Abstract
Background:
Recently, all-suture, all-inside meniscal repair devices—including devices containing flat sutures or tapes—have been introduced. Similar to those in suture anchors, these modifications may have different performance characteristics than conventional sutures and polyether ether ketone (PEEK)-anchored devices.
Purpose:
To compare the biomechanical characteristics of all-suture meniscal repair devices with those of a conventional PEEK-anchored device and an inside-out meniscal suture construct.
Study Design:
Controlled laboratory study.
Methods:
A total of 48 adult porcine menisci with simulated bucket-handle tears were included. Single-device repairs were performed with the SuperBall Meniscal Repair System, FiberStitch, and FAST-FIX 360 with 2 PEEK anchors, and a vertical mattress inside-out suture repair was performed using a Ti-Cron No. 2-0 braided polyethylene terephthalate suture. All specimens were preloaded (10 N) and cycled 200 times (between 10 and 50 N). Specimens surviving cyclic loading were then destructively tested. Endpoints included maximum failure load, stiffness, cyclic displacement, and failure mode. The goal was 12 successful tests in each group. Metrics between groups were compared using analysis of variance with post hoc tests to control for multiple comparisons.
Results:
The SuperBall (108.9 N) was significantly stronger than the FAST-FIX 360 (67.3 N) and Ti-Cron (75.2 N), and the FiberStitch (102.8 N) was significantly stronger than the FAST-FIX 360 (P≤ .01 for all). Cyclic stiffness increased during cyclic loading for all constructs (P < .001). The Ti-Cron was significantly stiffer than the SuperBall during 5 to 200 cycles (P < .001). Cyclic displacement significantly increased in all constructs during cycling (P < .001) but did not differ between devices. Failure mode varied by device: the Ti-Cron repairs failed because of suture breakage, the SuperBall and FAST-FIX 360 failed at the anchor, and the FiberStitch showed both failure modes.
Conclusion:
The all-suture, all-inside meniscal repair devices demonstrated superior strength to the PEEK-anchored device and the classic inside-out suture meniscal repair but no statistically significant difference in cyclic displacement.
The benefits of meniscal repair are based on the potential for successful meniscal healing.10,13,16,18 The improvement in knee biomechanics 20 is clear, and the earlier a tear can be repaired, the better the potential for meniscal healing. 16 Many different devices are available, and the all-inside meniscal repair technique is widely accepted, with no difference in failure rate compared with other repair approaches. 23 This technique reduces some potential complications of the inside-out technique—including wound problems with an additional incision, nerve injury, and posterior medial or posterior lateral scarring.2,11,14,25 The optimum suture repair configuration, regardless of the device, is a vertical mattress stitch across the longitudinal collagen fibers of the meniscus.5,7 This configuration secures the tissue better than a horizontal mattress stitch configuration. All-inside meniscal repair devices are typically based on sutures containing ultra-high molecular weight polyethylene (UHMWPE) attached to anchors of various materials secured to the capsule peripheral to the meniscus.
More recently, all-suture anchors were substituted for the polyether ether ketone (PEEK), poly-
The features of all-suture anchor designs have been applied to meniscal repair devices. While initial problems with inconsistent deployment, displacement during cyclic loading, and suboptimal failure loads were observed, more recent suture anchor iterations are notably better in clinical practice. 26 The trend of transitioning toward flat sutures in shoulder surgery has extended into the knee. Coreless FiberWire (Arthrex) is used in a meniscal repair device—the FiberStitch (Arthrex). A flat polyester mesh is included in the SuperBall Meniscal Repair System (Arcuro Medical). Considering these innovations and the fact that many existing devices have undergone significant updates along with the introduction of additional all-inside devices, revisiting the biomechanical behavior of these repairs seems warranted.
This study aimed to compare the biomechanical characteristics of all-suture meniscal repair devices with conventional anchored devices and inside-out meniscal sutures. The hypothesis was that different all-inside devices would not differ in structural properties and that while different in design philosophy and construction, these all-suture devices would have similar biomechanical properties under cyclic loading and destructive testing.
Methods
For this study, 60 skeletally mature (24 months old) Yorkshire/Berkshire crossbreed porcine knees (Advanced Tissue Concepts) and 48 intact and undamaged fresh-frozen medial menisci were harvested. Damaged, surplus, or otherwise unsuitable menisci were not used. A porcine meniscal model was used because it has been shown to demonstrate results comparable to those of a young adult human meniscus.15,22 Institutional review board approval was received for this study.
