Abstract
Background:
Meniscal repair is the gold standard for simple morphology tears. However, when the morphology and chronicity of the tear are less favorable, the success of the standard techniques is reduced.
Purpose/Hypothesis:
To compare meniscal repair augmented by a new bioresorbable implant (Meniscus Cap) versus a traditional simple suture technique and the currently available augmented repair collagen matrix meniscus wrapping technique. It was hypothesized that the Meniscus Cap suture technique would increase ultimate failure load and less displacement during cyclic loading.
Study Design:
Controlled laboratory study.
Methods:
A total of 80 fresh porcine menisci were harvested. Complex tears were created in 60 menisci, and 20 intact menisci were tested as the control group. Repairs were performed on the 60 meniscal tears using 1 of the 3 techniques (20 menisci each): an inside-out H-suture group (SS), the collagen matrix wrapping technique (CMW), and the Meniscus Cap bioresorbable implant group (CM). The menisci were subjected to 500 loading cycles from 4 to 20 N at a frequency of 1 Hz, and the total displacement was recorded. Then, the specimens underwent load to failure testing at a rate of 3.15 mm/s, and the failure mode was noted.
Results:
After 500 cycles of cyclic loading, there were no significant differences in displacement between the controls and CM group (0.524 vs 0.448 mm; P = .95). The displacement after the CM was significantly smaller compared with the CMW and the SS (0.448 vs 1.077 mm [P = .0009] and 0.448 vs 0.848 mm [P = .04], respectively). The ultimate load to failure was significantly greater for the controls and the CM group compared with the SS and CMW groups (controls, 1278.7 N and CM, 628.5 N vs CMW, 380.1 N and SS, 345.1 N; P < .05). The failure mode was suture breakage (suture failure) for all repairs.
Conclusion:
In a porcine specimen meniscal repair model, the biomechanical properties of a novel Meniscus Cap repair technique were superior to that of the simple suture and CMW techniques.
Clinical Relevance:
The results suggest that the Meniscal Cap repair technique may provide sufficient primary stability of the meniscal fixation even in the cases of complex meniscal tears.
Meniscus surgery is one of the most commonly performed orthopaedic procedures worldwide. In the United States, nearly 850,000 meniscus surgeries are performed annually, comprising 10% to 20% of the entirety of orthopaedic surgical procedures each year. 1 A simple suturing of the meniscus utilizing various devices and materials is the best-known surgical technique for meniscal repair, but its indications are limited.20,23 Among the key factors influencing the healing of the meniscus after repair are the mechanical properties of the device or the suturing technique.20,23 Biomechanical testing was performed for various meniscus sutures and device repair techniques to determine the mechanical strength and stiffness of the repaired area.2,8,9,11-13,15,28 Similar testing was performed for artificial meniscus scaffolds such as the Collagen Meniscus implant, marketed in Europe by Ivy Sports Medicine (also known as Menaflex) or Actifit polymer (polyurethane) meniscus implant (Orteq).14,17
Because of the known benefits of meniscal repair, there is a growing tendency to expand the indications for meniscal repair utilizing sutures.5,6,19,21,22,24 Such procedures commonly include biological factors to boost the regeneration and healing of the tissue—for example, fibrin clots or bone marrow products. In the past 20 years, various strategies have been developed for biologically supporting the meniscus healing, sometimes with augments.3,16 An example of such a strategy is the suturing method of Jacobi and Jakob, 18 which involves wrapping the meniscal repair site with a collagen membrane. While initial results were satisfactory, the technique proved difficult to recreate by