Abstract
Objective
Osteochondral defects develop into osteoarthritis without intervention. Costal cartilage can be utilized as an alternative source for repairing osteochondral defect. Our previous clinical study has shown the successful osteochondral repair by costal cartilage graft with integration into host bone bed. In this study, we investigate how cartilaginous graft adapt to osteochondral environment and the mechanism of bone-cartilage interface formation.
Design
Costal cartilage grafting was performed in C57BL/6J mice and full-thickness osteochondral defect was made as control. 3D optical profiles and micro-CT were applied to evaluate the reconstruction of articular cartilage surface and subchondral bone as well as gait analysis to evaluate articular function. Histological staining was performed at 2, 4, and 8 weeks after surgery. Moreover, costal cartilage from transgenic mice with fluorescent markers were transplanted into wild-type mice to observe the in vivo changes of costal chondrocytes.
Results
At 8 weeks after surgery, 3D optical profiles and micro-CT showed that in the graft group, the articular surface and subchondral bone were well preserved. Gait analysis and International Cartilage Repair Society (ICRS) score evaluation showed a good recovery of joint function and histological repair in the graft group. Safranin O staining showed the gradual integration of graft and host tissue. Costal cartilage from transgenic mice with fluorescent markers showed that donor-derived costal chondrocytes turned into osteocytes in the subchondral area of host femur.
Conclusion
Costal cartilage grafting shows both functional and histological repair of osteochondral defect in mice. Graft-derived costal chondrocytes differentiate into osteocytes and contribute to endochondral ossification.
Introduction
Articular cartilage covers and integrates to subchondral bone at the joint surface to reduce friction and to transfer load. Because of its avascular nature, articular cartilage has limited self-repair ability after injury.1,2 Osteochondral defect may cause cartilage degeneration and develop into osteoarthritis without intervention, which will lead to pain and disability and require a joint replacement at late stage.3,4 Many clinical methods have been developed to restore osteochondral defect, such as microfracture, osteochondral autograft transplantation, and cartilage tissue engineering. 5 Fibrocartilage forming, donor-site morbidity, limited cell source, and cost-effectiveness have been major barriers to their clinical use.6,7 Another major concern for all those autologous and tissue-engineered grafts is how they can maintain hyaline cartilage nature while integrate into host tissue to form a transition to subchondral bone, since the stability of osteochondral graft is vital for load bearing. 8
Costal cartilage is the most abundant source of hyaline cartilage in human body, which has similar composition to articular cartilage. 9 Studies have explored costal cartilage as alternative source for cartilage repair.10,11 In tissue engineering, costal chondrocytes have higher cell yield and proliferate faster than articular chondrocytes in vitro and they can be induced into articular cartilage phenotype under appropriate condition.11,12 Our previous clinical study of autologous costal cartilage grafting (ACCG) for osteochondral defect showed that ACCG improved hip function and quality of life in young patients within 3-year follow-up. 13 This promising long-term outcome may be due to the good integration between the recipient and the implanted costal cartilage graft, which was shown in delayed gadolinium-enhanced magnetic resonance imaging (dGEMRIC) of patients’ femoral head. 13 One of the most important characteristics that distinguishes costal cartilage from articular cartilage is that the human costal cartilage develops into highly hypertrophic type during puberty, and ossification of costal cartilage is arrested when reaching this highly hypertrophy status. 14 Moreover, at the end of puberty, the endochondral ossification of costal cartilage starts, takes place slowly, and lasts throughout whole lifespan,15,16 which might made ossification more inducible in costal cartilage by external environment.
Our study designs a clinically relevant costal cartilage graft surgery in mice model. We test its effect to repair osteochondral defect and explored the underlying mechanism of the formation of graft-host interface. We provide evidence that costal cartilage graft surgery form steady graft-host interface. Moreover, we traced down long-term transition of costal chondrocytes and observed that costal chondrocytes differentiate into osteocytes during osteochondral repair in the environment of subchondral area but not articular cartilage.