After being thawed to room temperature, the front leg was carefully dissected to expose the intact menisci on the tibia. Once exposed, a roughly 2 cm vertical longitudinal cut paralleling the periphery of each medial meniscus was made with a No. 15 scalpel, consistent with a previous study. 8 This cut, created while the meniscus was still attached to the tibia for ease of control, was not extended into the anterior and posterior meniscal horns until after the repair device was inserted to facilitate device insertion.
For each tissue sample, 1 of 4 repair devices was selected using a block randomization method. The chosen meniscal repair device was placed in each meniscus across the midpoint of the cut using a vertical stitch configuration to approximate the meniscal segments. The devices were inserted by 2 board-certified, fellowship-trained orthopaedic surgeons who were very experienced in meniscal repair (F.A.B. and P.A.D.). The vertical orientation of an all-inside device has less displacement, greater stiffness, and greater strength after cyclic loading than horizontal orientations. 19 In addition, it is well established that vertical suture orientation provides greater repair strength than horizontal repair sutures by more effectively capturing the circumferentially oriented collagen bundles.5,7
After inserting and tensioning (as felt by the surgeon) the repair device in accordance with the respective instructions for use, the remaining tissue bridges in the anterior and posterior horns were divided, creating 2 independent segments attached only by the repair device. Afterward, the meniscus was completely detached from the tibia. Only medial menisci were used for the tests to provide consistency in the size and location of repair placement, and only devices successfully deployed and tensioned were biomechanically evaluated.
Devices Tested
Four constructs were tested. Three single-device repairs were performed with the SuperBall Meniscal Repair System (n = 14; Arcuro Medical), FiberStitch (n = 16; Arthrex), and FAST-FIX 360 (n = 17; Smith+Nephew). The fourth construct involved a vertical mattress inside-out suture repair using a Ti-Cron No. 2-0 braided polyethylene terephthalate suture (n = 12; Covidien Medtronic).
SuperBall Meniscal Repair System
This device comprises 2 UHMWPE suture bundles for the anchors and locking suture. The fixation device contacting the meniscus is a flat, woven, coreless polyester mesh. The repair was placed using the low-profile crescent needle insertion device in a vertical mattress configuration spanning the meniscal cut, starting on the superior surface of the meniscus 3 mm inside the meniscal cut and in the middle of the superior surface of the peripheral meniscal segment to complete the repair. After mechanical tensioning of the repair construct, the device was locked by cinching a locking suture cluster “ball” on the peripheral capsule, leaving no knot in the joint space (Figure 1).

(A) The SuperBall meniscal repair system is composed of UHMWPE all-suture bundles (anchors) with a repair stitch created using a woven polyester mesh that lies flat on the meniscal surface (white arrow). A green looped tensioning suture (right side of the photograph) in combination with the single green actuating suture (left side of the photograph) creates the SuperBall construct. (B) A mechanical tensioning process built into the inserter reduces the meniscal tear with the repair suture (white arrow) and locks the tensioning with a “ball” on the peripheral capsule of the meniscus (black arrow), leaving no knot in the joint space. UHMWPE, ultra-high molecular weight polyethylene. (©F. Alan Barber, MD).
FiberStitch
This device has a coreless No. 2-0 FiberWire suture (Arthrex) containing a pretied, sliding locking knot attached to 2 braided polyester sleeves, which become anchors when tensioned (Figure 2). Once deployed and tensioned, the tensioning suture is cut flush to the meniscal surface, leaving 2 strands of the suture lying across the surface of the meniscus attached to 2 all-suture “balls,” which are the anchors. These anchors are created by bunched sleeves covering the underlying suture. The repair was placed using a curved needle insertion device in a vertical mattress configuration spanning the meniscal cut, starting on the superior surface of the meniscus 3 mm inside the meniscal cut and in the middle of the superior surface of the peripheral meniscal segment to complete the repair.

The FiberStitch repair device has a coreless No. 2-0 FiberWire suture containing a pretied, sliding locking knot attached to 2 braided polyester sleeves. When tensioned, these sleeves become anchors (black arrow), leaving 2 sutures lying on the surface of the meniscus (white arrow). (©F. Alan Barber, MD).
FAST-FIX 360
This device has 2 arrow-shaped PEEK anchors (the first is approximately 5 × 1 mm, and the second is approximately 5 × 1.5 × 0.7 mm) connected by a No. 2-0 braided UHMWPE suture with a pretied sliding locking knot 9 (Figure 3). The repair was placed using the curved needle insertion device in a vertical mattress configuration spanning the meniscal cut, starting on the superior surface of the meniscus 3 mm inside the meniscal cut and in the middle of the superior surface of the peripheral meniscal segment to complete the repair.