other surgeons. In 2010, this technique was modified by Piontek et al 26 into an arthroscopic procedure, placing a suture on the damaged meniscus and wrapping it with the collagen membrane, with bone marrow aspirate being injected between the collagen membrane and the meniscus. Follow-up results after 2 and 5 years demonstrated that the arthroscopic matrix meniscal repair (AMMR) technique is safe and that such augmented repair techniques may expand the indications for the repair of previously nonsalvageable menisci. However, AMMR is technically demanding, and the collagen membrane only provides limited early postoperative stabilization of the meniscal tear area.10.29
Despite efforts to encourage meniscal repair, there is still a difference of opinion between higher- and lower-volume surgeons on which meniscal tears are potentially repairable and need meniscectomy. 4 Perhaps more education is needed—as was the conclusion in the article by Bąkowski et al 4 —nonetheless, an alternative solution would be a simpler surgical technique for augmented repair. In our opinion, this approach requires a new implant to facilitate simple arthroscopic introduction over the damaged meniscus. The implant forms a scaffold for cells to regenerate the injured meniscus, potentially leading to a full recovery because of the restoration of meniscal function. The implant mechanically stabilizes the fragments of the injured meniscus immediately after the surgical procedure to physically support the healing processes and facilitate early rehabilitation. The Meniscus Cap (Meniscus Cap; Sp. Z o.o.) is a new collagen-polycaprolactone meniscus covering comprising a set of 2 wings in the shape of the lateral or medial meniscus. Each plate is a composite structure of collagen membrane and a bracing polymer (polycaprolactone) skeleton, connected by a flexible hinge along the inner curvature of the implant. The polycaprolactone skeleton incorporated into the collagen membrane contributes to the stability of the repaired construct to a far greater extent than just the collagen membrane. Theoretically, forces generated in the meniscal repair area may be partially transferred to the intact edges of the meniscus through the augmentation graft. This load shielding by the graft may directly protect the underlying repair from gap formation. The morphological structure of the collagen membrane, together with the polycaprolactone skeleton fibres, is shown in Figure 1.
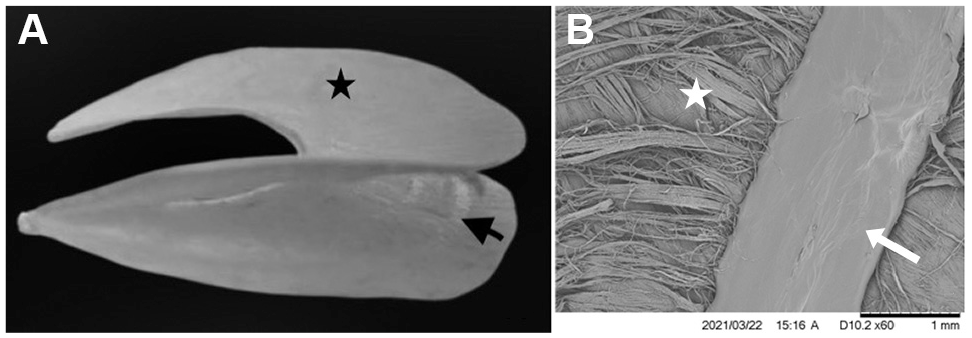
(A) A macroscopic view of the Meniscus Cap implant comprising a set of 2 wings in the shape of the meniscus, each in the form of a composite layer of collagen membrane (black star) and bracing polymer (polycaprolactone) skeleton (black arrow) connected by a flexible hinge along the inner curvature of the implant. (B) A scanning electron microscopy image showing the polycaprolactone fiber diameter of the Meniscus Cap scaffold (white arrow) and the collagen membrane (white star).
This study aimed to determine the biomechanical properties of the 3 meniscal repair techniques—simple suture, repair with collagen membrane, and repair with the novel Meniscus Cap implant—for a complex tear of the medial menisci compared with the intact meniscus (control group). In particular, we wanted to assess the biomechanical strength, stiffness, and ultimate mode of failure for each repair technique. It was hypothesized that the Meniscus Cap suture technique would provide increased ultimate failure load, increased stiffness, and less displacement during cyclic loading.
Methods
Overview of the Research Design
This was an in vitro biomechanical study on fresh human-sized porcine medial menisci aged 18 months old. The porcine knees were obtained from a local supplier. The trial was performed in accordance with all recommendations established by Good Laboratory Practice regulations. Ethical approval was not required for this study.
