Abstract
Background:
Primary anterior cruciate ligament (ACL) repair has gained renewed interest in select centers for patients with proximal or midsubstance ACL tears. Therefore, it is important to reassess contemporary clinical outcomes of ACL repair to determine whether a clinical benefit exists over the gold standard of ACL reconstruction (ACLR).
Purpose:
To (1) perform a meta-analysis of comparative trials to determine whether differences in clinical outcomes and adverse events exist between ACL repair versus ACLR and (2) synthesize the midterm outcomes of available trials.
Study Design:
Systematic review; Level of evidence, 3.
Methods:
The PubMed, OVID/Medline, and Cochrane databases were queried in August 2023 for prospective and retrospective clinical trials comparing ACL repair and ACLR. Data pertaining to tear location, surgical technique, adverse events, and clinical outcome measures were recorded. DerSimonian-Laird random-effects models were constructed to quantitatively evaluate the association between ACL repair/ACLR, adverse events, and clinical outcomes. A subanalysis of minimum 5-year outcomes was performed.
Results:
Twelve studies (893 patients; 464 ACLR and 429 ACL repair) were included. Random-effects models demonstrated a higher relative risk (RR) of recurrent instability/clinical failure (RR = 1.64; 95% confidence interval [CI], 1.04-2.57; P = .032), revision ACLR (RR = 1.63; 95% CI, 1.03-2.59; P = .039), and hardware removal (RR = 4.94; 95% CI, 2.10-11.61; P = .0003) in patients who underwent primary ACL repair versus ACLR. The RR of reoperations and complications (knee-related) were not significantly different between groups. No significant differences were observed when comparing patient-reported outcome scores. In studies with minimum 5-year outcomes, no significant differences in adverse events or Lysholm scores were observed.
Conclusion:
In contemporary comparative trials of ACL repair versus ACLR, the RR of clinical failure, revision surgery due to ACL rerupture, and hardware removal was greater for primary ACL repair compared with ACLR. There were no observed differences in patient-reported outcome scores, reoperations, or knee-related complications between approaches. In the limited literature reporting on minimum 5-year outcomes, significant differences in adverse events or the International Knee Documentation Committee score were not observed.
Acute traumatic rupture of the anterior cruciate ligament (ACL) or symptomatic chronic ACL deficiency has traditionally been treated with ACL reconstruction (ACLR), which currently remains the gold standard of surgical treatment.21,34 More recently, primary repair of ACL for proximal or midsubstance tear locations has gained renewed interest among knee surgeons given recent surgical and technological advancements. Proponents of primary ACL repair advocate that this procedure may result in several purported benefits compared with ACLR, including faster rehabilitation, an accelerated return to sports, preserved tibiofemoral proprioception, elimination of donor-site morbidity, and a reduction in adverse events given the less traumatic nature of the procedure.5,17 However, as ACL repair continues to be explored as a potential treatment approach in select patients, it is essential that such proposed advantages are empirically proven before abandoning the current gold standard of ACLR.
Numerous studies have investigated the clinical efficacy and survivorship of ACL repair,3,14,15 with several recent systematic reviews suggesting that primary repair can be an effective treatment modality with reasonable clinical outcomes when considered against ACLR.2,33,36 However, data from these studies also consider historical cohorts and may not reflect current practices. A systematic review of only 5 contemporary comparative studies reported no difference between these treatments as they pertain to clinical outcome scores, knee laxity, or graft rerupture rates. 24 Recent data from contemporary randomized controlled trials are also conflicting, with some authors reporting that ACL repair confers noninferior clinical outcomes compared with ACLR, whereas others suggest higher rates of adverse events and failure with ACL repair.13,22 The inconsistency observed in the literature as it pertains to the relative propensity for clinical outcome improvement, restoration of knee stability, and graft failure-free survival may partially be attributed to small sample sizes, historic practices, and heterogeneity in methodology. As such, a comprehensive synthesis of contemporary literature may allow for a robust quantitative assessment of treatment efficacy that is appropriately powered and may subsequently leverage enhanced insight into this ongoing clinical controversy.
Given the recent increase in the number of clinical trials comparing ACL repair and ACLR,13,23,25,26,29,32,36 it is essential that the data from these more recent investigations are synthesized and reviewed such that the advantages and disadvantages of both procedures are comprehensively and transparently understood. Furthermore, few trials have recently published the relative efficacy of these procedures at midterm follow-up,13,23,35 the results of which have not been systematically compiled and interpreted.
The aims of the current study were to (1) perform a systematic review and meta-analysis of available contemporary comparative trials including all to determine whether differences in clinical outcomes and adverse events exist between all types of ACL repair versus ACLR techniques and (2) synthesize the midterm outcomes of available trials. We hypothesized that ACL repair would not demonstrate clinical superiority in terms of reducing adverse events or improving clinical outcomes when compared with ACLR at both the short- and midterm follow-up.
Methods
Article Search Process
This systematic review and meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) statement. 28 Literature reporting on the clinical outcomes of ACL repair and ACLR was performed using the following indexing databases: (1) Cochrane Database of Systematic Reviews, the Cochrane Central Register of Controlled Trials, (2) PubMed, and (3) Ovid/MEDLINE. The query was performed in August 2023 using combinations of the following Boolean search and MESH terms: “ACL repair,”“ACL reconstruction,” and “Clinical outcomes.” This review was registered with the PROSPERO international prospective register of systematic reviews before initiation of data collection (PROSPERO identification: 455143).
Study Eligibility
Studies qualified for inclusion in this systematic review if the topic of study pertained to any investigation reporting on clinical outcomes of ACL repair versus ACLR that were published in the English language. Exclusion criteria consisted of (1) outcome studies not directly comparing ACL repair and ACLR; (2) editorials, abstracts, case reports, and surveys; (3) biomechanical or cadaveric studies. Furthermore, a time restriction of minimum 2-year follow-up was implicated in assessing study eligibility. The level of evidence of the studies was restricted to 1 to 3 (prospective and retrospective clinical trials), evidence Level 4 and 5 studies being excluded.
All articles identified by the search were independently screened by 2 reviewers (K.K., A.P.). Sequential screening of all articles was performed, which included assessment of duplicates, screening of article title, evaluation of content in the abstract, and full text review. References of identified articles were also explored and reconciled to minimize any risk of missing published literature. Furthermore, studies reporting on the same cohort at different follow-up points were identified through this systematic approach, and only the article with longer follow-up was included.
Data Procurement
All data were recorded in a custom spreadsheet using a modified information extraction table. 18 Categories for data collection for each full article included (1) article information, (2) ACL repair or reconstruction method, (3) patient information, (4) tear location, (5) adverse events, and (6) patient-reported outcome measures.
