Abstract
Background:
Delayed gadolinium-enhanced magnetic resonance imaging of cartilage (dGEMRIC) is an advanced imaging technique that is purported to quantify cartilage damage in acute and chronic joint disease and predict periacetabular osteotomy (PAO) outcomes. There is a paucity of literature relating dGEMRIC values to arthroscopic findings before PAO and postoperative outcomes after PAO.
Purpose:
To assess the utility and validity of dGEMRIC as a preoperative and prognostic assessment tool of cartilage status and integrity as it relates to intraoperative findings and midterm postoperative outcomes after PAO.
Study Design:
Case series; Level of evidence, 4.
Methods:
We analyzed a cohort of 58 patients (70 hips) with a median age of 30.1 years (range, 15-50) with hip dysplasia who underwent hip arthroscopy, followed by a PAO with preoperative dGEMRIC. The primary outcome measures were intraoperative assessment and correlation with cartilage damage (presence of cartilage flap, Outerbridge grade of the acetabulum and femoral head). Secondary outcome measures were postoperative patient-reported outcome (PRO) scores, including the International Hip Outcome Tool and Non-arthritic Hip Score. Correlation analyses were performed to determine the relationship between dGEMRIC values and (1) PROs and (2) intraoperative assessment of cartilage damage.
Results:
There were significant negative linear relationships between dGEMRIC values and the primary outcome measures: presence of a cartilage flap (coronal, P = .004; sagittal, P < .001), Outerbridge grade of acetabular articular cartilage lesion (coronal, P = .002; sagittal, P = .003), and Outerbridge grade of femoral head articular cartilage lesion (coronal, P = .001; sagittal, P < .001). Despite significant overall improvement in all patients, there was no significant correlation between preoperative dGEMRIC values and improvement in PROs from presurgery to latest postoperative follow-up (median, 2.2 years; range, 1.0-5.0 years).
Conclusion:
Although dGEMRIC values (sagittal and coronal) were significant predictors of the intraoperative presence of cartilage flaps and overall cartilage integrity, they were not associated with midterm outcomes after PAO.
Progressive cartilage damage of the hip joint commonly results from aberrant joint reactive forces associated with unfavorable anatomy, such as acetabular dysplasia and/or femoroacetabular impingement. 15,19 Left unaddressed, chondral damage is thought to progress to end-stage arthritic change and is associated with poor clinical and functional outcomes. 15 Periacetabular osteotomy (PAO) is a surgical intervention that corrects acetabular orientation in dysplastic cases to improve hip joint stability and subsequently joint biomechanics, which delays the development of secondary osteoarthritis and enhances patient function and activity. 25,26 Assessing articular cartilage damage before surgical intervention requires not only clinical information but also imaging for accurate identification, which is essential for selecting appropriate treatment strategies and surgical planning. 23 While preoperative plain radiographs are often used to analyze cartilage status via joint space measurements and osseous changes, 19 magnetic resonance imaging (MRI) and arthrogram provide more direct cartilage assessment but lack information regarding the biological characteristics and health status of cartilage. 25,26
Gadolinium is an anionic contrast agent acting as a functional measure of cartilage status by binding avidly to damaged cartilage with less anionic glycosaminoglycans as compared with healthy cartilage. 43 It has been proposed that 3-dimensional delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) improves specificity for identifying articular cartilage defects associated with hip pathologies, such as femoroacetabular impingement and dysplasia, via enhanced gadolinium binding and subsequent shortened T1 relaxation times. 13
Although a few studies have associated low dGEMRIC scores (indicative of worse cartilage status) with poor PAO outcomes, 13,18 there is a deficit of literature defining the relationship between dGEMRIC and both arthroscopic findings and clinical outcomes. This information would be useful to hip-preservation surgeons who may consider implementing the use of dGEMRIC into the standard preoperative imaging assessment, with the end goal of determining whether surgical success and/or failure can be predicted by dGEMRIC score.
