Abstract
Background:
The Micheli technique for anterior cruciate ligament (ACL) reconstruction (ACLR) has proven to be a reliable method with a minimal risk for growth disturbance among skeletally immature patients.
Purposes:
To evaluate the Micheli technique of iliotibial band (ITB) graft passage for ACLR using cadaveric knee models and to measure the distance between the surgical instrument tip and the neurovascular bundle in the posterior knee joint: specifically, the peroneal nerve, tibial nerve, and popliteal artery.
Study Design:
Descriptive laboratory study.
Methods:
Gross dissection was performed on 17 pediatric cadaveric knees (12 male and 5 female) aged between 4 and 12 years. To simulate ITB graft passage, we passed a curved-tip hemostat clamp through the posterior capsule, with the knee flexed from 90° to 100°. Next, clinical photographs were taken, and digital imaging software was used to measure the distance in centimeters from the clamp tip to each respective neurovascular structure.
Results:
The mean distances from the clamp tip to the tibial nerve, popliteal artery, and peroneal nerve were 0.875 cm (range, 0.468-1.737 cm), 0.968 cm (range, 0.312-1.819 cm), and 1.149 cm (range, 0.202-2.409 cm), respectively. Mean values were further calculated for age groups of ≤8, 9-10, and 11-12 years. The mean distance from the clamp tip to the peroneal nerve was 1.400 cm larger for 11- to 12-year-old specimens than for ≤8-year-old specimens (95% CI, 0.6-2.2 cm; P = .005).
Conclusion:
The neurovascular structures in the posterior knee were in close proximity to the path of graft passage, with distances <1 cm in many specimens in this study. When passing the graft through the knee for an over-the-top position, surgeons should consider these small distances between the path of graft passage and critical neurovascular structures.
Clinical Relevance:
As the incidence of ACL tears is continuously increasing within the pediatric population, there are a larger number of ACLR procedures being performed. Although neurovascular injuries during ACLR are rare, this study clarifies the close proximity of neurovascular structures during ITB graft passage using the Micheli technique of ACLR.
Higher rates of anterior cruciate ligament (ACL) injuries have led to an increase in the incidence of ACL reconstruction (ACLR) procedures among skeletally immature patients over the past 20 years. 3,8,15,36,37 The management of pediatric ACL tears remains controversial: Nonoperative activity modification, bracing, and physical therapy may be treatment options. 23,24,30 However, studies increasingly demonstrate that nonoperative management may lead to poor outcomes such as chondral injuries, fractures, and persistent instability. 6,7,12,15,18,21,29,30
In young, active athletes with ACL injuries, early reconstruction may be indicated. Techniques include all-epiphyseal (drill holes located in the tibial and femoral epiphyses), transphyseal (drill holes through the physes), or extraphyseal (graft passed without epiphyseal drill holes and avoidance of the physes) graft placement. 9,31 However, physeal injuries, physeal tethering, or premature physeal closure may be risks associated with the use of drill holes during epiphyseal and transphyseal reconstruction. 22 To minimize these risks for future growth, researchers have described extraosseous techniques for ACLR that avoid drilling through, or near, the physes completely. 6,11,14
For patients with significant growth remaining, the Micheli technique (iliotibial band [ITB] graft) of ACLR has proven to be a reliable method with a minimal risk for growth disturbance. 15 During this procedure, the ITB is harvested and sutured to the periosteum (outer surface) outside of the lateral femoral condyle. 15,42 The graft is then passed over the top of the femur, from the lateral aspect of the knee posteriorly around the lateral femoral condyle, and into the notch over the ACL footprint. The free end of the graft is passed through the front of the knee joint and sutured to the periosteum of the proximal tibia. 42 The proximity of the posterior knee neurovascular structures to surgical instrumentation during graft passage dictates following a safe technique during surgery.
Neurovascular complications during knee procedures are rare but have been described for both pediatric and adult populations. 2,5,17 A 2015 literature review on pseudoaneurysms after arthroscopic knee procedures reported popliteal artery involvement in 46% of cases, with ACLR comprising 23% of reported cases. 10 Injuries to the popliteal artery specifically during the over-the-top approach for ACLR have further been reported. 32 Neurological injuries have also been described for the sciatic and peroneal nerves during ACLR. 4,26,27,41
To our knowledge, there are no current studies that have reported direct measurements of proximity from surgical instruments to neurovascular structures when using the Micheli ITB graft technique for ACLR in the pediatric knee. The purposes of this study were to evaluate the over-the-top graft passage technique in pediatric cadaveric knees and measure the distance between the surgical instrument tip (ie, hemostat clamp) and the peroneal nerve, tibial nerve, and popliteal artery. Our hypothesis was that in children, the neurovascular structures lie relatively close to the clamp tip during ITB graft passage.