Assessment of Methodological Quality
The Methodological Index for Nonrandomized Studies (MINORS) checklist was used to evaluate the quality of nonrandomized comparative studies, 19 while the Cochrane Risk-of-Bias Version 2 (RoB-2) tool was used to evaluate randomized controlled trials. 31 The MINORS checklist involves 12 items to assess quality, of which only 4 are applicable to comparative studies. The 4 additional criteria specific to comparative groups were used to assess the bias present in articles when selecting cohorts. The maximum MINORS score is 16 for noncomparative studies and 24 for comparative studies.
Using the RoB-2 tool, potential study biases are classified in 1 of 3 ways: low risk of bias, high risk of bias, and some concern regarding potential risk of bias. The 5 potential types of biases and the method by which they are assessed include (1) bias arising from the randomization process, (2) bias due to deviations from the intended intervention, (3) bias due to missing outcome data, (4) bias in measurement of the outcome, and (5) bias in selection of the reported result. Two reviewers scored each study, and any discrepancies were adjudicated by consensus agreement or excluded altogether. Two independent observers (K.K., A.P.) assessed all included studies.
Statistical Analysis
Data from all trials comparing primary ACL repair versus ACLR were quantitatively explored using DerSimonian-Laird random-effects models to determine pooled effect sizes.6-8 For binary outcomes, the relative risk (RR) was calculated from 2 × 2 tables for each individual study. In accordance with previous recommendations and guidelines for performing meta-analyses from statistical methods literature, a minimum of 2 studies were incorporated into each meta-analysis as this number is efficient in drawing conclusions from resultant data.12,16 For continuous outcomes, the standardized mean difference (SMD) was calculated from mean postoperative patient-reported outcome measures. The pooled effect size was calculated as a weighted mean of the effects estimated in the individual studies, with weights representing the amount of information from each study. The 95% confidence interval (CI) was used to report all pooled statistics. Heterogeneity was assessed by the I2 statistic using random-effects models and was regarded as nonsignificant when I2 = 0%, possibly unimportant when the I2 value was <40%, moderate between 40% and 75%, and considerable when more than 75%. 20 Comparison-adjusted funnel plots were constructed to understand the potential for publication bias among treatment comparisons. 10 The Egger regression test of the intervention effect estimates on their standard errors weighted by their inverse variance were performed to statistically evaluate for the presence of publication bias when a minimum number of 5 studies reported on an outcome investigated quantitatively. 27
All statistical analyses were performed using the computing software R (Version 3.6.1) and RStudio (Version 1.2.5033, R Foundation for Statistical Computing). A 2-tailed P value of <.05 was considered to indicate statistical significance.
Results
Twelve studies (893 patients; 464 ACLR vs 429 ACL repair) published between 2016 and 2023 were included in the final quantitative analysis (Figure 1). § The characteristics of the included studies are outlined in Appendix Table A1. The mean age of patients who underwent ACLR was 25.2 ± 7.2 years, while that of patients who underwent ACL repair was 25.7 ± 7.7 years (P > .05). The mean time from injury to surgery for the ACL repair cohort was 24.3 ± 18.5 days, while that of the ACLR cohort was 42.5 ± 18.3 days (P = .07). A total of 5 (41.7%) studies utilized the dynamic intraligamentary stabilization (DIS) system for ACL repair,4,13,23,25,26 while 5 (41.7%) utilized suture anchors with internal bracing,1,9,11,32,35 and 2 (16.6%) utilized the bridge-enhanced ACL repair (BEAR) system.29,30 Five (41.7%) studies included patients with only proximal (Sherman grade 1 or 2) ACL tears,1,9,11,32,35 5 (41.7%) studies included both proximal and midsubstance tears,13,25,26,29,30 and 2 (16.6%) studies included tear types of any location.4,23 Four studies (33.3%) were Level 1 randomized controlled trials,13,23,26,29 2 studies (16.6%) were prospective nonrandomized trials,9,30 and the remaining 6 studies (51.1%) were retrospective cohort studies.1,4,11,25,32,35 The methodological quality of nonrandomized studies was high overall (Table 1). Likewise, the methodological quality of the 4 randomized trials was high (Figure 2).

Flowchart for study search and inclusion.
Methodological Bias Assessment of Studies Using MINORS Criteria 19

MINORS, Methodological Index for Non-randomized Studies.
Total score out of 24 possible points.

Cochrane Risk-of-Bias Version 2 tool assessment for the included randomized controlled trials. 31 (A) Traffic light plot of specific bias domains. (B) Summary plot demonstrating overall percentage of bias within studies.
Adverse Events: Recurrent Instability and Clinical Failure
Eleven studies reported the incidence of recurrent instability and clinical failure after the index procedure (Figure 3). ‖ The incidence of failure was 11.9% in the ACL repair cohort and 6.1% in the ACLR cohort. The RR of failure was significantly greater for participants who underwent primary repair (RR = 1.64; 95% CI, 1.04-2.57; P = .032). Publication bias was found to be a nonsignificant contributor to the study effect estimates (see Supplemental Figures S1-S5; all supplemental material is available separately).
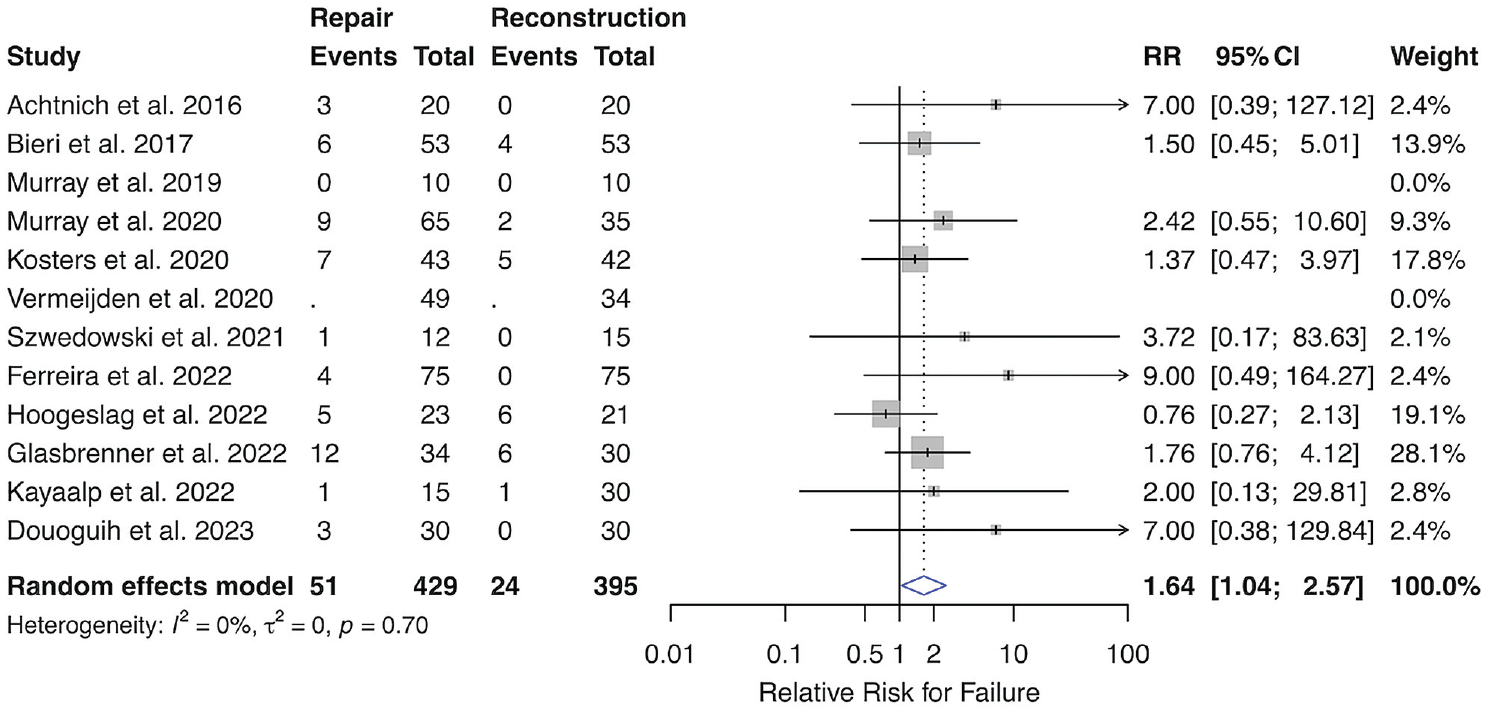
Forest plot depicting random-effects model for RR for clinical failure after ACLR after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in RR. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 0% (0%-46.8%) and was nonsignificant. ACL, anterior cruciate ligament; ACLR, ACL reconstruction; CI, confidence interval; RR, relative risk.
