Abstract
Background:
The Shoulder Pain and Disability Index (SPADI) is a widely used 13-item shoulder-specific patient-reported outcome measure for shoulder pain disorders. The English version of SPADI is easy to use and demonstrates excellent measurement properties for clinical and research settings.
Purpose:
To translate and culturally adapt an Indonesian version of SPADI (SPADI-IDN) and then validate its use in Indonesian patients.
Study Design:
Cohort study (Diagnosis); Level of evidence, 2.
Methods:
Through a forward and a backward translation process, the validity of the questionnaire was investigated. The study population was 100 patients with shoulder pain who were treated in a hospital in Indonesia. The patients were asked to fill out the SPADI-IDN during their visit. To evaluate the validity of SPADI-IDN, the patients were also asked to fill out the Medical Outcomes Study 12-Item Short-Form Health Survey and the Oxford Shoulder Score. To assess the test-retest reliability, the same patients were asked to fill out the SPADI-IDN questionnaire again 1 week later. The assessment of construct validity, test-retest reliability, internal consistency, floor and ceiling effect, and measurement of error followed the COSMIN (Consensus-based Standards for the selection of health Measurement Instruments) guidelines. The Bland-Altman method was used to explore absolute agreement.
Results:
Of the 100 eligible patients, all were used to assess construct validity, and 87 patients (87%) were used to assess test-retest reliability. Almost every established hypothesis about the correlations between SPADI-IDN and other questionnaires could be confirmed, implying good construct validity. No floor or ceiling effects were found. The intraclass correlation value was 0.99, indicating strong test-retest reliability. A Cronbach α was 0.95, indicating a good internal consistency. The Bland-Altman analysis did not reveal any bias. The standard error of measurement and the minimal detectable change at the individual and group levels were 2.65, 7.3, and 0.7, respectively.
Conclusion:
The SPADI-IDN represents a valid and reliable tool for measuring pain and disability in patients with shoulder pain disorders.
Keywords
The shoulder is one of the most common sites of musculoskeletal pain, with a 1-month to 1-year prevalence in the general population of 6% to 37%. 17 Rotator cuff disease is one of the most common shoulder pains. Rotator cuff disease can either be tendinopathy or partial or complete tears. 25 Age plays a significant role in these particular cases. 25 The incidence is 9.7% in patients aged ≤20 years and increases to 62% in patients aged ≥80 years, regardless of the presence of symptoms. 23 Shoulder pain is therefore associated with considerable personal and economic costs in relation to the individual sufferer and the health care system alike. 17
Recent advances have improved the outcome of shoulder pain treatment, either conservatively or surgically, which offers excellent results in both function and quality of life.7,13 However, the degree of recovery after shoulder pain treatment varies distinctly between patients. 7 One of the easiest ways to obtain information about shoulder pain and the outcome before and after treatment is through appropriately designed self-assessment questionnaires, which collect specific information from the participants and are also used as patient-reported outcome measures (PROMs).21,24
PROMs can be used to evaluate the treatment and provide valuable feedback on the progress of clinical conditions. 24 Different PROMs have been developed to measure shoulder disabilities, one of which is the Shoulder Pain and Disability Index (SPADI). SPADI is used widely for evaluating functional and quality of life outcomes after shoulder surgery since it has good reliability and validity.6,8,15,20
The SPADI is a self-administered questionnaire developed by a panel of experts to measure shoulder pain and disability in either inpatient or outpatient settings.2,20 Evidence has been reported supporting the reliability, validity, and responsiveness of the SPADI. The original English version has been shown to have excellent measurement properties in various shoulder conditions.2,20,21 It has subsequently been translated and cross-culturally adapted into multiple languages with good measurement properties.5,11,15,19,25,26 However, it has not been translated into Indonesian—the language of the most populous country in Southeast Asia. Furthermore, the Indonesian patient populace is not conversant with English; therefore, the Indonesian version of SPADI will lessen linguistic fencing and provide valid and reliable data or information for research and clinical intent. This study aimed to translate and culturally adapt an Indonesian version of the SPADI questionnaire and validate its use in Indonesian patients with shoulder pain. For that reason, we thoroughly investigated the SPADI scale in terms of construct validity and reliability in a population-based study of patients with self-reported symptoms related to their shoulder problems. We hypothesized that the Indonesian version of SPADI (SPADI-IDN) would be valid and reliable for Indonesians with shoulder pain disorders.
