Abstract
Background:
For combined reconstruction of both the anterior cruciate ligament (ACL) and the posterior cruciate ligament (PCL), there is no consensus regarding which graft should be tensioned and fixed first.
Purpose:
The purpose of this study was to determine which sequence of graft tensioning and fixation better restores normal knee kinematics. The hypothesis was that ACL-first fixation would more closely restore normal knee kinematics, graft force, and the tibiofemoral orientation in the neutral (resting) position compared with PCL-first fixation.
Study Design:
Controlled laboratory study.
Methods:
A total of 15 unpaired human cadaveric knees were examined using a robotic testing system under the following 4 conditions: (1) 89.0-N anterior tibial load at different knee angles; (2) 89.0-N posterior tibial load at different knee angles; (3) combined rotational 7.0-N·m valgus and 5.0-N·m internal rotation load (simulated pivot shift) at 0°, 15°, and 30° of flexion; and (4) 5.0-N·m external rotation load at 0°, 15°, and 30° of flexion. The 4 evaluated knee states were (1) intact ACL and PCL (intact), (2) ACL and PCL deficient (deficient), (3) combined anatomic ACL-PCL reconstruction fixing the ACL first (ACL-first), and (4) combined anatomic ACL-PCL reconstruction fixing the PCL first (PCL-first). A 9.0 mm–diameter quadriceps tendon autograft was used for the ACL graft, tensioned with 40.0 N at 30° of flexion. A 9.5 mm–diameter hamstring tendon autograft (gracilis and semitendinosus, quadrupled loop, and augmented with an additional allograft strand if needed), tensioned with 40.0 N at 90° of flexion, was used for the PCL graft.
Results:
There were no statistically significant differences between ACL-first and PCL-first fixation regarding knee kinematics. ACL-first fixation restored anterior tibial translation to the intact state at all tested knee angles, while PCL-first fixation showed higher anterior tibial translation than the intact state at 90° of flexion (9.05 ± 3.05 and 5.87 ± 2.40 mm, respectively; P = .018). Neither sequence restored posterior tibial translation to the intact state at 30°, 60°, and 90° of flexion. At 15° of flexion, PCL-first fixation restored posterior tibial translation to the intact state, whereas ACL-first fixation did not.
Conclusion:
There were no differences in knee laxity between ACL-first and PCL-first fixation with the ACL graft fixed at 30° and the PCL graft fixed at 90°.
Clinical Relevance:
This study showed that there was no evidence to support the use of one tensioning sequence over the other in single-stage multiligament knee reconstruction.
Multiligament knee injuries represent around 0.2% of all orthopaedic injuries and 11% to 20% of all knee ligament sprains. 10,15,47 They occur mainly as a result of high-energy trauma, but they can also be the result of low-energy injury mechanisms, especially in less active people who have a higher body mass index. 4,40,46,62 Being a relatively uncommon injury, there is a paucity of evidence regarding the treatment aspects of this wide spectrum of injury patterns.
A combined anterior cruciate ligament (ACL)–posterior cruciate ligament (PCL) injury presents one of the most common multiligament injury patterns. 31 The Schenck classification of knee dislocations (KDs) is most widely used for multiligament knee injuries, as it is both the most relevant to surgical management and has the highest predictive value when it comes to associated neurovascular injuries. 38,42 Although a combined ACL-PCL injury (classified as KD II) only represents only about 5% of all multiligament knee injuries, injuries involving at least the ACL and PCL (ie, both cruciate ligaments torn in isolation or in conjunction with 1 or both of the collateral complexes and/or a periarticular fracture; KD II-V) represent almost 99% of all knee dislocations. 40 The management of these injuries depends on many factors, such as associated neurovascular compromise and the patient’s preinjury activity level. 23,26,60 Most bicruciate ligament injuries are managed by simultaneous reconstruction of both ligaments during a single surgical procedure. 6,24,34,44,66 The surgical techniques used vary widely among surgeons and are generally determined by the surgeon’s preference for isolated ligament reconstruction. As a result, the literature shows a wide range of reconstruction techniques as to graft type 57 (allografts, autografts, artificial ligaments), number of bundles reconstructed (single vs double bundle for either or both cruciate ligaments), knee flexion angle during graft tensioning, and the order in which the grafts are fixed. 16,23,25,66
In clinical studies looking at the outcomes of multiligament knee reconstruction, most authors seem to prefer to fix the PCL first, usually at around 90° of flexion. 6,16,18,43,55 The ACL graft is often fixed afterward at a low knee flexion angle (0°-30°). However, there is a lack of underlying research to support this graft tensioning and fixation preference. The few biomechanical studies available are heterogeneous with regard to the methodology such as graft type, surgical technique, tensioning method, force applied during graft tensioning, fixation angle, and testing system that is used. 19,37,39,67 In addition, most studies only measured displacement in a single direction, examined only 1 knee flexion angle, or simulated a physical examination test. 19,37,39
The purpose of the current study was to determine which sequence of graft tensioning better restores normal knee kinematics during single-stage combined ACL-PCL reconstruction. The secondary aims of the study were to determine (1) which sequence of graft tensioning and fixation would give an in situ graft force closest to that of the native cruciate ligaments and (2) which graft fixation sequence more closely reproduces the knee’s neutral tibiofemoral position. The hypothesis was that tensioning and fixation of the ACL graft first would more closely restore normal knee kinematics, in situ graft force, and the tibiofemoral orientation in the neutral (resting) position compared with fixing the PCL graft first.