Methods
Experimental Overview
In this experiment, a total of fifty-seven 8-week-old adult male C57BL/6J mice were used based on the published protocol.17-23 8-week-old C57BL/6J mice is considered adult.21,24 In our study, we used osteochondral defect mice model in trochlear groove, trochlear groove of 8-week-old mice is mature and of adequate size. 18
The costal cartilage graft was either from male C57BL/6J mice of same age or B6/J mGmT mice. Five wild-type mice and 1 transgenic fluorescent mouse were sacrificed at surgery as resource of transplanted costal cartilage. Mice that received surgery were divided into 3 experimental groups (sham, defect, and graft) and sacrificed at 3 time point (2 weeks, 4 weeks, and 8 weeks after surgery). Five mice were used for each experimental group of 2 weeks and 4 weeks, 7 mice for sham group of 8 weeks, 7 mice for defect group of 8 weeks, and 9 mice for graft group of 8 weeks. For recipient of costal cartilage from transgenic mice with fluorescent markers, another 3 mice were used and sacrificed at 8 weeks. 2 weeks, 4 weeks and 8 weeks after surgery, specimens were collected. 8 weeks after surgery, gait analysis was performed on the animals. A postsurgery period of 8 weeks was chosen based on previous publications.19-23,25,26
Animals and Animal Procedures
C57BL/6J mice and B6/J mGmT mice were purchased from GemPharmatech Co., Ltd. All animal experiments were conducted according to the regulations and with approval of the Animal Care and Use Committee of Shanghai Sixth People’s Hospital (DWSY2021-0938). The animals were housed 3 per cage in the animal experimentation facility under a normal 12-hour light-dark cycle with free access to food and water.
Mouse Model of Osteochondral Defect and Costal Cartilage Graft
The mouse model of osteochondral defect was adapted from previous described ones.17,20,27 Eight-week-old male C57BL/6J mice were anesthetized with sodium pentobarbital (1% wt/vol) intraperitoneally injected. The right knees were shaved and disinfected with 70% ethanol. The skin was cut with fine scissors and separated from the underlying tissue by blunt dissection. Medial para-patellar arthrotomy was made and the articular surface of distal femur was exposed by placing the knees at flexion position. The incisions of mice in sham group were sewed up layers by layers. For mice in defect group, a full-depth osteochondral defect (0.5 mm in diameter and 2 mm in depth) was made by a G25 needle at the middle of patellar groove. The syringe needle was placed vertical to the articular surface and then was gently rotated and advanced until its bevel was hidden into the cartilage surface. Then normal saline was used to irrigate the joint cavity to remove debris. The incision was carefully closed as was done in sham group. For murine model of costal cartilage graft, allogenic costal cartilage grafts were harvested from cartilaginous part of fifth to sixth ribs of 8-week-old male C57BL/6J mice. Perichondrium was carefully removed by microscopic tweezers and costal cartilage fragments ready for use were kept in phosphate buffer saline (PBS) on ice. An osteochondral defect was made as described above. A costal cartilage fragment of suitable size was picked out and pressed into the defect, and then trimmed to fit the articular surface. After surgery, all mice were allowed to move freely inside the cages without load restriction.
Gait Analysis
Gait behaviors of mice at 8 weeks after surgery were collected by video-based Catwalk gait analysis system (MobileDatum Co., Ltd), which is composed of an enclosed walkway, a light source, a high-speed camera, and an analysis software. Briefly, each mouse was placed individually in the walkway and allowed to walk freely from one side to the other side after adaptive training. Footprints and walking time were recorded by high-speed camera and gait parameters were analyzed by the Catwalk software. According to previous references, print area, duty cycle, and stride length were adopted as main parameters in this study.28,29
Specimen Preparation
Mice were sacrificed under anesthesia at 2, 4, and 8 weeks after surgery. Specimen of costal cartilage and knee joint were collected and then fixed with 4% paraformaldehyde for 24 hours. For HE, SO, and immunohistochemical staining, specimen was decalcified in 10% ethylenediaminetetraacetic acid (EDTA) in 37°C for 2 days and prepared for 6-μm-thick paraffin embedded sagittal sections. For Goldner’s trichrome staining, specimen was prepared for 20-μm-thick hard tissue sections. For immunofluorescence staining, specimen was also decalcified in 10% EDTA in 37°C for 2 days and then incubated at 4°C with 15% sucrose for 24 hours and 30% sucrose for another 24 hours. They were placed in optimal cutting temperature (OCT) compound (Sakura) and frozen at −20°C until they were sectioned by cryotome (Leica) to generate 10-μm sections.
3D Optical Profiles and Micro-Computed Tomography Analysis (Micro-CT)
For 3D optical profiling, skin and muscles of knee joint were removed, and the articular surface of distal femur was exposed. 3D optical profiles of articular surface were scanned by 3D optical profiler ZeGageTM Pro (ZYGO) to evaluate the articular cartilage smoothness precisely. Graph describing articular surface quantitively was drawn by Microsoft Excel according to the width and depth provided by the results of 3D optical profiles for 3 different samples from sham, defect and graft group respectively. For micro-CT, knee joints were stored in 70% ethanol after fixed. All samples were scanned at 9 µm resolution by a micro-CT scanner (SkyScan 1176, Kontich). Three-dimensional reconstructed images were visualized using CTVox software (Bruker MicroCT, Kontich). The sagittal images of the knee joints were obtained by DataViewer (Bruker MicroCT, Kontich).