Methods
This study was approved by the institutional review board of the University of Riau (No: B/089-I/UN19.5.1.1.8/UEPKK/2021) before data collection and conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent before enrollment in the study.
Translation Procedure
SPADI-IDN translation followed Guillemin and Beaton’s guidelines for forward and backward translation.3,12 The process is divided into 5 main phases: initial translation, translation synthesis, back translation, committee review, and pretesting of the translated version. Subsequently, all written reports were submitted to expert committee review to evaluate and finalize the end version of the translated score. Two independent Indonesians with proficient speaking, reading, and understanding of the English language initially completed both the literal (word-to-word translation) and conceptual (the principle of the disease related to each item of the PROM) translation of SPADI into Indonesian (T1 and T2). Two professional translators (BT1 and BT2) translated the synthesized version (T12) back into English based on feedback from the 2 original translations to ensure there were no discrepancies with the original version. An expert panel consisting of 3 orthopaedic surgeons specializing in sports medicine (R.D., I.W.A., T.N.U.), 1 methodologist who is an expert in the methodology applied in this study (F.F.A.), and 1 translator reviewed the Indonesian translation (A.D.) After the expert panel review, 1 of the authors (R.D.) changed the questionnaire to a prefinal version. This prefinal translated version was then sent to 10 patients with rotator cuff disease. The same author (R.D.) documented difficulties patients experienced when completing the questionnaire. The expert panel examined the documentation and made necessary modifications to the questionnaire before it was included in the final SPADI-IDN version.
Patients and Procedure
A total of 100 consecutive patients who spoke Indonesian as a primary language, complained of shoulder pain, and were treated for shoulder pain and rotator cuff disease at a hospital in Indonesia between July 2022 and June 2023 were invited to participate in the validity and reliability study. Patients were asked to fill out the first set of questionnaires (part A) during their visit to the outpatient clinic. Patients were then asked to come after 1 week and fill out the second set of questionnaires (part B).
Part A consisted of SPADI-IDN and 2 other questionnaires, that is, the Medical Outcomes Study 12-Item Short-Form Health Survey (SF-12) and the Oxford Shoulder Score (OSS).10,27 Part B consisted of only the SPADI-IDN questionnaire. To ascertain whether the shoulder function and health status of the patients remain the same when filling out parts A and B, an additional question to evaluate the Global Rating of Change (GRC), “Has your status changed since filling out the initial questionnaire?” was added to the part B questionnaire. The 3 options for answers were (1) no; (2) yes, the complaint changed for the better; and (3) yes, the complaint changed for the worse. Only patients who reported no change in their shoulder symptoms were included in the test-retest analysis. Patients who did not fill out part B were excluded from the test-retest analysis.
