Abstract
Background:
Quadriceps tendon autograft (QTA) has recently gained popularity in the treatment of anterior cruciate ligament (ACL) ruptures in pediatric patients. The addition of lateral extra-articular tenodesis (LET) to an ACL reconstruction (ACLR) has been found to reduce the risk of ACL retear in high-risk patients.
Purpose:
To (1) compare ACL graft maturity using signal intensity ratios (SIRs) on magnetic resonance imaging (MRI) scans in skeletally immature patients undergoing ACLR with QTA either with or without concomitant LET and (2) evaluate LET safety by calculating the physeal disturbance-related reoperation rate in the ACLR+LET group.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
The records of patients aged ≤18 years who underwent ACLR between 2015 and 2021 were reviewed retrospectively. Patients undergoing ACLR with QTA who had open distal femoral and proximal tibial physes on MRI scans and a minimum 2-year follow-up data were included. SIR values were measured on sagittal MRI scans by averaging the signal at 3 regions of interest along the ACL graft and dividing by the signal of the posterior cruciate ligament at its insertion. Statistical analysis was performed to evaluate differences in SIR values at 6 months, 1 year, and 2 years postoperatively in patients who underwent ACLR alone versus ACLR+LET.
Results:
Overall, 29 patients were included in the study: 16 patients in the ACLR+LET group and 13 patients in the ACLR-only group. There were no significant differences in SIR values between groups at the 6-month or 1-year postoperative timepoints. At 2 years postoperatively, the median SIR of the ACLR+LET group was significantly lower than that of the ACLR-only group on both univariate (1.33 vs 1.86, respectively, P = .0012) and multivariate regression analyses adjusting for both sex and surgical technique (β = -0.49 [95% CI, -0.91 to -0.05]; P = .029). There were no cases of reoperation for physeal disturbance in patients who underwent ACLR+LET.
Conclusion:
The addition of LET to an ACLR with QTA was associated with lower average SIR values and thus improved graft maturity at 2 years postoperatively compared with ACLR alone in skeletally immature patients. The addition of LET to an ACLR was found to be safe in skeletally immature patients.
Keywords
After anterior cruciate ligament (ACL) reconstruction (ACLR), the tendon graft begins a complex process of revascularization and ligamentization. 2 Graft ligamentization can be further stratified into 4 stages: early (repopulation), remodeling, maturation, and quiescent. 29 The maturation process takes anywhere from 6 months to 3 years to complete.5,10,29 Understanding the timing and processes underlying ACL graft maturation allows patients to progress through the postoperative rehabilitation process and return to sports at the appropriate time without placing undue risk on the healing graft and potentially compromising graft integrity.
The role of magnetic resonance imaging (MRI) in the evaluation of ACL graft maturation is an expanding area of recent investigation.15,20,27,36 Once the process of ligamentization begins, signal-to-noise quotients (SNQs) and signal intensity ratios (SIRs) can be used to evaluate water content, a surrogate for graft maturity, on MRI scans. Lower SNQs/SIRs, indicative of improved graft maturity, are associated with improved biomechanical properties, including higher load to failure, increased stiffness, and higher tensile strength. 34 Previous ACLR graft maturity studies in adults have found that quadriceps tendon autografts (QTAs) with a bone block are more mature at 6 months postoperative compared with hamstring tendon autografts (HTAs). 22 Studies in skeletally immature patients have found that QTAs are more mature on 1-year postoperative MRI scans compared with HTA. 1
Lateral extra-articular tenodesis (LET) is often used in cases of revision ACLR as a means by which to add extra stability to the knee. This procedure can also be added to primary ACLR in patients with risk factors for ACL retear, including high-grade pivot shift (3+), ligamentous laxity, > 10° recurvatum, age <25 years, and patients who play high-risk pivoting sports (eg, soccer, basketball).12,33 The addition of LET to an ACLR has been associated with lower graft retear rates at short-, mid-, and long-term follow-up13,25,32 and improved graft maturity on MRI scans at 2 years postoperatively in skeletally mature patients. 35 The impact of LET on ACL graft maturity in skeletally immature persons has yet to be elucidated.
However, the addition of LET to an ACLR in a skeletally immature patient is not without risks. Given the proximity of the proximal LET fixation site to the distal femoral physis, there is risk of growth disturbance after LET in skeletally immature patients.7,17 Only 1 previous study has evaluated the rate of growth disturbance after ACLR+LET in skeletally immature patients. 11 Those authors found that 5.4% of patients experienced femoral overgrowth at minimum 3.5-year follow-up after ACLR with HTA and LET.