Histology
The paraffin embedded sections of knee joints were stained with hematoxylin–eosin (HE) and safranin O (SO)/fast green. The cellularity and morphology of cartilage and subchondral bone were examined by 2 independent experienced researchers in a blinded manner. The histological assessment of cartilage defect was evaluated by International Cartilage Repair Society (ICRS) scores. 30
Immunohistochemical Staining
For immunohistochemistry (IHC) staining, sections were treated with citric acid antigen repair solution (Ribiology) in micro-oven, and then treated with 3% H2O2 at room temperature for 25 minutes. Nonspecific binding sites was blocked by 10% bovine serum albumin for 30 min at room temperature. The sections were then incubated with the primary antibody (Cell signaling technology) overnight at 4°C. Finally, the sections were incubated with biotinylated secondary antibody (DAKO) and visualized by DAB solution (DAKO) for IHC. The sections were also counterstained with hematoxylin. For Goldner’s trichrome staining, the sections were embedded in collodion after deplasticization by 2-methoxyethylacetate. Collodion were fixed by 80% ethanol. Then the sections were stained by iron-hematoxylin for 15 min, acid fuchsin for 15 minutes, Orange G for 5 minutes, and light green for 10 minutes. Xylene was used to remove the collodion.
Immunofluorescence Staining
The frozen sections were treated with 0.1% Triton X-100 for 10 minutes and blocked by 3% bovine serum albumin (BSA) for 1 hour. The sections were incubated with goat anti-CD31 (R&D) in 0.2% BSA at 4°C overnight. Then they were incubated with donkey anti-goat secondary antibody in 0.2% BSA at room temperature