Anecdotally, it was observed that patients who underwent DIS may experience higher rates of adverse events compared with their counterparts. When removing only patients who underwent DIS from the analysis and keeping all other participant data constant, the rate of failure after ACL repair decreased to 7.7% while that of ACLR was 6.1% (P = .46). However, results from a formal leave-one-out sensitivity analysis, removing all studies comparing DIS with ACLR, to better understand the influence of the DIS technique on clinical failure, demonstrated that the RR of failure was more pronounced than the initial analysis (failure rate: ACL repair, 7.7% vs ACLR, 0.9%; RR = 3.96; 95% CI, 1.4-11.3; P = .0098).
Adverse Events: Graft Failure Necessitating Revision ACLR
Eleven studies reported the incidence of revision ACLR due to graft rupture after the index procedure, differing from clinical failure as not all patients meeting criteria for clinical failure went on to revision surgery (Figure 4). ‖ The incidence of revision surgery was 11.4% in the ACL repair cohort and 5.8% in the ACLR cohort. The RR of revision surgery was significantly greater for participants that underwent primary ACL repair (RR = 1.63; 95% CI, 1.03-2.59; P = .039). Publication bias was found to be a nonsignificant contributor to the study effect estimates (Supplemental Figure S2).

Forest plot depicting random-effects model for RR for revision ACLR after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in RR. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 0% (0%-46.9%) and insignificant. ACL, anterior cruciate ligament; ACLR, ACL reconstruction; CI, confidence interval; RR, relative risk.
A secondary analysis excluding patients who underwent DIS was subsequently performed. When removing only patients who underwent DIS from the analysis and keeping all other participant data constant, the revision rate of ACL repair decreased to 7.7% while that of ACLR was 5.8% (P = 0.38). However, results from a formal leave-one-out sensitivity analysis, removing all studies comparing DIS with ACLR, to better understand the influence of the DIS technique on revision ACLR, demonstrated that the RR of failure was more pronounced than the initial analysis (revision rate: ACL repair, 7.7% vs ACLR, 0.9%; RR = 3.96; 95% CI, 1.4-11.3; P = .0098).
Adverse Events: Reoperations (Nongraft Related)
Eleven studies reported the incidence of nongraft-related reoperations following the index procedure (Figure 5). ‖ The incidence of reoperations was 15.4% in the repair cohort and 11.9% in the ACLR cohort. The RR of reoperations was greater for participants who underwent primary repair (RR = 1.24; 95% CI, 0.73-2.10; P = 0.43). Publication bias was found to be a nonsignificant contributor to the study effect estimates (Supplemental Figure S3).

Forest plot depicting random-effects model for nongraft-related reoperations after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in relative risk. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 42% (0%-72.5%) and was moderate. ACL, anterior cruciate ligament; CI, confidence interval; RR, relative risk.
Adverse Events: Complications (Knee-Related)
Ten studies reported the incidence of complications that were knee-related but excluded graft rupture (Figure 6). ¶ The incidence of complications was 5.6% in the ACL repair cohort and 6.6% in the ACLR cohort. The RR of complications was lower for participants who underwent ACL repair (RR = 0.88; 95% CI, 0.53-1.46; P = .61) and was not statistically significant. Publication bias was found to be a nonsignificant contributor to the study effect estimates (Supplemental Figure S4).

Forest plot depicting random-effects model complications after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in relative risk. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 0% (0%-37.7%) and was insignificant. ACL, anterior cruciate ligament; CI, confidence interval; RR, relative risk.
Adverse Events: Hardware Removal
Ten studies reported the incidence of hardware removal specifically after ACL repair and ACLR (Figure 7). ¶ The incidence of hardware removal was 6.7% in the repair cohort and 1.0% in the ACLR cohort. The RR of hardware removal was greater for participants who underwent primary ACL repair (RR = 4.94; 95% CI, 2.10-11.61; P = .0003). Publication bias was found to be a nonsignificant contributor to the study effect estimates (Supplemental Figure S5).