Specimen Preparation
A total of 80 specimens were harvested intact from young adult pigs by resecting the tissue at the meniscocapsular junction and with the 2 insertional roots. All the medial menisci were inspected and exhibited no macroscopic signs of meniscal tear or degeneration. The resected menisci were wrapped with normal saline-soaked gauze before testing.
Complex tears were then formed in 60 of the menisci with a No. 11 surgical blade in the midbody section equidistant from the anterior and posterior horns. The radial tears extended from the central margin to 1 mm from the peripheral meniscus rim. Next, the vertical tear was created perpendicular to the radial tear, extended for 10 mm, and crossed with the radial tear halfway. For reproducibility of the radial and vertical tears pattern, a template was prepared to define the position of the meniscal transection (Figure 2, A and B). Finally, the horizontal tear was created on both sides of the radial tear, and the depth of the horizontal tear was 5 mm. The tear was a defect in 3 planes, through the 3 vascular involvement zones1-3 of the meniscus, and was thus designated a complex tear (Figure 2C).
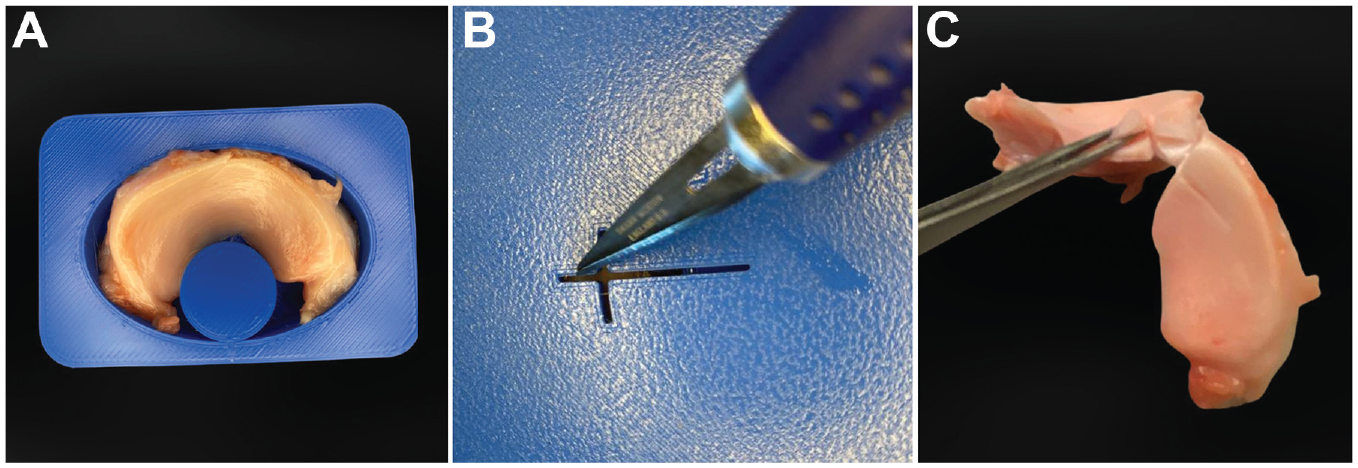
Template of a meniscal tear. (A) meniscal samples: a medial meniscus placed in the template. (B) The template of radial and vertical tears created with a No. 11 surgical blade. (C) A cross-section of the complex meniscal tear created in 3 planes across vascular involvement zones 1 to 3, indicating that the tear crossed the varying vasculature of the meniscus.
Randomization, Allocation, and Blinding
After preparation, the 80 meniscal specimens were randomly assigned to 4 equal groups of 20 with 1 control group and 3 groups of different meniscal repair techniques—the control group of intact porcine meniscus; the H-suture without scaffold group (SS); the suture with collagen matrix group (CWM); and the suture with Meniscus Cap group (CM). For all specimens, the sutures were tied manually in an open fashion (Figure 3).

Meniscal specimens. (A) The medial meniscus harvested intact from adult pigs without intervention (control group). (B) The medial meniscus repaired without scaffolding by the “H” suture technique. (C) The medial meniscus repaired with a collagen membrane with the same suture pattern. (D) The medial meniscus repaired with a Meniscus Cap scaffold with the same suture pattern.