Methods
Institutional review board approval was granted for this human cadaveric study, and all knee specimens were procured from institutionally approved tissue suppliers. Included were 15 unpaired, fresh-frozen cadaveric specimens of human knees, 9 male and 6 female, with a mean age of 59 years (range, 48-67 years). All specimens were stored at –20°C and then thawed for 24 to 30 hours before testing. A gross physical examination of each specimen was conducted to check for malalignment and instability, followed by diagnostic arthroscopic surgery to confirm intact cruciate ligaments, menisci, and articular cartilage. All knee specimens were prepared by removing all soft tissue 10 cm from the knee joint. The femur and tibia were then potted in epoxy and mounted in custom aluminum cylinders.
Surgical Procedure
Single-stage, arthroscopic, anatomic single-bundle ACL-PCL reconstruction was performed. For the ligament-deficient state, the native ACL and PCL were resected, and the remnants of the ligaments were debrided, with the insertion sites marked, to create an ACL- and PCL-deficient knee.
Anatomic ACL reconstruction was performed by creating 9-mm tibial and femoral tunnels in the center of the ACL insertion site on both the tibia and the femur. A 9.5-mm femoral tunnel was placed at the center of the PCL 41 footprint using an inside-out freehand technique through the anterolateral portal. Fluoroscopy (BV Pulsera Mobile C-arm; Philips) was used along with a 70° scope and a guide pin to mark the location of the PCL insertion site on the tibia, 5 and then a 9.5-mm tibial tunnel was drilled.
Soft tissue autografts 57 were harvested from the tested knee, with the quadriceps tendon used for ACL 3,11 reconstruction (sized to 9.0 mm), while for the PCL, a hamstring tendon autograft (semitendinosus and gracilis, quadrupled loop) was used in the majority of cases, and an additional allograft strand was added, if needed, to reconstruct the PCL 36,51 (sized to 9.5 mm) (Figure 1).

Arthroscopic view (through the anterolateral portal) of a left knee specimen: (A) intact, (B) anterior cruciate ligament (ACL)–posterior cruciate ligament (PCL) deficient, and (C) ACL-PCL reconstruction. LFC, lateral femoral condyle; MFC, medial femoral condyle. ** indicates ACL ligament/graft ; * indicates PCL ligament/graft in the figure.
For preparation of the hamstring tendon graft, the gracilis and semitendinosus strands were looped through a closed-loop extracortical button suspensory device 3,11,45,53 and then doubled and whip-stitched together using braided polyester No. 2 suture (Arthrex). The full-thickness quadriceps tendon was split into superficial and deep layers from distal to proximal, keeping the most proximal 1 cm of the graft conjoined. Then, the closed-loop extracortical button suspensory device was passed between the split layers and securely sutured to the closed loop, and both layers were whip-stitched together using braided polyester No. 2 suture. Both grafts were fixed first on the femoral side with the extracortical button, then each tensioned at 40 N using a tensiometer (Meira) at 30° of flexion for the ACL graft and 90° of flexion for the PCL graft, and fixed on the tibia with a screw and spike washer. 2,35,52,56,59,64,67
All specimens were randomized to have either the ACL graft fixed first and PCL graft fixed second (ACL-first) or the PCL graft fixed first and the ACL graft fixed second (PCL-first). After robotic testing of the specimens in their randomized fixation sequence, they all underwent graft retensioning and refixation using the opposite graft fixation sequence and subsequently underwent robotic retesting.