for 2 hours followed by phalloidin 647 (Invitrogen, 1:250) in PBS at room temperature for 1 hour and DAPI (Servicebio) for 10 minutes. The stained tissue sections were observed and imaged by LSM710 microscope (Zeiss). Osteocytes dendrites were recorded by overexposure of phalloidin.
Statistical Analysis
Data were analyzed and presented using GraphPad Prism version 8.0. All results were shown as means ± SEM of independent individuals. Statistical significances were calculated by Student’s t test for 2 group comparisons and analysis of variance (ANOVA) for multiple group comparisons as indicated in figure legends. p-values were considered significant at p < .05.
Results
Establish a Costal Cartilage Graft Mouse Model to Repair Articular Osteochondral Defect
Taking advantage of costal cartilage, we designed a costal cartilage graft mouse model to study the endochondral ossification process during the repairment of articular osteochondral defect (
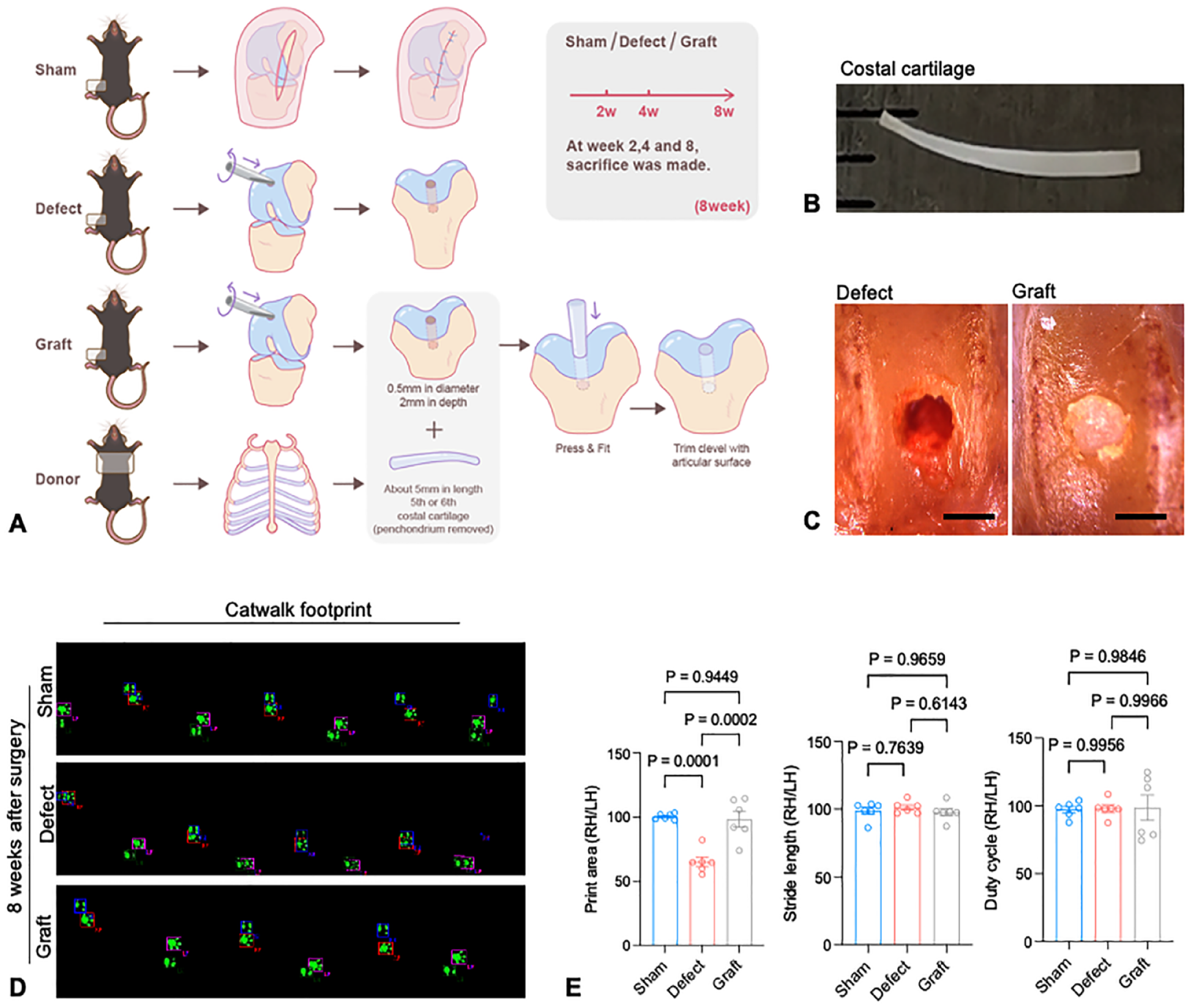
Costal cartilage graft in C57BL/6J mice model. (
To assess the repair effect of costal cartilage graft on knee joint function, gait analysis was carried out by Catwalk system at 8 weeks after surgery. The defect group showed a decrease in print area compared with sham group, indicating a weightbearing shift from the defected right knee to left knee (
Repaired Cartilage Surface and Subchondral Area
To further study repair effect of costal cartilage graft in the defected knee joint, the articular surface of patellar groove was scanned by 3D optical profiles. The articular surface in the graft group remains smooth and continuous at 8 weeks as shown in 3D optical profiles (