Patient-Reported Outcome Measures
SPADI is a 13-item shoulder-specific PROM. It consists of 2 subscales: a 5-item subscale of pain representing pain severity in the last week and an 8-item disability subscale representing shoulder function in activities of daily living. The total score ranges from 0 to 100, with higher scores representing greater pain intensity and disability. Test-retest reliability for the SPADI has been reported as being good to excellent (intraclass correlation coefficient [ICC] > 0.75 ).2,16,20
A general score for creating a health profile is the SF-12. It has 8 scaled scores and is used to determine the patient’s physical and mental health (MH). Each subscale is converted into a scale from 0 to 100 directly: physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social function, emotional role functioning, and MH. In addition, the sum of the physical functioning, physical role functioning, bodily pain, and general health perceptions subscales generates a physical component summary score (PCS), and the sum of the vitality, social function, emotional role functioning, and MH subscales generates a mental component summary score (MCS). Standardized scores range from 0 to 100, with higher scores indicating better health status. 27
The OSS is a shoulder-specific questionnaire for assessing patients with shoulder pain. It has 12 items, consisting of 4 items about pain and 8 questions about daily shoulder function. Each item is assessed based on 5 categories of answers, from 0 (worst) to 4 (best condition). The total score of the OSS ranges from 0 to 48.9,10
Validity
Validity refers to how well a score captures its intended meaning or measures what it is meant to measure. By calculating the correlation of the SPADI-IDN total score with the total score on the OSS and the PCS and MCS of the SF-12, the construct validity of the SPADI-IDN was evaluated. Predetermined hypotheses on the strength of the relationships between the SPADI-IDN, the OSS, the PCS, and the MCS were developed in accordance with the Consensus-based Standards for the selection of health-Measuring Instruments (COSMIN) guidelines. 18 Since SPADI-IDN and OSS questionnaires are shoulder-specific PROMs, whereas SF-12 is not, we predicted that the SPADI-IDN would correlate better with the OSS. The correlation between the Chinese SPADI and the OSS was 0.68 in a previous study. 26 Based on this, a correlation of 0.6 or greater between the SPADI-IDN and the OSS was hypothesized. The Persian version of SPADI showed the correlations between the total SPADI score and the PCS and MCS of the 36-Item Short-Form Health Survey (SF-36) were 0.34 and 0.28, respectively. 11 This led to the hypothesis that correlations between the SPADI-IDN and the PCS should be ≥0.3, whereas correlation between the SPADI-IDN and the MCS should be <0.3. The SPADI-IDN was designed primarily to assess physical functioning, as opposed to social or emotional aspects. As a result, it was predicted that there would be stronger correlations between the SPADI-IDN and the PCS instead of the MCS. The discovery of at least 75% of these hypotheses indicates good construct validity. 22
Floor and Ceiling Effects
It was determined how frequently floor and ceiling impacts occurred. If >15% of respondents have the lowest or highest possible score, it indicates that these effects are present. 22
Reliability
Reliability refers to how well people can be identified from one another despite measurement flaws. Reliability was evaluated using the COSMIN criteria for measurement error, test-retest reliability, and internal consistency. Measurement error is the measure of systematic error in a patient’s score that is not brought on by real variations in the measured construct; test-retest reliability is the degree to which patients’ scores remain consistent across multiple assessments; internal consistency is the degree to which a questionnaire’s subscales are related. 18 Absolute agreement, or the degree of agreement across repeated measurements, was investigated using the Bland-Altman analysis. 11
Statistical Analysis
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 26.0 (IBM Corp). Means and standard deviations for continuous variables and frequencies (percentages) for categorical variables were used to characterize the study population’s characteristics and the questionnaire scores. Spearman rho correlation coefficients between the SPADI-IDN scores and the results of the other questionnaires were computed to assess construct validity. The Spearman rho values were then categorized into the following: strong (r > 0.6), moderate (0.6 < r < 0.3) (0.6 > r > 0.3), or weak (r < 0.3).14,24 Cronbach α was used to evaluate the internal consistency, with a value between 0.70 and 0.95 indicating a good internal consistency. 22
The ICC between the test and retest SPADI-IDN scores was computed to evaluate test-retest reliability. The calculation of ICC is based on a 2-way mixed-effects model and absolute agreement. ICC values <0.5, 0.5-0.75, 0.76-0.9, and >0.90 indicate poor, moderate, good, and excellent reliability, respectively. 16 To evaluate measurement error, 2 metrics were computed: standard error of measurement (SEM) and minimal detectable change (MDC). The calculation of SEM involved increasing the pooled standard deviation by √(1 − r), where r is the ICC. The formula 1.96 × SEM × √2 was used to determine the MDC at the individual (MDCind) and group (MDCgrp) level by dividing MDCind by √n. 22
Bland-Altman plots were used to evaluate absolute reliability. When the 0 value was within the 95% CI of the mean difference between the test and retest scores, it indicated the absence of systematic bias. To evaluate the variability and identify the outliers, the limits of agreement (LOA) were calculated by using the following equation:
where SDdiff is the standard deviation of the mean difference between the first and second administration of the SPADI-IDN. 4 A significance threshold of 5% was used in all statistical analyses.