Methods
Specimen Preparation
Gross dissection was performed on 17 skeletally immature cadaveric knees aged between 4 and 12 years; the specific ages were 4 years (n = 2), 8 years (n = 4), 9 years (n = 2), 10 years (n = 3), 11 years (n = 4), and 12 years (n = 2). There were 12 male and 5 female specimens examined in this study: 10 right and 7 left knees, including 6 pairs. Tissue samples were grouped by age ≤8, 9 to 10, and 11 to 12 years to have an equal distribution of specimens per group. Before conducting the present study, we consulted the institutional review board. As the dissections in this study were performed on cadaveric tissue and no genetic information or identifiers were analyzed, institutional review board approval was unnecessary. All specimens were provided by an allograft harvest facility (AlloSource).
Specimens were prepared for visualization of simulated ITB graft passage utilizing the Micheli ACLR technique. A 5.5-inch (14-cm) curved-tip hemostat clamp was used to create a path through the posterior capsule after the posterior neurovascular structures had been identified (peroneal nerve, tibial nerve, and popliteal artery and vein). Based on a standard trajectory of the described technique, the tip of the hemostat was passed from an anteromedial portal through the notch posterosuperiorly toward the proximal lateral capsule, with the knee flexed from 90° to 100°. The tip of the instrument was kept adjacent to the posterior and lateral cortexes of the femur (Figure 1).

Lateral view shows the clamp position after passage through the intercondylar region of a posterior knee joint in a 3-dimensional pediatric knee model.
Once passed, the clamp was positioned in the popliteal fossa and native surrounding soft tissue remained intact to avoid a distorted anatomy or tissue tension. Clinical photographs were taken using a digital camera (24.2-megapixel D3500; Nikon) from the posterior aspect of the dissected knee, visualizing the tip of the clamp and the neurovascular structures. When performing the Micheli ACLR technique, authors A.G. and M.S. positioned the hemostat directly along the lateral condyle. For the purpose of visualizing and measuring the actual 3-dimensional distance on a 2-dimensional image, the clamp tip remained in the posterior capsule but was purposely exposed from surrounding soft tissue.
Image Analysis and Measurements
A metric ruler was included in all photographs of the dissected specimens. ImageJ (Version 1.41; National Institutes of Health), an open-source image analysis software shown to have 98.4% accuracy, 1 was used to examine and interpret the photographs. 34,35 Distance was assessed from the tip of the clamp to the most proximal edge of each respective structure. To evaluate the distance from the surgical instrument tip to the neurovascular bundle, we used the length in centimeters. By default, all JPEG image files imported into ImageJ had a resolution of 72 dots per inch (DPI). Using standard DPI units, we measured 1 cm on the ruler in the photographs. To convert DPI to centimeters, we selected the “Set Scale” function under the “Analyze” tab. “Known Distance” was then set to 1 cm, and DPI units were converted to centimeters. The distance from the clamp tip to each respective neurovascular structure was then measured. Measurements were verified by a fellowship-trained sports/pediatric orthopaedic surgeon.
As displayed in Figure 2, distances to the tibial nerve (blue line), popliteal artery (red line), and peroneal nerve (green line) were recorded. It should be noted that in Figure 2, the clamp tip is in plane with the most superficial structures in the posterior knee joint.
