Abstract
Background:
Microfracture is the first-line treatment for cartilage defects; however, the suboptimal quality of the repaired cartilage remains an issue.
Purpose/Hypothesis:
The aim of this first in-human study was to compare the clinical efficacy and safety of a combination of particulated costal allocartilage and microfracture versus microfracture alone in treating knee cartilage defects. We hypothesized that the particulated costal allocartilage with microfracture would result in superior cartilage repair quality and better clinical outcomes at 48 weeks postoperatively.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
Patients with cartilage defects were allocated randomly to the treatment group (particulated costal allocartilage with microfracture) and control group (microfracture alone). Magnetic resonance imaging (MRI) outcomes of cartilage repair (the primary outcome measure) were evaluated at the 48-week follow-up using the Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) score. Patient-reported clinical outcomes (visual analog scale [VAS] pain score, Knee injury and Osteoarthritis Outcome Score [KOOS], and International Knee Documentation Committee score) and adverse events were evaluated at 12, 24, and 48 weeks postoperatively.
Results:
Overall, 88 patients were included (44 patients each in the treatment and control groups). The total MOCART score at 48 weeks postoperatively was significantly higher in the treatment group than in the control group (P < .001). Among the 9 MOCART variables, 6 were significantly superior in the treatment versus the control group: degree of repair and defect filling (P < .001), integration to the border zone (P < .001), surface (P = .006), structure (P = .011), signal intensity of the repair tissue (P < .001), and subchondral lamina (P = .005). There were significant between-group differences in KOOS-Pain (P = .014), KOOS-Activities of Daily Living (P = .010), KOOS-Sports (P = .029), and KOOS-Symptoms (P = .039) at 12 weeks postoperatively and in VAS pain (P = .012) and KOOS-Pain (P = .005) at 24 weeks postoperatively. At 48 weeks postoperatively, clinical outcomes were comparable between the groups.
Conclusion:
Microfracture augmented with particulated costal allocartilage resulted in superior cartilage repair quality compared with microfracture alone in terms of MRI evaluation of the knee joint cartilage defect at the 48-week follow-up. Functional outcomes were favorable for both treatments at final follow-up.
Registration:
KCT0004936 (Clinical Research Information Service [CRiS] of the Republic of Korea).
The innate healing capacity of injured articular cartilage is limited owing to its lack of blood supply and low cellularity. 15 Marrow stimulation techniques, such as microfracture, are reasonable options for the repair of injured cartilage in the knee joint because of their relatively low cost, ease of implementation, low risk of complications, and satisfactory short-term outcomes. 24,38 Nonetheless, the mechanically unstable “super clot” at the defect site and suboptimal quality of the repaired fibrocartilage rather than the hyaline cartilage, which consequently influence durability and long-term outcomes, continue to be areas of concern. 27,45 Various types of scaffolds have been proposed to manage these issues by providing mechanical and biochemical support as well as a suitable environment for cartilage regeneration. 37
Decellularized hyaline cartilage is potentially an ideal scaffold for cartilage regeneration due to its mechanical, biochemical, and structural properties, which resemble those of native hyaline cartilage. 28 The extracellular matrix (ECM) scaffold, derived from native articular hyaline cartilage, is capable of inducing in vitro chondrogenic differentiation in progenitor cells without external stimuli. 5 Similar to the articular cartilage, the costal cartilage represents another emerging source of hyaline cartilage scaffold for the treatment of cartilage defects via promotion of chondrogenesis. 10,34 However, only a few recent observational studies have reported favorable clinical outcomes after microfracture augmented with decellularized hyaline cartilage, 4,6 and no previously published work has presented the outcomes of microfracture augmented with costal cartilage. Therefore, evaluating the efficacy of decellularized costal cartilage in treating knee cartilage defects is imperative.
MegaCarti (L&C BIO Co) is a newly developed hyaline cartilage-derived ECM scaffold harvested from the costal cartilage of cadavers under the age of 45 years; it undergoes particulation and decellularization processes. In this first in-human study, we aimed to compare the clinical efficacy and safety between particulated costal allocartilage with microfracture and microfracture alone in treating knee cartilage defects. We hypothesized that particulated costal allocartilage with microfracture would have (1) superior quality of repaired cartilage based on magnetic resonance imaging (MRI) results and (2) better clinical outcomes than that of microfracture alone at 48 weeks postoperatively.
