of the ITB, it might be variably related to the magnitude and mechanism of the trauma, leading to these injuries.
Conclusion
The results demonstrated that native knee stability was restored after a combined ALLR/ACLR, whereas the isolated ACLR demonstrated residual increased ATT and tibial IR in ALS/ACL-deficient knees, especially during the pivot-shift test. Because of the time-zero findings and the limitations of synthetic graft longevity, further clinical studies are needed to confirm the safety and efficacy of combined artificial ALLR/ACLR.
Footnotes
Final revision submitted October 10, 2024; accepted November 18, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding for this study was received from the National Outstanding Youth Science Fund Project of the National Natural Science Foundation of China (31972924, 32371367, 81972060), the Science and Technology Commission of Shanghai Municipality (22S31906000, 21DZ2208200, 23S31901000), the Fundamental Research Funds for the Central Universities (AF0820060), the Pudong Science Technology and Economy Commission (210H1147900), and the Shanghai Jiao Tong University “New Young Teachers Startup Plan” (23X010501995). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine (ref No. SH9H-2023-T124-1).