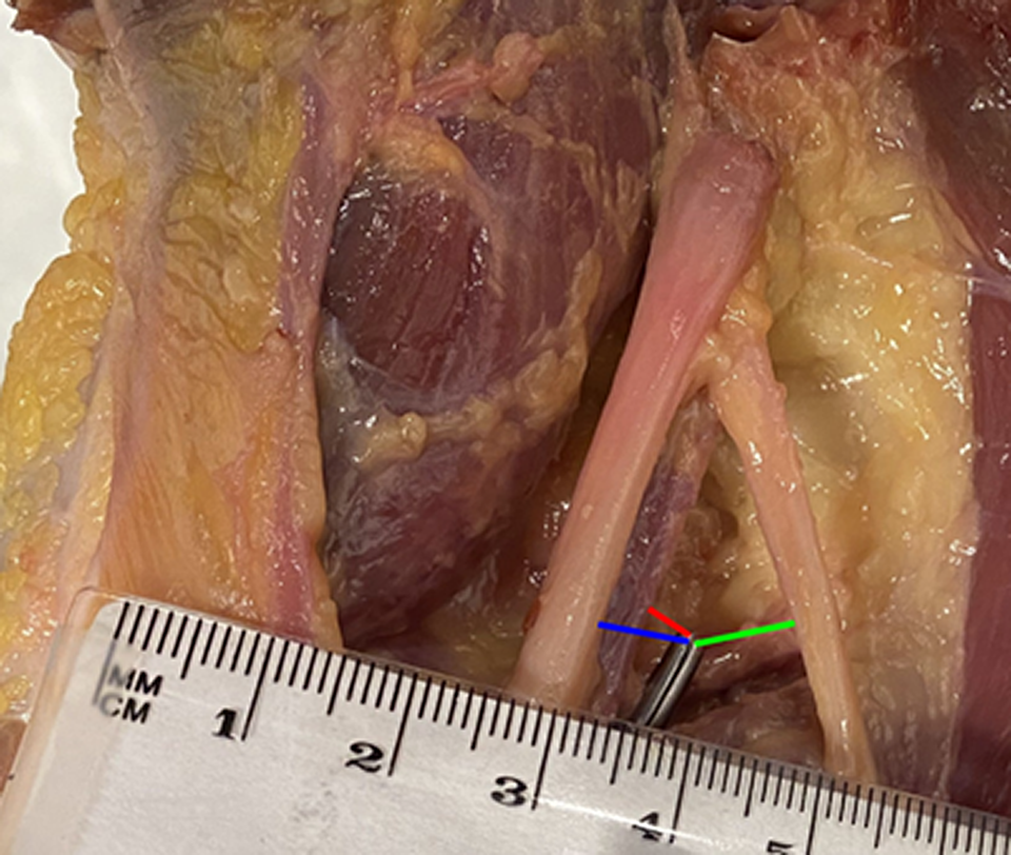
Measurements in a right knee from the clamp tip to the proximal edge of the neurovascular bundle: popliteal artery (red line), tibial nerve (blue line), and peroneal nerve (green line).
Measurement Reliability
Images (n = 17) were selected randomly, and measurements were recorded on separate occasions by 2 researchers (N.D.T. and S.A.) (“raters”; k = 2); the same measurements were repeated in random order by both raters 2 weeks later. The reliability of measurements was determined using intraclass correlation coefficients (ICCs). ICC estimates and their 95% CIs were calculated using SPSS Statistics (Version 27; IBM) based on a mean-rating (k = 2), absolute-agreement, 2-way mixed-effects model with the 2 raters across the 17 specimens. According to published guidelines, 16 an ICC >0.7 is indicative of moderate reliability, >0.8 indicates optimal reliability, and >0.9 indicates excellent reliability.
Results
In all specimens studied, the relative position of the clamp tip to the neurovascular structures was reproducible, and no damage to any neurovascular structure from graft passage was noted. The obtained ICC values were 0.983 (95% CI, 0.969-0.990; P < .001) for interrater reliability and 0.987 (95% CI, 0.976-0.993; P < .001) for intrarater reliability, indicating excellent reliability for all measurements. Mean distances were calculated for all 3 age groups (≤8, 9-10, and 11-12 years) and are shown in Table 1. Multivariable linear regression models for the 3 different age groups were run for each distance variable, with results presented in Table 2.
Distance From Clamp Tip to Neurovascular Bundle by Age Group a

a Data are presented as mean ± SD.
Multivariable Regression Results for Neurovascular Bundle Distances

a Statistically significant difference (P < .01). Unit of measure is cm.
Measured distances from the clamp tip to the neurovascular bundle in each specimen are listed in Table 3. Notably, 3 independent measurements (distance to the popliteal artery in a 4-year-old specimen and to the peroneal nerve in 8- and 12-year-old specimens) were indiscernible because of decreased tissue integrity or damage during dissection. However, the remaining tissue was preserved, and distances to the other respective structures were successfully measured and included in all analyses. The mean distance from the clamp tip to the peroneal nerve was 1.400 cm (95% CI, 0.6-2.2 cm) larger for 11- to 12-year-old specimens compared with ≤8-year-old specimens after adjusting for sex (P = .005). No significant differences in the mean distance were found between sexes or between the other age groups (Figure 3).
Distance From Clamp Tip to Neurovascular Bundle by Specimen a

a ≠, distance indiscernible on photograph; F, female; L, left; M, male; R, right.

Mean distance from the clamp tip to the neurovascular bundle by age group. Error bars represent 95% CI. **Statistically significant difference (P < .01). Illustration depicts two dimensional example of hemostat clamp placement in a right knee specimen.