Methods
Study Design and Population
The present study was a multicenter, prospective, randomized, participant- and rater-blinded trial conducted across 4 hospitals. This study was approved by the Korean Ministry of Food and Drug Safety and the institutional review board of each involved hospital, and it was prospectively registered at the Clinical Research Information Service (KCT0004936), which is a primary registry in the World Health Organization registry network and is recognized by the International Committee of Medical Journal Editors. All participants were provided with detailed information about the trial; written informed consent was obtained from each participant before screening and enrollment.
The inclusion criteria were as follows: age of 19 to 65 years, focal cartilage defects measuring <10 cm2 in size, International Cartilage Repair Society (ICRS) grade 3 or 4, and knee joint symptoms attributable to cartilage defects. The exclusion criteria were as follows: a history of cartilage-related surgery in the 1 year before to screening, body mass index ≥30 kg/m2, inflammatory arthritis, arthritis associated with autoimmune disease, intra-articular injection in the 3 months before screening, intake of systemic steroid medications in the 1 month before screening, current pregnancy or breastfeeding, and systemic or localized infection.
After assessing 101 patients for eligibility between April 1, 2020 and January 31, 2021, 90 patients were randomized in a balanced ratio to either the treatment or control group using the block randomization method (Figure 1). Sealed opaque envelopes containing information about group assignment were provided just before surgery and were managed by independent personnel unrelated to the medical institutions. Ultimately, 44 patients were allocated to the treatment group (particulated costal allocartilage with microfracture) and 46 were assigned to the control group (microfracture alone). The patients and independent raters were blinded to the group allocation until 48 weeks postoperation, unless serious adverse reactions were suspected.

Consolidated Standards of Reporting Trials (CONSORT) flow diagram showing the inclusion and exclusion process.
Operative Procedures
Four surgeons specializing in sports medicine (M.J., K-M.J., S-H.P., and S-H.K.) performed the operation at 4 different hospitals. Diagnostic arthroscopy was conducted on each compartment of the knee to localize the cartilage defect and check for the presence of other pathologies. Following confirmation of cartilage defects, the nonfunctional unstable cartilage flaps were meticulously debrided using an arthroscopic shaver, gouge, or curette until the defects were surrounded by healthy stable vertical margins. The residual calcified layer of the cartilage defects was completely removed without violating the subchondral bone by either arthroscopy or mini-open arthrotomy. The microfracture procedure involved creating as many holes as possible using drills; each hole had a depth >2 mm and was spaced approximately 3 to 4 mm apart. 13,38,39
In the treatment group, paste-type particulated costal allocartilage was augmented on the microfractured cartilage defect. Particulated costal allocartilage (MegaCarti) with a size of 200 to 1000 µm and weight of 1.5 g was prepared in a prefilled 3-mL syringe (Figure 2). It was prepared in the form of a viscous paste by adding sodium hyaluronate (Hyundai Bioland) crosslinked with sodium carboxymethyl cellulose (Sigma-Aldrich). Before applying the particulated costal allocartilage, the defect site was dried by removing any intra-articular fluid using suction, gauze, or cotton swabs. Subsequently, the push stick of the syringe was gently pressed so that the particulated costal allocartilage was driven into the defect site. Fibrin glue (Greenplast; Green Cross, Yongin, Korea) was applied after adjusting the inserted particulated costal allocartilage to the same height as or lower height than the surrounding healthy cartilage. At 5 minutes after setting of the fibrin glue, the knee was moved manually throughout the range of motion (ROM) to confirm the stability of the inserted particulated costal allocartilage.

(A) Particulated costal allocartilage (MegaCarti; L&C BIO Co) with a size of 200 to 1000 µm and weight of 1.5 g was prepared in a prefilled 3-mL syringe. (B) The particulated costal allocartilage was made into a viscous paste by adding sodium hyaluronate crosslinked with sodium carboxymethyl cellulose.