The purpose of this study was to assess the utility and validity of dGEMRIC as a preoperative and prognostic assessment tool of cartilage status and integrity as it relates to intraoperative findings and postoperative outcomes after PAO.
Methods
After institutional review board approval, a prospective longitudinal cohort study was performed involving 70 hips (58 patients) treated with the University of Colorado PAO 28 between November 2015 and June 2018. All patients were prospectively enrolled in an institutional review board–approved hip registry. All procedures were performed by the senior author (O.M.-D.). Inclusion criteria were (1) persistent hip pain refractory to nonoperative management lasting at least 6 months, (2) reproducible clinical examination findings suggestive of intra-articular pain and instability, (3) a joint-space width >3 mm on all radiographic views, and (4) radiographic findings consistent with frank or borderline hip dysplasia. 23
Patients with borderline dysplasia who exhibited substantial signs of instability 20 (Beighton Hypermobility Score 40 >6, excessive femoral and/or acetabular version) were typically advised that the risk of failure of hip arthroscopy alone was high, although this was presented as a valid first-line treatment option. When these patients elected to proceed with a PAO after a previously failed arthroscopic-only treatment, hip arthroscopy was again performed before the PAO to address any new chondrolabral pathology that may have developed since the previous arthroscopic surgery or residual cam pathology. Patients with concomitant excessive femoral ante/retrotorsion were offered a derotational femoral osteotomy in addition to hip arthroscopy and PAO.
Clinical diagnosis of acetabular frank or borderline dysplasia was determined according to accepted pathomorphologic signs and measurements. History of hip pain, positive findings on provocative hip tests indicating a labral tear, radiographic evidence of hip dysplasia (lateral center-edge angle <25°, sourcil angle >10°), excessive acetabular version and/or femoral antetorsion, and interruption of the Shenton line on the anteroposterior pelvic radiograph, as well as MRI findings of labral hypertrophy, 16,21,32 articular cartilage thickening, 1 or a ligamentum teres tear, 41 all aided in establishing a diagnosis of symptomatic hip instability. Measurements were performed by a hip-preservation fellow (J.H.L.) and verified by the senior author. Patients selected for surgery underwent preoperative computed tomography and MRI to assess acetabular version, femoral torsion, and femoral head sphericity as well as cartilage, labral, and subchondral bone integrity. Coronal and sagittal dGEMRIC indices of the hip joint were obtained during preoperative imaging.
Before the PAO operation, all patients underwent routine hip arthroscopy 3 to 10 days prior for direct visualization of cartilage status and to address intra-articular pathology. Hip arthroscopy was performed before PAO, rather than during the same anesthetic, for the following reasons: (1) to avoid prolonged anesthesia time, (2) to prevent fluid from hip arthroscopy in the surgical field during the PAO, (3) to reduce the rate of capsulolabral adhesions by enabling stationary bicycle use before PAO, and (4) to allow the surgeon and patient to discuss possibly opting out of the PAO if substantial articular cartilage damage is seen during arthroscopy. 28 Cartilage status during arthroscopy was assessed and subsequently graded using the Outerbridge grading scale. 30 Cartilage flaps were identified and confirmed with use of an arthroscopic probe. Acetabular chondral flaps were often “inside-out” lesions with an intact chondrolabral junction. 22 Any cartilage flaps identified at the time of arthroscopy were addressed with debridement, with or without microfracture, and subsequent limitations in weightbearing to allow viable fibrocartilage to develop into the former hyaline cartilage defect.
Imaging Protocol
The dGEMRIC protocol was based on established literature-supported parameters. 2 –4,8 –10 Sequences of the hips were obtained on a single 1.5-T MRI scanner optimized for dGEMRIC. Coronal and sagittal dGEMRIC sequences of the hip were obtained 1 hour postinjection of a double weight–based dose (0.4 mL/kg) of gadobenate dimeglumine (Multihance). T1 map images were created by fitting saturation recovery curves to varying image intensity, and 3-dimensional variable flip angle images were then used to create the color dGEMRIC map on which regions of interest (ROIs) were drawn. Coronal ROI measurements were taken over the weightbearing femoroacetabular joint cartilage in 3 positions (anterior, middle, and posterior) (Figure 1A). Sagittal ROI measurements were taken across the anterior superior quadrant in 3 positions (medial, central, and lateral) (Figure 1B). Measurements were obtained by a single musculoskeletal radiologist (M.K.J.).