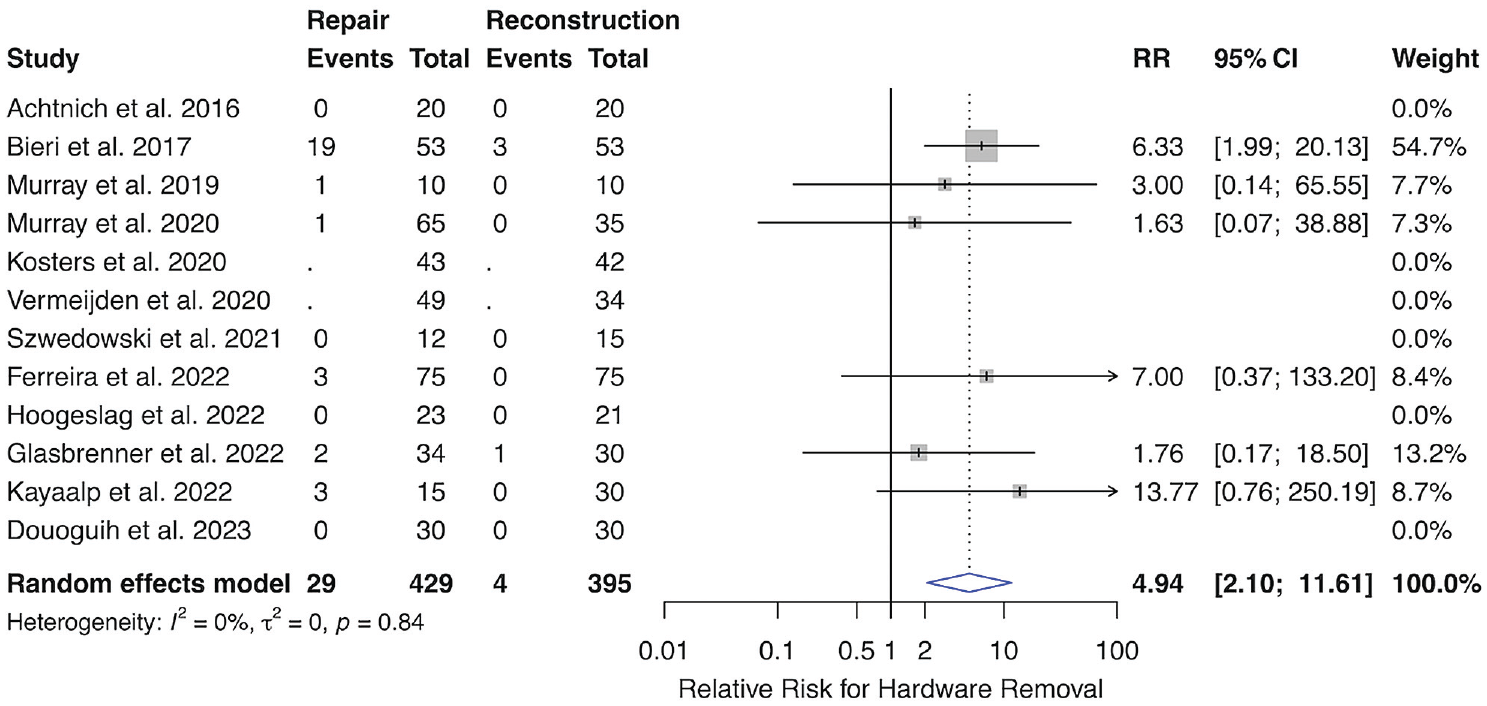
Forest plot depicting random-effects model hardware removal after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in relative risk. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 0% (0%-61.3%) and was insignificant. ACL, anterior cruciate ligament; CI, confidence interval; RR, relative risk.
Anecdotally, it was also observed that the rate of hardware removal may be influenced by those undergoing DIS given the properties of the implant system. When removing only DIS patients from this analysis, the rate of hardware removal was no longer statistically significant (ACL repair, 1.9% vs ACLR, 1.0%; P = .69). When performing a formal leave-one-out meta-analysis removing all studies examining DIS versus ACLR, the lack of a statistically significant difference was maintained (ACL repair, 1.9% vs ACLR, 0%; RR = 3.37; 95% CI, 0.6-19.7; P = .18).
Clinical Outcomes: International Knee Documentation Committee Score
Seven studies reported the mean postoperative International Knee Documentation Committee (IKDC) score at latest follow-up (Figure 8).13,23,25,26,29,30 The random-effects model favored ACLR (SMD = 0.08; 95% CI, -0.23 to 0.39; P = .60), although it did not reach statistical significance. Publication bias was found to be a nonsignificant contributor to the study effect estimates (Supplemental Figure S6).

Forest plot depicting random-effects model for the IKDC score after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in relative risk. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 56.9% (0%-81.4%) and was moderate. ACL, anterior cruciate ligament; CI, confidence interval; IKDC, International Knee Documentation Committee; SMD, standardized mean difference.
Clinical Outcomes: Lysholm Score
Six studies reported the mean postoperative Lysholm score at latest follow-up (Figure 9).9,11,13,25,26,32 The random-effects model favored the ACLR group (SMD = 0.15; 95% CI, −0.04 to 0.34; P = .12), though it did not reach statistical significance. Publication bias was found to be a nonsignificant contributor to the study effect estimates (Supplemental Figure S7).

Forest plot depicting random-effects model for the Lysholm score after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in relative risk. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 0% (0%-59.9%) and insignificant. ACL, anterior cruciate ligament; CI, confidence interval; SMD, standardized mean difference.
Clinical Outcomes: Tegner Score
Five studies reported the mean postoperative Tegner score at latest follow-up (Figure 10).9,11,23,25,26 The random-effects model favored the ACLR group (SMD = 0.09; 95% CI, -0.11 to 0.29; P = .40), though this did not reach statistical significance. Publication bias was found to be a nonsignificant contributor to the study effect estimates (Supplemental Figure S8).
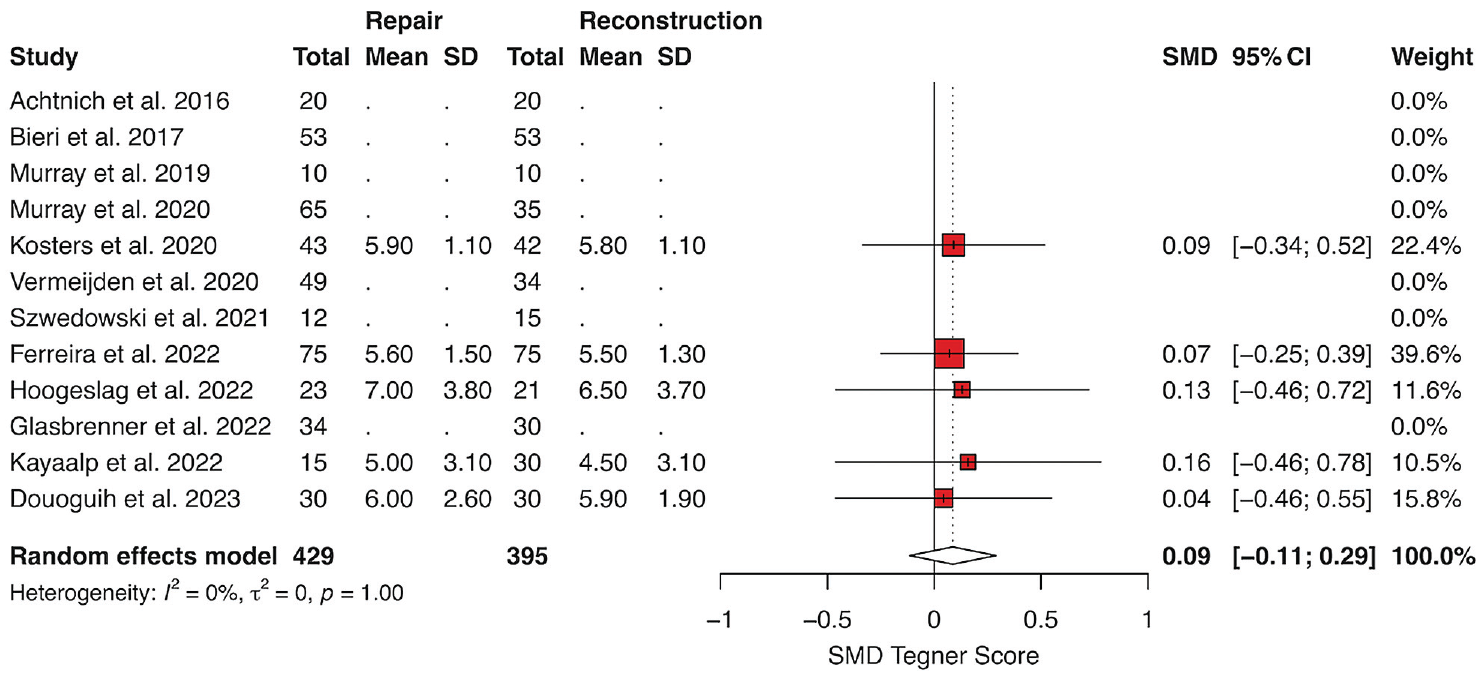
Forest plot depicting random-effects model for the Tegner score after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in relative risk. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. The heterogeneity was I2 = 0% (0%-0%) and was insignificant. ACL, anterior cruciate ligament; CI, confidence interval; SMD, standardized mean difference.