The aim of the present study was to compare the SIR of QTAs in skeletally immature patients with and without LET at 6 months, 1 year, and 2 years postoperatively. The secondary aim of the study was to evaluate the safety of LET in skeletally immature patients. We hypothesized that QTAs in skeletally immature patients with LET would be more mature (lower average SIR values) across all postoperative timepoints compared with QTAs in patients without LET and that the addition of LET would not be associated with any physeal disturbance-related reoperations, including distal femur hemi-epiphysiodesis or epiphysiodesis, at a minimum 2-year follow-up.
Methods
Study Participants
After institutional review board approval of the study protocol, we performed a retrospective review of 74 patients who underwent ACLR with QTA by the senior authors (D.W.G. and F.A.C.) between 2015 and 2021 at a single institution. Patients undergoing primary ACLR with open distal femoral and proximal tibial physes were included in the study if they had an MRI ordered for physeal monitoring at a minimum of 2 years postoperatively. Patients were excluded from the study if they had a graft other than QTA (either HTA, bone-patellar-bone autograft, or iliotibial band [ITB] autograft, n = 33) or were undergoing revision ACLR (n = 1) or if the knee MRI scan was obtained for a new injury (n = 11) (Figure 1). Demographic information, surgical information, MRI scan data, return-to-sport data, and new injury data were collected.

Flowchart with inclusion and exclusion criteria for the cohort. ACL, anterior cruciate ligament; ACLR, ACL reconstruction; BTB, bone-tendon-bone; HS, hamstring; ITB, iliotibial band; QTA, quadriceps tendon autograft.
Surgical Technique
QTA Harvest
A 5 cm–longitudinal incision was made from the superior pole of the patella, extending proximally. Dissection was carried to the level of the quadriceps tendon, and a double-bladed scalpel, with blades set 10 mm apart, was used to divide the tendon to a length of approximately 60 to 65 mm for all-epiphyseal ACLR or 70 to 75 mm for complete transphyseal ACLR. The full-thickness tendon graft was dissected sharply from the patella (without a bone plug), and the remaining quadriceps tendon defect was reapproximated with a combination of interrupted No. 1 Vicryl sutures (Ethicon).
The target diameter of the graft was 9 to 10 mm. The proximal and distal ends of the graft were secured with a nonabsorbable #2 suture using a modified Krakow technique (combination of locking and nonlocked running suture) to a depth of 20 mm and the TightRope implants (Arthrex) were included proximally and distally during graft preparation.
All-Epiphyseal ACLR
After standard diagnostic arthroscopy, debridement, and notch microfracture, the femoral tunnel was prepared, leaving 2 to 3 mm of the back wall remaining. A 1-cm incision was made just anterior to the lateral epicondyle, and a guide pin was inserted under fluoroscopic guidance from a position just anterior to the lateral epicondyle, aimed medially toward the intercondylar notch, with care to avoid the distal femoral physis. The guide pin was then overdrilled with a FlipCutter (Arthrex) from lateral to medial until the ACL footprint in the intercondylar notch was reached. Fluoroscopy was then used again to confirm FlipCutter position outside of the physis. A mallet was used to place a drill sleeve through the lateral femoral cortex to a depth of 7 mm to ensure that a 7-mm bone bridge would remain between the proximal end of the femoral tunnel and lateral cortex. The FlipCutter was then deployed and reamed retrograde to a depth of 25 mm. The FlipCutter was removed and replaced with a FiberStick (Arthrex) for later graft passage.
The tibial footprint was then prepared, and a tibial ACL guide was used to place a guide pin through the anteromedial portal in the epiphysis under fluoroscopic guidance to confirm position outside of the physis. The tibial guide pin was then similarly overdrilled with a FlipCutter, a drill sleeve was placed through the proximal medial tibial cortex to ensure a 7-mm bone bridge remains, and the FlipCutter was deployed and drilled retrograde. A FiberStick was similarly used for graft passage.
The graft was passed all-inside, first through the femoral tunnel, using a FiberStick. The TightRope was passed through the femoral cortex, flipped, and secured to the lateral femoral cortex. The tibial end of the graft was then shuttled through the tibial tunnel using a FiberStick. The whipstitched sutures were then passed through a button and secured to the proximal medial tibial cortex. The graft was tensioned in full extension, first on the femoral side and then the tibial side.
