Abstract
Background:
Surgical and anesthetic techniques have enabled a shift to the ambulatory setting for the majority of patients with anterior cruciate ligament (ACL) tears. While this change likely reflects improvements in acute pain management, little is known about national trends in pediatric perioperative pain management after ACL reconstruction (ACLR).
Purpose:
To describe recent trends in the United States in perioperative pain management for pediatric ACLR.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
Pediatric patients (age, ≤18 years) who underwent ACLR with peripheral nerve blocks between January 2008 and December 2017 were identified in the Pediatric Health Information System database. We modeled the use of oral and intravenous analgesic medications over time using Bayesian logistic mixed models. Models were adjusted for patient age, sex, race, primary payer, and treatment setting (ambulatory, observation, or inpatient).
Results:
The study criteria produced a sample of 18,605 patients. Older children were more likely to receive intravenous acetaminophen, intravenous ketorolac, and oral and intravenous opioids. Younger children were more likely to receive ibuprofen. In our adjusted logistic model, treatment setting was found to be an independent predictor of the utilization of all medications. We found an increase in the overall utilization of oral acetaminophen (adjusted odds ratio [adj OR], 1.14 [95% CI, 1.04-1.23]), intravenous acetaminophen (adj OR, 1.42 [95% CI, 1.22-1.65]), and oral opioids (adj OR, 1.16 [95% CI, 1.06-1.28]) over the study period at a typical hospital. We found significant heterogeneity in medication use across hospitals, with the most heterogeneity in intravenous acetaminophen. Other studied descriptive variables did not appear to predict practices.
Conclusion:
After adjusting for patient characteristics and treatment settings, pain management strategies varied among hospitals and over time. Patient age and treatment setting predicted practices. Regional anesthesia, opioid medications, and intravenous ketorolac remained the mainstays of treatment, while intravenous acetaminophen emerged in use over the course of the study period. The variability in the pain management of pediatric patients undergoing ACLR suggests that further study is necessary to establish the most effective means of perioperative pain management in these patients.
Keywords
Anterior cruciate ligament (ACL) rupture remains a common injury among active youth. 25 To reduce its short- and long-term complications, 3,7,15,19 surgical reconstruction has become widespread among pediatric patients with ACL injury. 4,29 More recently, multimodal strategies, including peripheral nerve blocks (PNBs) have permitted the shift to the ambulatory setting for ACL reconstruction (ACLR) 1,27 and other arthroscopic procedures. However, little is known about perioperative practices in this population. Identifying national trends in perioperative pain management strategies in pediatric ACLR may help to further reduce opioid overreliance and improve outcomes by identifying effective emerging strategies and outlier behavior.
We conducted this study to generate meaningful data on perioperative pain management practices for pediatric ACLR nationally using an administrative data set of tertiary care pediatric hospitals. Study aims were as follows: (1) evaluate national utilization trends of common analgesics, (2) estimate hospital-level variability in practice across the study sample, and (3) identify differences in analgesic utilization based on patient characteristics. We hypothesized that (1) opioid utilization would decline over the study period, (2) pain management practice would vary among hospitals without a standardized strategy present, and (3) race-related differences would exist in analgesic combinations utilized in the perioperative period. 20,24
Methods
Data Source
We analyzed data from the Pediatric Health Information System (PHIS) database—a comparative pediatric clinical and administrative database of >49 freestanding, not-for-profit, tertiary care pediatric hospitals in the United States. The PHIS can be queried for case inpatient, ambulatory, emergency department, and observation unit encounters for patient characteristics, diagnostic information, medication, and procedural treatment data. As data from the PHIS are considered not readily identifiable, the current study was determined to be exempt from institutional review board approval.
Population and Selection Criteria
Pediatric patients (age, ≤18 years) undergoing any ACLR procedure between January 2008 and December 2017 were included in the study. We used the International Classification of Diseases (ICD) 9th Revision, the ICD 10th Revision, and the Current Procedural Terminology codes for old disruption of the ACL, a knee cruciate ligament sprain, or sprain of the ACL to identify those patients eligible for the study. We used a similar approach to include patients who received a PNB. The vast majority of patients who underwent ACLR received a PNB (96.7%). Because the majority of the patients were managed with a PNB in our cohort, we elected to exclude patients without a PNB from our analyses. Additionally, we excluded patients who also had a congenital absence of the ACL, had a congenitally short femur, had a posterior cruciate ligament tear, or were undergoing tibial spine fixation. Patients with missing descriptive data (<1%) were also excluded in the models, as evidence suggests that this proportion is not large enough to meaningfully bias the results. 26
Clinical Variables
All analgesic medications administered during the inpatient encounter were collected and used as the outcome of our models. Medications used in models included oral and intravenous acetaminophen, oral ibuprofen, oral celecoxib, intravenous ketorolac, and oral and intravenous opioids. Indomethacin was used 14 times over the study period (<0.01% of cases) and was not modeled. We included date of encounter by month, setting of the encounter (ambulatory, observation, or inpatient), patient sex (male vs female), race (White, Black, or other), age, payer (commercial vs noncommercial), and hospital as predictors. “Observation” status is an administrative label applied by the hospital to patients who do not meet the criteria for inpatient admission. Differences may exist among hospitals in how this label is applied to patients admitted postoperatively. 17 For the purposes of the analyses performed, we maintained the administrative labels with the understanding that overlap likely exists in the patients assigned to the observation and inpatient settings.