(A) The FAST-FIX 360 device was placed using a curved needle insertion device in a vertical mattress configuration on the superior surface of the meniscus. This device has 2 arrow-shaped PEEK anchors (black arrow) connected by a No. 2-0 braided UHMWPE suture with a sliding locking knot, which leaves the suture lying on the surface of the meniscus (white arrow). (B) A deployed but not inserted anchor demonstrates the features of the device. PEEK, polyether ether ketone; UHMWPE, ultrahigh molecular weight polyethylene. (©F. Alan Barber, MD).
Ti-Cron Suture
A single vertical suture of No. 2-0 Ti-Cron (nonabsorbable braided polyethylene terephthalate coated with silicone) was used. Both arms of this suture were swaged onto their own meniscal repair needle. The first suture arm was inserted 3 mm inside the meniscal cut into the bucket-handle portion, extending from the superior surface across the cut and into the inferior portion of the peripheral meniscal segment. The second suture needle was inserted into the superior surface of the peripheral meniscal segment to exit the periphery of the meniscus. These 2 strands were hand tied on the peripheral meniscus capsule using 6 alternating half hitches, leaving a single suture on the meniscal surface, simulating an inside-out repair (Figure 4).

A single vertical suture of No. 2-0 Ti-Cron (nonabsorbable braided polyethylene terephthalate coated with silicone) was used to repair the meniscal cut and hand tied on the capsular side of the meniscus to approximate the 2 meniscal fragments (©F. Alan Barber, MD).
Testing Protocol
To be consistent with previous studies,6,8,9 the target sample size of this test protocol was 12 successful tests of each repair device studied. A surplus number of devices was obtained from the manufacturers in case of technical failures. Testing was considered complete once 12 specimens of each device had successfully been created or all the available devices had been used.
For testing, the prepared repair constructs were mounted on a servohydraulic test system (Instron model 1331 load frame with model 8800 controller) equipped with a 1-kN load cell. Both portions of the repaired meniscus were securely held by two 5.25-inch (13.34-cm) towel forceps that, in turn, were attached to fixtures specially designed and created for this test configuration6,8 (Figure 5). The meniscus was oriented with the bucket-handle portion in the inferior position and the peripheral rim portion in the superior position. This clamping configuration allowed the consistent application of force on the repair device by evenly distributing the load between all 4 clamps.

(A) Testing was conducted on a mechanical testing machine, with each meniscal segment securely held by (B) 2 towel forceps attached to apply a distractive force to the repair device. (©F. Alan Barber, MD).
An initial 10-N preload was applied for 60 seconds along the axis of the repair (transverse to the fiber orientation and arc of the meniscus) to pretension the repairs to a consistent reference load. Next, cyclic loading between 10 and 50 N at 1 Hz for 200 cycles was applied using a triangle waveform. After cyclic testing, those surviving specimens were subjected again to a 10-N preload, and destructive testing was performed6,8 at a rate of 5 mm/min. Actuator displacement and force were recorded at 1 kHz throughout testing. The tissue was kept moist with a manual saline spray until the testing concluded.
Specimens were analyzed for cyclic and ultimate failure properties, and those that failed during cyclic testing were documented according to the cycle and load at which they failed. Cyclic displacement (mm) between the start and end of testing was recorded as cyclic stiffness (N/mm), which was defined as the maximum tangent slope on the linear region of the force-displacement curve at cycles of interest (Figure 6). In contrast to overall displacement, the ultimate failure displacement occurred when the construct could no longer support higher loading forces and showed a distinctive drop in the loading curve with increasing displacement. The ultimate failure displacement for specimens that survived 200 cyclic loads was recorded.

Schematic of cyclic stiffness (N/mm), defined as the maximum tangent slope on the linear region of the force-displacement curve.
The failure mode was determined using visual inspection and a consensus of all investigators after removal from the testing apparatus. The first repair anchor inserted was referred to as “anchor 1” and the second as “anchor 2.” The endpoints for this test were as follows:
Maximum failure load after 200 cycles.
Cyclic displacement: the difference between the mean trough value for the last 10 cycles and the mean trough value for the first 10 cycles; displacement between the initial 10 cycles and 100 cycles; and displacement between 100 cycles and final 10 cycles (200 cycles).
Mode of failure: anchor failure, suture breaking, or tissue failure.