Transphyseal ACLR
Standard diagnostic arthroscopy was performed, and the tibial and femoral footprints were debrided. A guidewire was placed at the femoral footprint, with 2 to 3 mm of backwall remaining, and drilled through the femur, crossing the physis and out of the lateral femoral cortex. The femoral socket was then reamed with a FlipCutter. A guidewire was placed at the tibial footprint, and a reamer was used to drill the tibial tunnel outside-in. FiberStick sutures were used to assist with graft passage. The graft was first passed through the femoral tunnel, followed by the tibial tunnel. The TightRope on the femoral side was deployed first and tensioned. The tibial side of the graft was secured with an adjustable button system loop (Arthrex), and the TightRope was then deployed and tensioned. Fluoroscopy was used to confirm deployment of the TightRopes and buttons.
Lateral Extra-articular Tenodesis
A 5-cm incision was made over the ITB at the lateral aspect of the knee. A 10-mm strip of ITB was harvested over the central third of the tendon starting 8 cm from Gerdy’s tubercle and working proximally to the level of the lateral epicondyle, leaving the ITB attached at Gerdy’s tubercle. The ITB was passed deep to the lateral collateral ligament and secured proximal and posterior to the femoral insertion of the lateral collateral ligament with either a SwiveLock anchor (Arthrex) or a FiberTak anchor (Arthrex) with the knee at 30° of flexion and neutral tibial rotation. The ITB graft was then passed superficial to the lateral collateral ligament and secured to itself with No. 1 Vicryl suture.
Postoperative Rehabilitation
Postoperatively, patients were placed in a hinged knee brace and made partially weightbearing (50%) with crutches for 4 weeks postoperatively if they had ACLR with or without LET and 6 weeks postoperatively if they had a concomitant meniscal repair. Range of motion was 0° to 90° for the first 4 to 6 weeks, after which time patients were allowed to begin full range of motion and a gradual increase in weightbearing as tolerated. Clearance to return to sports was considered at 9 to 12 months postoperatively depending on patients’ progress with physical therapy.
Signal Intensity Measurements
MRI examinations were obtained using a scanner with a 1.5-T or 3.0-T magnet with a dedicated knee coil (General Electric). As part of the routine knee examination, nonfat suppressed sagittal images were obtained using fast-spin echo sequences with a 3.5-mm slice thickness, a bandwidth of 31.25 to 62 kHz, a 16-cm field of view, a matrix of 512 × 320 to 480, an echo train length of 11 to 16, a relaxation time of 3500 5000 ms, and an echo time of 20 to 36 ms.
Measurements were performed using picture archiving and communication system software (Sectra IDS7). The single sagittal slice that demonstrated the majority of the intra-articular ACL graft and the insertion of the posterior cruciate ligament (PCL) onto the tibia was chosen, and 3 circular regions of interest (ROI) were chosen on the intra-articular graft (proximal, middle, and distal) (Figure 2). An additional ROI was chosen at the PCL insertion as the PCL signal intensity. The average value of the 3 ROIs from the intra-articular graft was recorded as the ACL graft signal intensity. The SIR was then calculated from the following previously described formula1,30:
The signal intensity measurements were performed by 2 blinded raters: an orthopaedic surgery resident (J.S.R.) and a research assistant trained by a musculoskeletal radiologist (D.C.). Two-way mixed-effects intraclass correlation coefficients were calculated for interrater reliability of the SIR values at each timepoint.

Sagittal MRI scan in a patient at 24.9 months after ACLR+LET. ROI circles were placed at 3 locations along the graft (proximal, middle, and distal) and at the PCL insertion. ACL, anterior cruciate ligament; ACLR, ACL reconstruction; LET, lateral extra-articular tenodesis; MRI, magnetic resonance imaging; PCL, posterior cruciate ligament; ROI, region of interest.
Physeal Disturbance–Related Reoperation Rate
Chart review was used to obtain information regarding physeal disturbance–related reoperations for the ACLR+LET group, including distal femoral hemi-epiphysiodesis or epiphysiodesis.
Statistical Analysis
Data analysis was performed using STATA (Version 17.0, StataCorp). Discrete variables were reported as percentages. Shapiro-Wilk tests were performed to evaluate for normality of data distribution, and continuous variables were reported as means and standard deviations for normally distributed data and medians and interquartile ranges (IQRs) for non-normally distributed data. Univariate analyses were performed with either unpaired 2-tailed t tests or Wilcoxon rank-sum tests for continuous variables, and Fisher exact tests were used for categorical variables. Multivariate linear regression analysis was performed to control for confounding factors, and statistically significant variables from univariate analysis were used as inputs to the model. Statistical significance was evaluated at the α < 0.05 level.