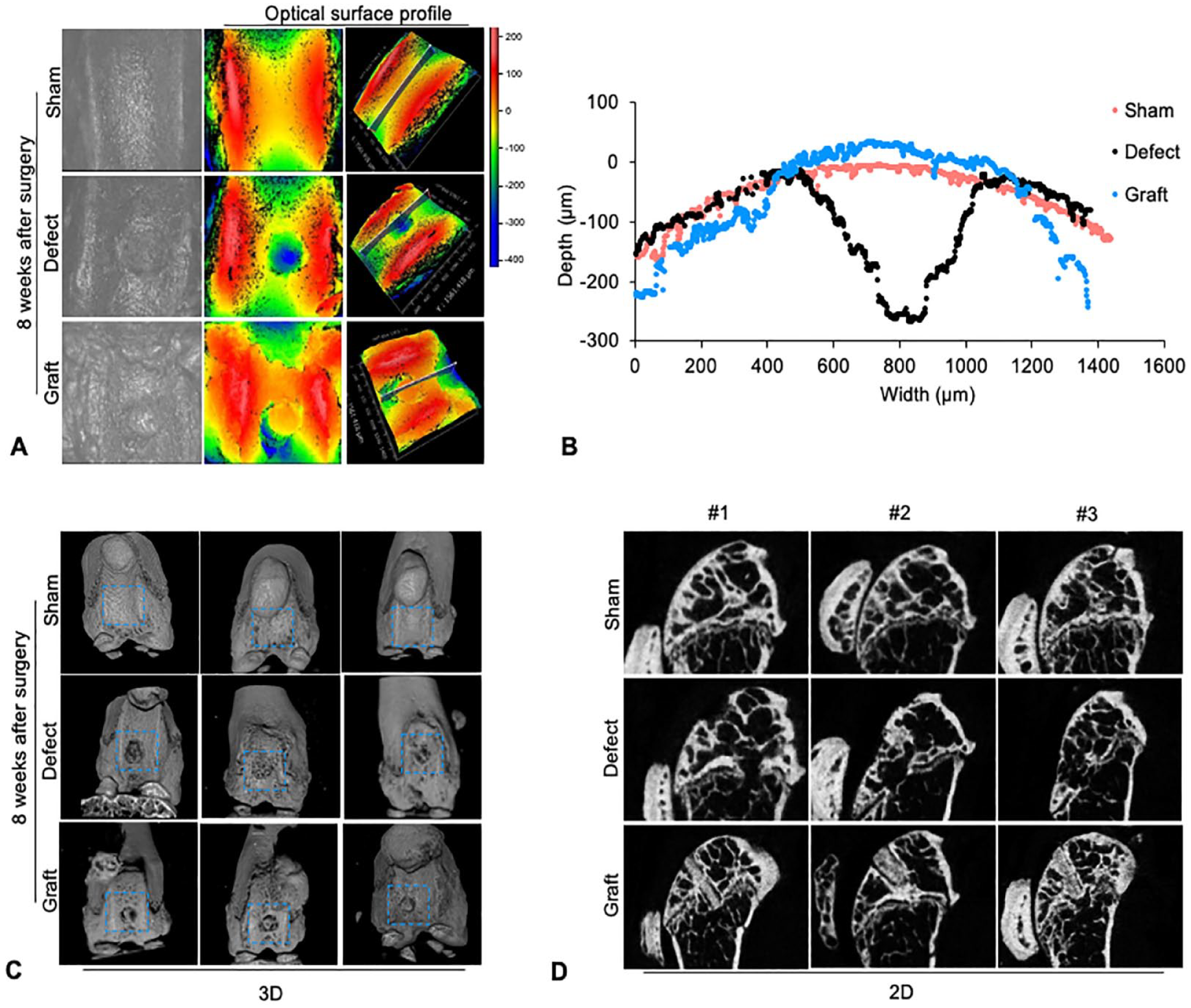
Costal cartilage graft restores both articular cartilage surface and subchondral area. (
To explore the nature of repaired cartilage and subchondral bone, histological analysis was performed. At 8 weeks after surgery, HE and SO staining demonstrated that in graft group, cartilage defect was fitted with costal cartilage graft, which show similar quality to articular cartilage (
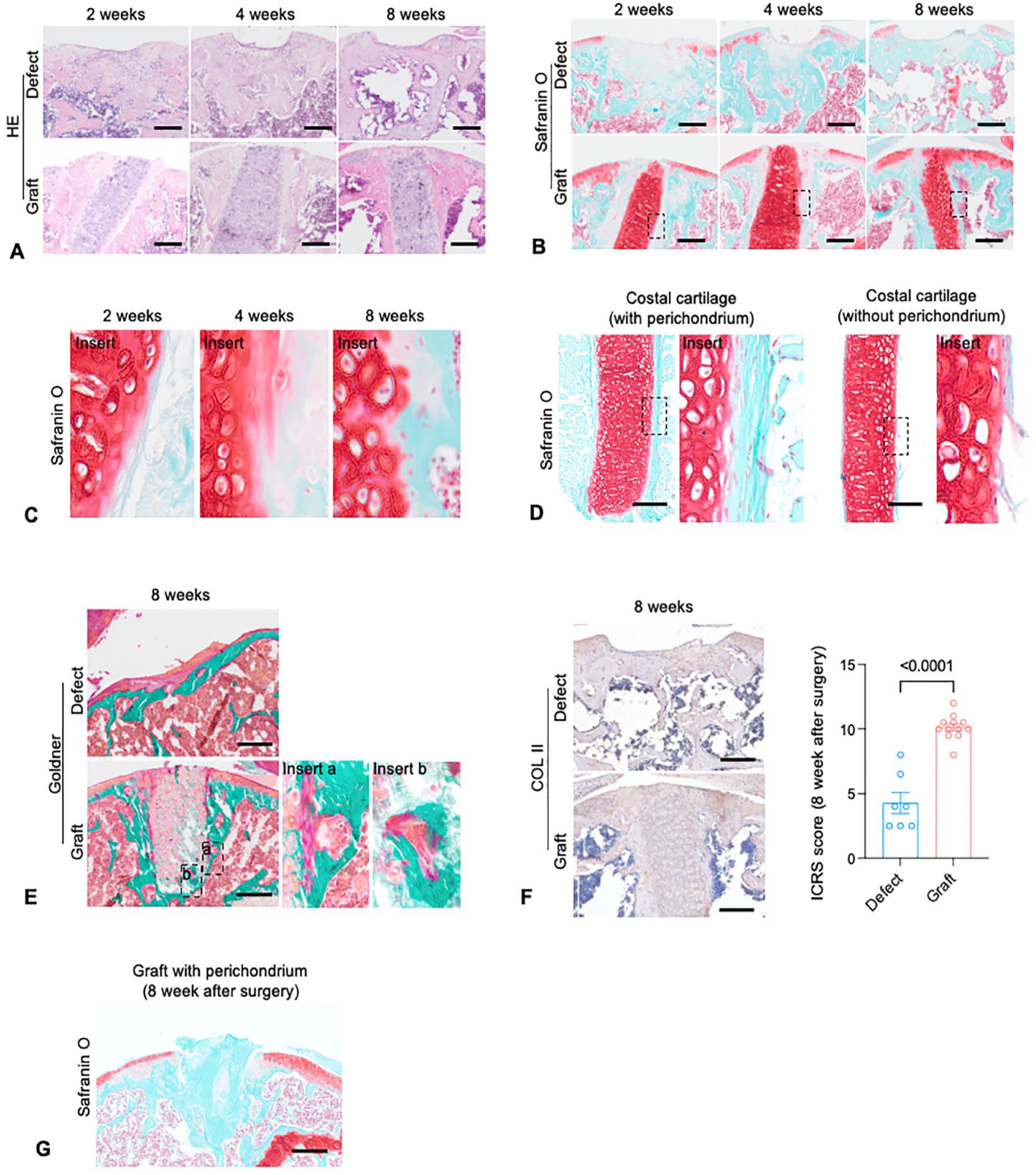
Formation of graft-cartilage and graft-bone interfaces in costal cartilage graft model. (
Formation of Graft-Cartilage and Graft-Bone Interfaces
In addition to overall repair effect, a stable graft-cartilage interface and graft-bone interface gradually formed from 2 weeks to 8 weeks after surgery (
Intriguingly, at subchondral area in graft group, new osteoid was found between mineralized bone and costal cartilage as transition, which was recognized by Goldner’s trichrome staining (
Ossification of Costal Cartilage at Graft-Bone Interface
To elucidate the mechanism behind biological interface formation of costal cartilage graft, mGmT transgenic mice were used as donor of costal cartilage to trace the fate of costal chondrocytes in graft. At 8 weeks after surgery, confocal imaging on serial sections observed maintained nuclei integrity in most of the chondrocytes, indicating most cells from graft were survived (
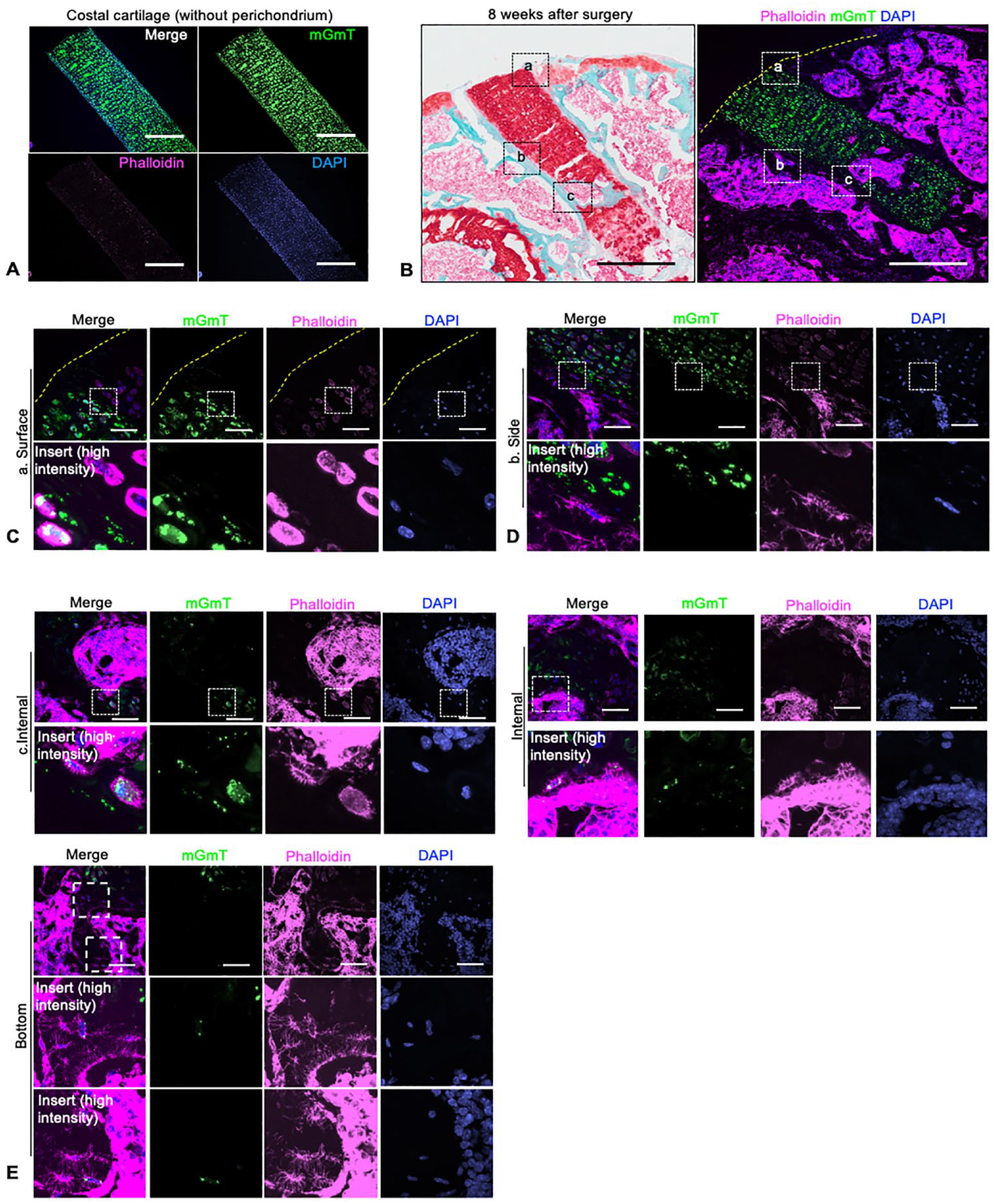
Chondrocytes in graft costal turn into osteocytes at graft-bone interface, but not in graft-cartilage interface. (
At the graft-bone interface, like histology results, fluorescence results demonstrated that bone and bone marrow like structures formed surround the graft at subchondral area (