SS Group
A rip-stop meniscal technique was employed using an “H-suture configuration” performed with 2-0 absorbable meniscal sutures (PDS No 2.0) (Figure 3B). The radial elements were performed first analogous to an inside-out technique in knee surgery, with vertical mattress suture configuration serving to reduce both vertical and horizontal components. These vertical radial sutures function as a rip-stop style reinforcement for the 2 horizontal mattress sutures that follow, performed as an inside-out technique, perpendicular to and over the top of the vertical mattress sutures at the radial tear area. One horizontal suture was placed on the femoral surface of the meniscus and another on the tibial surface.
CMW Group
The tear area was wrapped with a wet collagen matrix (Evolution; Osteobiol-Tecnoss) before suturing. The suture configuration was exactly as described for the SS group (Figure 3C).
CM Group
The tear area was covered with a wet Meniscus Cap before suturing. The suture configuration was exactly as described for the SS group (Figure 3D). The specimen to be tested was then mounted on a dedicated, custom-made test device that was connected to a material testing machine (Figure 4). Distracting loads were applied perpendicular to the radial meniscal tear to simulate a worst-case scenario of maximal traction across the tear/repair zone, which may be experienced at maximal axial loading in physiological conditions.

(A) Mechanical testing setup. (B) A repaired meniscus securely fastened to the dedicated, custom-made universal tissue clamps with a textured surface to prevent tissue slippage.
Assessments
Before cyclic testing, each repaired specimen was inspected to check for any suture slippage or tissue damage that may have been incurred during the repair and mounting process. Specimens with visible suture damage or unsecured knots were discarded; 3 of 60 were excluded.
Cyclic Load Testing
The intact and repaired menisci were securely fastened to the dedicated, custom-made universal tissue clamps with textured surfaces to prevent tissue slippage. The menisci were aligned perpendicular to the radial tear and subsequently mounted to a mechanical testing system (Insight 50 KN; MTS Systems) (Figure 4). Because of the wide measuring range of the standard Insight 50 KN testing machine header, the pilot tests were performed with the simultaneous use of a Hottinger Messtechnik sensor; its measuring range made it possible to measure the traction force on the meniscus with the required degree of accuracy and precision. During pilot testing, the specimens were tested and subject to the above-described roughening technique to confirm negligible slippage at the interface between the tissue and the clamps.
Primary Outcomes
The primary outcomes of this study were the amount of displacement during cyclic loading and ultimate failure load. After a preload of 2 N was applied to the specimen, cyclic loading from 4 to 20 N was performed at 1 Hz. Both the intact and repaired menisci are characterized by different rigidity; therefore, to obtain the deformation frequency at the level of 1 Hz, the displacement velocity of the testing machine traverse was set at 2 to 5 mm/s. The load and frequency were chosen based on previous studies and reflect in vivo postsurgical rehabilitation.2,8,9,11,12,14,15,17,28 Specimens underwent 500 submaximal loading cycles, and the MTS device was programmed to execute a 45-second pause at 500 cycles to facilitate data collection. Normal saline was applied to preserve the moisture of the specimens. The displacement value and corresponding load were recorded continuously in the software (Test Work 4.0; MTS Systems). Gap formation—the increased distance between the clamps—was measured and recorded as the mean distance across the tear at 20 N at 500 cycles. Cycle 0 was a reference point for reporting the displacement in the subsequent 500 cycles. After completion of cyclic loading, load to failure testing was performed at a rate 15 of 3.15 mm/s.
Secondary Outcomes
The mode of failure was documented after careful inspection. The 3 possible failure modes were tissue failure (suture pulled through the tissue), suture failure (breakage of the suture material), or knot failure (usually knot slippage).
Statistical Analysis
One-way analysis of variance, together with post hoc Tukey analysis, was performed to determine the mean differences in the load to failure across the 4 groups and between any 2 groups. A similar analysis was performed for the results of specimen stiffness measurement during the load cycles (500 cycles total) to determine the differences between specimen groups. Data were analyzed using Statistica software Version 13.3 (TIBCO Software Inc). All comparisons were 1-tailed tests, and P < .05 was considered statistically significant.
