Abstract
Background:
Some patients with proximal rectus femoris (PRF) avulsions require surgical treatment after failed nonoperative treatment. There is no consensus on the superiority of suture anchor repair with the suture-bridge repair (SBR) technique versus tenodesis repair (TR) for PRF avulsions.
Purpose:
To compare the failure load and elongation at failure between SBR and TR and to compare the stiffness of these 2 repair techniques versus the native state.
Study Design:
Controlled laboratory study.
Methods:
Seven pairs of human cadaveric hemipelvises were dissected to the PRF and sartorius origins. Each specimen underwent preconditioning followed by a distraction test to determine the stiffness of the native specimen. One specimen of each pair received one of the repair methods (SBR or TR), while the other specimen in the pair received the other repair technique. After repair, each specimen underwent preconditioning followed by a pull to failure. The failure load, elongation at failure, stiffness, mode of failure, and stiffness as a percentage of the native state were determined for each repair.
Results:
The SBR group exhibited a stronger failure load (223 ± 51 N vs 153 ± 32 N for the TR group; P = .0116) and significantly higher stiffness as a percentage from the native state (70.4% ± 19% vs 33.8% ± 15.5% for the TR group; P = .0085). While the stiffness of the repair state in the SBR group (41.5 ± 9.4 N/mm) was not significantly different from that of the native state (66.2 ± 36 N/mm), the stiffness of the repair state in the TR group (20.3 ± 7.5 N/mm) was significantly lower compared with that of the native state (65.4 ± 22.1 N/mm; P < .001) and repair state in the SBR group (41.5 ± 9.4 N/mm; P = .02). The SBR group primarily failed at the repair site (71%), and the TR group primarily failed at the suture-sartorius interface (43%) and at the muscle (29%).
Conclusion:
SBR and TR specimens were significantly weaker than the native tendon. The stiffness of the SBR was equivalent to that of the native tendon, while TR was significantly less stiff than the native tendon. The SBR was superior to TR in terms of failure load, stiffness, and percentage stiffness from the native state.
Clinical Relevance:
SBR may be a better surgical option than TR to optimize failure load and stiffness for PRF avulsions.
Proximal rectus femoris (PRF) avulsion has been reported to occur primarily in high-level athletes involved in sports that demand repetitive kicking and sprinting.3,7 Injuries in the PRF remain rare. However, the prevalence of PRF injuries is 0.5% to 1.5% of hip injuries that occur in athletes. 17 As the prevalence of reported PRF injuries is low, PRF injuries are often overlooked and undertreated, leading to chronic injuries with sustained symptoms and additional time lost from sports for the athletes.9,1,6
Traditionally, PRF injuries in high-level athletes have been treated nonoperatively, utilizing a combination of stretching, ice, manual therapies, and gradual strengthening, with multiple studies reporting favorable clinical outcomes.5,6,19 However, nonoperative treatment is often unsuccessful in high-level athletes for the reasons of not restoring the native anatomy of the PRF and scarring of the tendon, resulting in the recurrence of symptoms, diminished strength, decreased function, sustained pain, and cramping.7,9,20,21 For these reasons, surgical repair is generally recommended for high-level athletes with PRF injuries.4,7,15,18,21,23 For PRF injuries, suture anchor repair and tenodesis are 2 common repair techniques.4,12,15,21 Clinical studies have reported satisfactory postoperative results for both techniques,1,7,13,15,18,21-23 and there is no consensus on which technique is superior.15,17 Furthermore, no biomechanical studies have evaluated the time-zero strength of these repair techniques for PRF injuries. As a result, these treatment plans are not optimized for postoperative rehabilitation, as they are performed with little biomechanics support.
This study aimed to compare the failure load and elongation at failure between the 2 repair techniques (suture anchor repair with suture-bridge technique and tenodesis repair [TR] technique) and compare the stiffness among these 2 repairs and the native state. We hypothesized that suture anchor repair with the suture-bridge technique would be stronger than the TR technique and exhibit equivalent stiffness to the native tendon.