Results
Patient Characteristics
Of the 100 patients who completed the initial set of questionnaires, 92 (92%) also completed the second set of questionnaires. Five patients (5%) were excluded as they reported better shoulder function when completing the second questionnaire. Therefore, internal consistency and validity assessment included data from 100 patients, whereas test-retest reliability was evaluated using data from 87 patients (87%) (Figure 1).

Flow chart of the study population selection.
Most patients had graduated from high school and had a mean age of 50.3 ± 12.9 years. Table 1 provides a detailed description of the demographic features of patients. The first and second assessments of the SPADI-IDN mean scores were 64.6 ± 17.7 and 77.5 ± 10.6, respectively. The PROM scores are listed in Table 2.
Patient Characteristics (N = 100)

Mean ± SD.
Patient-Reported Outcome Measures a

MCS, mental component summary score; OSS, Oxford Shoulder Score; PCS, physical component summary score; SF-12, 12-Item Short-Form Health Survey score; SPADI, Shoulder Pain and Disability Index; SPADI-IDN, Indonesian version of SPADI score.
Validity
Out of the 6 predetermined hypotheses on the strength of correlations between the SPADI-IDN and the OSS, PCS, or MCS, 5 (83%) were validated. The SPADI-IDN had a strong correlation with the OSS (r = 0.71) and a weak correlation with the PCS (r = 0.26) and MCS (r = 0.02). The results of Table 3 indicate that the SPADI-IDN has a stronger correlation with the OSS than with PCS and MCS and with PCS more so than with MCS. Floor and ceiling effects were absent.
Spearman Rho Correlation Coefficients Between SPADI-IDN, OSS, PCS, and MCS a
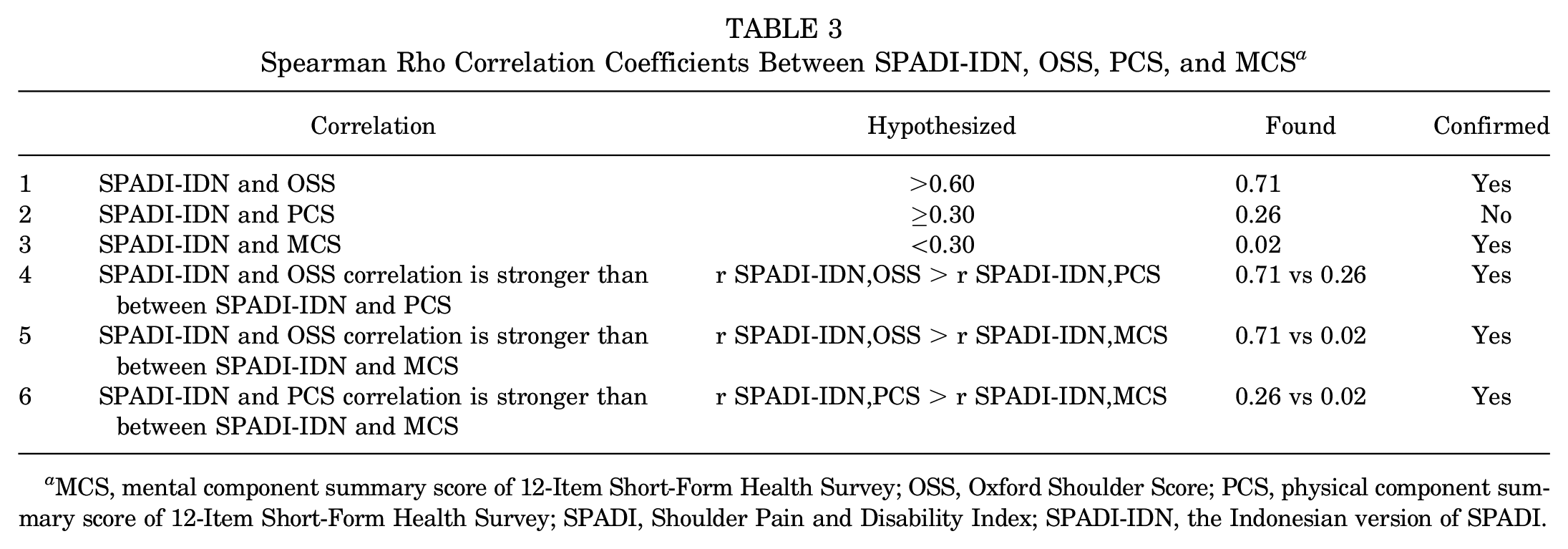
MCS, mental component summary score of 12-Item Short-Form Health Survey; OSS, Oxford Shoulder Score; PCS, physical component summary score of 12-Item Short-Form Health Survey; SPADI, Shoulder Pain and Disability Index; SPADI-IDN, the Indonesian version of SPADI.