Distance to Tibial Nerve
The mean distance from the clamp tip to the tibial nerve was 0.875 ± 0.38 cm (range, 0.468-1.737 cm). The closest distance to the nerve was 0.468 cm in an 8-year-old specimen, and the farthest was 1.737 cm, also in an 8-year-old specimen.
Distance to Popliteal Artery
The mean distance from the clamp tip to the popliteal artery was 0.968 ± 0.41 cm (range, 0.312-1.819 cm). The closest distance to the artery was 0.312 cm in an 11-year-old specimen, while the farthest was 1.819 cm, also in an 11-year-old specimen.
Distance to Peroneal Nerve
The mean distance from the clamp tip to the peroneal nerve was 1.149 ± 0.60 cm (range, 0.202-2.409 cm). The closest distance to the nerve was 0.202 cm in an 8-year-old specimen, while the farthest was 2.409 cm in a 12-year-old specimen.
Discussion
This pediatric cadaveric study is the first to demonstrate direct measurements of the proximity of surgical instrumentation to neurovascular structures during ITB graft passage utilizing the Micheli ACLR technique. In the majority of our specimens, the clamp tip was found to lie within 1 cm of the tibial nerve (70%) and peroneal nerve (53%), and 44% were within 1 cm of the popliteal artery. This close relationship was seen in pediatric knee specimens aged 4 to 12 years, which is representative of the population in which extraosseous techniques are indicated to avoid physeal injuries during ACLR. By understanding the relationship between surgical instrumentation and the popliteal artery, tibial nerve, and peroneal nerve, the surgeon may utilize techniques that minimize the risk of injuries to these structures during posterior graft passage for physeal-sparing ACLR.
ACLR within the pediatric population is both increasing in incidence and evolving. As procedures become more refined, the neurovascular structures of the knee should be specifically considered. Although relatively rare, damage to these structures has serious consequences. Damage to the peroneal nerve 11,25 and popliteal artery 13,32 during ACLR has been previously documented, but these injuries were reported primarily in adult populations using conventional repair techniques. A recent meta-analysis reviewing clinical outcomes after pediatric ACLR between 1985 and 2016 reported that 3.7% developed angular deformity and 7.5% had at least 1 cm of limb-length discrepancy, but no data were reported on neurovascular injuries. 44 While documentation of neurovascular damage during pediatric ACLR is scarce in the literature, 5 surgeons should be mindful that children have smaller distances between posterior knee neurovascular structures and surgical instrumentation used for extraosseous ITB graft passage.
Previous pediatric cadaveric studies have focused on ligament development 31,36,38 and the distance between soft tissue structures 33 in the posterior compartment of the knee. 39,45 Similar to findings from previous knee development studies, the present cadaveric cohort exhibited statistically significant variations in distance from the clamp tip to the neurovascular bundle with age. Among the 11- to 12-year-old age group, there was an increased distance from the clamp tip to the peroneal nerve. When the graft is passed through the posterior knee, there may be a larger zone of safety for graft passage at the lateral cortex among older patients who are nearing the age of physeal closure. During ACLR, the tip of the clamp is directed laterally to allow the ITB graft to pass around the posterior lateral aspect of the femur. This orientation of the tip may direct surgical instrumentation more closely to the peroneal nerve and away from the more central positions of the popliteal artery and tibial nerve during graft passage. This may explain the incidence of peroneal nerve injuries during ACLR in prior literature. 4,26,27
There are a number of physeal-sparing ACLR procedures that either avoid the physis completely (ie, extraphyseal [Micheli] 14,15 and all-epiphyseal 9 ) or utilize only a minor portion of the physis (transphyseal 19,40 ). 8,12 Recent studies have shown similar postoperative outcomes after extraphyseal and all-epiphyseal procedures, with overall complication rates, including graft failure, ranging from 6% to 14%. 28 The transphyseal technique has a higher overall rate of complications but demonstrates no difference in the rate of graft failure (7.2% vs 6.6%, respectively) in comparison with the extraphyseal or all-epiphyseal technique. 20,28,42 While all 3 respective techniques have displayed excellent patient-reported outcomes, future studies that assess long-term outcomes are needed to further assist physicians in selecting the appropriate ACLR technique.
We utilized an inside-out approach for ITB graft passage, which was based on the original extraphyseal description. Other modifications have described an outside-in approach to graft passage, specifically to optimize the position of the ITB within the notch of the knee. 43 Several authors have also used a dual inside-out and outside-in approach to facilitate graft passage. 9,20 For this combined approach, the first instrument is introduced anteriorly into the knee joint, and the tip is passed slightly beyond the posterior knee capsule to create an opening. A second instrument, a curved clamp, is then introduced posteriorly through the opening over the back of the lateral femoral condyle. The tips of the instruments are joined together, guiding the posterior instrument and allowing the ITB graft to pass through the knee joint. Although not compared within this cadaveric study, the safest trajectory remains unclear.