Statistical Analysis
Descriptive statistics were calculated. Outcome variables were tested between groups using a 1-way ANOVA analysis of variance with the Tukey honestly significant difference correction for multiple comparisons. For comparisons within groups, paired t tests were used to compare metrics at a given cycle (ie, cycle 1) to the initial conditions to determine whether the relative change reached statistical significance. Statistical significance was placed at P < .05. All data were normally distributed, as verified by the Shapiro-Wilk test. Statistical software used was SPSS (v20, IBM, Armonk, NY, USA).
Results
The mode and timing of device failure influenced the ultimate failure. Some devices failed during cyclic loading after successfully reaching a maximum load of 50 N during the initial cycle, while others survived 200 cycles loaded to 50 N before the destructive testing was initiated. Thus, failure status was considered in 2 subgroups (those completing all 200 cycles and those failing during cycling) (Table 1). The first analysis includes the ultimate failure force of only those tests successfully completing 200 cycles. The second analysis reports the ultimate failure forces from all tests, including those specimens that did not successfully complete all 200 cycles and failed during cyclic testing.
Maximum Failure Load and Implant Testing Survival Rates a

Data are reported as mean ± SD unless otherwise indicated.
Significant differences, SuperBall versus FAST-FIX 360 and SuperBall versus Ti-Cron (P≤ .039).
Significant differences, FiberStitch versus FAST-FIX 360 and FiberStitch versus Ti-Cron (P≤ .001).
Significant differences, SuperBall versus FAST-FIX 360 and SuperBall versus Ti-Cron (P≤ .024).
Significant differences, FiberStitch versus FAST-FIX 360 and FiberStitch versus Ti-Cron (P≤ .001).
Significant differences, SuperBall versus FAST-FIX 360 and SuperBall versus Ti-Cron (P≤ .01).
Significant differences, FiberStitch versus FAST-FIX 360 (P≤ .01).
Considering all tests performed, not just those completing 200 cycles, the mean ultimate failure forces were highest for the SuperBall and FiberStitch constructs (Table 1). Notably, the repairs in the FiberStitch group showed a 2 to 3 times higher variance and larger range in failure loads than any other device. There was no significant difference in ultimate failure force between the SuperBall and FiberStitch constructs (P = .939). While a significantly higher ultimate failure force was seen in the SuperBall group than in the FAST-FIX 360 and Ti-Cron constructs, the ultimate failure force in the FiberStitch construct was significantly higher than in the FAST-FIX 360 construct (Table 1).
All constructs demonstrated statistically significant increases in cyclic stiffness between cycle 1 and cycle 200 (P < .001). The beginning of a plateau in stiffness was observed after 150 cycles (Figure 7). The only significant difference was with the Ti-Cron group, which was higher than the SuperBall group for cycles 5 to 200 (P < .001) (Table 2).
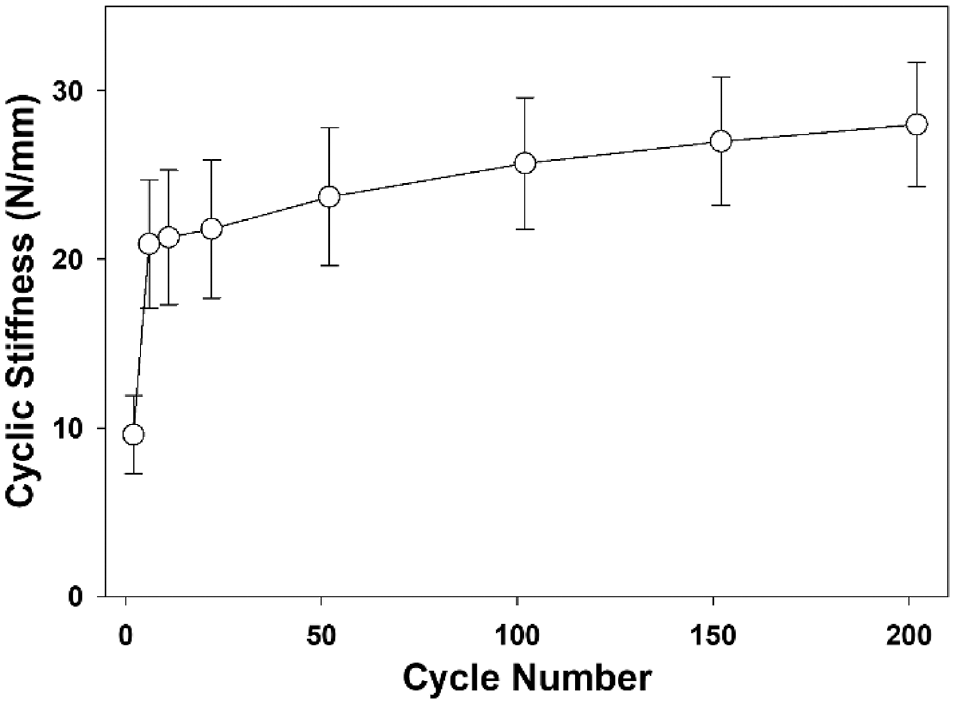
Cyclic stiffness (N/mm) as a function of cycle number. Note the initiation of a plateau after approximately 150 cycles. For clarity, only the data for the FiberStitch construct is shown. Error bars represent standard deviations.