Internal Consistency, Test-Retest Reliability, and Measurement Error
The internal consistency was good, as indicated by the Cronbach α of 0.95. The ICC [95% CI] was 0.998 [0.997-0.999] (P < .001). The SEM, MDCind, and MDCgrp were 2.65, 7.3, and 0.7, respectively. The first and second SPADI-IDN assessments had a mean difference of 0.2 (95% CI, −0.09 to 0.44; 95% LOA, −44.3 to 44.6) (Figure 2). Since zero was inside the 95% CI of the mean difference between the test and retest scores, this indicates that there was no systematic bias.
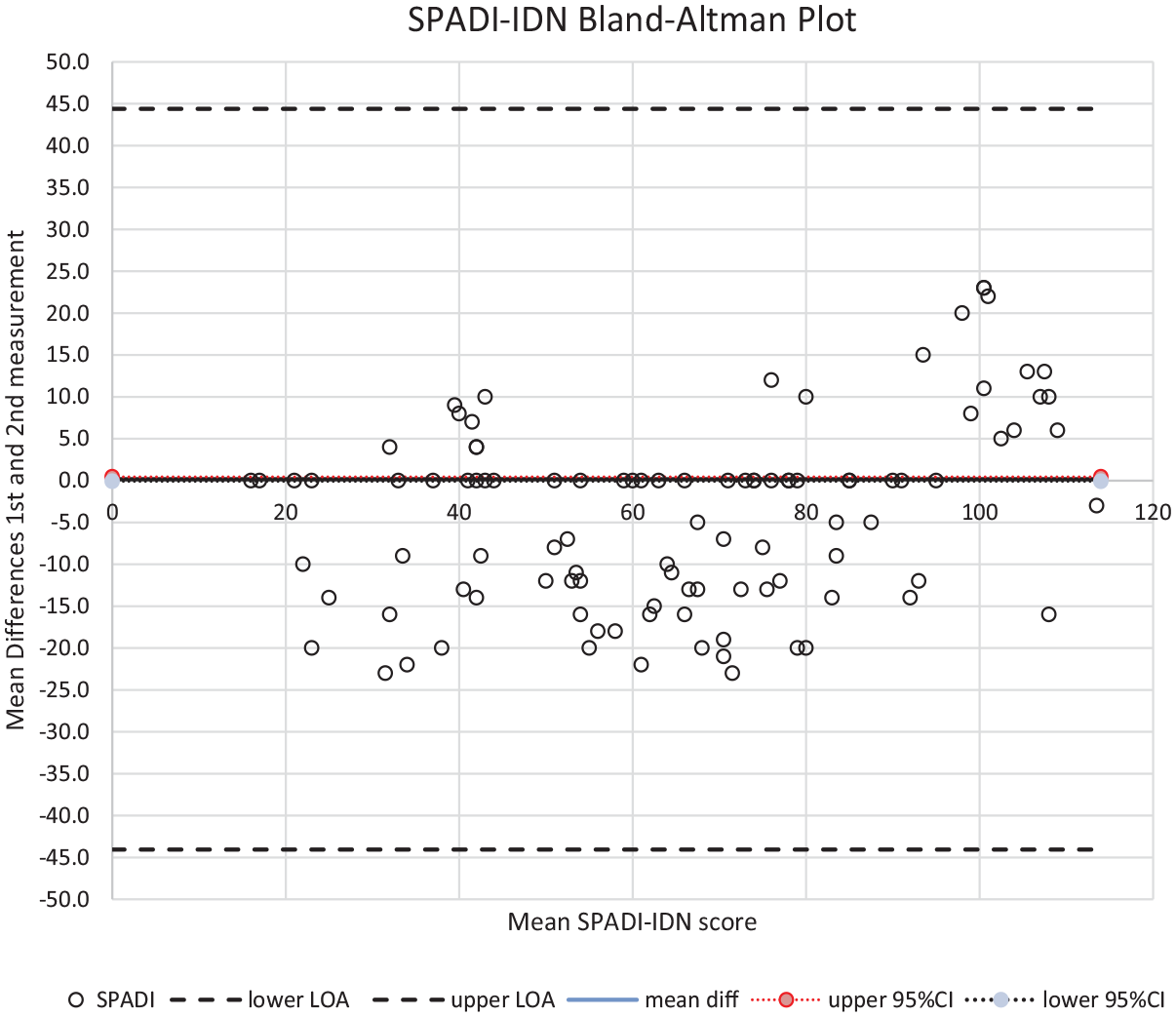
Bland-Altman plot displaying the mean difference between the initial and subsequent measurements of the SPADI-IDN. Open circles, datapoints; black line, mean difference; dotted lines, 95% CIs; dashed lines, boundaries of agreement. LOA, limits of agreement; mean diff, mean difference; SPADI, Shoulder Pain and Disability Index; SPADI-IDN, Indonesian version of the SPADI.
Discussion
Our study demonstrated that >75% of predefined hypotheses on correlations between the SPADI-IDN and other scores, indicating good construct validity; an ICC value of 0.99 indicated good test-retest reliability; a Cronbach α of 0.95 indicated good internal consistency; and no floor or ceiling effects were found.
This study aimed to translate the SPADI into Indonesian and explore the reliability and validity of this questionnaire in a community of shoulder pain sufferers who speak Indonesian. SPADI was chosen for this study because of high psychometric assessment to assess outcomes in patients with shoulder pain disorders, especially rotator cuff disease. 6 Based on the results, SPADI-IDN can be considered valid and reliable for use with patients who speak Indonesian.