Robotic Testing
All knees were tested on a robotic testing system as previously described, 1,15,63 consisting of a robotic manipulator (CASPAR Stäubli; Orto MAQUET) with the robotic arm having ±0.02 mm of motion repeatability at each joint. The universal force/moment sensor (Model 4015; JR3) has a force and moment accuracy of ±0.2 N and ±0.1 N·m, respectively, as provided by the manufacturer. A custom MATLAB programming environment with a multitask operating system (MathWorks) was used to perform control and data acquisitions; the computer program controlled the displacement and measured the force/moment in all 6 degrees of freedom while performing data acquisition. The passive path of the intact knee was determined by the robotic testing system from full extension (FE) to 90°, in 0.5° increments, which was performed by minimizing the applied forces and moments about the joint at each increment. 49 The robotic testing system used a Cartesian coordinate system with defined axes in the anteroposterior, mediolateral, and proximodistal directions of the tibia, based on the method described by Fujie et al. 20
Each knee was tested in the intact, ACL- and PCL-deficient (deficient), ACL-first, and PCL-first states under the following loads:
89.0-N anterior tibial load, 13
combined rotational 7.0-N·m valgus 27 and 5.0-N·m internal rotation load (simulated pivot shift), 29 and
5.0-N·m external rotation load.
The anterior tibial load and posterior tibial load were applied at FE and at 15°, 30°, 60°, and 90° of flexion, while the simulated pivot-shift and external rotation loads were applied at FE and at 15° and 30° of flexion. Because the passive path provides a reference position at each flexion angle to which external loads are applied, the passive path was determined for the intact knee and after each fixation sequence. Knee kinematics was calculated by comparing the tibial position after each loading condition with the reference position.
By removing the ACL, PCL, and grafts, in situ tissue forces were determined using the principle of superposition, 33,48 in which the difference in the force data recorded during the same path of motion of the intact ACL/PCL versus the cut ACL/PCL was used to calculate the in situ force for each. The same was applied to the soft tissue grafts used for ACL reconstruction and PCL reconstruction.
Position Measurement
To measure the relative tibiofemoral position of each knee state at different flexion angles, we used a 3-dimensional digitizer (FaroArm Platinum; Faro Technologies) with a manufacturer-reported accuracy of 0.025 mm. 67 The FaroArm unit was mounted to the base of the robotic testing system, where the femur of the knee joint was mounted and did not move. Registration screws were placed on the femur and tibia according to the method of Wang et al, 61 with 3 screws in the anterior aspect of the metaphyseal-diaphyseal portion of the distal femur and 3 screws in the anterior aspect of the tibia, distal to the tibial tubercle. The screws in the bones of the femur and tibia were digitized before (in the neutral position) and after each kinematic load to measure displacement of the tibia (mounted to the end effector of the robotic testing system) relative to the femur (fixed to the robotic testing system base). The position of the femoral and tibial screws was measured and used to calculate the relative position of the tibia to the femur, which allowed any change in the neutral position from the intact knee to be determined.
Statistical Analysis
With knee state as the factor, the following measurements were compared using 1-way repeated-measures analysis of variance at each flexion angle: knee kinematics between intact, deficient, ACL-first, and PCL-first states; the in situ force in the ACL and PCL native tissues and grafts; and the relative tibiofemoral position. All results are reported as the mean and standard deviation, and statistical significance was set at P < .05. A Bonferroni correction was applied to account for the multiple comparisons (SPSS Version 25; IBM). The post hoc P values were adjusted in the software so that significance remained at P < .05.
An a priori sample size calculation was performed using data from a previous similar study, 1 which indicated that a sample size of 15 specimens would be sufficient to achieve a power of >0.9 for the primary outcomes. A post hoc power analysis was also conducted using G*Power 17 (Version 3.1.9.6; Heinrich Heine University Düsseldorf) to confirm this calculation. Indeed, with the 15 specimens tested, all kinematic outcomes achieved a power of 0.99.