Methods
Specimen Preparation
Seven pairs of fresh-frozen human cadaveric hemipelvises (N = 14 specimens [mean age, 60.4 ± 5 years; age range, 51-65 years]; 6 men and 1 woman) with no evidence of previous injury or abnormality were acquired. A priori power analysis using G*Power 3.1 (Universität Düsseldorf, Germany) based on the data from a previous study 10 showed the total required sample size is 7 per group (effect size, 1.99; α = .05; power, 0.8; No. of groups, 3). The specimens used were donated to a tissue bank for medical research and then purchased by our institution. Using cadaveric specimens does not require institutional review board approval at our institution.
Specimens were placed in a supine position, and exposure of the PRF and sartorius origins was achieved using an open approach. The rectus femoris and sartorius were followed proximally to their respective origins in the anterior inferior iliac spine (AIIS) for the rectus femoris and the AIIS for the sartorius. Dissected specimens were stored at −20°C until the day before testing. Specimens were thawed 24 hours before biomechanical testing, and the posterior half of the iliac wing was potted in a cylindrical mold using polymethyl methacrylate (Fricke Dental).
Each pair of specimens was randomly assigned to undergo 2 repair methods. One specimen of the pair was repaired utilizing a suture bridge repair technique (suture-bridge group), and the other specimen of the pair was repaired using a soft tissue tenodesis technique (tenodesis group).
Biomechanical Testing of the Native State
The potted iliac wing was secured within a custom fixture that was clamped to the base platform of a dynamic tensile testing system (Instron ElectroPuls E10000; Instron Systems) with the musculotendinous portion of the rectus femoris fixed within a custom clamp fixed to the actuator (Figure 1). Pilot testing was performed to verify that slippage at the clamps did not occur, especially at applied loads exceeding 300 N. Potential slippage was monitored by marking a line on the specimen at the clamp and by tracking displacement and force data during and after the testing period. If the displacement of the mark was >0.5 mm or if the force/displacement data recorded a shift in the value of >10% of the previous value, then slippage was deemed to have occurred. To minimize the damage of clamping to the specimen, the clamped region of the specimen was supported with wraps and sutures. Specimens were clamped between 3 and 7 cm distal to the anterior superior iliac spine enthesis. Applied loads were representative of in vivo loads, 16 with the applied loading direction resulting in a shear force between the rectus femoris entheses and the anterior superior iliac spine.

(A) The front view and (B) the side view of the biomechanical testing set up. The iliac wing was potted and secured to the base of the dynamic tensile testing system, and the rectus femoris was fixed within a clamp secured to the end effector of the dynamic tensile testing system.
Initially, all specimens were nondestructively tested in the native state. Specimens were preconditioned with cyclic loads representative of a progressive postoperative rehabilitation protocol. A previous study reported a mean maximum net force on the rectus femoris during walking to be approximately 500 N. 16 Recognizing the time-zero nature of the study, a 1-Hz cyclic loading protocol was applied to the specimen with loads cycling from 25 N to a maximum load that increased by 75 N every 50 cycles. Native stiffness was calculated by pulling the specimen at 1 mm/s for 20 seconds and then returning the applied load to 25 N. This way, native stiffness could be calculated, and specimen damage could be avoided. Also, 325 N, 65% of the 500 N in vivo rectus femoris load, 16 was selected to account for the reduced time-zero loading observed in the clinic.
Surgical Techniques
The surgeries using the suture-bridge technique or the tenodesis technique in the present study were performed following the technique of previous studies.4,21 All surgeries were performed by 2 fellowship-trained orthopaedic surgeons (H.N. and K.Y.). For both surgeries, the injury state was recreated via transection of the rectus femoris from its insertion. For the suture-bridge technique, the soft tissue around the AIIS was excised to expose the subchondral bone and prepare for anchor placement. First, 2 all-suture, single-loaded anchors (Q-Fix; Smith & Nephew) were used to establish the distal row. The suture strands from the anchor were passed through the tendon from the deep side to the superficial side. After that, 2 holes were drilled for the second anchors (SwiveLock; Arthrex), proximal to the first anchors. Two strands were passed through each second anchor, and the tissue tension was evaluated. The surgeon introduced the second anchors into the holes, and adequate tension was obtained. For the soft-tissue tenodesis technique, the periosteum was excised, and multiple interrupted nonabsorbable sutures (No. 2 FiberWire; Arthrex) were used to suture the muscle-tendon complex of the rectus femoris in a side-to-side fashion without tension to the surrounding muscle (proximal site of vastus lateralis), sartorius fascia, and rectus bed. Both sides of the rectus femoris were repaired by creating 10 stitches with continuous interlocking sutures on each side. In addition, 2 single sutures were added to repair the rectus femoris with the rectus bed. The images for each surgery are shown in Figure 2.