The construct validity of SPADI-IDN can be considered good, with >75% of predefined hypotheses confirmed. As hypothesized, SPADI-IDN showed a strong correlation with the OSS (r = 0.71). The strong correlation between SPADI and OSS could be related as both SPADI-IDN and OSS questionnaires are shoulder-specific PROMs. This finding lends evidence to the validity of SPADI-IDN as a measure of shoulder pain disorders. This is comparable with previous studies of the Chinese and Italian versions that showed a strong correlation between their translated SPADI and OSS (r = 0.68 and 0.77, respectively).5,26 As hypothesized, SPADI-IDN showed a weak correlation with the MCS (r = 0.02). This result aligns with the studies on the Persian and Chinese versions of the SPADI.11,26 In addition, the SPADI-IDN showed a stronger correlation with the PCS than the MCS domains. This finding lends evidence to the view that the SPADI-IDN was designed primarily to assess physical functioning, as opposed to social or emotional aspects. However, contrary to our hypothesis, the correlation of the SPADI-IDN with PCS was <0.3. This was probably caused by the difference in characteristics of the participants and the variation in the patients’ diseases among those studies. Besides that, different general health measures may cause contrary results. This comparative study uses SF-36, while this study uses a subset of SF-36, namely SF-12. The SF-12 is a shortened version of the SF-36 created to reduce the response burden.