Results
There was no statistical difference in anterior tibial translation (ATT) under anterior tibial loading between the ACL-first state and the intact state at all flexion angles. In the PCL-first state, there was also no statistically significant difference compared with the intact state, except at 90° of flexion (9.05 ± 3.05 and 5.87 ± 2.40 mm , respectively; P = .018) (Figure 2). The approximate difference in ATT and posterior tibial translation (PTT) between the intact and fixation states could be up to 3 mm. There was no statistical difference in ATT or PTT between the 2 fixation sequences.
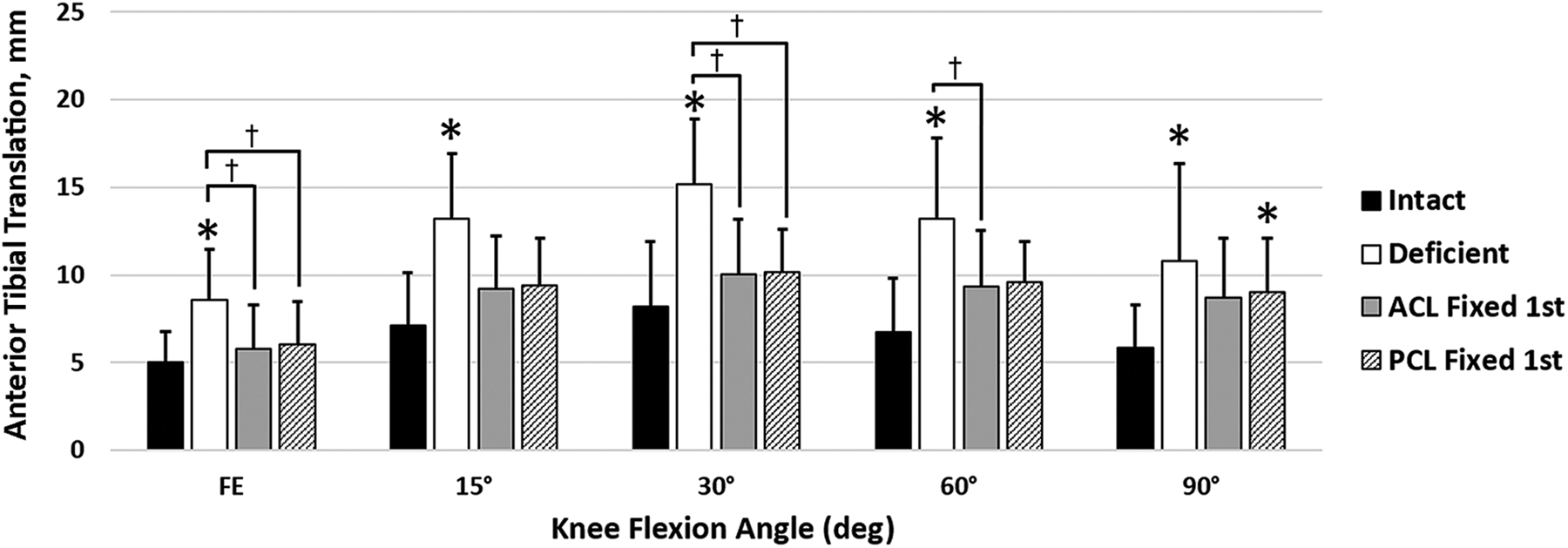
Anterior tibial translation as a function of knee state and flexion angle. *P < .05 vs intact. † P < .05 between groups. ACL, anterior cruciate ligament; FE, full extension; PCL, posterior cruciate ligament.
Under posterior tibial loading, PTT with PCL-first fixation was not significantly different from the intact knee at FE and 15° of flexion, but there was a difference at 30° (P = .001), 60° (P < .001), and 90° (P < .001), with PCL-first fixation resulting in more PTT than the intact state. ACL-first fixation resulted in increased PTT compared with the intact state at all flexion angles, except at FE in which there was no difference (Figure 3).