Results
The records of 73 patients were initially reviewed, and after application of exclusion criteria, 29 patients (39.7%) who underwent ACLR with QTA with or without concomitant LET remained for analysis. Of this cohort, 16 patients (55.2%) were included in the ACLR+LET group, and 13 patients (44.8%) underwent ACLR only. The mean age of all patients was 13.57 years, 41.4% were female, 37.9% of the affected knees were right knees, and 58.6% underwent transphyseal ACLR. With regard to concomitant procedures, 34.5% underwent medial meniscal repair, 48.3% underwent lateral meniscal repair, and 6.9% underwent partial lateral meniscectomy. The average follow-up was 3.3 ± 1.0 years. Further patient and surgical characteristics, alone and according to study group, are shown in Table 1. The ACLR+LET group had significantly more female patients (62.5% vs 15.4%, P = .022) and underwent ACLR via transphyseal technique more often (81.3% vs 30.8%, P = .010) than the ACLR-only group. Otherwise, there were no significant between-group differences (Table 1).
Demographic and Surgical Characteristics Overall and by Study Group a

Data are reported as mean ± SD or n (%). Boldface P values indicate statistically significant difference between groups (P < .05). ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; LET, lateral extra-articular tenodesis; LMR, lateral meniscal repair; MMR, medial meniscal repair; PLM, partial lateral meniscectomy.
The mean time to 6-month, 1-year, and 2-year MRI scan for the entire cohort was 6.15 ± 1.12, 11.66 ± 1.19, and 25.15 ± 4.12 months, respectively (Table 2). Overall, 90% of patients had a 6-month postoperative MRI scan, 72% had a 1-year postoperative MRI scan, and all patients had a 2-year postoperative MRI scan. There were no differences with regard to demographic factors between those with versus those without MRI scan at 6 months and 1 year, respectively (Table 3). There were no differences with regard to the proportion of patients who had MRI scan available or MRI magnet strength (1.5-T vs 3.0-T) between groups at each timepoint (Table 2).
MRI Information Overall and by Study Group a

Data are reported as mean ± SD (range) or n (%). ACLR, anterior cruciate ligament reconstruction; LET, lateral extra-articular tenodesis; MRI, magnetic resonance imaging; NA, not applicable.
Differences in Characteristics Between Patients With Versus Without 6-Month and 1-Year Postoperative MRI Scan a

Data are reported as mean ± SD or n (%). BMI, body mass index; LMR, lateral meniscus repair; MMR, medial meniscus repair; MRI, magnetic resonance imaging; PLM, partial lateral meniscectomy; PMM, partial medial meniscectomy.
The 2-way mixed effects intraclass correlation coefficients were 0.75 for the 6-month postoperative SIR values, 0.65 for the 1-year postoperative SIR values, and 0.78 for the 2-year postoperative SIR values, indicating moderate-to-good interrater reliability. 18
At 2 years postoperatively, the SIR was significantly lower in the ACLR+LET group (median, 1.33; IQR, 1.14-1.56) compared with the ACLR-only group (median, 1.86, IQR: 1.68-2.09; P = .0012) (Table 4, Figure 3). There were no differences in SIR values by MRI magnet strength (1.5-T vs 3.0-T) at 6 months, 1 year, and 2 years postoperatively (P = .34, .85, and .36, respectively).
Univariate Analysis Comparing Postoperative SIR values at 6 Months, 1 Year, and 2 Years Postoperatively by Study Group a

Data are reported as mean ± SD or median [interquartile range]. Boldface P value indicates statistically significant difference between groups (P < .05). ACLR, anterior cruciate ligament reconstruction; LET, lateral extra-articular tenodesis; SIR, signal intensity ratio.
Wilcoxon rank-sum test.
Unpaired 2-tailed t test.