Cyclic Stiffness a

Data are reported as mean ± SD.
Significant differences, SuperBall versus Ti-Cron for cycles 5 to 200 (P < .001).
Considering only specimens that completed both cyclic and ultimate failure tests, there were no differences in displacement between the SuperBall and FiberStitch groups, but both groups had greater displacement compared with the FAST-FIX 360 and Ti-Cron groups. All constructs demonstrated statistically significant increases in cyclic displacement between cycle 1 and cycle 200 (P < .001) (Table 3). No significant differences were detected between the groups at any cycle (P = .134).
Construct Displacement Versus Initial Length After Preloading a
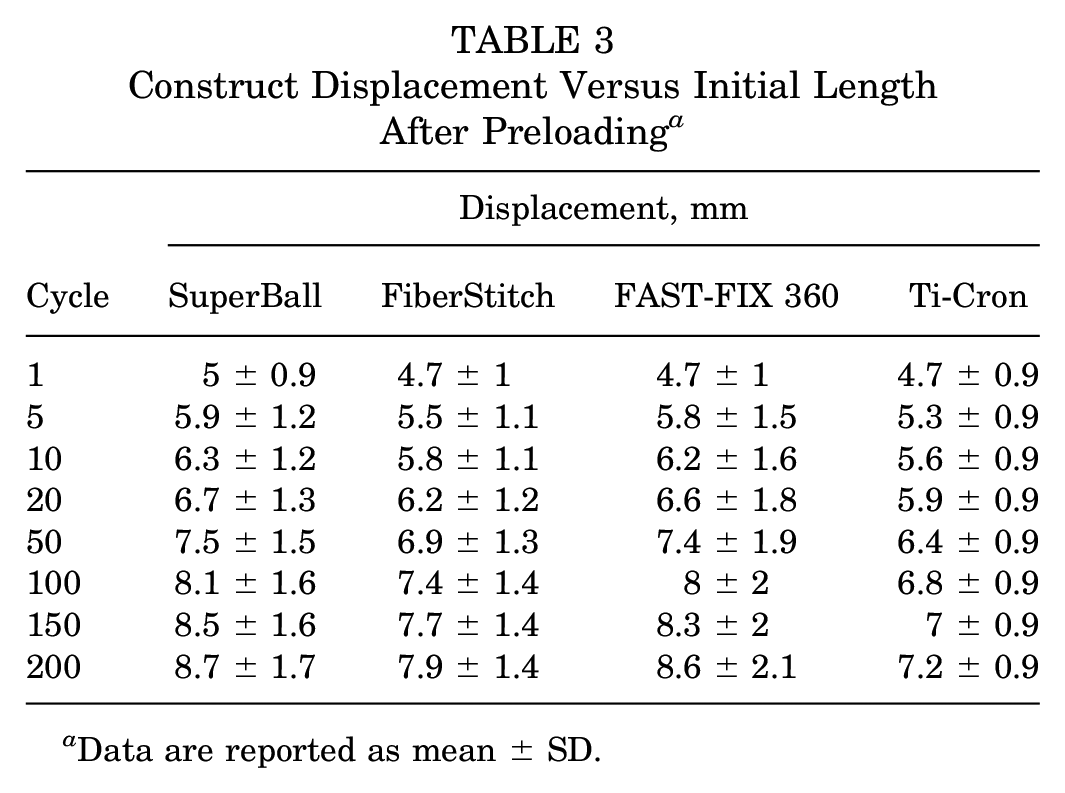
Data are reported as mean ± SD.
The increase in displacement between the mean of the initial 10 cycles and the 100th cycle was significantly greater than the displacement increase in the final 100 cycles for all devices tested (P < .001) (Table 4).
Comparison of Construct Displacement at 10, 100, and 200 Cycles a

The greatest increase occurred during the initial 100 cycles (P < .001).
The first 12 of 14 SuperBall devices were successfully placed, leaving 2 unused. There were 16 FiberStitch devices available for testing. After the insertion of all 16 devices, only 10 samples were available for failure testing. Of the 17 FAST-FIX 360 devices available, all were required to create the 12 constructs used for failure testing. The first 12 Ti-Cron constructs were successfully inserted and available for failure testing.