Clinical Outcomes: Knee Injury and Osteoarthritis Outcome Score
Knee injury and Osteoarthritis Outcome Score (KOOS) subscale values were reported by 3 studies (Figures 11 and 12).11,23,30 The random-effects model favored ACLR for KOOS-Pain (SMD = 0.38; 95% CI, -0.07 to 0.83; P = .097), KOOS-Symptoms (SMD = 0.13; 95% CI, −0.41 to 0.66; P = .63), and KOOS-Quality of Life (QoL) (SMD = 0.13; 95% CI, -0.21 to 0.47; P = .45), although these models failed to reach statistical significance (Figure 11). For the KOOS-Activities of Daily Living (ADL), the model was equivocal (SMD = -0.01; 95% CI, -0.28 to 0.26; P = .93), while the model for KOOS-Sport and Recreation (Sport) favored ACL repair although this was not significant (SMD = -0.23; 95% CI, -0.90 to 0.45, P = .51) (Figure 12). Heterogeneity for KOOS-Sport was considerable (I2 = 75%; 95% CI, 17.1%-92.5%), while that for KOOS-Pain (I2 = 46.8%; 95% CI, 0%-84.4%) and KOOS-Symptoms (I2 = 61.6%; 95% CI, 0%-89%) was moderate. Heterogeneity for KOOS-QoL was considered possibly unimportant (I2 = 22.1%; 95% CI, 0%-91.9%), while heterogeneity for KOOS-ADL was nonsignificant (I2 = 0%; 95% CI, 0%-0%). The Egger test was not performed in this analysis due to the limited number of studies.

Forest plots depicting random-effects model for (A) KOOS-Pain, (B) KOOS-Symptoms, and (C) KOOS-ADL scores after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in RR. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. ACL, anterior cruciate ligament; ADL, activities of daily living; CI, confidence interval; KOOS, Knee injury and Osteoarthritis Outcome Score; RR, relative risk.

Forest plots depicting random-effects model for (A) KOOS-Sport and (B) KOOS-QoL scores after primary repair versus reconstruction of ACL tears. The x-axis depicts incremental changes in RR. Gray boxes represent the weighted contribution of each study, with the horizontal black lines representing the 95% CI of the treatment estimate. ACL, anterior cruciate ligament; CI, confidence interval; KOOS, Knee injury and Osteoarthritis Outcome Score; QoL, quality of life; RR, relative risk; SMD, standardized mean difference.
Subanalysis: 5-Year Outcomes
A secondary analysis of studies reporting minimum 5-year outcomes was performed to determine whether differences in adverse events or clinical outcomes existed at midterm follow-up. Two of these studies were 5-year follow-up studies of randomized controlled trials,13,23 while the remaining study was a retrospective cohort study. 35 This study reported only the means and standard deviations of the Forgotten Joint Score 12-item questionnaire, with the ACL repair group demonstrating higher scores (thought less about their operated knees) compared with the ACLR group (85.3 ± 14.2 vs 74.3 ± 23.3, P = .022).
Data on adverse events (clinical failure, revision ACLR, and nongraft-related reoperations) as well as clinical outcomes (IKDC score) was available from 2 randomized controlled trials. The random-effects models of randomized controlled trials favored the ACLR cohort but failed to reach statistical significance (Supplemental Figures S9-S12).
Subanalysis: Return to Sport and Progression of Osteoarthritis
A single study reported on the propensity to return to sport after ACL repair and ACLR, 11 while a single study reported on the progression of osteoarthritis after ACL repair and ACLR. 23 In the study examining return to sports, the authors reported that no statistically significant differences were observed in time to return (ACL repair, 10.2 months vs ACLR, 11.2 months; P = .44), proportion of patients who returned to sport (ACL repair, 58% vs ACLR, 49%; P = .14), or proportion of patients who returned at the same level (ACL repair, 56% vs ACLR, 45%; P = .078). In the study examining osteoarthritis progression, no significant differences were observed at final follow-up (P = .25), with 90.9% of ACL repair patients and 77.8% of ACLR patients demonstrating Kellgren-Lawrence grade 0, and 9.1% of ACL repair patients and 22.2% of ACLR patients demonstrating Kellgren-Lawrence grade 1. There were no patients with osteoarthritis classified as Kellgren-Lawrence grades 2 to 4.
Discussion
The principal findings of the current study are as follows: (1) the RR of incurring clinical failure with recurrent instability, necessitating revision ACLR, and undergoing hardware removal was significantly greater for patients undergoing ACL repair compared with ACLR; (2) the SMDs for 8 patient-reported outcome measures were not significant and therefore did not meaningfully favor either treatment; and (3) in limited data from randomized controlled trials with minimum 5-year outcomes after ACL repair versus ACLR, no significant differences in adverse events or IKDC score were observed.