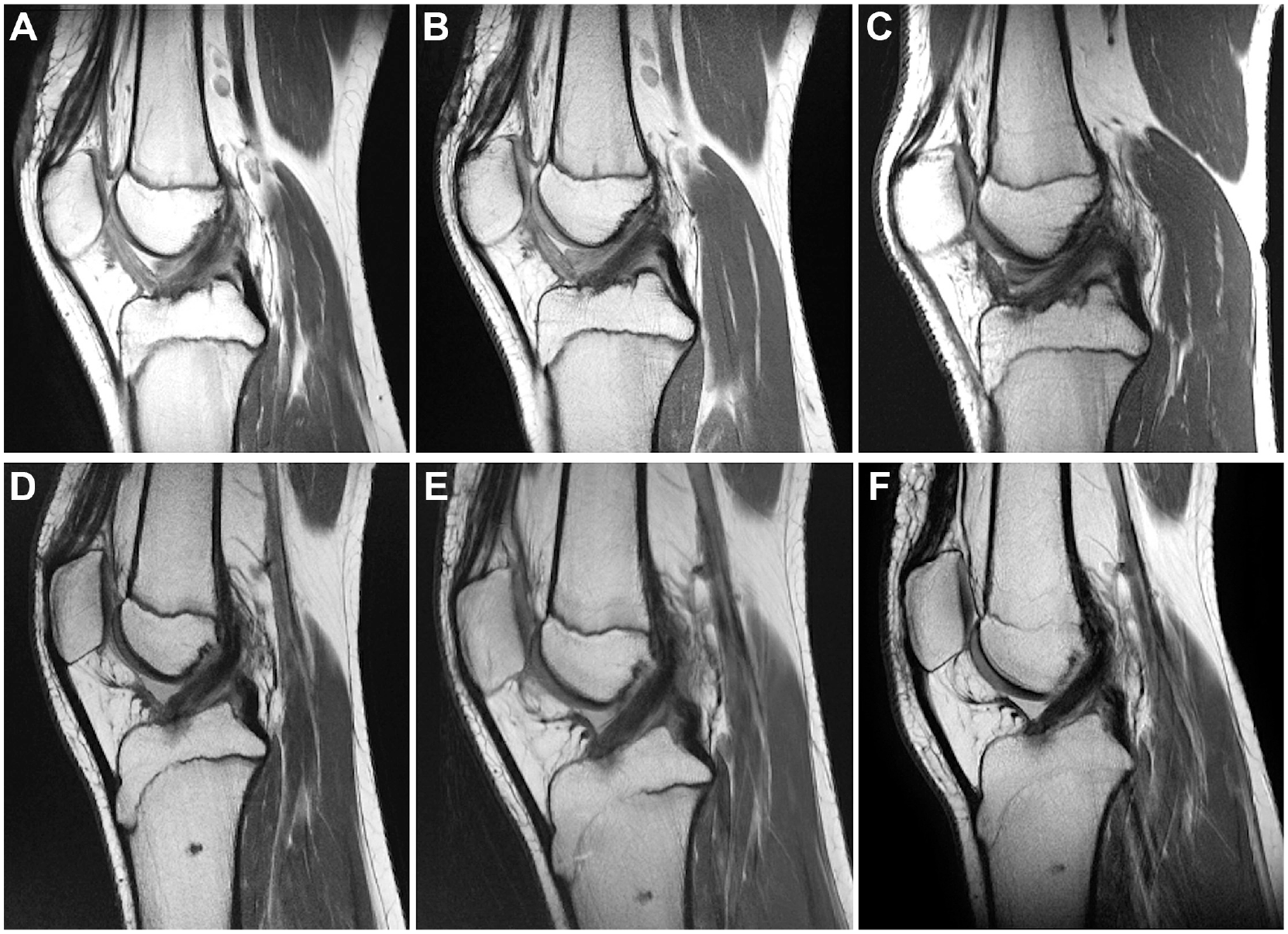
Sagittal MRI scans at 6 months, 1 year, and 2 years postoperatively in patients who underwent (A-C) ACLR only and (D-F) ACLR+LET. ACLR, anterior cruciate ligament reconstruction; LET, lateral extra-articular tenodesis; MRI, magnetic resonance imaging.
The statistically significant variables from the univariate analysis, including presence or absence of LET, surgical technique, and sex, were used as inputs to the multivariate linear regression analysis model. Results indicated that presence of LET was associated with lower SIR values (β = -0.49; 95% CI, -0.91 to -0.05; P = .029) after adjusting for surgical technique (β = -0.28; 95% CI, -0.70, 0.15; P = .19) and sex (β = -0.10; 95% CI, -0.51 to 0.32; P = .63) (Table 5).
Multivariate Regression Analysis Controlling for Confounding Variables a

Boldface P value indicates statistical significance (P < .05). LET: lateral extra-articular tenodesis.
The majority of patients undergoing LET underwent fixation with a polyether ether ketone (PEEK) SwiveLock anchor (13, 81%, Table 6). There were 3 instances of peri-implant cystic changes with the PEEK SwiveLock anchors (Figure 4), but no cystic changes with either the FiberTak anchor or fixation using interrupted nonabsorbable sutures to the periosteum of the lateral femoral condyle. For all patients with cystic changes, changes first appeared at the 6-month postoperative MRI scan and persisted at most recent (minimum 2-year postoperative) MRI scan. The majority of patients (n = 14, 88%) were skeletally mature at most recent MRI follow-up, and 2 patients (12%) had partial closure of the distal femoral and proximal tibial physes at most recent MRI (Table 6).