For patients with varus malalignment of the affected lower extremity, open-wedge high tibial osteotomy (HTO) was also performed in the treatment and control group. The goal of alignment correction was set as the mechanical axis line passing through the Fujisawa point, which was determined preoperatively using the Miniaci method. 18
Postoperative Rehabilitation
The postoperative rehabilitation protocol for the treatment group was identical to that for the control group. Immediately after surgery, passive patellar mobilization, ROM exercises, and isometric quadriceps-strengthening exercises were encouraged. Both active and continuous passive motion machine-assisted ROM exercises were recommended daily for 4 to 6 weeks. Ambulation with crutch-assisted partial weightbearing was allowed from 4 weeks postoperatively, with a gradual increase in intensity. Full weightbearing was permitted at 8 to 12 weeks postoperatively.
Magnetic Resonance Imaging Evaluation
The quality of the cartilage repair tissue, which was the primary efficacy endpoint, was evaluated by MRI using a 3-T scanner. Two independent musculoskeletal radiologists with >5 years of experience evaluated the MRI scans acquired from all participants at 48 weeks postoperation in a blinded manner. Standardized, semiquantitative morphological evaluation based on the Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) score, rated from 0 as the worst to 100 as the best, was performed. 26 The 2 independent radiologists recorded the MOCART scores of the variables separately, and the final scores were calculated as the mean of the 2 scores.
Clinical Evaluation
Clinical outcomes, which were the secondary study endpoint, were evaluated using patient-reported outcome measures, such as the visual analog scale (VAS) pain score, 30 Knee injury and Osteoarthritis Outcome Score (KOOS), 3 and International Knee Documentation Committee (IKDC) score. 14,17 Blinded independent raters collected data on clinical outcomes at 12, 24, and 48 weeks postoperatively.
Safety Evaluation
Patients were monitored for safety parameters at 12, 24, and 48 weeks postoperatively. Any adverse event that was not present before the start of treatment or that was already present but worsened after the treatment was reported, regardless of whether it was relevant to the intervention. Subsequently, adverse events related to the intervention were identified.
Statistical Analysis
The sample size was calculated a priori based on previous studies comparing other cartilage restoration procedures and microfracture. 2,8,21,29 In previous studies, the mean MOCART score at 48 weeks postoperatively was estimated to be 65.5 and 56.1 in the investigational and control groups, respectively. In addition, in previous studies, the highest value of standard deviation of the MOCART score was estimated to be 14.1. Sample size calculation was performed based on a two-sided independent t test with a significance level (α) of 5% and power (1 - β) of 80%. Accordingly, the minimum required sample size was 36 per group. Considering a possible dropout rate of 20%, 90 patients were planned to be included in the trial.
The full cohort of patients completing the study was used for baseline demographics and clinical efficacy of treatment, after excluding those who did not fit the inclusion and exclusion criteria. All patients who were enrolled in the trial and attended a subsequent visit were included in the safety analysis. Statistical analyses were performed using SAS software Version 9.4 (SAS Institute Inc). For all analyses, statistical significance was set at P < .05. Continuous variables were compared between the 2 groups using the independent t test or Wilcoxon rank-sum test, depending on the results of the Shapiro-Wilk test. Categorical variables were compared between the 2 groups using the chi-square test or Fisher exact test. Changes in continuous variables before surgery and at every follow-up visit were compared within each group using repeated-measures analysis of variance, and Bonferroni correction was performed for post hoc analysis.
Results
Patient Characteristics
In the control group, 1 patient was lost to 48-week follow-up, and 1 patient was excluded from analysis, leaving 44 patients each in the treatment and control groups. Other than the distribution of age >50 years being higher in the treatment group (P = .031), the baseline patient characteristics were comparable between the study groups (Table 1).
Baseline Patient Characteristics a

a Values are presented as mean ± SD or No. of patients (%). Boldface P value indicates statistically significant difference between groups (P < .05). HTO, high tibial osteotomy; ICRS, International Cartilage Repair Society.