The current study compiled the largest aggregation of data comparing ACL repair and ACLR to date, with a study population approaching 900 patients. These data were derived from several recent investigations comparing ACLR and ACL repair, reflecting currently applied methods and more contemporary outcomes than previous analyses. Furthermore, this systematic review restricted follow-up to a minimum of 2 years, reflecting longer potential for improvement compared with studies examining follow-up at <1 year. Utilizing these data, it was found that several random-effects models favored ACLR as it pertains to reducing the risk of experiencing recurrent instability and clinical failure, necessitating revision ACLR due to graft failure, or a reoperation for hardware removal. When excluding studies comparing DIS, these effects were pronounced in favor of ACLR, with RR estimates for failure and revision ACLR exceeding 300% for patients undergoing ACL repair. When including DIS, ACL repair patients experienced approximately double (11.9% vs 6.1%) the rate of clinical failure/recurrent instability compared with ACLR patients. Furthermore, undergoing ACLR resulted in 64% and 63% reductions in the risk of failure and revision ACLR, respectively, while it conferred an almost 500% reduction in the need for hardware removal; however, risk for hardware removal was diminished significantly when removing DIS patients.
The profound difference in rates of adverse events may be attributed to the heterogeneous number of ACL repair systems employed among studies, including the DIS, BEAR system, and suture anchor repair with internal brace stabilization. For example, Bieri et al 4 reported a 35.8% rate of hardware removal for patients undergoing ACL repair with the DIS system at 2-year follow-up. This may be explained by the need for use of a monobloc spring screw with DIS that is larger than interference screws used ACLR and subsequently greater symptoms attributed to this - an issue that is generally not encountered with ACLR. In terms of failure rates, Glasbrenner et al 13 reported 35.3% and 29.4% rates of failure and revision ACLR, respectively, with the DIS system at 5-year follow-up. Interestingly, when performing a sensitivity analysis removing DIS patients while keeping all participants constant, the pooled rate of failure in patients undergoing ACL repair (7.7%) approaches that of ACLR (6.1%); however, when removing all studies comparing DIS versus ACLR and performing a formal leave-one-out meta-analysis, the difference in failure was more pronounced than the original pooled analysis (ACL repair, 7.7% vs ACLR, 0.9%; P = .0098), suggesting a high rate of failures for both treatment cohorts in these select studies. Perhaps moving forward, implementing primary repair with internal bracing or the BEAR system should be reserved for proximal ACL tears in select patients, as opposed to the DIS system, to avoid excessive failure rates and other adverse events.
None of the random-effects models were found to be statistically significant as it pertains to comparing postoperative patient-reported outcome measures. Although the models favored ACLR for the primary endpoints of the mean IKDC, Lysholm, Tegner, KOOS-Pain, KOOS-Symptoms, and KOOS-QoL, the models favored ACL repair for KOOS-ADL and KOOS-Sport, none of which were significant nor represented clinically important differences. This is plausible, as although outcomes after ACL repair had traditionally been reported as poor, advancements in patient indications, postoperative rehabilitation protocols, functional retraining, pain control regimens, and return to sports testing may now help to partially mitigate these differences. Recently, Douoguih et al 9 compared clinical outcomes of 30 patients who underwent ACL repair with internal bracing for proximal ACL avulsions or high-grade partial tears and 30 patients who underwent ACLR with bone-patellar tendon-bone or quadriceps tendon autograft as well as allograft in select cases. At a minimum of 2-year follow-up, they reported comparable rates in achievement of the MCID for the IKDC and the KOOS subscales, with no significant differences in mean patient-reported outcome scores. Findings such as these suggest that ACL repair may be used appropriately in select patients with proximal ACL tears, although the methodologically heterogeneity in these studies remains concerning and longer-term data are needed. Moving forward, it will also be important for studies to routinely assess the maximal outcome improvement and define metrics of clinically meaningful outcome improvement, such as the patient acceptable symptom state, to fully understand the propensity for improvement and satisfaction with each procedure. However, at this point, the current study cannot provide sufficient data to suggest that the current standard of ACLR can be abandoned for proximal ACL tears when considering clinical outcomes and adverse events.
Limited data were available on the midterm outcomes of trials comparing ACL repair and ACLR, with only 2 of the 3 studies presenting data amenable to a formal meta-analysis. Both of these trials randomized patients to ACL repair using the DIS system and patients to ACLR using 4-strand hamstring tendon autograft. In the study by Hoogeslag et al, 23 the location of ACL tears was proximal in 83.3% of repairs, midsubstance in 12.5% of repairs, and distal in 4.2% of repairs. In the study by Glassbrenner et al, 13 the location of ACL tears was proximal in 91.2% of repairs midsubstance in 8.8% of repairs, and no repairs were for distal tears. Hoogeslag et al 23 reported a higher rate of clinical failure and revision surgery for the ACLR cohort (27.2% versus 20.8%), while Glassbrenner et al 13 reported a higher rate of failure (35% versus 20%) and revision ACLR (29% versus 17%) for the ACL repair cohort. It is unclear as to why results from these randomized controlled trials are in opposition but may be related to differences in demographic characteristics, concomitant lesions such as meniscal tears, and rehabilitation protocols. For example, Hoogeslag et al 23 excluded patients with concomitant ligamentous lesions, a history of contra- or ipsilateral knee surgery, meniscal lesions needing surgical intervention, full-thickness cartilage lesions, and osteoarthritis seen on the preoperative (weightbearing) radiographs, while Glassbrenner et al 13 noted that debridement or partial resection of meniscal lesions was performed in 12 patients in the ACL repair group and 6 in the ACLR group. Regardless, additional data of similar follow-up is needed to draw meaningful conclusions about these procedures at mid- and long-term follow-up.
Currently available data from the literature suggest that, when considering adverse events, ACL repair may be an inferior treatment option than ACLR. Despite a renewed interest in ACL repair as a treatment approach for patients with select ACL tear patterns, patients undergoing ACL repair may be at an increased risk of failure after this surgery in the short-term and should weigh this risk in the context of proposed benefits of ACL repair. Future investigations are necessary to further refine the indications for ACL repair; specifically, it is necessary to better understand when it is reasonable, based on patient age, ACL tear pattern, physical activity, and expectations, to perform primary ACL repair with suture augmentation, ACL repair with the DIS system, or BEAR. Furthermore, prospective comparisons of different ACL repair techniques may be of interest to determine whether associations exist between repair technique, clinical outcomes, and adverse events.
Limitations
The results of the current systematic review are important to consider in the context of several limitations. First, the majority of studies were of evidence Levels 2 and 3, and therefore, some bias was inherently present given the nature of the study design. However, methodological bias was examined and considered to be low. Second, ACL repair and ACLR techniques differed across studies and may therefore contribute to observed differences in study results. Third, only 3 studies were identified at the time of this study that examined minimum 5-year outcomes between ACL repair and ACLR, with only 2 of these studies reporting data amenable to pooling in meta-analyses for adverse events and clinical outcomes. Therefore, additional homogenous data are necessary to draw meaningful conclusions on the relative efficacy of these procedures at longer-term follow-up. Fourth, heterogeneity in inclusion criteria across studies was observed. For example, the timing from injury to surgery ranged between 3 weeks and 12 months, tear location varied within and across studies, and concomitant injuries and their treatment also differed. Fifth, outcome data relative to ACL repair that may be of interest to compare with ACLR, such as joint proprioception and related measures, were not reported, while the risk of osteoarthritis progression and return to sports metrics were only reported in 1 study each.11,23 Finally, given the limited quantitative data available, additional analyses stratified by ACL repair technique, ACLR technique, or ACL tear location could not be performed. Future studies are warranted to properly investigate whether differences in outcomes exist between the various ACL repair techniques and applied to specific tear locations in comparison with the gold standard of ACLR.