The bulk dGEMRIC values were the mean of 3 consecutive images obtained from the following regions of interest: (A) the weightbearing articular cartilage in the anterior, middle, and posterior coronal views (outlined in yellow) and (B) the anterolateral quadrant in the medial, central, and lateral sagittal views (outlined in yellow). All images were obtained from a patient who exhibited a cartilage flap intraoperatively. dGEMRIC, delayed gadolinium-enhanced magnetic resonance imaging of cartilage.
Outcome Measures
All patients enrolled in the study completed preoperative questionnaires and detailed demographic data sheets. Collected data included age, clinical diagnosis, sex, height, weight, body mass index, duration of pain at initial presentation, laxity using the Beighton Hypermobility Score, prior hip arthroscopy, radiographic parameters measured on anteroposterior pelvis radiographs, 39 and hip range of motion. The primary outcome measures were arthroscopic intraoperative assessment and correlation with cartilage damage. The secondary outcome measures were the Non-arthritic Hip Score (NAHS) 12 and the International Hip Outcome Tool (iHOT-12). 17 Patient-reported outcome (PRO) scores were measured pre- and postoperatively at 6 and 12 weeks, 6 and 12 months, and 2 and 5 years.
Statistical Analysis
Patient demographics and outcome variables (baseline, intraoperative, PROs, and other outcomes) were summarized using descriptive summary measures expressed as means with standard deviations or medians with ranges for continuous variables, depending on the distribution, and as number (percentage) for categorical and ordinal variables. In patients with at least 12 months of follow-up, Wilcoxon tests were used to compare PROs (iHOT-12 and NAHS) between presurgery and latest postoperative follow-up. A Pearson correlation coefficient was computed to determine the relationship between the dGEMRIC values and the PAO outcome (defined as raw improvement in PROs from presurgery to latest postoperative follow-up). Additionally, biserial or polyserial correlations were computed for quantitative variables (dGEMRIC values) and a dichotomous variable (binary or ordinal; presence of cartilage flap, Outerbridge grade of the acetabulum, or Outerbridge grade of femoral head). A distribution-based method was used to calculate the minimal clinically important difference (MCID) of both PROs. 29 Linear regression analysis was performed using ordinary least squares regression to determine whether prior hip arthroscopy, lower preoperative scores, or duration of symptoms predicted latest postoperative PROs in patients with at least 12-month follow-up. All analyses were performed in R Statistical Software. 34 For all tests, statistical significance was set at P < .05.
Results
Demographic Data
The demographic characteristics for the 70 hips (58 patients) are outlined in Table 1, and radiographic parameters are presented in Table 2. The mean dGEMRIC coronal value was 420 ± 101, and the mean dGEMRIC sagittal value was 397 ± 108.
Baseline Patient Demographics (70 Hips in 58 Patients) a

Radiographic Parameters a

a COTAV, combined index of femoral torsion and acetabular version; dGEMRIC, delayed gadolinium-enhanced magnetic resonance imaging of cartilage.
b The Shenton line was divided into interrupted and broken based on the degree of stepoff.
Intraoperative Findings
Intraoperative findings are presented in Table 3. Direct visualization of cartilage status during hip arthroscopy before the PAO demonstrated an acetabular cartilage flap in 34 hips (Outerbridge grade 4; 48.6%), while 78.6% of hips had Outerbridge grade 0 of the femoral head. The majority of hips (62.9%) exhibited cartilage damage that extended from 10% to 30% of the rim to the acetabular fossa distance (width of damage was measured according to hours of the clock).