MRI Outcomes
The total MOCART score at 48 weeks postoperatively, which was the primary outcome measure in this study, was significantly higher in the treatment group than in the control group (56.0 ± 10.5 vs 43.0 ± 17.4; P < .001) (Table 2). Among the 9 variables of the MOCART score, 6 were significantly different between the groups: degree of repair and defect filling (P < .001), integration to the border zone (P < .001), surface (P = .006), structure (P = .011), signal intensity of the repair tissue (P < .001), and subchondral lamina (P = .005). Variables regarding the subchondral bone, adhesions, and effusion were not significantly different between the groups.
MRI Outcomes Evaluated Using the MOCART Score at the 48-Week Follow-up a

a Values are presented as mean ± SD or No. of patients (%). Boldface P value indicates statistically significant difference between groups (P < .05). MOCART, Magnetic Resonance Observation of Cartilage Repair Tissue; MRI, magnetic resonance imaging.
b Values in brackets indicate the number of points possible. Each MOCART variable was evaluated by 2 independent radiologists, and the MOCART score was calculated from the mean of the 2 reviewers’ scores.
Subgroup analyses of the total MOCART score at 48 weeks postoperatively revealed that the quality of cartilage repair was better in the treatment group than in the control group regardless of stratification according to size (≤4 cm2, P = .015; >4 cm2, P = .002) and ICRS grade (grade 3, P = .038; grade 4, P < .001) of the cartilage defects (Table 3 and Figure 3). The subgroup of patients who were >50 years of age (P < .001) or who underwent concurrent HTO (P < .001) showed statistically better tissue quality in the treatment group than in the control group, whereas those who were ≤50 years of age (P = .076) or who underwent operation without HTO (P = .075) did not.
MRI Outcomes Evaluated Using the Total MOCART Score According to Subgroups at the 48-Week Follow-up a

a Values are presented as mean ± SD. Boldface P value indicates statistically significant difference between groups (P < .05). HTO, high tibial osteotomy; ICRS, International Cartilage Repair Society; MOCART, Magnetic Resonance Observation of Cartilage Repair Tissue.

Preoperative (left) and postoperative (right) MRI scans. (A) A 48-year-old woman with a trochlear cartilage defect (arrows) and (B) successful cartilage repair at 48 weeks after treatment with particulated costal allocartilage combined with microfracture. (C) A 50-year-old woman with a medial femoral cartilage defect (arrows) and (D) successful cartilage repair at 48 weeks after treatment with particulated costal allocartilage combined with microfracture. (E) A 65-year-old woman with a medial femoral cartilage defect (arrows) and (F) successful cartilage repair at 48 weeks after treatment with a combination of particulated costal allocartilage, microfracture, and HTO. HTO, high tibial osteotomy; MRI, magnetic resonance imaging.
Clinical Outcomes
With respect to the VAS pain score, IKDC score, and KOOS, the clinical outcomes progressively improved in each group at the 48-week follow-up (Figure 4). Between-group comparison indicated that the preoperative functional scores were not significantly different between the groups (Table 4). However, there were significant between-group differences in KOOS-Pain (P = .014), KOOS-activities of daily living (ADL) (P = .010), KOOS-Sports (P = .029), and KOOS-Symptoms (P = .039) at 12 weeks postoperatively and in VAS pain score (P = .012) and KOOS-Pain (P = .005) at 24 weeks postoperatively. At 48 weeks postoperatively, patient-reported clinical outcomes were comparable between the groups.