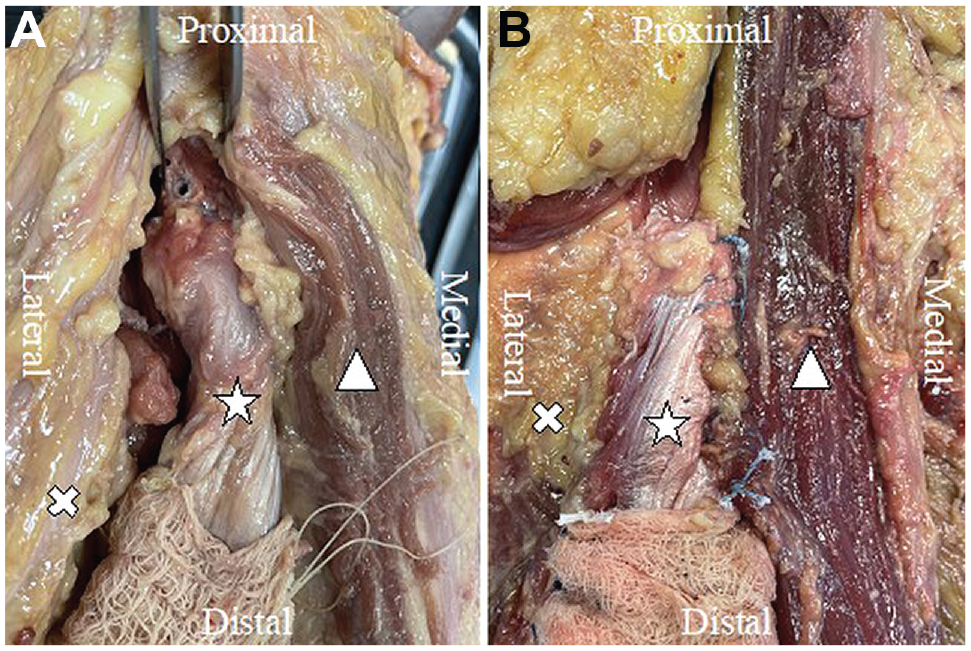
Surgical repair for the rectus femoris using (A) the suture-bridge technique and (B) the tenodesis technique on right hips. The white star, the triangle, and the cross indicate the rectus femoris, sartorius, and lateral vastus of the quadriceps, respectively.
Biomechanical Testing of the Surgical States
After performing the repairs, a 1-Hz cyclic loading protocol was applied to the specimen, with loads cycling from 25 N to a maximum load that increased by 75 N every 50 cycles. If the specimen did not fail after reaching 325 N, then the specimen was pulled at 1 mm/s to failure. The failure load, elongation at failure, stiffness, and failure mode were recorded for each repair technique. Stiffness as a percentage of the stiffness of the native state was calculated for both study groups.
Statistical Analysis
Statistical analyses were performed using MATLAB (Mathworks), and data were expressed as means and standard deviations. Normal distribution of ultimate failure load, elongation at failure, and stiffness data were verified using Shapiro-Wilk test. Paired t tests were used to compare ultimate failure load and elongation at failure between the suture-bridge and the tenodesis groups. Stiffness data were compared between the groups and between the repair state and the native state within each repair group using paired t tests. Statistical significance was defined as P = .05.