Complications After LET by Fixation Type a
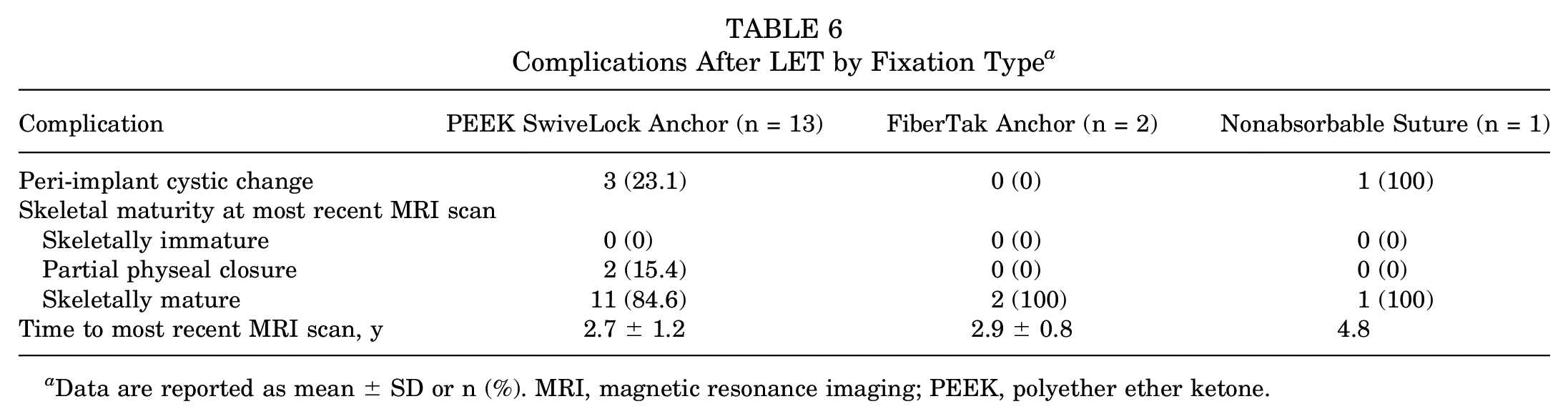
Data are reported as mean ± SD or n (%). MRI, magnetic resonance imaging; PEEK, polyether ether ketone.
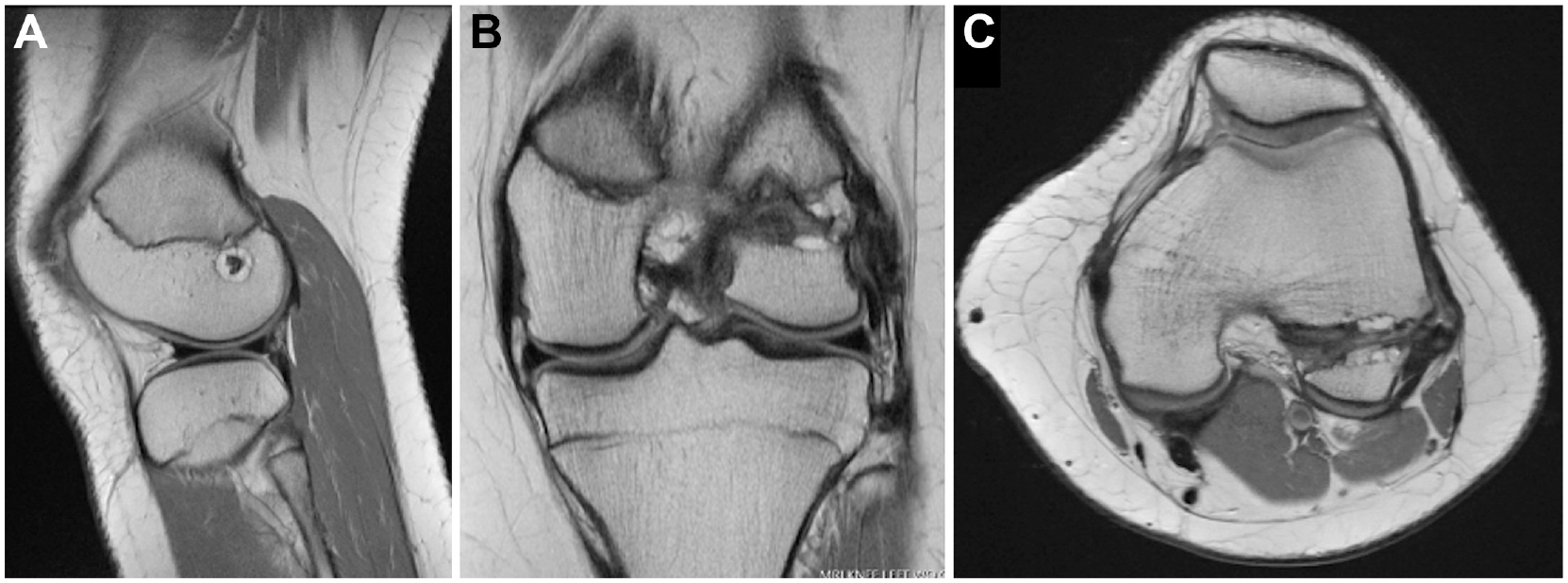
(A) Sagittal, (B) coronal, and (C) axial MRI sequences demonstrating peri-implant cystic changes in a patient after ACLR+LET with SwiveLock anchor. ACLR, anterior cruciate ligament reconstruction; LET, lateral extra-articular tenodesis; MRI, magnetic resonance imaging.