Statistical Analysis
Descriptive statistics of the patient variables and analgesic medications in our sample were reported. The rate of use for each medication was estimated using a Bayesian logistic mixed model framework. This approach allowed us to test and account for the influence of hospitals on drug choice. By allowing the intercept to vary by hospital, we were able to test whether drugs were used at different rates on average. By allowing the time slope to vary, we were able to test whether the use of different drugs changes at different rates across hospitals. We reported the SDs of the intercept and the slope of each medication’s time trend as an estimate of hospital-specific variation. Effects of patient sex, race, primary payer, and treatment setting were also included. Because of a large number of statistical tests performed, which increased the probability of a false-positive result, a Bonferroni correction was applied for each predictor. 10,21 Significance was originally defined at P<.05, with P<.007 after adjustment. Data analysis was performed using R Version 3.5.3.
Results
The final study sample consisted of 18,605 patients across 51 hospitals, the majority of whom were male (53.6%) and White (62.2%). The mean age of the patients was 14.2 years. Patient characteristics are further detailed in Table 1. Unadjusted overall medication trends are presented in Figure 1A. All estimated models converged.
Sample Characteristics (N = 18,605)

(A) Overall rates of medication use by year. (B) Adjusted rates of medication use at a typical hospital. CE, celecoxib; IA, intravenous acetaminophen; IK, intravenous ketorolac; IO, intravenous opioid; OA, oral acetaminophen; OI, ibuprofen; OO, oral opioid.
Aims 1 and 2: Nationwide Trends and Variation in Perioperative Practices
Within our cohort, we identified the adjusted increase in the utilization of oral acetaminophen (adjusted odds ratio [adj OR], 1.14 [95% CI, 1.04-1.23]), intravenous acetaminophen (adj OR,1.42 [95% CI, 1.22-1.65]), and oral opioids (adj OR, 1.16 [95% CI, 1.06-1.28]) during the study period. Adjusted trends for the typical hospital are presented in Figure 1B. The oral acetaminophen and oral opioid trends for a typical hospital, however, seem to contradict their overall trends in Figure 1A. As the overall trends are volume-weighted means of individual hospital trends, the utilization patterns at large-volume centers may mask typical hospital trends. Using Tukey outlier criteria, 30 we excluded the 3 highest-volume centers and found that the overall trends for oral acetaminophen (unadjusted OR, 1.02 [95% CI, 1-1.04] and oral opioids (unadjusted OR, 1.14 [95% CI, 1.12-1.17) reflected the trends for a typical hospital. There was no evidence for an overall change in utilization for the other medications studied.
Significant variability in the utilization of all medications was identified between hospitals in 2008 (minimum P < .001) and 2017 (minimum P < .001). As intravenous acetaminophen was approved in 2010, we first noted use and significant variability in 2011 (χ2[39] = 282; P < .001). After adjustment, we found that intravenous acetaminophen (intercept SD, 2.76 [year OR SD, 0.61]) demonstrated the most variation in typical use and change in use across hospitals. We also noted that even the medication that varied the least, ibuprofen (intercept SD, 0.70 [slope SD, 0.14]), still demonstrated meaningful variability among hospitals.
Aim 3: Descriptive and Setting Differences
Older children were more likely to receive intravenous acetaminophen (adj OR, 1.07 [95% CI, 1.05-1.09]), celecoxib (adj OR, 1.17 [95% CI, 1.06-1.31]), intravenous ketorolac (adj OR, 1.07 [95% CI, 1.06-1.08]), oral opioids (adj OR, 1.11 [95% CI, 1.09-1.13]), and intravenous opioids (adj OR, 1.17 [95% CI, 1.16-1.20]). Younger children were more likely to receive ibuprofen (adj OR, 0.89 [95% CI, 0.87-0.90]). We found no significant effect of sex or race after multiplicity correction. We did find that treatment setting appeared to contribute most broadly to the variation in medication utilization (Table 2).