None of the SuperBall or Ti-Cron constructs failed during the 200 cycles, whereas 3 FiberStitch and 6 FAST-FIX 360 constructs did not complete the cyclic testing regimen (Table 5). These specimens failed after as few as 1 cycle up to as many as 165 cycles. Of those constructs that completed both cyclic loading and failure testing, the SuperBall construct primarily failed at anchor 1, the FiberStitch failed primarily via suture breakage, the FAST-FIX 360 failed primarily at anchor 2, and the Ti-Cron always failed with suture breakage.
Modes of Failure a
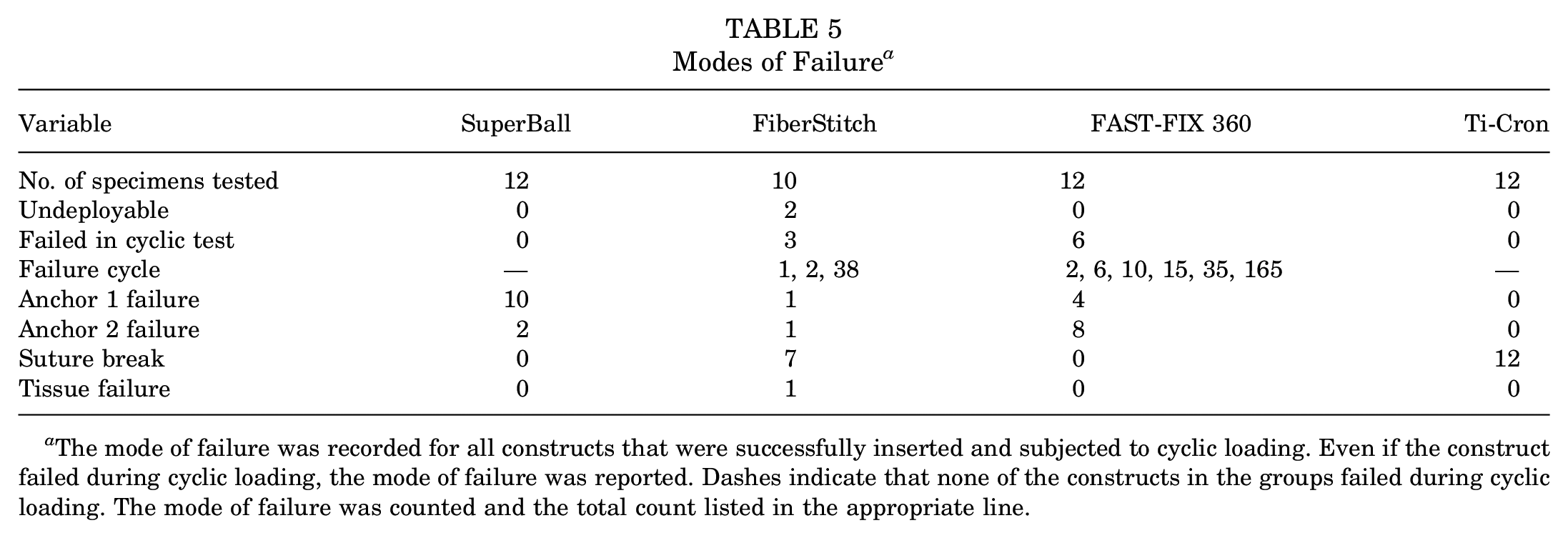
The mode of failure was recorded for all constructs that were successfully inserted and subjected to cyclic loading. Even if the construct failed during cyclic loading, the mode of failure was reported. Dashes indicate that none of the constructs in the groups failed during cyclic loading. The mode of failure was counted and the total count listed in the appropriate line.
Discussion
The 2 devices with the highest loads during destructive testing after cyclic loading—the SuperBall (108.9 N) and FiberStitch (102.8 N)—were stronger than the FAST-FIX 360 (67.3 N) and a classic inside-out meniscal repair using a No. 2-0 Ti-Cron suture (75.2 N) (P≤ .01). All constructs demonstrated statistically significant increases in stiffness over the course of the 200 cycles (P < .001). This stiffness started to plateau after 150 cycles. The suture repair showed greater stiffness (24.1 N/mm increasing to 31.1 N/mm) than the SuperBall (18.8 N/mm increasing to 25.2 N/mm) during the cyclic loading (P < .001). Similarly, all constructs demonstrated statistically significant increases in displacement during cyclic loading over 200 cycles (P < .001). As with previous studies,4,6 displacement was significantly greater during the initial 100 cycles than the final 100 cycles for all devices tested (P < .001). The cyclic loading phase did not demonstrate any statistically significant differences in displacement among the 4 groups tested. Ultimate failure modes varied by construct, with the principal modes being anchor pullout (SuperBall and FAST-FIX 360) and suture breakage (FiberStitch and Ti-Cron). The hypothesis that these suture-based devices would not differ in any biomechanical property under cyclic and failure loading from a standard inside-out suture repair was not confirmed. These all-inside repair devices demonstrated some improved biomechanical characteristics.