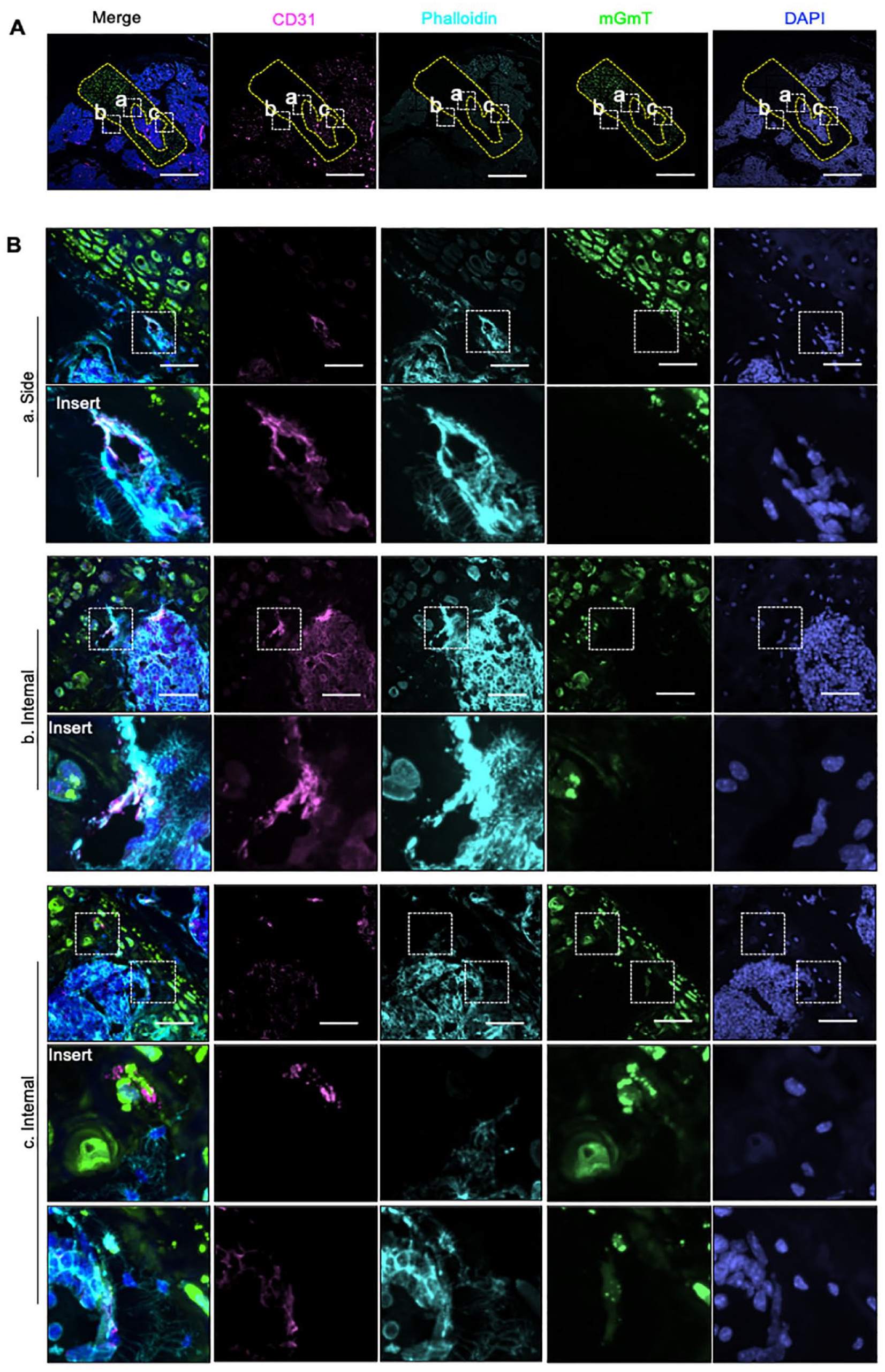
Blood vessels at the graft-bone interface mediate the ossification of costal cartilage graft. (
Discussion
Osteochondral defect left untreated leads to pain and disability of patients and has placed heavy economic burden on society. The compositional similarity of articular cartilage and costal cartilage as well as the hypertrophic nature of costal cartilage make costal cartilage an ideal alternative for osteochondral repair and studying the late differentiation of chondrocytes under different conditions.11,31 Thus, we proposed a costal cartilage transplantation model, in which hypertrophic costal cartilage graft is transplanted into osteochondral defect in mice knee (
The malleable nature of costal cartilage made it a 1-step repair strategy for the multi-layered osteochondral complex. On one hand, the endochondral ossification highly relies on the intrusion of vascular structure, which was enriched in subchondral area.32-34 In the subchondral bone, we observed costal cartilage penetrated by vessels (
Our study on transdifferentiation of hypertrophic chondrocytes during osteochondral repair besides endochondral ossification during development provides insight for clinical treatment. Establishing a stable interface between graft and host has long been a problem for osteochondral repair.8,36 The osteogenic potential of hypertrophic chondrocytes makes it a good candidate for osteochondral repair since our study show the changes of the graft are subject to its environment. The graft undergoes endochondral ossification under the influence of subchondral area, so that the graft could integrate into subchondral area and form a biological interface, while the graft near articular surface doesn’t lose its cartilage nature, which is appropriate for cartilage repair.