Results
Failure Mechanics
The failure load and mode of failure of the study groups are reported in Table 1. The suture-bridge group exhibited significantly stronger mean failure load (223 ± 51 N) than the tenodesis group (153 ± 32 N; P = .0116) (Figure 3). Elongation at failure of the suture-bridge group (17 ± 7.4 mm) was not significantly different from that of the tenodesis group (27.6 ± 16.1 mm; P = .0783) (Figure 3).
Failure Load and Mode of Failure Data for 7 Pairs of Specimens a

Data are presented as mean (SD) or n.
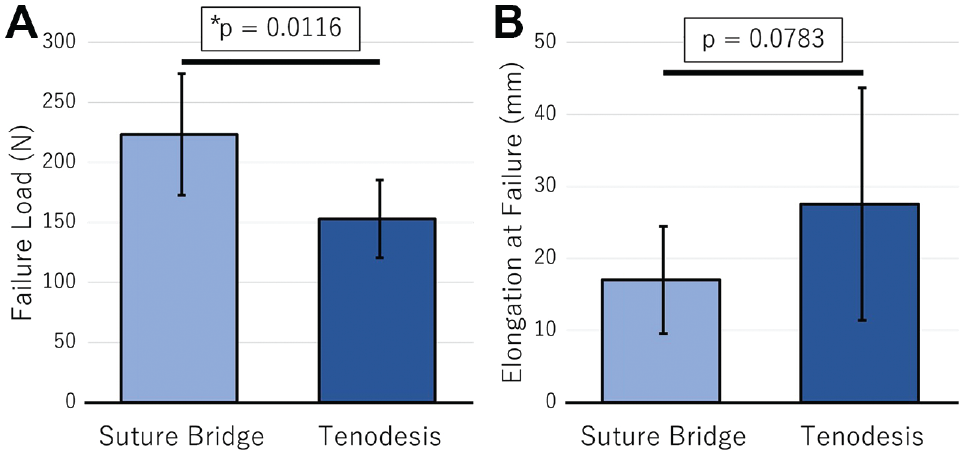
(A) The mean failure load and (B) elongation at failure for the suture-bridge and tenodesis groups. Error bars represent standard deviations. *A statistically significant difference between groups (P = .05).
Failure in the suture-bridge group occurred at the repair site in 5 specimens (71%), at the clamp in 1 specimen (14%), and via the anchor pullout in 1 specimen (14%). Failure in the tenodesis group was at the suture-sartorius interface in 3 specimens (43%), at the muscle in 2 specimens (29%), at the clamps in 1 specimen (14%), and at the suture-vastus lateralis interface in 1 specimen (14%) (Table 1).
Stiffness
Data for native and repair stiffnesses of both groups are reported in Table 2. The native stiffness of the suture-bridge group (66.2 ± 36 N/mm) was not significantly different from the native stiffness of the TR group (65.4 ± 22.1 N/mm; P = .999). The repair stiffness of the suture-bridge group (41.5 ± 9.4 N/mm) was not significantly different from the native stiffness of the suture-bridge group (P = .127). The repair stiffness of the TR group (20.3 ± 7.5 N/mm) was significantly lower than the native stiffness of the TR group (P < .001). The repair stiffness of the suture-bridge group was significantly higher than the repair stiffness of the TR group (P = .02) (Figure 4). Percentage stiffnesses from native of the suture-bridge group (70.4% ± 19%) was significantly higher than that of the TR group (33.8% ± 15.5%; P = .0085) (Figure 5).
Stiffness of Native and Repair States for 7 Pairs of Specimens a

Data are reported as mean ± SD or n. Postop, postoperative.