To perform the analysis of variance with the Shapiro-Wilk test, the hypothesis assuming a normal distribution of ultimate failure load test results was verified. The Shapiro-Wilk result for each examined group (n = 20 per group), with a P value > .95, proved the hypothesis that test results in each population followed a normal distribution. In the next step, the Brown-Forsythe test was used to test the equality of group variance. The P value was > .05, confirming the assumption that there was equality of result variance across groups. Successful confirmation of the initial hypotheses then allowed analysis of variance employing the post hoc Tukey test. The resulting variance between individual populations was statistically relevant, excluding the SS and CMW group pairing.
Analogous to the description provided earlier, the test results for extension after cyclic loading were analyzed in a similar fashion. In the Shapiro-Wilk test, the W parameter value had a P value of >.90 for populations = 20, whereas the Brown-Forsythe test had a P value > .29. Thus, the test results confirmed the hypotheses that the populations had a normal distribution and that group variance was uniform. In the next step, the post hoc Tukey test was performed.
Results
Primary Outcomes
Table 1 and Figure 5 show the mean ultimate failure load value for the given populations, with minimum and maximum values, and first, second, and third quartile values. The P values for each pairing of ultimate failure load in the compared populations are recorded in Table 2.
Ultimate Failure Load by Study Group a

Data are reported in N.CM, Meniscus Cap bioresorbable implant; CMW, collagen matrix wrapping technique; SS, inside-out H-suture.