Further, the establishment of mouse cartilage transplant model enables us to take advantage of transgenic mice for characterizing the molecular mechanisms of endochondral ossification in osteochondral repair. Numerous studies have reported the molecular mechanisms of chondrocyte maturation and hypertrophy.37-40 But only recently a few studies focus on the intrinsic mechanisms of transdifferentiation of hypertrophic chondrocytes into osteocytes.41,42 The molecular basis and driving force of osteogenic transdifferentiation have not been fully elucidated yet. Intriguingly, among those studies, diabetic pregnancy of mice can interfere with the chondrocytes transdifferentiation through NF-κB signaling in the situation of embryonic long-bone development, 42 which indicates that metabolic environment might play a role. Based on our findings, the presence of subchondral area including trabecular bone, bone marrow cells and vasculature but not articular cartilage induce the transdifferentiation of hypertrophic chondrocytes to osteocytes in vivo.
This study has several limitations. First, many factors affect osteochondral repair, such as age, sex, and degree or location of osteochondral defect, our study only investigate adult male animals of 8-week-old and the osteochondral defect of non-weightbearing area. Although 8-week-old mice were widely used as adult mice to study cartilage repair,17-23 mice over 3-month-old is generally considered skeletally mature in previous study.43-45 8-week-old mice haven’t achieved