Intraoperative Findings (70 Hips)

In the 34 hips (48.6%) with a cartilage flap present, the mean dGEMRIC coronal value was 391.0 ± 99.1, and the mean dGEMRIC sagittal value was 360.0 ± 110.0 (Table 4). In the 36 hips (51.4%) without a cartilage flap present, the mean dGEMRIC coronal value was 447.0 ± 96.8, and the mean dGEMRIC sagittal value was 431.0 ± 96.2.
Intraoperative Cartilage Status: dGEMRIC Values a
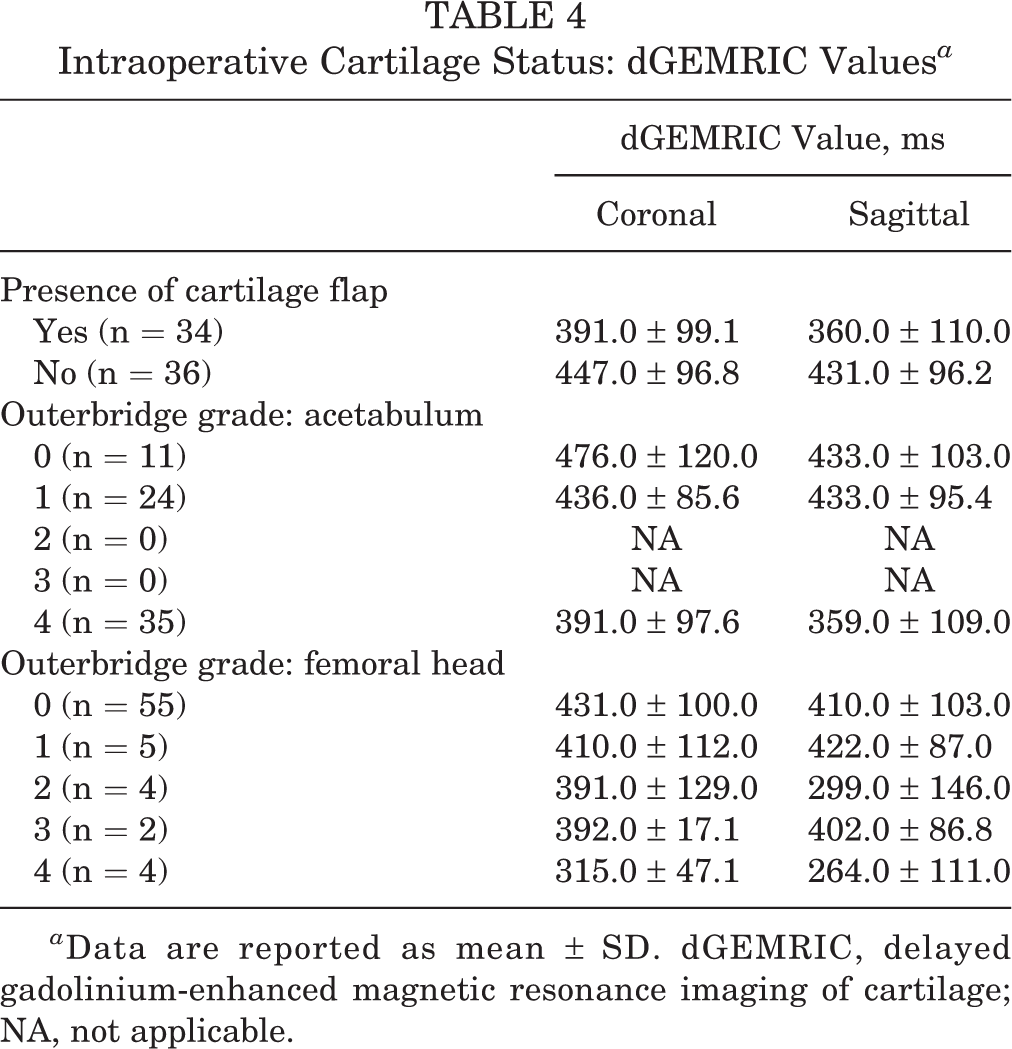
a Data are reported as mean ± SD. dGEMRIC, delayed gadolinium-enhanced magnetic resonance imaging of cartilage; NA, not applicable.