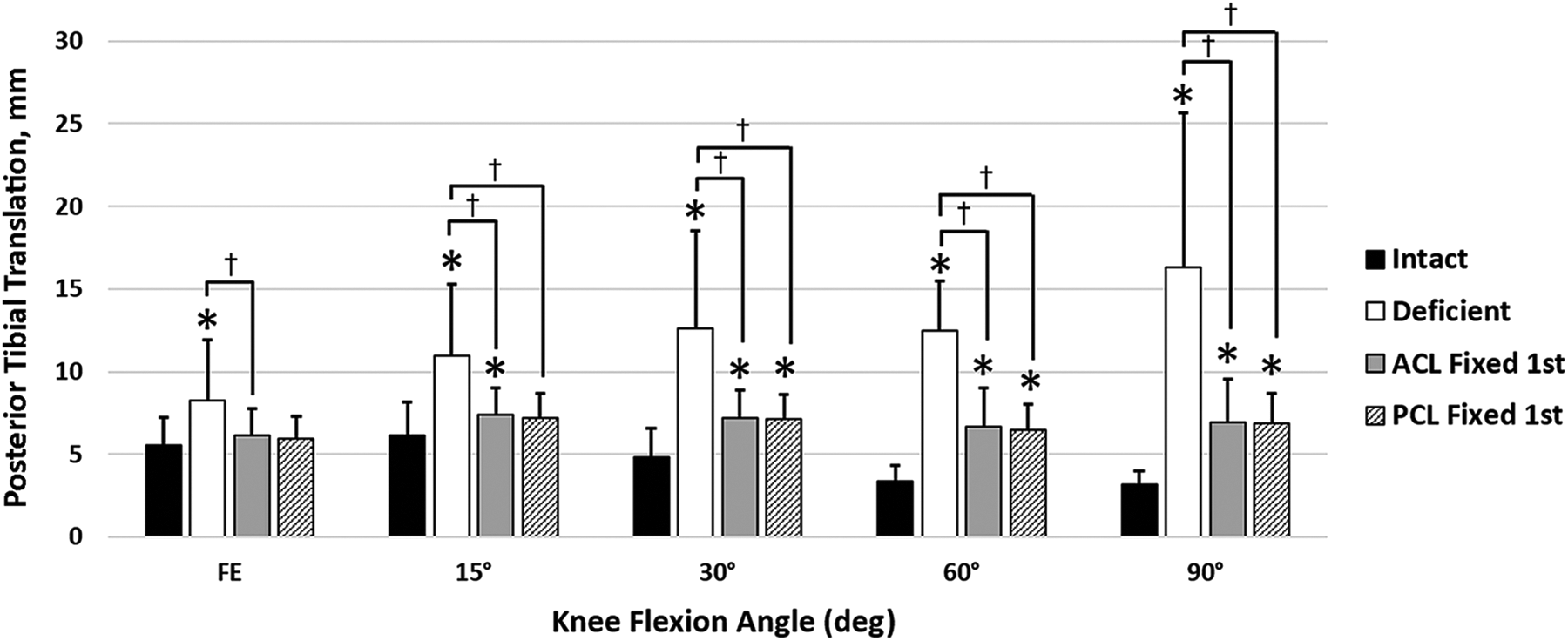
Posterior tibial translation as a function of knee state and flexion angle. *P < .05 vs intact. † P < .05 between groups. ACL, anterior cruciate ligament; FE, full extension; PCL, posterior cruciate ligament.
Under a simulated pivot shift, ATT in both fixation sequences was statistically different compared with the intact knee at all the tested flexion angles: ACL-first fixation at FE (P = .002), 15° (P < .001), and 30° (P < .001), and PCL-first fixation at FE (P = .003), 15° (P = .001), and 30° (P = .004). Other than compared with the intact state, there were no other significant differences (Figure 4).