We did not see any floor or ceiling effects in this study. No floor or ceiling effects occurred because no single participant reached the best or lowest score (Figure 2). This finding is similar to the other-language versions, such as the Chinese version. 26 There was a chance that a ceiling effect would have happened, especially in patients whose adhesive capsulitis was healing and who had taken physiotherapy adequately. However, this was not the case in this study. This may occur because the patients in this study underwent treatment that was not yet completed. Still, the absence of floor or ceiling effect indicates good content validity. 22 In addition, the internal consistency of SPADI-IDN is considered good, as indicated by a Cronbach α of 0.95. This is comparable with the Greek (0.93), Persian (0.94), Chinese (0.91), and German (0.95) versions.1,11,25,26
The SPADI-IDN showed excellent test-retest reliability (ICC = 0.99). This is comparable with the Greek (0.93), Nepali (0.95), and German (0.94) versions.1,15,25 All showed excellent test-retest reliability with the test-retest time interval of 1 to 2 weeks. Following COSMIN guidelines, the interval between the test and retest should be sufficiently long to avoid recall bias yet short enough to ensure that the patients’ characteristics relevant to the measured construct remain unchanged.18,22 In this study, a 1-week time interval was used. This study also used the GRC question, and the exclusion of those patients who changed, which may have resulted in a high ICC value compared with other studies.1,15,25 The application of GRC in the reliability study is good practice and results in better insight into reliability.
The SEM of SPADI-IDN was 2.65, MDCgrp was 0.7, and MDCind was 7.3. These values are comparable with those of the Nepali version of the SPADI (SEM = 2.1, MDCind = 5.7). 15 The results indicated sufficient capability of the SPADI-IDN for comparisons at the group level, as only small values are needed to detect change. To identify a statistically significant change in group SPADI-IDN scores, a difference of >2.65 is required since values less than the SEM cannot be consistently distinguished from measurement error. The difference between the 2 measurements should be greater than the MDCind value in individual patients and SEM to verify that an actual change happened and to separate it from measurement error. Given that the MDCind is quite low (7.3), this indicates sufficient capability of SPADI-IDN as an appropriate tool enabling long-term tracking of certain patients.
Limitations
The study has certain limitations. Some patients did not come back to fill in the second questionnaire, and this may cause a bias. Nevertheless, according to COSMIN guidelines, at least 100 participants are needed to evaluate the validity, and 50 participants are needed for test-retest reliability, 18 and the number of our study participants for both the validity and test-retest reliability evaluation met that requirement. Another limitation was that almost all patients in this study were well educated, while more than half of Indonesian people have below graduate level education. There could thus be some issues with the generalizability of this study. An additional possible drawback was that the SPADI-IDN was compared with only 1 other disease-specific questionnaire—the OSS—and 1 general health-related quality of life questionnaire, SF-12. Consequently, the number of hypotheses was lower than advised in COSMIN guidelines. Nevertheless, almost all predefined hypotheses were confirmed, indicating a good validity of the SPADI-IDN. Future research should explore the responsiveness of SPADI-IDN as well as the minimal clinically important difference (MCID). MCID should be determined to examine whether a measured difference is also clinically important as perceived by the patient. For the Nepali version of SPADI, the MCID was 12.3. 15 However, these results do not apply to the Indonesian version. Thus, future studies should evaluate the MCID of SPADI-IDN. The information on the reproducibility and responsiveness of SPADI-IDN to detect change over time is required to interpret these scores when used in longitudinal research and clinical practice to monitor Indonesian-speaking patients over time.
Conclusion
The SPADI-IDN was adapted into Indonesian. It can be considered valid and reliable as a useful tool for measuring shoulder pain in the Indonesian population.
Footnotes
Final revision submitted June 21, 2024; accepted July 2, 2024.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Kakultas Kedokteran Universitas Riau (B/089-1/UN19.5.1.1.8/UEPKK/2021).