Primary Outcome Measure
In the overall cohort of patients, there was a weak negative linear significant relationship between the dGEMRIC coronal value and the presence of a cartilage flap during hip arthroscopy (r = –0.34; P = .004) (Table 5). A moderate negative linear significant relationship occurred between the dGEMRIC sagittal value and the presence of a cartilage flap during hip arthroscopy (r = –0.41; P < .001).
Correlation Between dGEMRIC Values and Parameters Related to the Arthroscopic Intraoperative Assessment of Cartilage Damage a

a All correlations, P < .05. dGEMRIC, delayed gadolinium-enhanced magnetic resonance imaging of cartilage.
In the overall cohort of patients, the dGEMRIC coronal value and the Outerbridge grade of acetabulum during hip arthroscopy demonstrated a weak negative linear significant relationship (r = –0.36; P = .002). There was a weak negative linear significant relationship between the dGEMRIC sagittal value and the Outerbridge grade of acetabulum during hip arthroscopy (r = –0.35; P = .003) and between the dGEMRIC coronal value and the Outerbridge grade of femoral head during hip arthroscopy (r = –0.38; P = .001). The dGEMRIC sagittal value and the Outerbridge grade of femoral head during hip arthroscopy had a moderate negative linear significant relationship (r = –0.45; P < .001).
PRO Scores
Fourteen hips were lost to follow-up. In the remaining 56 hips with a minimum follow-up of 1 year (median, 2.2; range, 1.0-5.0), significant improvements in the iHOT-12 (P < .001) and NAHS (P < .001) were achieved from presurgery to latest postoperative follow-up (Table 6). Using the distribution-based method, the MCID was 9.7 for the iHOT-12 and 9.0 for the NAHS. At the latest postoperative follow-up, 85.7% (n = 48) of patients reached the MCID for the iHOT-12, and 67.9% (n = 38) of patients reached the MCID for the NAHS. There was no relationship between dGEMRIC values and improvement in PROs from presurgery to the latest postoperative follow-up (P > .05 for all). Additionally, linear regression models demonstrated that prior hip arthroscopy, lower preoperative scores, and duration of symptoms did not predict latest postoperative PROs.
Patient-Reported Outcome Scores (n = 56) a

a Data are reported as median [range]. Each pre- and postoperative comparison, P < .05. iHOT-12, International Hip Outcome Tool; NAHS, Non-arthritic Hip Score.
Discussion
Our results were demonstrative of dGEMRIC value being a significant predictor of cartilage damage in a population of patients that required PAO surgery for bony instability. Decreasing dGEMRIC scores on both views (coronal and sagittal) were related to the presence of a cartilage flap. Additionally, dGEMRIC values negatively correlated with Outerbridge score of the femoral head and the acetabulum graded at the time of hip arthroscopy. Although surgical intervention alone resulted in significant improvements in PROs (iHOT-12 and NAHS) from presurgery to latest postoperative follow-up, there was no relationship between preoperative dGEMRIC values and improvement in PROs.
Because there was no significant relationship between the dGEMRIC value and either the iHOT-12 or the NAHS at follow-up, this calls into question the utility of the test as a predictor of failure after PAO. Therefore, dGEMRIC may not be a useful diagnostic adjuvant in determining whether a patient with dysplasia is a candidate for hip preservation surgery vs adult reconstruction. Although a lower dGEMRIC score was correlated with the presence of cartilage flaps, it was not correlated with clinical outcomes in our cohort. The presence of chondral injury can be assessed on preoperative MRI without the use of dGEMRIC. Thus, our study does not support the routine use of dGEMRIC in the preoperative assessment of patients undergoing PAO.