A box-and-whisker plot of the ultimate failure load according to study group. The X and the horizontal line represent the mean and the median, the top and bottom of the box are the first and third quartiles, and error bars represent the range. CG, control group; CM, Meniscus Cap bioresorbable implant; CMW, collagen matrix wrapping technique; SS, inside-out H-suture.
P Values for Each Pairing of Ultimate Failure Load Between Study Groups a

Bold P values indicate a statistically significant difference between groups (P < .05). CM, Meniscus Cap bioresorbable implant; CMW, collagen matrix wrapping technique; SS, inside-out H-suture.
Table 3 and Figure 6 provide each study group’s mean displacement after cyclic loading. P values for each pairing of mean displacement values are provided in Table. 4. The differences in results between individual populations were statistically significant for the following pairings: Control versus CMW, CM versus CMW, and CM versus SS.
Displacement After Cyclic Loading by Study Group a

Data are reported in mm. CM, Meniscus Cap bioresorbable implant; CMW, collagen matrix wrapping technique; SS, inside-out H-suture.
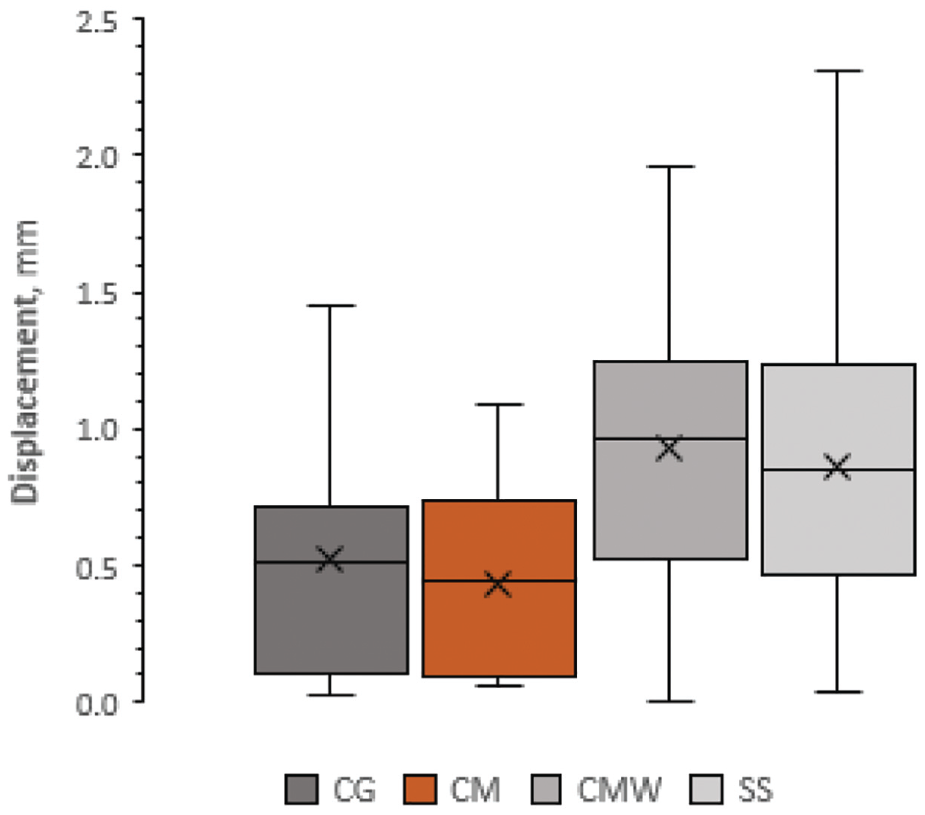
A box-and-whisker plot of displacement after cyclic loading. The X and the horizontal line represent the mean and the median, the top and bottom of the box are the first and third quartiles, and error bars represent the range. CG, control group; CMW, collagen matrix wrapping technique; CM, Meniscus Cap bioresorbable implant; SS, inside-out H-suture.
P Values for Each Pairing of Displacement After Cyclic Loading a
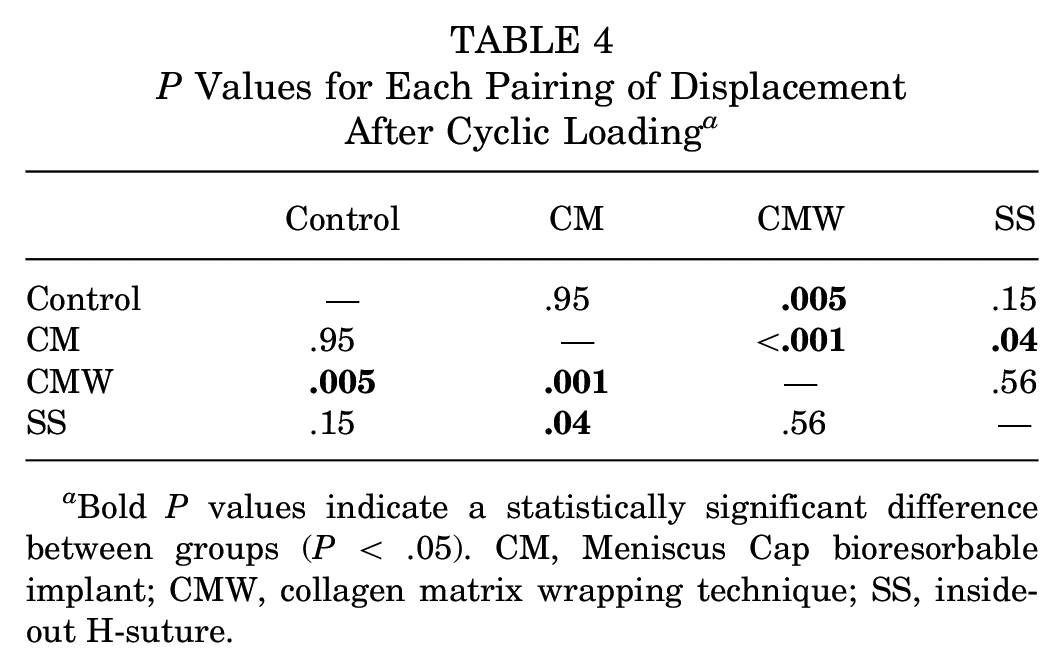
Bold P values indicate a statistically significant difference between groups (P < .05). CM, Meniscus Cap bioresorbable implant; CMW, collagen matrix wrapping technique; SS, inside-out H-suture.