Measurements on photographs have limitations, as this is a 2-dimensional analysis of a 3-dimensional anatomic relationship. After passage, the clamp tip was in plane with the most superficial neurovascular structures. A photographic measurement does not account for the trajectory of the clamp or the slight variation in depth from the clamp tip to each respective structure. Furthermore, with the Micheli ACLR technique, while keeping surgical instruments adjacent to the lateral cortex of the femur (hugging the lateral cortex) during graft passage, the clamp may not penetrate as far posterior, thus positioning the surgical instruments farther away from the neurovascular bundle.
Despite these limitations, the current study provides an assessment of the proximity of the instrument to critical structures. A future study that conducts measurements during dissection with surgical calipers to account for depth and compares measurements made via imaging software (ie, ImageJ) can further assess reliability. Also, while we do not anticipate significant variations in clamp placement, arthroscopic fluid insufflation during the actual procedure may allow for slightly greater working room between the posterior knee capsule and neurovascular structures during graft passage. Last, freezing and thawing of specimens allows for normal elasticity of tissue, but dissection may lead to slightly altered positioning of the neurovascular structures with respect to the over-the-top position of the clamp tip. Although access to pediatric cadaveric tissue is severely limited, a larger number of specimens per group could increase the precision of these measurements.
Conclusion
The findings of this study demonstrated the proximity of posterior knee neurovascular structures to the instrument tip during the Micheli ACLR technique. As the incidence of ACL tears is increasing within the pediatric population, there are inevitably a larger number of ACLR procedures being performed. While this extraosseous technique has proven to have the least risk of growth complications, care must be taken when performing this procedure to avoid damage to the neurovascular structures within the popliteal fossa region.
Authors
Nicholas D. Thomas, MS (Stanford University School of Medicine, Stanford, California, USA; Florida State University College of Medicine, Tallahassee, Florida, USA); Salvador Ayala, BS (Stanford University School of Medicine, Stanford, California, USA; University of Illinois College of Medicine, Chicago, Illinois, USA); Matthew Rohde, BS (Stanford University School of Medicine, Stanford, California, USA); Anshal Gupta, MS (Stanford University School of Medicine, Stanford, California, USA); Mark Sanchez, MD (Stanford University School of Medicine, Stanford, California, USA); Henry Ellis, MD (Texas Scottish Rite Hospital for Children, Dallas, Texas, USA); Marc Tompkins, MD (University of Minnesota Medical School, Minneapolis, Minnesota, USA); Phil Wilson, MD (Texas Scottish Rite Hospital for Children, Dallas, Texas, USA); Seth Sherman, MD (Stanford University School of Medicine, Stanford, California, USA); Daniel Green, MD (Hospital for Special Surgery, New York, New York, USA); Theodore J. Ganley, MD (Children’s Hospital of Philadelphia, Philadelphia, Pennsylvania, USA); Curtis VandenBerg, MD (Children’s Hospital Los Angeles, Los Angeles, California, USA); Yi-Meng Yen, MD, PhD (Boston Children’s Hospital, Boston, Massachusetts, USA); and Kevin G. Shea, MD (Stanford University School of Medicine, Stanford, California, USA).
Footnotes
Final revision submitted April 17, 2022; accepted May 17, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: H.E. has received education payments from Pylant Medical and speaking fees from Smith & Nephew. M.T. has received grant support from DJO and hospitality payments from Aesculap. P.W. has received education payments from Pylant Medical. S.S. has received grant support from DJO; education payments from Elite Orthopaedics and Evolution Surgical; consulting fees from Bioventus, JRF Ortho, Flexion Therapeutics, Olympus Americas, Smith & Nephew, and Vericel; speaking fees from Arthrex, ConMed Linvatec, Smith & Nephew, Synthes, and Vericel; honoraria from Flexion Therapeutics, JRF Ortho, and Vericel; and royalties from ConMed Linvatec. D.G. has received speaking fees from Arthrex and Synthes and royalties from Arthrex. T.J.G. has received education payments from Arthrex and is a paid associate editor for The American Journal of Sports Medicine. C.V. has received education payments from Arthrex and hospitality payments from Smith & Nephew. K.G.S. has received education payments from Evolution Surgical and hospitality payments from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