Odds of Pain Medication Utilization by Patient Characteristics a
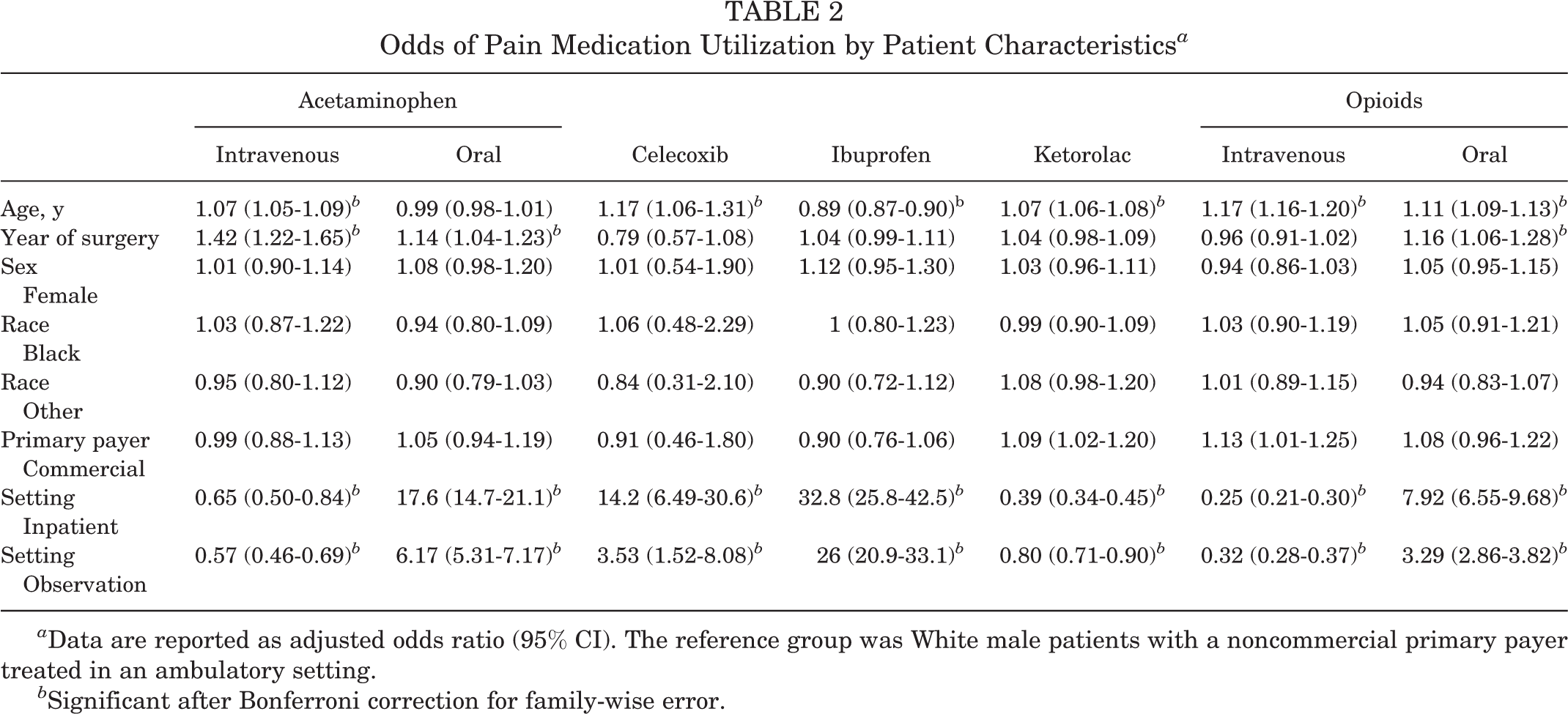
a Data are reported as adjusted odds ratio (95% CI). The reference group was White male patients with a noncommercial primary payer treated in an ambulatory setting.
b Significant after Bonferroni correction for family-wise error.
Discussion
This study examined perioperative pain management practices in pediatric ACLR using a nationwide administrative database of pediatric hospitals. We identified significant temporal trends in the use of multiple analgesics over the studied decade (2008-2017) without a clear decrease in reliance on intravenous opioid medications during this time. Interhospital variability was also found for all studied medications. Finally, we found a significant effect of patient age and treatment setting on the combination of analgesics used perioperatively, while sex, race, and insurance status appeared to have almost no effect on these practices.
Nationally, we noted an increase in the use of oral and intravenous acetaminophen and oral opioids at the typical hospital. While there remains limited evidence demonstrating the superiority of ketorolac over other analgesics for the management of postoperative pain in children and adolescents, 18 our findings demonstrated that it is widely utilized as an adjuvant medication for perioperative pain management during pediatric ACLR. Intravenous acetaminophen, which gained US Food and Drug Administration approval in 2010, increased in use over the latter portion of the study period.