While load to failure is a key datum and often the principal focus of biomechanical testing, the mode of failure may be clinically more significant as it relates to meniscal repair. A device that fails after deployment by pulling an anchor into the joint can result in articular cartilage damage, which may have worse long-term implications than the failure of a meniscal repair to heal. A loose body composed of a piece of plastic or a ball of knotted suture can do irreparable harm to a joint in a relatively short time.
Reviewing the ultimate failure load ranges for those tests surviving 200 cycles, the FiberStitch construct had by far the widest range (98 N [73-171 N]). This contrasts with the much smaller failure load ranges for SuperBall (32 N [93-125 N]), FAST-FIX 360 (34 N [68-102 N]), and the suture repair (20 N [69-89 N]). A wide range of 3 times as great as the other constructs tested suggests considerable inconsistency in device performance.
All-suture, all-inside meniscal repair devices were developed to take advantage of the all-suture technology used in shoulder suture anchors. Anchoring an all-suture anchor into bone is different than placing 1 in meniscal material, and some performance differences are to be expected. The initial all-suture meniscal repair device was the MaxFire (Biomet Sports Medicine) made of UHMWPE suture covered by 2 polyethylene sleeves, which bunched up to become anchors after tensioning compressed them against the tissue peripheral to the meniscus. 8 During a cyclic loading test, the device failed because of the suture breaking in 3 specimens and the anchors pulling out of the meniscal tissue in 9 specimens. 8 This contrasts sharply with the device's mode of failure with an uncycled single static pull test (11 suture breaks and only 2 anchor pullouts) and suggests that it is important that the mode of tensile and failure testing be clinically relevant.
The MaxFire was tested in human meniscal specimens using a cyclic loading/destructive testing protocol similar to that used in the present study. 6 It demonstrated the poorest performance of all devices tested by cyclic loading, surviving a mean of only 37 out of 200 cycles. Those data created concerns about applying all-suture anchoring technology to meniscal repair. This latest generation of all-suture, all-inside meniscal repair devices appears to have addressed these concerns.
The FiberStitch was compared with other devices by Bachmaier et al. 1 Notably, their protocol used cyclic loading in a range of 2 to 20 N in human cadaveric menisci. The displacement data reported in that study is not comparable with that in the present report since the upper-level stress during cyclic loading was less than half of the 50-N load used in the present study. In the present study, the FiberStitch device was pulled from the meniscus at 38 N and twice at 50 N. These pullout failures would not have been detected in the previous study with the lower cyclic load. The principal failure mode reported by Bachmaier et al 1 was suture breaking, consistent with the present study. Bachmaier et al also reported that the ultimate failure load of the FiberStitch was significantly greater than that of the FAST-FIX 360, which is also consistent with the findings of the present study.
Other all-suture, all-inside meniscal repair devices are currently available but were not included in this test for logistical reasons—including the limited number of biologic specimens available, funding restraints, and laboratory limitations. These devices include the NovoStitch PRO meniscal repair system (Smith+Nephew) and JuggerStitch and Cross Fix 2 meniscal devices (Zimmer Biomet). The all-inside meniscal repair device is a repurposed shoulder suture anchor modified for meniscal repair.
A comparison of similarly tested single-loaded all-suture anchors in porcine bone subjected to cyclic loading followed by destructive testing demonstrated that the Q-Fix (Smith+Nephew) was stronger (319 N), showed less displacement after 100 cycles (1.03 mm) and at 200 cycles (1.22 mm), and failed by suture breaking compared with other single loaded all-suture anchors. 4 The unique design of the all-suture Q-Fix device is a radially expanding anchor created by an internal tensioning mechanism that does not rely on the surgeon placing traction by hand on the suture to create the distinctive ball, which serves as an anchor. This mechanism is in sharp contrast to other all-suture anchor constructs, made by surgeon-applied suture traction, which bunches the overlying sleeve to create an anchoring mass of the suture. This mechanical approach to anchor creation may explain the superior performance of the “ball” anchor. While not having identical mechanisms, it is notable that the SuperBall device also has a mechanically created anchor, which results in a robust anchor ball on the periphery of the meniscus (Figure 1B). While a direct correlation cannot be made between a suture anchor and a meniscal repair device, this may explain why the SuperBall was consistently successful during deployment and cyclic loading compared with the FiberStitch and FAST-FIX 360.