Conclusion
In contemporary comparative trials of ACL repair versus ACLR, the RR of clinical failure, revision surgery due to ACL rerupture, and hardware removal was greater for primary ACL repair compared with ACLR. There were no observed differences in patient-reported outcome measures, reoperations, and knee-related complications between approaches. In the limited literature reporting on minimum 5-year outcomes, significant differences in adverse events or the IKDC score were not observed. Additional data comparing mid- to long-term outcomes of ACL repair and ACLR are needed.
Supplemental Material
sj-pdf-1-ojs-10.1177_23259671241253591 – Supplemental material for Clinical Results of Primary Repair Versus Reconstruction of the Anterior Cruciate Ligament: A Systematic Review and Meta-analysis of Contemporary Trials
Supplemental material, sj-pdf-1-ojs-10.1177_23259671241253591 for Clinical Results of Primary Repair Versus Reconstruction of the Anterior Cruciate Ligament: A Systematic Review and Meta-analysis of Contemporary Trials by Kyle N. Kunze, Ayoosh Pareek, Benedict U. Nwachukwu, Anil S. Ranawat, Andrew D. Pearle, Bryan T. Kelly, Answorth A. Allen and Riley J. Williams in Orthopaedic Journal of Sports Medicine
Footnotes
Appendix
Characteristics of the Included Studies a
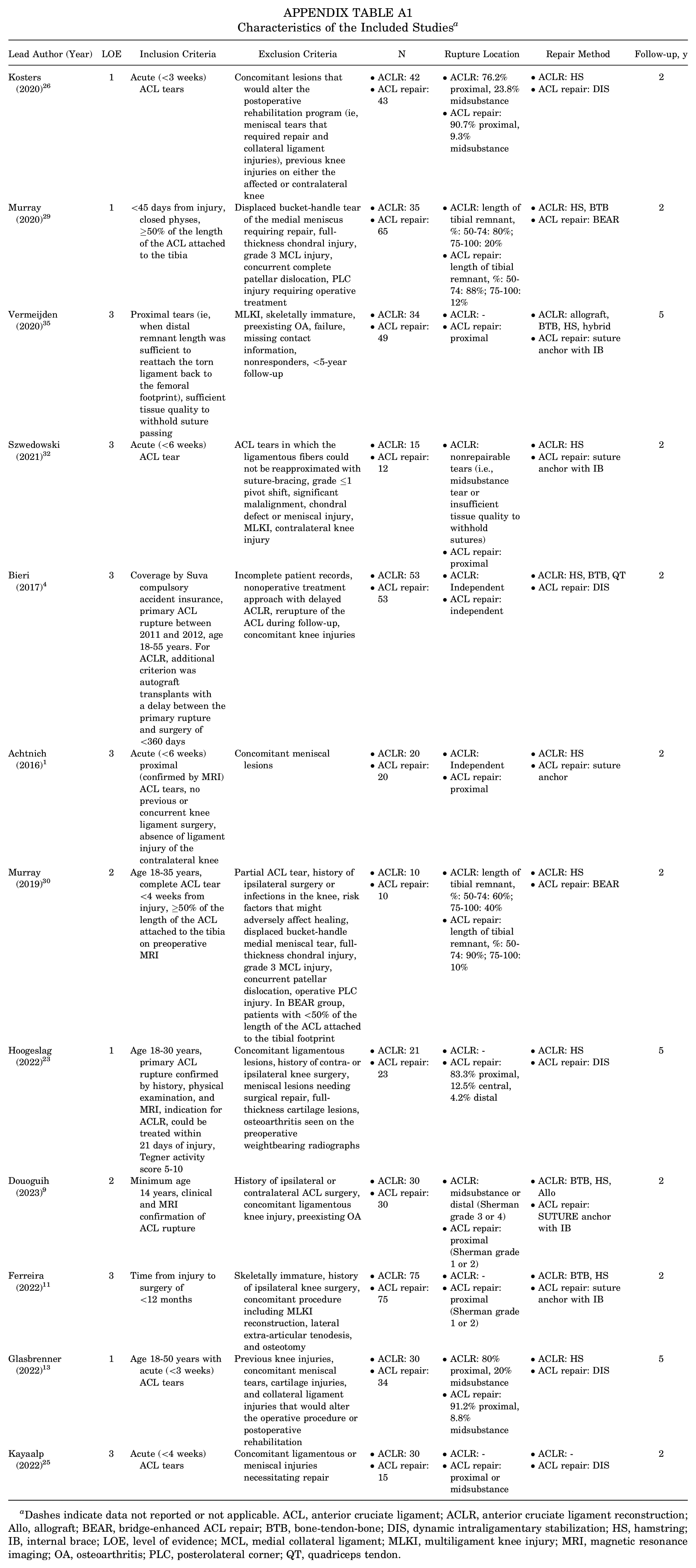
| Lead Author (Year) | LOE | Inclusion Criteria | Exclusion Criteria | N | Rupture Location | Repair Method | Follow-up, y |
|---|---|---|---|---|---|---|---|
| Kosters(2020) 26 | 1 | Acute (<3 weeks) ACL tears | Concomitant lesions that would alter the postoperative rehabilitation program (ie, meniscal tears that required repair and collateral ligament injuries), previous knee injuries on either the affected or contralateral knee | • ACLR: 42 • ACL repair: 43 |
• ACLR: 76.2% proximal, 23.8% midsubstance • ACL repair: 90.7% proximal, 9.3% midsubstance |
• ACLR: HS • ACL repair: DIS |
2 |
| Murray(2020) 29 | 1 | <45 days from injury, closed physes, ≥50% of the length of the ACL attached to the tibia | Displaced bucket-handle tear of the medial meniscus requiring repair, full-thickness chondral injury, grade 3 MCL injury, concurrent complete patellar dislocation, PLC injury requiring operative treatment | • ACLR: 35 • ACL repair: 65 |
• ACLR: length of tibial remnant, %: 50-74: 80%; 75-100: 20% • ACL repair: length of tibial remnant, %: 50-74: 88%; 75-100: 12% |
• ACLR: HS, BTB • ACL repair: BEAR |
2 |
| Vermeijden(2020) 35 | 3 | Proximal tears (ie, when distal remnant length was sufficient to reattach the torn ligament back to the femoral footprint), sufficient tissue quality to withhold suture passing | MLKI, skeletally immature, preexisting OA, failure, missing contact information, nonresponders, <5-year follow-up | • ACLR: 34 • ACL repair: 49 |
• ACLR: - • ACL repair: proximal |
• ACLR: allograft, BTB, HS, hybrid • ACL repair: suture anchor with IB |
5 |
| Szwedowski(2021) 32 | 3 | Acute (<6 weeks) ACL tear | ACL tears in which the ligamentous fibers could not be reapproximated with suture-bracing, grade ≤1 pivot shift, significant malalignment, chondral defect or meniscal injury, MLKI, contralateral knee injury | • ACLR: 15 • ACL repair: 12 |
• ACLR: nonrepairable tears (i.e., midsubstance tear or insufficient tissue quality to withhold sutures) • ACL repair: proximal |
• ACLR: HS • ACL repair: suture anchor with IB |
2 |
| Bieri(2017) 4 | 3 | Coverage by Suva compulsory accident insurance, primary ACL rupture between 2011 and 2012, age 18-55 years. For ACLR, additional criterion was autograft transplants with a delay between the primary rupture and surgery of <360 days | Incomplete patient records, nonoperative treatment approach with delayed ACLR, rerupture of the ACL during follow-up, concomitant knee injuries | • ACLR: 53 • ACL repair: 53 |
• ACLR: Independent • ACL repair: independent |
• ACLR: HS, BTB, QT • ACL repair: DIS |
2 |
| Achtnich(2016) 1 | 3 | Acute (<6 weeks) proximal (confirmed by MRI) ACL tears, no previous or concurrent knee ligament surgery, absence of ligament injury of the contralateral knee | Concomitant meniscal lesions | • ACLR: 20 • ACL repair: 20 |
• ACLR: Independent • ACL repair: proximal |
• ACLR: HS • ACL repair: suture anchor |
2 |