Among hospitals, we noted meaningful variability in pain management. This national variability indicated that, despite the widespread adoption of PNBs and the broad use of intravenous ketorolac, differences in approach still exist. The trends for a typical hospital differed from the overall utilization trend, underscoring the importance of modeling hospital-specific variation. Specifically, the most variability was seen in intravenous acetaminophen and celecoxib use. The variation in intravenous acetaminophen use may be because of its recent introduction across a number of institutions. Although intravenous acetaminophen use has been associated with reduced opioid consumption and overall cost in other areas of orthopaedics, 8 cost concerns and questions regarding its superiority over oral acetaminophen use may have limited its broader adoption. 9,11,28 Further work is necessary to delineate the optimal approach to perioperative pain management in this patient cohort.
Our study also demonstrated significant variability in medication use based on the treatment setting. Oral acetaminophen, ibuprofen, celecoxib, and oral opioids were more often utilized for patients in nonambulatory settings, while intravenous acetaminophen, ketorolac, and intravenous opioids were more often used at ambulatory sites. This may be the result of different pain management strategies based on patient or procedural complexity, which is likely to guide treatment settings. This difference may also stem from a higher volume in ambulatory centers, which may lead to protocols that maximize multimodal analgesia in an effort to allow for same-day discharge. However, the PHIS database does not include outpatient prescription data, which limits our comparison of medication utilization in the perioperative period for ambulatory and observation/inpatient procedures over a comparable time (ie, through postoperative day 1).
We found that younger patients were less likely to receive oral and intravenous opioids, intravenous acetaminophen, and intravenous ketorolac, but they were more likely to receive oral ibuprofen. While age-related findings were limited by the epidemiology of this surgical procedure (ie, a small proportion of patients aged <10 years in our study sample), practice differences based on age may still have some clinical relevance. On one hand, the extra physeal techniques 13,14 utilized commonly in preadolescent and early adolescent patients may be less painful because of the reduced tunnel drilling or the lack of bone-based harvest. On the other hand, past research has shown that pain may be undertreated in younger children. 2,22 As the PHIS administrative coding does not enable the identification of the ACLR technique, future work is needed to identify the source of these differences.
We identified no effect of sex, race, or insurance status on perioperative pain management practices for pediatric ACLR. Past work has demonstrated disparities in analgesia based on race, sex, and insurance status potentially because of unconscious bias. 6,12,16,23 Although there have been demographic-based differences noted in other phases of ACL patient management, 5 we did not find evidence of a difference in perioperative pain medication. Looking at the other medications, we attribute this finding to institutional protocols of perioperative pain management adopted for common pediatric procedures like ACLR. These policies may minimize the effect of biases that otherwise could cause disparities in care.
Limitations
The limitations of this study are similar to those of any study that relies on administrative data. Although we had data on in-hospital medication use, the PHIS does not collect information on patients’ reported pain, psychological traits, or metrics other than length of stay. As such, work outside of the limitations of this database would be better suited to explore whether these differences in practice patterns actually affect patient outcomes. Similarly, although statistical adjustments were made for relevant descriptive variables contained in the PHIS, there were notable patient and surgical factors that may have affected anesthetic management (eg, patient weight, medical comorbidities, and operative time) that were not available for inclusion. It is possible that the adjustments made were unable to control for meaningful factors that might drive the interhospital differences observed in the study. Nevertheless, we suspect that variability due to provider training and preferences, as well as internal policy, is more likely to drive differences than the unmeasured population heterogeneity of these additional patient characteristics. Finally, because of database constraints, we were unable to evaluate postoperative medication prescribing practices and utilization after patient discharge. Although after-hospital pain management was not the primary focus of this study, we do recognize that these prescribing practices may have an influence on strategies utilized before discharge to prepare patients for their anticipated disposition.
Conclusion
In this administrative database study, we found that age and setting, but not the other patient characteristics, predicted perioperative pain management practices in pediatric ACLR. The use of PNBs is near ubiquitous, perioperative opioid utilization is still the norm, and ketorolac use is common at many hospitals. The variability across hospitals and time in terms of analgesic combinations suggests that providers are actively refining their approaches, and it warrants further exploration of how these differences in pain management drive outcomes.
Footnotes
Final revision submitted September 1, 2021; accepted October 7, 2021
One or more of the authors has declared the following potential conflict of interest or source of funding: L.W. has received speaking fees from Synthes GmbH. B.A.W. has received education payments from Arthrex and hospitality payments from K2M and Liberty Surgical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