The biomechanical endpoints measured in this study were failure load (strength), stiffness, displacement, and failure mode. This bench test provides data that may be compared with data in previous reports. While no ideal level of meniscal repair stiffness has been established and no evidence exists that stiffness affects clinical healing, greater strength logically must be considered. Greater strength is why braided polyester suture has been replaced by UHMWPE suture. The SuperBall and FiberStitch constructs demonstrated greater strength, but that does not mean that better healing will occur when they are used in a meniscal repair. Insertion of the second anchor was problematic for both the FiberStitch and FAST-FIX 360 constructs. Three FiberStitch failures occurred when the second anchor would not deploy if even a slight flex in the insertion needle developed while passing through the bucket-handle tissue. Three additional failures occurred when the deployed second anchor pulled out during the stepwise tensioning of the individual loops to achieve final repair tensioning. Consequently, only 10 samples of the fully deployed FiberStitch were available for testing. The 5 unsuccessful FAST-FIX 360 insertions occurred when the second anchor failed to remain in the meniscal tissue during tensioning. This may be due to the larger inserter needle size. Construct strength is important when tightening the repair, and surgeons can undoubtedly relate to the frustration of placing a stitch perfectly only to have the suture break when it is tensioned. Also, the mode of failure can have implications. A meniscal suture that breaks and separates the bucket handle from the periphery is frustrating; nonetheless, a plastic anchor or suture ball migrating into the joint can result in catastrophic articular cartilage damage.3,12,17 Knowing the most likely mode of failure for any particular device is important.
Displacement should also be considered. While statistically significant increases in displacement occurred with all 4 devices over 200 cycles (reaching nearly 4 mm), there were no statistically significant differences in displacement among the 4 groups tested. Moreover, the type of displacement commonly tested may not reflect the clinical condition. Displacement (or “gapping”) may be more relevant for meniscal repair than ultimate failure load. Published studies24,25 have reported that a meniscal tear is compressed rather than distracted during knee flexion and extension. Distraction forces were not reported during knee cycling. A sheer stress is the most likely force at the meniscal repair site. The current testing method cannot be assumed to mirror the behavior of longitudinal meniscal segments in the postoperative setting. This methodology certainly does not mirror the behavior of a radial or complex meniscal repair.
Finally, meniscal repair success can occur with incomplete healing. In 1991, Morgan et al 21 reported that during second-look arthroscopy, 19% of meniscal repairs demonstrated a stable but only partially healed meniscus measuring up to 50% of the original repair length or depth with no additional meniscal tearing. This suggests that partial healing may be tolerated clinically.
Limitations
This study has several limitations. This was a time-zero, nonarthroscopic bench test, performed at room temperature, in a nonaqueous environment, and using porcine tissue. Porcine tissue, while similar to human tissue, may not respond in the same manner as the clinically repairable human meniscus. The observed results cannot be assumed to apply to clinical cases in human patients. This test used a cyclic distractive force and not a compressive, axial, rotational, or shear stress, which is more likely to replicate the in vivo mechanism of meniscal repair failure. These data cover only some aspects of repair device performance, and no correlation exists with clinical healing. The number of samples tested, while consistent with similar tests, is limited. Only 4 different repair devices were tested, and these data cannot be considered representative of other devices on the market that were not tested. No a priori power analysis was performed since benchmark data for these respective comparisons were not available. Only 1 repair stitch was used for the test. The repair characteristics for a specific device could be impacted if multiple stitches were used.
Conclusion
The present study's findings indicated that all-suture meniscal repair devices had higher ultimate failure loads than a conventional anchored meniscal repair device or an inside-out meniscal suture; nonetheless, the cyclic properties were similar for all devices tested.
Footnotes
Final revision submitted September 25, 2023; accepted October 11, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: Support for this study was provided by Arcuro Medical, Musgrave Israel, and DePuy Mitek. F.A.B. has received grant support from Arcuro Medical and nonfinancial support from Arcuro Medical and DePuy Mitek. P.A.D. has received grant support and nonfinancial support from Arcuro Medical and has an equity position in Arcuro Medical. H.B.H. has received grant support from Arcuro Medical, Stryker, DePuy Synthes, and Wright Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Utah (ref No. 11755).