All patients returned to sports at the most recent follow-up (Table 7). A total of 11 patients (37.9%) underwent reoperation, and the average time to reoperation was 2.47 ± 0.85 years for the entire cohort; 1 patient in the ACLR-only group sustained an ACL retear and underwent revision ACLR+LET at 5.2 years postoperatively. In the entire cohort, 4 patients sustained a contralateral ACL tear for which they underwent ACLR+LET. There were no differences with regard to reoperation rate and time to reoperation between the 2 groups (Table 7). No patients in the ACLR+LET group underwent distal femoral epiphysiodesis or hemi-epiphysiodesis. One patient in the ACLR-only group developed a physeal bar in the posterolateral femoral physis for which they underwent a distal femur varus/extension osteotomy at 2.5 years postoperatively. None of the removal of hardware procedures or suture granuloma excisions were related to the LET incision or LET suture anchors.
Return to Sport, Complications, and Reoperations Overall and by Study Group a

Data are reported as mean ± SD or n (%). ACLR, anterior cruciate ligament reconstruction; LET, lateral extra-articular tenodesis; LLD, leg length discrepancy; MRI, magnetic resonance imaging; NA, not applicable.
Discussion
We found that LET is associated with improved graft maturity 2 years after ACLR with QTA in skeletally immature patients after controlling for both surgical technique and sex. Although there was no statistically significant difference between groups at 6 months and 1 year postoperatively, SIR values tended to be lower in the ACLR+ LET group compared with the ACLR-only group at both timepoints. We speculate that improved graft maturity seen on MRI scan with the addition of LET to an ACLR may be due to the protective effect of the LET on the ACL graft. Previous clinical studies have shown that the addition of LET to an ACLR is associated with lower rerupture rates compared with ACLR alone.13,25,32 Biomechanical studies have shown that some of the forces through the ACL graft are transferred to the LET, which may offload the graft as it undergoes the process of ligamentization,9,23 and contribute to the conferred stability to the graft with the addition of LET. 8 The findings from the present study provide important insight into the potential biological impact of LET on ACL graft strain and, thus, ACL graft maturity.
Several previous studies have used MRI scans to evaluate ACL graft maturity. In a small MRI study of 17 patients who underwent ACLR, Lutz et al 21 evaluated HTA maturity on MRI at 1 and 2 years postoperatively using SNQs. Although the authors found that graft maturity was not correlated with clinical outcomes postoperatively, they did find that improved graft maturity was associated with an increased likelihood of return to the preinjury level of sport. By contrast, Weiler et al 34 found that lower ACL graft SIR values on MRI scan were associated with higher load to failure, stiffness, and tensile strength of the ACL graft in sheep. In addition, previous studies have shown that improved ACL graft maturity on MRI scan is associated with higher Lysholm scores 3 , and decreased ACL graft maturity on MRI scan is associated with lower Tegner scores. 34
Previous studies evaluating ACL graft maturity, such as the study by Lutz et al 20 described above, have used the formula SNQ = (signal [ACL graft] - signal [PCL])/signal (background). 3 This formula is advantageous in that it diminishes the impact of the heterogeneity of different MRI techniques and machines by accounting for variable background signal intensities. However, in the present study, the scarring induced by the QTA harvest would dramatically change the background signal measurements and make our measurements less reliable. Therefore, SIR was used in place of SNQ in this study. The technique used in the present study is similar to that used by several other authors.1,31
Two previous studies have evaluated the impact of LET on ACL graft maturity.4,35 Cavaignac et al 4 found that the addition of LET to HTA was associated with improved graft maturity at 1 year postoperatively, whereas Ye et al 35 found improved graft maturity at 2 years postoperatively in patients undergoing ACLR with HTA+LET compared with those without concomitant LET.