Prior studies utilizing dGEMRIC value thresholds have likewise demonstrated mixed results in diagnostic and predictive utility. The standard dGEMRIC value of healthy cartilage in patients (mean age, 37 years) has been reported to be 570 ± 90 ms, while values <390 ms are associated with osteoarthritis as well as increased risk of failure after pelvic osteotomy. 18 Similarly, Cunningham et al 13 found a mean dGEMRIC index of 370 ± 88 ms to be the greatest predictor of PAO failure at a follow-up, ranging from 2 years to 3 years 10 months. By contrast, Chandrasekaran et al 11 reported significantly greater PROs at 2 years in nondysplastic cases of hip arthroscopy with a dGEMRIC >323 ms, but they were unable to establish an actual correlation between dGEMRIC and PROs. The inconsistencies in dGEMRIC thresholds and subsequent variability in postoperative outcomes in these studies reinforce the concept that value thresholds are population-based and therefore limited in their predictive utility outside the population included in each study. The mean coronal and sagittal dGEMRIC values for our cohort were greater for patients without a cartilage flap than with a cartilage flap (447.0 and 431.0 ms vs 391.0 and 360.0 ms, respectively). Although our mean sagittal dGEMRIC value of 360 ms in patients with a cartilage flap falls below the 390 ms, 18 suggestive of increased risk of PAO failure, this was not borne out clinically in our cohort. This further suggests that employing a dGEMRIC threshold may have a more limited role than previously demonstrated.
It is possible that the use of dGEMRIC bulk values as detailed in the protocol may be slightly confounded in a dysplastic population, which may have clinical implications. A recent study evaluated the use of dGEMRIC in assessing human cartilage adaptation to exercise. It called into question the validity of dGEMRIC bulk values, which may be misleadingly elevated in individuals with inherently thicker cartilage (ie, elite athletes with cartilage hypertrophy) and subsequently higher precontrast T1values from which the bulk values are primarily derived. 36 In a dysplastic population in which cartilage thickness is significantly greater 1 than a nondysplastic cohort as measured by lateral center-edge angle, there exists potential for similar confounding in the dGEMRIC values. The implications of a falsely elevated dGEMRIC secondary to increased cartilage thickness as it relates to postoperative outcomes suggest that bulk values may be misleadingly high in a dysplastic population regardless of cartilage viability.
While there may be a small role for dGEMRIC in localizing isolated acetabular chondral lesions in preoperative patients, 24,31 arthroscopic surgical intervention in and of itself for appropriate pathologies necessitating hip-preservation procedures seems to be a better predictor, albeit an uncontrolled one, of prognosis than dGEMRIC score, as demonstrated by our study and others. 31,35 Schmaranzer et al 35 found significant decreases in preoperative vs minimum 1-year postoperative dGEMRIC scores in operative and nonoperative groups. Despite a more pronounced negative difference (decrease) in operative group dGEMRIC score after surgery, all PROs (Western Ontario and McMaster Universities Osteoarthritis Index, Hip disability and Osteoarthritis Outcome Score, modified Harris Hip Score) improved significantly in the operative group, and only 1 (modified Harris Hip Score) improved in the nonoperative group. This begets further skepticism of the predictive capabilities of dGEMRIC with respect to clinical outcome.
In our dysplastic cohort, the relationship between dGEMRIC score and cartilage flap was marginally stronger for sagittal (r = –0.41) over coronal (r = –0.34) sequences. This is consistent with our initial expectations, as cartilage flaps are better identified and characterized on sagittal sequences because of the primarily anterolateral orientation of cartilage flaps in a dysplastic cohort. Conversely, the finding that the relationship between dGEMRIC and acetabular Outerbridge grade was marginally stronger for coronal (r = –0.36) over sagittal (r = –0.35) views was less consistent with expectations, especially given that all patients with acetabular grade 4 cartilage damage had a cartilage flap identified at the time of arthroscopy. It is possible that hyaline cartilage flaps, which represent the thickest area of cartilage and are best visualized on coronal sequence, may occasionally present with higher dGEMRIC scores by virtue of thickness alone, as discussed earlier, 36 thus confounding the relative relationships observed. Even so, this study differentiates itself by utilizing coronal and sagittal views. By contrast, Palmer et al 31 utilized only sagittal imaging, while Lerch et al 24 utilized radial imaging volumetrically reconstituted for their studies.