peak bone parameters such as bone density and bone strength, which might affect the repair effect. Further study will be conducted on skeletally mature mice to verify our conclusion. Secondly, since the costal cartilage have the potential of endochondral ossification, a longer period than 8 weeks postsurgery might be needed for observation of repair effect and outcome of the graft. Further study with longer observation will be conducted to explore the mechanism determining the differentiation of costal cartilage. Moreover, we used mice as the animal model. As the walking load differs between human legs and mice hind limb, the process of osteochondral repair may be different from that in humans.
In conclusion, costal cartilage grafting shows both functional and histological repair of osteochondral defect in mice. Graft-derived costal chondrocytes differentiate into osteocytes and contribute to endochondral ossification. Our findings support the idea that costal cartilage graft as feasible treatment for osteochondral defect and show how cartilaginous graft dynamically adapt to local environment and add to the knowledge of the mechanisms underlying osteochondral repair and graft-host interaction.
Footnotes
Acknowledgments and Funding
This work was supported by National Natural Science Foundation of China (81820108020 to C.Z.; 82002339 to J.G.) and Shanghai Frontiers Science Center of Degeneration and Regeneration in Skeletal System (BJ1-9000-22-4002).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
All animal experiments were conducted according to the regulations and with approval of the Animal Care and Use Committee of Shanghai Sixth People’s Hospital.