Our study builds on the existing literature by examining a population of skeletally immature patients undergoing ACLR with QTA only. QTA and HTA are not equivalent or interchangeable graft types. Pennock et al 28 found that skeletally immature patients with HTA were 5 times more likely to sustain an ACL reinjury compared with those with QTA. With regard to functional outcomes, Nyland et al 26 found that QTA has less pivot-shift laxity and lower failure rates than HTA. On MRI scan, Aitchison et al 1 found that QTA had improved graft maturation at 1 year postoperatively compared with HTA in adolescent athletes. Therefore, an assessment of graft maturation between QTA and HTA should be considered separately, as there are inherent differences in the structural and functional properties of HTA and QTA, which may impact the timing of the process of graft ligamentization, and, thus, appearance of graft maturity on MRI.
The present study also builds on the existing literature describing the safety of LET by evaluating reoperation rates for physeal disturbance-related complications, including angular deformity and leg length discrepancy. We found that LET is a safe procedure in skeletally immature patients, as there were no cases of reoperation for physeal disturbance at most recent follow-up in the ACLR+LET group, with the majority of patients at or near skeletal maturity. We did find 3 cases of peri-anchor cystic changes surrounding the PEEK anchors in our cohort. Peri-anchor cyst formation has been described previously with polylactic acid enantiomer biodegradable suture anchors in patients undergoing rotator cuff repair and arthroscopic shoulder labral repair,6,16,19,24 although peri-anchor cyst formation has not been shown to affect clinical or functional outcomes postoperatively. None of the peri-PEEK anchor cysts in our cohort were symptomatic or required removal of hardware. Importantly, none were associated with physeal disturbance. It should be noted that we now employ an all-suture 2.6-mm FiberTak anchor in an attempt to preclude the development of peri-anchor cysts. Thus far, we have not identified peri-anchor cysts to be present in our postoperative imaging, but further follow-up is necessary.
While we employ the use of a Quality of Movement Assessment before clearance for return to sport, 14 patients are often eager to progress through the postoperative rehabilitation process and return to sport as soon as possible after ACLR. However, even with progressive strength and conditioning and agility training through the Quality of Movement Assessment, grafts must be mature for patients to minimize their risk of ACL reinjury upon return to sport. It is important for surgeons to understand the timing and processes underlying ACL graft maturation so that patients can progress through the postoperative rehabilitation process and return to sports at the appropriate time without placing undue risk on the healing graft and potentially compromising graft integrity. The results from the present study suggest that LET is a safe procedure in skeletally immature patients, and the addition of LET to a QTA ACLR improves graft maturity 2 years postoperatively. Additional longitudinal studies are needed to determine how graft maturity at 2 years postoperative correlates with future risk of ACL rerupture in skeletally immature patients with and without LET.
Limitations
There are several limitations of the present study. Given that this was a retrospective study, the addition of LET to an ACLR was not randomized and, thus, our results are subject to treatment bias. There was also heterogeneity in the types of procedures performed (transphyseal vs all-epiphyseal), although results of our multivariate regression analysis suggest that the presence of LET is associated with decreased SIRs 2 years postoperatively. Multivariate regression analysis did now show a significant independent effect of surgical technique used or patient sex.
Although there was a mixture of different MRI magnet strengths in each group (1.5 T vs 3.0 T), there were no statistically significant differences with regard to magnet strength by group at each time point (ACLR+LET vs ACLR only). In addition, there was no association between magnet strength and SIR values at each timepoint. In addition, not every patient had complete MRI scan information available at 6 months and 1 year postoperatively, although there were no differences in demographic factors between the patients with versus without imaging available at these timepoints. There were no cases of reoperation for physeal disturbance in the ACLR+LET group at minimum 2-year follow-up. All but 2 patients in this group were skeletally mature at final follow-up; therefore, it is possible that these 2 patients could experience physeal disturbance before skeletal maturity, albeit unlikely given that both patients were nearly skeletally mature at most recent MRI scan.
Conclusion
The addition of LET to an ACLR with QTA was associated with lower average SIR values and thus improved graft maturity 2 years postoperatively compared with ACLR alone in skeletally immature patients. The addition of LET to an ACLR was found to be safe in skeletally immature patients.
Footnotes
Final revision submitted June 1, 2023; accepted June 20, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: F.A.C. has received nonconsulting services from Arthrex, royalties from Arthrex, and consulting fees from Arthrex. D.W.G. has received consulting fees from OrthoPediatrics and Arthrex, royalties from Arthrex and Pega Medical, nonconsulting fees from Arthrex and Synthes. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Hospital for Special Surgery (ref No. 2015-366).