Studies have touted dGEMRIC as a quantitative molecular assessment of cartilage integrity via preferential binding of glycosaminoglycans, 3,37 but it adds significant burden and risk to patients, including time, pain, and unstandardized billing ranging from US$2300 to US$24,000. 27 Patients must adhere to specific timed protocols to obtain requisite imaging for optimal dGEMRIC values. 33 Additionally, the use of gadolinium contrast agents, specifically those belonging to the group I subtype, is reported to have significant risk of systemic fibrosis in patients with renal disease 38,42 and may cause nonrenal acute reactions, such as anaphylaxis and local necrosis at the site of injection. 6 Therefore, dGEMRIC must demonstrate clinical benefits that outweigh these potential harms before this advanced imaging modality can be adopted by hip preservation surgeons as a routine imaging supplement.
There exist multiple noncontrasted alternate advanced imaging modalities that demonstrate noninferior and superior diagnostic and/or predictive capabilities as compared with dGEMRIC that do not expose patients to the aforementioned factors. 5 Beaulé et al 5 identified strongly significant correlations between T1ρ and T2ρ MRI mapping (noncontrasted studies) and dGEMRIC values in a cohort of patients with developmental dysplasia of the hip. Furthermore, Bittersohl et al 7 found dGEMRIC to have weak correlation with intraoperative findings of cartilage damage among those undergoing surgical intervention for femoroacetabular impingement, while standard MRI evaluation alone had a moderate correlation. Additional quantitative magnetic resonance techniques, such as T1 rho imaging, quantitative T2 mapping, and sodium MRI, may provide similar diagnostic capabilities without the use of contrast. 33
Limitations
The limitations of this study should be noted. First, the sample size was low and limited our ability to quantify dGEMRIC values based on each Outerbridge grade of the acetabulum and femoral head. In addition, there is a lack of a clinically significant threshold dGEMRIC value, which could provide a guideline. We did not include this grouping because a significant relationship was demonstrated but still not predictive of outcome, thus lending little clinical utility to such a value. All cartilage flaps were included in the same category, though the size and location of these defects may affect outcomes. In addition, microfracture and debridement of chondral flaps at the time of hip arthroscopy may have affected follow-up PROs differently from prior research 13 in which intra-articular procedures were not performed before PAO. All procedures and intraoperative diagnoses were performed by a single surgeon, which may introduce some observer bias. The median follow-up time for PROs was 2.2 years with a range of 1 to 5 years in our study cohort. Finally, because distribution-based MCIDs are typically sample dependent, 14 application of this MCID may be limited to populations with similar characteristics. We did not stratify the data based on specific patient demographics or characteristics to determine MCIDs for these subpopulations, as this was the not the purpose of the present study.
Future Directions
Further study is warranted to determine the utility of dGEMRIC in predicting long-term outcomes after PAO. Additional investigation is necessary to determine if various noncontrasted methods of imaging are noninferior to dGEMRIC with regard to diagnostic and prognostic value. Finally, future studies may seek to determine if cartilage thickness based on MRI or plain radiographs (in terms of joint space width) correlates with dGEMRIC values.
Conclusion
Although dGEMRIC values (sagittal and coronal) were significant predictors of the intraoperative presence of cartilage flaps and overall cartilage integrity, these findings were not associated with midterm outcomes after PAO.
Footnotes
Final revision submitted March 21, 2022; accepted May 12, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: T.G. has received consulting fees from Stryker. M.K.J. has received consulting fees and speaking fees from Medtronic USA and hospitality payments from Medicrea USA. O.M.-D. has received consulting fees from Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Colorado Denver (CRV006-1).