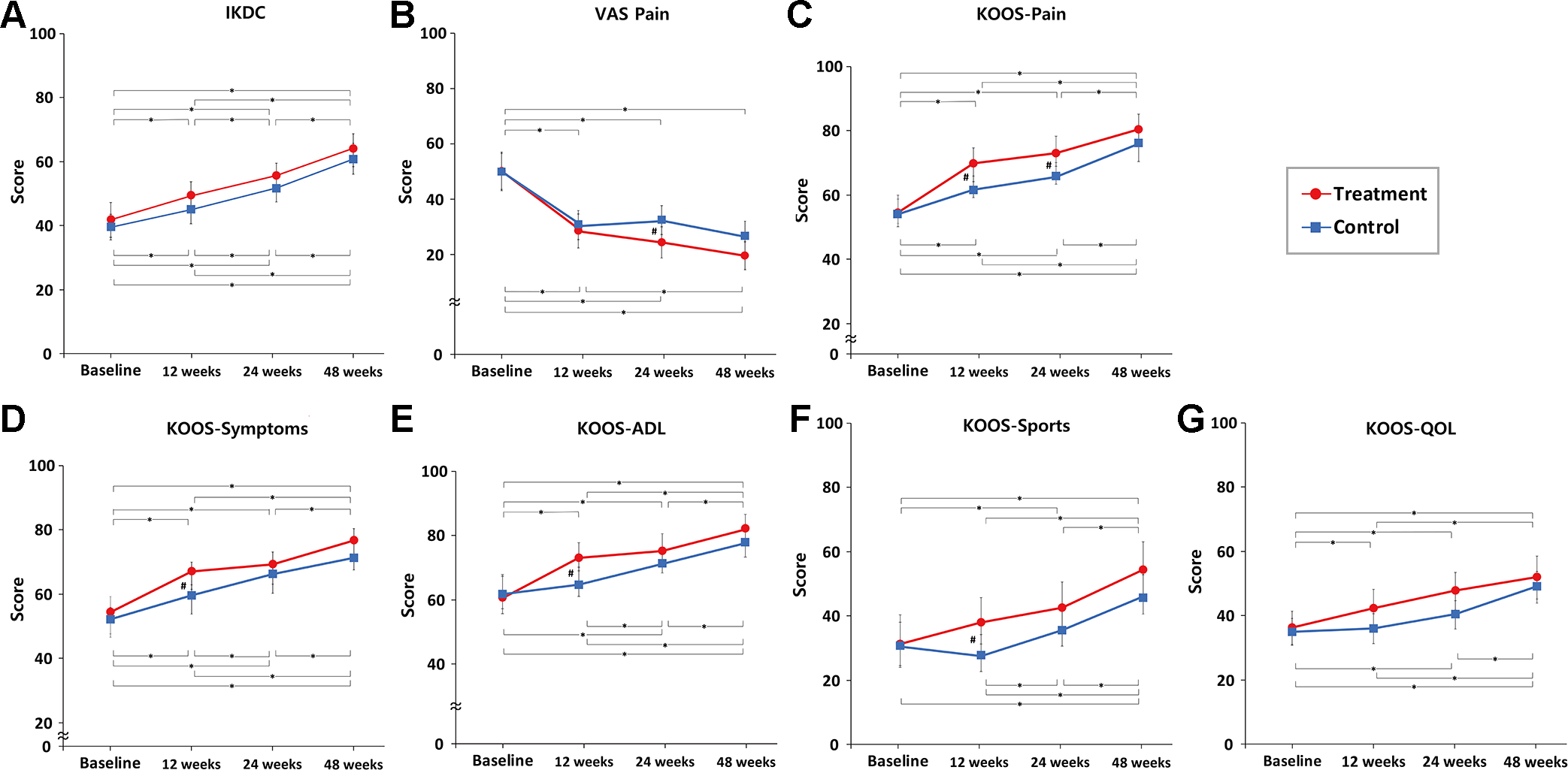
Changes in patient-reported functional outcomes from the preoperative baseline to 12, 24, and 48 weeks postoperatively for the treatment and control groups: (A) IKDC, (B) VAS pain, (C) KOOS-Pain, (D) KOOS-Symptoms, (E) KOOS-ADL, (F) KOOS-Sports, and (G) KOOS-QOL. Statistically significant differences #between groups and *between assessment times (P < .05). ADL, activities of daily living; KOOS, Knee injury and Osteoarthritis Outcome Score; IKDC, International Knee Documentation Committee; QOL, quality of life; VAS, visual analog scale.
Patient-Reported Functional Outcomes Between the Groups at Each Follow-up a

a Values are presented as mean ± SD. Boldface P value indicates statistically significant difference between groups (P < .05). VAS, visual analog scale; KOOS, Knee Injury and Osteoarthritis Outcome Score; IKDC, International Knee Documentation Committee; ADL, activities of daily living; QOL, quality of life.
Safety Outcomes
Overall, 16 patients (36.4%, 27 cases) in the treatment group and 14 patients (31.8%, 20 cases) in the control group experienced adverse events postoperatively (P = .551). None of the adverse events were related to the operation. No infection was observed.