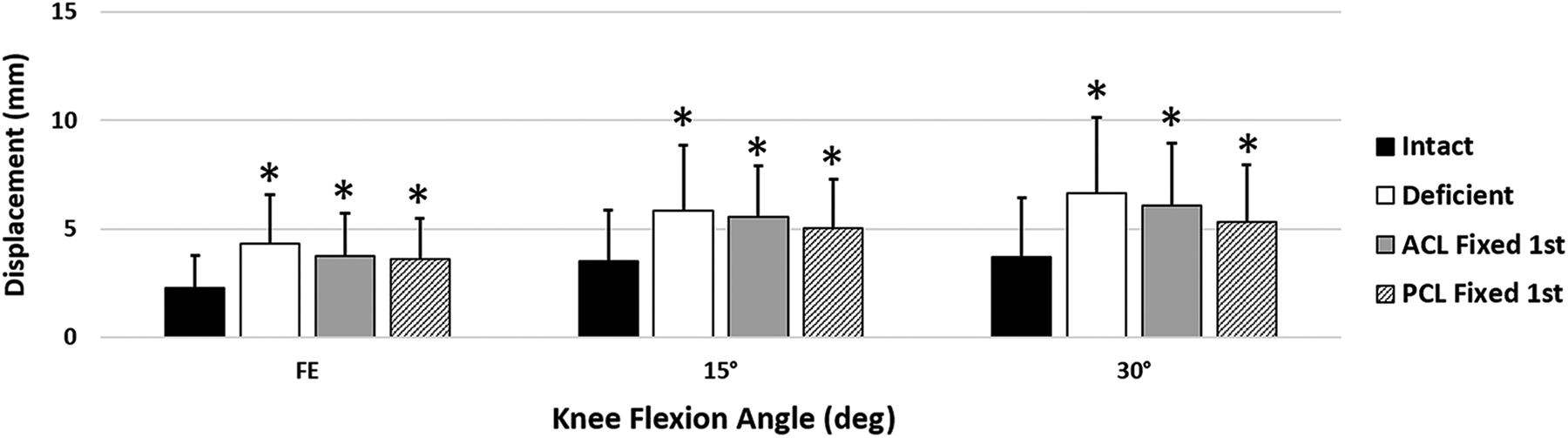
Anterior tibial displacement under simulated pivot-shift loading as a function of knee state and flexion angle. *P < .05 vs intact. ACL, anterior cruciate ligament; FE, full extension; PCL, posterior cruciate ligament.
No statistically significant differences were found between the intact, ACL-first, and PCL-first states for external rotation (Figure 5).

External rotation as a function of knee state and flexion angle. † P < .05 between groups. ACL, anterior cruciate ligament; FE, full extension; PCL, posterior cruciate ligament.
Under an anterior tibial load, in situ ACL graft forces in both fixation sequences were not statistically different from the native ACL force, except at 90° of flexion in which PCL-first fixation had a significantly lower ACL graft force than the native ACL (P = .039). There were no significant differences between both fixation sequences and the native knee, or between the fixation sequences, for in situ force in the PCL graft under anterior tibial loading (Figure 6).

Anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL) graft forces under anterior tibial loading as a function of knee state and flexion angle. *P < .05 vs intact. FE, full extension.
Under a posterior tibial load, in situ ACL graft forces in both fixation sequences, ACL-first and PCL-first fixation, were not statistically different from the native ACL force (Figure 7). However, the in situ force in the PCL graft was significantly lower than the native PCL at all flexion angles with ACL-first fixation: FE (P = .001), 15° (P < .001), 30° (P < .001), 60° (P < .001), and 90° (P < .001). With PCL-first fixation, the in situ force in the PCL graft was lower than the intact state at all flexion angles, except at FE (P = .227): 15° (P = .019), 30° (P = .001), 60° (P = .003), and 90° (P = .001) (Figure 7).

Anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL) graft forces under posterior tibial loading as a function of knee state and flexion angle. *P < .05 vs intact. FE, full extension.
There was a large variation in the tibiofemoral resting position, and no statistically significant differences could be found between both sequences (ie, ACL-first and PCL-first fixation) (Figure 8).

Relative change in the unloaded, neutral tibiofemoral position compared to the intact state (shift) for anterior cruciate ligament (ACL)–first and posterior cruciate ligament (PCL)–first fixation at each flexion angle. FE, full extension. (*P <.05)
Discussion
The most important finding of the present study was that there were no statistically significant differences in the order of ACL-first or PCL-first fixation with regard to knee kinematics. This study aimed to determine the effect of tensioning and fixing the ACL graft first versus PCL graft first in single-stage multiligament knee reconstruction. ACL-first fixation restored ATT to the intact state at all tested knee flexion angles, while PCL-first fixation showed higher ATT than the intact knee only at 90° of flexion. Both graft fixation sequences failed to restore PTT to the intact state at 30°, 60°, and 90° of flexion. PCL-first fixation restored PTT to the intact state at 15° of flexion, whereas ACL-first fixation did not. In addition, neither sequence restored ATT under simulated pivot-shift loading.