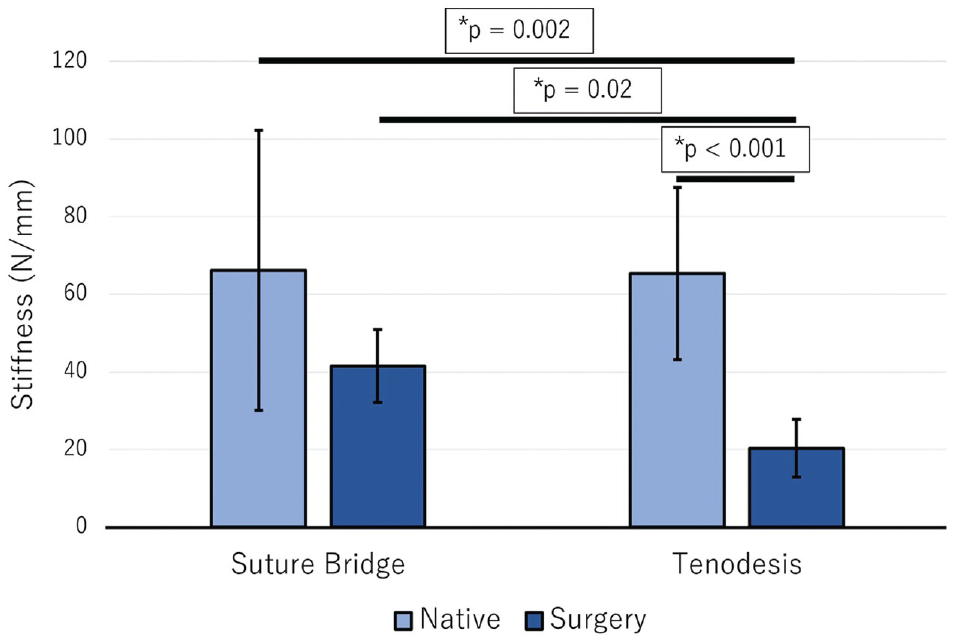
The mean stiffness values of the native and repair states for the suture-bridge and tenodesis groups. Error bars represent standard deviations. *A statistically significant difference between groups (P = .05).

The mean stiffness values of percentage from native for the suture-bridge and tenodesis groups. Error bars represent standard deviations. *A statistically significant difference between groups (P = .05).
Discussion
The findings of this study indicated that the failure load of the suture-bridge technique was significantly higher than that of the tenodesis technique. Furthermore, the stiffness of the tenodesis technique was significantly lower than that of its native condition and the suture-bridge technique. In contrast, the suture-bridge technique proved to be equivalent to its native condition. The percentage stiffness from native of the suture-bridge technique was significantly higher than that of the tenodesis technique. Although previous clinical studies have not reached a consensus on superiority or inferiority between these 2 repair techniques, the results of this study demonstrated the biomechanical superiority of the suture anchor repair using the suture-bridge technique.
Although there is a paucity of literature reporting the postoperative outcomes after suture anchor repair using the suture-bridge technique, previous clinical studies have reported satisfactory postoperative results for both tenodesis and suture anchor repair techniques using the single row technique1,4,7,13,15,18,20-23 (Table 3). Kayani et al 15 compared the postoperative clinical outcomes of tenodesis and suture anchor repair techniques for PRF injuries in professional athletes and reported that the tenodesis group had a shorter time to return to play at previous competitive level and a lower recurrence rate than the suture anchor repair group. Although our results showed that suture-bridge repair (SBR) was superior to TR at time zero in terms of the failure load and stiffness, their results suggest the importance of further investigation into how each repair procedure is coordinated with the other quadriceps during the subsequent healing process. Ishoi et al 14 reported that the mean quadriceps force among senior soccer players was 737.3 N. Since this value is much higher than the failure load after these 2 repair techniques, further clinical studies to investigate postoperative quadriceps muscle strength after these 2 repair methods are required.
Previous Clinical Studies of PRF Repair Surgery a

LOE, level of evidence; NA, not applicable; PRF, proximal rectus femoris.
In addition, there is no standard protocol for rehabilitation after surgical repair for PRF injury, and postoperative limits on range of motion (ROM) and weightbearing are left to the surgeon's discretion. As our results showed that the stiffness of SBR was not significantly different from that of the native state at time zero of repair, postoperative restriction of ROM with a brace or setting a nonweightbearing period may be avoided. However, Hale et al 9 reported that estimated rectus femoris muscle forces during squatting and lunging were both 400 N, which were higher than the mean failure load in the suture-bridge group. Therefore, it is suggested that the movements that put stress on the quadriceps muscle need to be avoided for a while after surgery. A careful postoperative follow-up is still necessary as outcomes from this study are recorded at time zero and do not account for structural healing of the repaired site. Furthermore, it may be beneficial to accelerate postoperative rehabilitation in terms of ROM exercises and weightbearing depending on patient symptoms.