Discussion
The primary finding of this randomized controlled trial was that the quality of cartilage repair yielded by the particulated costal allocartilage with microfracture was superior to that by the microfracture alone in terms of objective structural evaluation using the MOCART score. Compared with the controls, patients treated with the costal allocartilage showed significantly better patient-reported functional outcomes in some endpoints at 12 and 24 weeks of the relatively early postoperative period; however, the outcomes were comparable between the groups at the 48-week follow-up.
The allogeneic hyaline cartilage used in this study served as a cartilage ECM scaffold augmented on the microfractured cartilage defect. The ECM derived from the decellularized hyaline cartilage could retain the natural structure and main components of the native ECM, including type 2 collagen and glycosaminoglycans. 46 This could induce progenitor cells to develop chondrogenic direction via migration, adherence, proliferation, and differentiation. 25,32,46 Intrinsic biological elements or bioactive proteins from the ECM could also support chondrogenesis by regulating cartilage homeostasis. 7,46 Several in vitro and in vivo studies have established the chondroinductive potential of this hyaline cartilage ECM. 19,41,42 A case series of 6 patients treated with dehydrated micronized allogeneic articular cartilage combined with microfracture reported that the quality of the restored cartilage was similar to that of the adjacent native cartilage, as evaluated by T2 mapping. 4 More recently, a prospective case series of 49 patients with cartilage defects reported that the articular allocartilage showed favorable functional and MRI outcomes at the 24-month follow-up. 6
Costal cartilage, which is a type of hyaline cartilage, can serve as a source of native ECM instead of articular cartilage. Similar to articular cartilage, costal cartilage contains a high amount of type 2 collagen and glycosaminoglycans in the ECM. 35 However, costal and articular cartilages exhibit some differences in their mechanical and biochemical properties. 16,36 Despite these differences, costal cartilage ECM can induce chondrogenesis, similar to articular cartilage. 10 Setayeshmehr et al. showed that in vitro culture of adipose-derived mesenchymal stromal cells on human costal cartilage ECM could induce chondrogenic differentiation. 34 A few recent studies have reported the outcomes of using costal cartilage as a cell source for autologous chondrocyte implantation. 12,43,44 Nevertheless, the exclusive use of decellularized costal cartilage as a scaffold alone for the treatment of cartilage defects is difficult to find in the literature. To the best of our knowledge, the present study is the first human study that evaluated the outcomes of using costal allocartilage as scaffold augmentation over microfracture for the treatment of knee joint cartilage defects.
The results of our study highlight the superior cartilage repair quality on MRI achieved with microfracture augmented with costal allocartilage over that attained with microfracture alone. Microfracture remains the first-line treatment for cartilage defects; nonetheless, the unpredictable quality of the repair cartilage is a concern, leading to inferior durability that may result in the deterioration of clinical outcomes at 2 to 3 years after isolated microfracture. 27 Furthermore, lesion size of >4 cm2 and age of >40 to 50 years are considered to be factors related to worse outcomes. 20,23 In our study, the total MOCART score at 48 weeks postoperation was significantly higher in the costal allocartilage group than in the isolated microfracture group (56.0 ± 10.5 vs 43.0 ± 17.4; P < .001). Moreover, subgroup analyses according to lesion size and patient age showed that costal allocartilage augmentation yielded better quality results even in patients aged >50 years or those with a defect size >4 cm2. This finding implies that costal allocartilage augmentation could overcome the durability issue of microfracture and be applied in cases of larger lesion sizes and older patient age than previously indicated. A recent meta-analysis of randomized controlled trials comparing the outcomes after microfracture treatment with and without augmentation reported significant improvements in the postoperative MOCART score after microfracture with augmentation (mean, 64.3) as compared with that after isolated microfracture (mean, 46.1). 11 However, this meta-analysis included only 3 studies owing to a lack of high-quality randomized controlled comparison studies, of which 2 used porcine-derived collagen as an augment. Our study adds further evidence that augmentation with microfracture could improve the quality of the repaired cartilage, as compared with isolated microfracture.