Secondary Outcomes
In all repairs, the failure mode was suture breakage (suture failure).
Discussion
The present study is the first to report on the biomechanical behavior of menisci repaired with the Meniscus Cap implant compared with intact menisci and with menisci repaired with simple sutures or collagen matrix wraps. The most important finding was that after 500 cycles of cyclic loading, there were no significant differences in displacement between the intact meniscus and the Meniscus Cap repair method. We found that the mechanical characteristics of the complex meniscal tear repair can be significantly improved by using an additional Meniscus Cap scaffold. Consequently, the mechanical characteristics of the complex meniscal tear repaired and augmented with Meniscus Cap implant can be similar to the biomechanical properties of the intact porcine medial meniscus.
The primary stability of the repaired meniscus is the most important goal when aiming for a successful meniscal repair.7,20,23,26 To this end, numerous suture techniques have been developed and biomechanically tested. It was proven that sutures oblique to circumferential collagen fibrils showed better fixation than those parallel to circumferential fibrils. The current literature emphasizes the importance of suture orientation, the number of suture loops, and the location of fixation sutures or devices in the repair of meniscal lesions. 2,8,9,11-13,15,27,28,30 Buckley et al, 9 in a cadaveric comparison of 3 radial repair techniques, showed no particular benefit of 1 technique; nevertheless, the use of vertical mattress sutures—as a rip-stop device—significantly reduced the likelihood of the sutures pulling through the meniscal tissue during ultimate failure testing in any radial repair method. This is an important part of any radial repair technique but is particularly relevant for complex or 3 zone radial tears or where any meniscal adjuncts are being considered, perhaps in patients with suboptimal meniscal tissue. Furthermore, as opposed to previous studies of radial repair techniques, which reported failure strengths ranging from 62 N to 250 N,2,8,9,11-13,15,28 all 3 repair techniques tested in this study had failure strengths of >267 N. The results obtained for our SS study group (simple suture) samples are promising in the context of mechanical stabilization of complex (3-zone radial) meniscal tears.
The second possible way to enhance the mechanical properties of the suturing area is the augmentation of the treated area with scaffolds—such a technique was initially proposed by Henning et al. 16 This technique entails wrapping the meniscus with autologous fascia harvested from the pes anserinus area. This procedure was further developed to employ the collagen membrane as the material for meniscus wrapping, leading to what is presently referred to as the AMMR procedure.10,18,25,26 Although it seems possible to alter the mechanical characteristics of the sutured area by employing a scaffold, no scientific evidence is available to confirm this. The double-sided scaffold-augmented repair technique that we propose in this study has integrated an additional structural element to the collagen membrane, creating a stronger repair construct, which is proven in our biomechanical testing. Sutures supported with scaffolds offered better primary stability and improved the strength of the repair construct. The Meniscus Cap scaffold has greater potential for biomechanical enhancement of meniscal suturing than the simple collagen wrap (CMW), which is employed in the previously reported AMMR technique. The superior strength of the Meniscus Cap and suture construct was also proven after 500 loading cycles. Perhaps the most striking result of our study was that the Meniscus Cap and suture group was improved even compared with the intact meniscus group in relation to displacement; in other words, the length change of an experimentally divided medial meniscus that was then repaired with an H suture and a Meniscus Cap was less than, but in the same order as, the stretch of an intact meniscus. The necessary failure strength to resist displacement at the repair site to allow for an optimal healing environment for the meniscus has not been determined. However, the enhanced apposition of each meniscal tear end is likely beneficial, and a stronger construct that can resist displacement is likely to be favorable, as long as the construct is not too stiff. Given that the overall length changes are similar in the control group and the Meniscus Cap group, we are cautiously optimistic that the tensile strength of the Meniscus Cap is in the correct therapeutic range that when applied to a damaged meniscus produces a physiological response of the composite construct to loading.