| Murray(2019) 30 | 2 | Age 18-35 years, complete ACL tear <4 weeks from injury, ≥50% of the length of the ACL attached to the tibia on preoperative MRI | Partial ACL tear, history of ipsilateral surgery or infections in the knee, risk factors that might adversely affect healing, displaced bucket-handle medial meniscal tear, full-thickness chondral injury, grade 3 MCL injury, concurrent patellar dislocation, operative PLC injury. In BEAR group, patients with <50% of the length of the ACL attached to the tibial footprint | • ACLR: 10 • ACL repair: 10 |
• ACLR: length of tibial remnant, %: 50-74: 60%; 75-100: 40% • ACL repair: length of tibial remnant, %: 50-74: 90%; 75-100: 10% |
• ACLR: HS • ACL repair: BEAR |
2 |
| Hoogeslag(2022) 23 | 1 | Age 18-30 years, primary ACL rupture confirmed by history, physical examination, and MRI, indication for ACLR, could be treated within 21 days of injury, Tegner activity score 5-10 | Concomitant ligamentous lesions, history of contra- or ipsilateral knee surgery, meniscal lesions needing surgical repair, full-thickness cartilage lesions, osteoarthritis seen on the preoperative weightbearing radiographs | • ACLR: 21 • ACL repair: 23 |
• ACLR: - • ACL repair: 83.3% proximal, 12.5% central, 4.2% distal |
• ACLR: HS • ACL repair: DIS |
5 |
| Douoguih(2023) 9 | 2 | Minimum age 14 years, clinical and MRI confirmation of ACL rupture | History of ipsilateral or contralateral ACL surgery, concomitant ligamentous knee injury, preexisting OA | • ACLR: 30 • ACL repair: 30 |
• ACLR: midsubstance or distal (Sherman grade 3 or 4) • ACL repair: proximal (Sherman grade 1 or 2) |
• ACLR: BTB, HS, Allo • ACL repair: SUTURE anchor with IB |
2 |
| Ferreira(2022) 11 | 3 | Time from injury to surgery of <12 months | Skeletally immature, history of ipsilateral knee surgery, concomitant procedure including MLKI reconstruction, lateral extra-articular tenodesis, and osteotomy | • ACLR: 75 • ACL repair: 75 |
• ACLR: - • ACL repair: proximal (Sherman grade 1 or 2) |
• ACLR: BTB, HS • ACL repair: suture anchor with IB |
2 |
| Glasbrenner(2022) 13 | 1 | Age 18-50 years with acute (<3 weeks) ACL tears | Previous knee injuries, concomitant meniscal tears, cartilage injuries, and collateral ligament injuries that would alter the operative procedure or postoperative rehabilitation | • ACLR: 30 • ACL repair: 34 |
• ACLR: 80% proximal, 20% midsubstance • ACL repair: 91.2% proximal, 8.8% midsubstance |
• ACLR: HS • ACL repair: DIS |
5 |
| Kayaalp(2022) 25 | 3 | Acute (<4 weeks) ACL tears | Concomitant ligamentous or meniscal injuries necessitating repair | • ACLR: 30 • ACL repair: 15 |
• ACLR: - • ACL repair: proximal or midsubstance |
• ACLR: - • ACL repair: DIS |
2 |
Dashes indicate data not reported or not applicable. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; Allo, allograft; BEAR, bridge-enhanced ACL repair; BTB, bone-tendon-bone; DIS, dynamic intraligamentary stabilization; HS, hamstring; IB, internal brace; LOE, level of evidence; MCL, medial collateral ligament; MLKI, multiligament knee injury; MRI, magnetic resonance imaging; OA, osteoarthritis; PLC, posterolateral corner; QT, quadriceps tendon.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.P. has received education payments from Smith+Nephew and hospitality payments from Medical Device Business Services. B.U.N. has received grant support from Arthrex, education payments from Arthrex and Smith+Nephew, consulting fees from Stryker, and hospitality payments from Medical Device Business Services and Zimmer Biomet. A.S.R. has received education payments from Gotham Surgical; consulting fees from Anika Therapeutics, Bodycad, Smith+Nephew, Xiros, Stryker, Flexion Therapeutics, Arthrex, and Heron Therapeutics; and nonconsulting fees from Arthrex and Smith+Nephew. A.S.P. has received consulting fees from Smith+ Nephew, Zimmer Biomet, DePuy Synthes, Exactech, and Stryker; nonconsulting fees from Smith+Nephew; royalties from Smith+Nephew and Zimmer Biomet; and has stock/stock options in Smith+Nephew. B.T.K. has received education payments from Arthrex; consulting fees from Arthrex; nonconsulting fees from Arthrex and Synthes; royalties from Arthrex; and hospitality payments from Stryker and Smith+Nephew. R.J.W. has received royalty or license from Arthrex; consulting fees and nonconsulting fees from Arthrex; and has stock/stock options in Smith+Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Final revision submitted November 15, 2023; accepted November 21, 2023.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