In previous studies, augmented microfracture showed either equivalent or better results with respect to patient-reported functional outcomes, as compared with microfracture alone. In a systematic review of 18 studies by Arshi et al, 1 all included studies demonstrated significant improvement in each reported functional outcome for patients with combined microfracture and augmentation at a mean follow-up of 2 to 5 years. However, a subjective synthesis performed with 4 randomized controlled trials noted that 2 trials showed significantly greater improvements in the KOOS or IKDC score after combined microfracture and augmentation at the 2-year follow-up; conversely, the other 2 studies showed no differences when compared with microfracture alone. Recently, Fortier et al 11 performed a systematic review and meta-analysis of 744 patients within 14 studies comparing augmented microfracture using various types of adjuvants and isolated microfracture. They reported a significant improvement in the Lysholm score for patients who underwent augmented microfracture, but similar improvement in VAS, IKDC, or Western Ontario and McMaster Universities Arthritis Index (WOMAC) scores compared with patients who underwent only microfracture. In our study, the patient-reported clinical outcomes significantly improved from baseline but were comparable between the groups at the 48-week follow-up. The comparable functional outcomes at the final follow-up might be attributed to the excellent short-term results after isolated microfracture, which could be equivalent to those obtained with other advanced cartilage restoration procedures. 31 Nevertheless, patients treated with costal allocartilage demonstrated better functional outcomes at 12 weeks (KOOS-Pain, P = .014; KOOS-ADL, P = .010; KOOS-Sports, P = .029; KOOS-Symptoms; P = .039) and 24 weeks (VAS pain, P = .012; KOOS-Pain, P = .005) than those treated with isolated microfracture. This may indicate that costal allocartilage improves functional outcomes more quickly than isolated microfracture. We believe that subsequent long-term results will clarify the beneficial effects of augmented costal cartilage with microfracture on functional outcomes.
Limitations
This study has several limitations. First, the study had a relatively short follow-up period (48 weeks). The cohort in this study was designed to be followed up for 5 years; therefore, subsequent studies with longer follow-up periods will allow us to establish a definitive conclusion regarding the efficacy of costal allocartilage with microfracture. Second, the quality of cartilage repair was determined solely by the MOCART score calculated from MRI images. Although histological analysis remains the gold standard for evaluating the morphology of the repaired cartilage tissue, articular biopsy for humans is invasive and destructive. 33 Moreover, the MOCART score is a widely used and valid tool for assessing the morphology of the repaired articular cartilage. 40 Third, the results of the subgroup analyses could be underpowered, which could cause false-negative results because a previous sample size calculation accounted for overall groups instead of subgroups. 9 Fourth, although the age distribution of patients according to >50 years and ≤50 years was not different between the groups, the mean age in the treatment subgroup of patients aged >50 years was significantly higher than that in the control subgroup. Nevertheless, the total MOCART score in the subgroup of patients aged >50 years was higher in the treatment group than in the control group. Fifth, approximately 45% of patients included in the study underwent concurrent HTO. Simultaneous HTO could contribute to the restoration of cartilage, especially in the medial compartment of the knee joint, and could influence clinical results. 22
Conclusion
In the current study, microfracture augmented with particulated costal allocartilage resulted in superior cartilage repair quality compared with microfracture alone in terms of MRI evaluation of the knee joint cartilage defect at the 48-week follow-up. The functional outcomes were favorable for both treatments and comparable between the treatments at the final follow-up.
Footnotes
Acknowledgment
The authors thank Sungjun Kim, MD, PhD, and Min-Yung Jang, MD, for their assistance in the MRI evaluation.
Final revision submitted March 1, 2023; accepted April 13, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: The work was supported by a Korea Medical Device Development Fund grant funded by the Korean government (Ministry of Science and ICT, Ministry of Trade, Industry and Energy, Ministry of Health and Welfare, and Ministry of Food and Drug Safety) (project number: 1711138451, KMDF_PR_20200901_0217). L&C BIO Co also provided funding for this study by supporting a research grant, study protocol, and material (particulated costal allocartilage). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Yonsei University, Gangnam Severance Hospital (No. 3-2019-0428 and 1-2020-0003), Korea University Anam Hospital (No. 2020AN0056), and Ilsan Hospital (No. NHIMC 2020-01-006).