The failure mode in the present study did not differ across all 4 groups. The loading force and the number of cycles—as adopted from similar designed previous studies—have produced suture ruptures as a common failure mode in all 3 groups.2,8,9,11-13,15,28
There is a contrast with the above findings and the failure mode of currently available meniscal scaffolds. In all specimens tested by Gwinner et al, 14 the failure mode of the Collagen Meniscus Scaffold was a complete disruption of the scaffold integrity. The same results were observed by Hoburg et al 17 for the Actifit implant. The mechanical stabilization of both artificial meniscus implants depends on the suture materials and the biological environment of the knee joint. The main load to failure was 36.2 ± 13.1 N for the Collagen Meniscus Scaffold and 53.3 ± 6.5 N for the Actifit implant. Consequently, their primary stability was not favorable when employed in the meniscus as a bridging technique (in study by Gwinner et al 14 ) was not favorable compared with the Meniscus Cap and direct meniscal suture (in this study).
Limitations
There are several limitations to the present study. Although porcine menisci are similar in shape and function to human tissue, they are not perfect surrogates. Porcine menisci are thicker, denser, and smaller than human menisci and therefore may not serve to accurately mimic human meniscal biomechanical properties. However, 1 benefit was that porcine menisci were harvested from same-aged pigs, allowing them to test their biomechanical behavior in a standardized fashion, without the confounding factors of highly variable degenerative menisci harvested from cadaveric donors. Porcine menisci were used in previous studies and found to be a good biomechanical model.11,12,15,30
Although human meniscus tissue would be the most representative explant model for meniscus injury healing, live tissue specimens from human sources are very limited in availability and would never be available at the same time as a fresh specimen for biomechanical testing, meaning that there would be an additional variable for freezing, storing, and thawing. Typical human patients who sustain meniscus injuries are members of the young and active population. Only fragmented explant is likely available from young patients undergoing partial meniscectomy. Whole meniscus explants, possibly obtained from total knee joint replacement surgeries, are likely undergoing an age- or osteoarthritis-associated degradation process. Therefore, menisci obtained from various animals are the primary sources for explant injury healing models. Menisci harvested from bovine, porcine, canine, equine, and caprine specimens are commonly used as explants to study injury repair and healing. 29
This study aimed to determine the biomechanical properties of repair techniques for complex meniscal lesions. To eliminate confounding factors, complete radial, horizontal, and longitudinal tears were made with axial force applied perpendicular to the radial tear. However, such a setup did not fully reflect the actual physiological conditions in which compression, tension, and shear forces are applied to the meniscus simultaneously. Furthermore, the repair knots were tied manually, in an open fashion, and uniformly for all specimens, leading to very small gaps forming after cyclic loading. This is just not achievable in the arthroscopic all inside or inside-out suture techniques that are contemporary knee surgical practice today. This study design simulates the immediate postsurgical rehabilitation, where there is no healing and the repair is more vulnerable to damage. Since the authors could not find studies on suture strength in complex meniscal lesions in the literature, test protocols for radial lesions were adopted for this study.2,15,28 The results were also compared with the studies on the biomechanical evaluation of sutures available on the market for meniscal scaffolds.14,17 However, as a control group, we used undamaged porcine menisci harvested from the same specimen series to test the suturing methods. In our opinion, such an approach allowed us to draw scientifically valid and clinically relevant conclusions regarding the tested methods of meniscal suture with or without augmentation by a scaffold. Despite all 3 techniques demonstrating significantly improved strength and stiffness, it is still unknown to what degree the strength and stiffness of the repaired construct contribute to the ideal healing environment to achieve the best clinical outcome.
Conclusion
In a porcine specimen meniscal repair model, the biomechanical properties of a novel Meniscus Cap repair technique were superior to those of the simple suture and CMW techniques. Future studies of biomechanical and clinical outcomes in human meniscal repairs with this device are warranted to explore whether this repair method is valuable to clinical practice and patient outcomes.
Footnotes
Acknowledgements
The authors thank Marcin Chyla and Maciej Cybulski for their assistance with the preparation of the Meniscus Cap implant.
Final revision submitted September 13, 2023; accepted October 6, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: K.C.-G. has an international patent for the Meniscus Cap implant. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