Although the results in this study demonstrated that suture anchor repair using the suture-bridge technique was stronger and stiffer than TR, suture anchor repair is not a perfect approach. Suture anchors are not recommended to use for cases with residual epiphyseal line because of the possibility of physeal injury and subsequent growth disturbance or in cases of hypersensitivity to anchor materials and inadequate bone stock. 8 In addition, to utilize the suture anchor repair, the tendon of PRF must be able to be reduced to its footprint. Considering these factors, TR is a suitable option for cases lacking epiphyseal line closure or cases where direct repair with a suture anchor is difficult because of a long history of injury or scarring. In addition, TR is a lower-cost procedure compared with suture anchor repair, as it does not require any hardware. Clinical studies will be needed to compare the clinical results of these 2 techniques.
Limitations
This study has several limitations that could have potentially affected the conclusions of this study. In this study, we used the transection model of PRF. We chose this method because there is a paucity of previous biomechanical studies on PRF avulsion in the literature, and a native-to-avulsion model was not feasible. Therefore, the model may differ from the actual PRF injury in vivo, but similar transection models have been used in studies about proximal hamstring injuries and Achilles tendon injuries.2,11 In the present study, we dissected the PRF from the AIIS attachment site as much as possible, which we believe mimics the clinical model as closely as possible. Accordingly, we could not evaluate the failure load of the native state. As an alternative, we assessed the stiffness of 2 surgical techniques and the native state. In addition, using cadaveric specimens could potentially alter the quality of the biological bone and tendon tissues. The age of specimens was relatively older than that of patients with PRF injury. Therefore, the quality of the rectus femoris in cadaveric specimens may not be representative of muscle quality in vivo. Furthermore, the sample size was small, despite the power analysis indicating adequate power with 7 pairs. Also, most samples were male specimens, which may have affected the quality of the bones. Furthermore, there was a wide variation in native biomechanical outcomes and large side-to-side differences in some specimens, which should be considered in interpreting the results. Moreover, the biological effects of healing and the physiological effects of loading on the repair site are unknown. This study was performed at time zero, immediately after surgical repair, when the healing process had not occurred.
Conclusion
In this study, suture-bridge and TR techniques were significantly weaker than the native tendon. The stiffness of the suture-bridge technique was equivalent to that of the native tendon, while the soft tissue TR was significantly less stiff than the native tendon. The suture-bridge technique was superior to the soft tissue TR in terms of failure load, stiffness, and percentage stiffness from the native state. Based on the findings in this biomechanical study, the suture-bridge technique may be a better surgical option than soft tissue tenodesis to optimize failure load and stiffness. Results from this study may inform clinical research evaluating postoperative ROM exercises and weightbearing protocols.
Footnotes
Final revision submitted June 12, 2023; accepted June 21, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.U. has received consulting fees from Smith & Nephew and CONMED. M.J.P. has received education payments from Smith & Nephew, CONMED, Linvatec, Ossur, Arthrex, and Siemens Medical Solutions; consulting fees from Smith & Nephew, Arthrosurface, Bledsoe, CONMED, Linvatec, DJO, Slack, Elsevier, MIS, Olatec, and NICE Recovery Systems; nonconsulting fees from Synthes GmbH, Smith & Nephew, Arthrosurface, Bledsoe, CONMED, Linvatec, DJO, Slack, Elsevier, MIS, Olatec, and NICE Recovery Systems; and royalties from Smith & Nephew, Arthrosurface, Bledsoe, CONMED, Linvatec, DJO, Slack, and Elsevier and is a shareholder in Arthrosurface, MJP Innovations, Vail Valley Surgery Center, Vail MSO Holdings, MIS, EFFRx, Olatec, iBalance, Manna Tree Partners, Stryker, Trimble, 3M, Bristol Myers, Squibb, Pfizer, AbbVie, and Johnson & Johnson. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