These findings are largely consistent with the existing literature. Residual posterior laxity is not uncommon after PCL reconstruction in basic science studies as well as the clinical setting, both with isolated PCL reconstruction and with PCL reconstruction as part of multiligament knee surgery. 22,30,32,39 Lenschow et al 32 assessed knee joint kinematics and in situ forces after isolated anatomic PCL reconstruction using a robotic testing system. They showed that although reconstruction of the PCL resulted in reduced PTT, it neither restored kinematics to that of the PCL-intact knee nor restored the in situ force to that of the native PCL. 32 Clinically, the results are similar, with at least 1-grade difference between the native knee and the reconstructed knee for PTT. 22,30 The graft used for PCL reconstruction in the present study was a hamstring tendon autograft with a diameter of 9.5 mm, and a single-bundle technique was used to ensure that the graft type and size were reproducible and identical in all the tested specimens. However, in clinical practice, the use of a larger graft size (ie, when allograft tissue is available) with additional graft fixation and the use of a double-bundle reconstruction technique are not uncommon. 5,7,9,36,66 Although these technical modifications may result in knee laxity measurements closer to those of the native knee, they may still have varying degrees of residual PTT. In addition, these options may not be readily available depending on the geographic location, are associated with higher costs, and are more technically challenging, especially in the setting of multiligament knee reconstruction. 8,21,50
The present study also found residual laxity during simulated pivot-shift testing, regardless of the order of graft tensioning and fixation. Residual knee laxity with only ACL reconstruction under pivot-shift loading is also commonly seen in existing biomechanical studies 28,39,52,67 as well as clinical studies, 12,54,58 but the reason for this is still poorly understood.
For the ACL graft in the present study, a 9.0 mm–diameter graft was used in all specimens to allow for a reproducible graft size across specimens. In addition, the specimens in this study were older (mean, 59 years) than patients who generally undergo knee ligament surgery as well as older than the donor age for allograft material routinely used.
The present study showed that ACL-first fixation had an in situ ACL graft force that was not statistically different from the native ACL force, which is consistent with the findings of Markolf et al. 37 In their study, the authors prioritized normal ACL graft force over normal PCL graft force (because of the known difficulty in restoring both). They found no difference in knee kinematics and graft forces between ACL-first and PCL-first graft tensioning and fixation. Similar to the present study, they were unable to find a tensioning and fixation sequence that was superior at restoring anteroposterior laxity and graft forces throughout the entire knee range of motion. 37
At the lower knee flexion angles, the present study showed that ACL-first fixation was not different from PCL-first fixation with regard to the anteroposterior resting position of the tibia relative to the femur. This is different from the study by Franciozi et al, 19 who found that PCL-first fixation, after a simultaneous tensioning protocol, was able to achieve 30% less combined anteroposterior translation. However, there are methodological differences between the 2 studies when it comes to the graft tensioning protocol, loads applied during ATT and PTT, and graft sizes. 19 A porcine study by Zheng et al 67 assessed 5 different graft tensioning and fixation combinations during simultaneous ACL and PCL reconstruction, and the authors were also unable to find a single method that best restored the anteroposterior resting position to that of the intact knee. In another study by Moatshe et al, 39 the effect of different tensioning sequences on the neutral tibiofemoral orientation during combined ACL, PCL, and posterolateral corner reconstruction was assessed. Overall, 4 different orders of graft tensioning were tested, but no single method restored normal knee kinematics. 39
Limitations
This study has several limitations. This was a time-zero study, and it is not known how knee laxity and in situ forces in the grafts change as tissue undergoes healing in the bone tunnels and ligamentization. Tensioning and retensioning can induce some inherent laxity in the graft; this laxity is removed when the graft is retensioned. Other limitations were that this study did not evaluate other autograft types and size options and did not evaluate other possible fixation angles and tensions. In this study, the different graft fixation sequences resulted in different resting positions of the knee joint, and further investigation of graft tensioning is warranted to better restore the native knee position. Finally, there are several different types of fixation used for grafts on both the tibia and the femur; however, this study only investigated 1 type of fixation.
Conclusion
This cadaveric, biomechanical, single-stage ACL-PCL reconstruction study found no differences in knee laxity between fixing the ACL or PCL graft first to support the use of one tensioning sequence over the other with the ACL graft fixed at 30° and the PCL graft fixed at 90°.
Footnotes
Acknowledgment
In memoriam and appreciation of Dr Freddie H. Fu (1950-2021).
Final revision submitted April 26, 2022; accepted May 17, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding was received from the Albert B. Ferguson Jr MD Orthopaedic Fund of the Pittsburgh Foundation. C.E. has received grants from DJO and Zimmer Biomet and education payments from Arthrex and Smith & Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Committee for Oversight of Research and Clinical Training Involving Decedents (CORID) at the University of Pittsburgh (CORID ID: 940).
