Abstract
Background:
Apoptosis and autophagy are known to be correlated with the extent of damage in torn rotator cuffs, and there is no biological evidence for self-recovery or healing of the rotator cuff tear.
Purpose:
To establish in a rat model of partial- and full-thickness rotator cuff tears how a glycogen synthase kinase 3β (GSK-3β) inhibitor affects the expression of apoptotic and autophagic markers.
Study Design:
Controlled laboratory study.
Methods:
Twelve-week-old Sprague Dawley rats were divided into 3 groups (n = 16 per group). Group 1 acted as the control, with no treatment; group 2 received partial-thickness (right side) and full-thickness (left side) rotator cuff tears only; and group 3 received the same rotator cuff injuries, with GSK-3β inhibitor injected afterward. The tendons from each group were harvested 42 days after surgery. Evaluation of gene expression, immunohistochemistry, and TUNEL staining (terminal deoxynucleotidyl transferase–mediated dUTP nick end labeling) were performed for the following markers: caspases 3, 8, and 9 as well as Bcl-2 (B-cell lymphoma 2); BAX (Bcl-2-associated X protein); beclin 1; p53; and GSK-3β; which represented apoptotic and autophagic reactions. Statistical analysis was performed using 1-way analysis of variance.
Results:
In the group 2 rats with partial- and full-thickness tears, there were significant increases in the mRNA levels (fold changes) of all 8 markers as compared with group 1 (control). All these increased markers showed significant downregulation by the GSK-3β inhibitor in partial-thickness tears. However, the response to the GSK-3β inhibitor in full-thickness tears was not as prominent as in partial-thickness tears. The number of TUNEL-positive cells in group 2 (partial, 35.08% ± 1.625% [mean ± SE]; full, 46.92% ± 1.319%) was significantly higher than in group 1 (18.02% ± 1.036%; P < .01) and group 3 (partial, 28.04% ± 2.607% [P < .01]; full, 38.97% ± 2.772% [P < .01]), and immunohistochemistry revealed increased expression of all the markers in group 2 as compared with control.
Conclusion:
The apoptotic and autophagic activity induced in a rat model of an acute rotator cuff tear was downregulated after treatment with a GSK-3β inhibitor, particularly with partial-thickness rotator cuff tears.
Clinical Relevance:
A GSK-3β inhibitor may be able to modulate deterioration in a torn rotator cuff.
Rotator cuff tears are common musculoskeletal diseases that may reduce quality of life as the result of shoulder pain. Other than simple external factors, such as trauma or impingement, rotator cuff tears are influenced by intrinsic factors, including degeneration, inflammation, and apoptotic or autophagic cell death, and are known to progress gradually from tendinopathy. 39
Apoptosis, the highly regulated physiological process of programmed cellular suicide, has been reported to be involved in the pathogenesis associated with rotator cuff tears. 20,21,28 Proinflammatory cytokines, which also drive apoptosis, were found to be significantly upregulated in human rotator cuff tears. 25 Autophagy is a programmed process that degrades long-lived cellular proteins and damaged organelles to maintain cellular homeostasis. 32,33,36,43 Autophagy plays an important role in remodeling cells in response to outer stimuli. 32 Although its mechanism is not fully understood, autophagy is known to interact with apoptosis in a complex manner. 2,22,27,43 Apoptosis and autophagy correlate positively with the extent of damage sustained by the extracellular matrix (ECM) in human rotator cuff tears. 39 These ongoing processes surely do not work in a positive manner on torn rotator cuffs, as there is no biological evidence for self-recovery or healing of the rotator cuff tear yet.
Glycogen synthase kinase 3 (GSK-3) is a serine/threonine protein kinase that phosphorylates and inactivates glycogen synthase. 4,8,15 Among the isoforms of GSK-3, GSK-3β is involved in stress-induced cellular apoptosis, and GSK-3β activation occurs before the initiation of the caspase cascade on apoptosis. 1,40 However, despite evidence that apoptosis and autophagy are observed in the pathology of rotator cuff injuries, no attempt has been made to regulate them by modulating the activities of GSK-3.
Just because apoptosis and autophagy have been shown to correlate with the extent of ECM degeneration in biopsies taken from rotator cuff tears, this does not mean that either or both are the cause of ECM degeneration. Apoptosis and autophagy are observed in biopsies of chronically torn tendons, but that is as much as we can say in cross-sectional studies. 20,39 However, regarding the positive correlation of apoptosis and autophagy to the extent of cuff damage, we thought that there could be a chance to modulate disease progression by the regulation of the confirmed process. In fact, given its involvement in various cellular events, modulation of GSK-3 activity has been examined in various clinical areas, including nervous system disorders, diabetes, inflammatory diseases, cancer, and bone diseases. 3,5,8,23
On a practical level, research on apoptotic activity in human rotator cuff tears contains a chronic component, and it has always been challenging to implement chronic tears in an animal model. Despite clear evidence that apoptosis takes place with acute injury of the rotator cuff in humans and animals, we decided to use a rat model of partial- and full-thickness rotator cuff tears to evoke apoptotic and autophagic reactions. 21,24,26 The purpose of this study was to examine the effect of a GSK-3β inhibitor on rotator cuff tears in rats and elucidate how the expression of apoptotic and autophagic markers was affected. We hypothesized that there would be an increase in the expression of these markers after rotator cuff tears and that this increased activity would be downregulated after treatment with a GSK-3β inhibitor.
Methods
This study was approved by the Institutional Animal Care and Use Committee of our institution. Twelve-week-old Sprague-Dawley rats (N = 48) with a mean weight of 400 to 500 g were used. The rats were randomly assigned to 1 of 3 groups (n = 16 for each group): Group 1 received no treatment and acted as the control; group 2 received partial- and full-thickness rotator cuff tears (right and left, respectively); and group 3 received the same rotator cuff injuries, plus an injection of GSK-3β inhibitor after the injury. All rats were sacrificed 6 weeks (42 days) later, and we performed gene expression, immunohistochemistry, and TUNEL staining (terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling) analysis of apoptotic and autophagic activity between the groups (Figure 1).

Diagram showing group allocation and study design. BAX, Bcl-2–associated X protein; Bcl-2, B-cell lymphoma 2; GSK-3β, glycogen synthase kinase 3β.
Surgical Procedures and Tissue Harvesting
The rats allocated to groups 2 and 3 were anesthetized with an intraperitoneal injection of ketamine (75-95 mg/kg) and xylazine hydrochloride (5 mg/kg). All operations were performed using a sterile technique. After shaving and sterilization, an incision was made at the shoulder with deltoid splitting. The anterior margin of the supraspinatus was identified adjacent to the biceps tendon, and the posterior margin was determined by the junction with the infraspinatus tendon fibers. On the right shoulder, the tendon was scratched using a scalpel blade on the bursal side to make a partial-thickness tear. A partial tear of the rat rotator cuff was made according the process of Gereli et al 10 by sharply detaching 50% of the tendon, but during our pilot study, this method tended to lead to a full-thickness tear. So rather than making indistinguishable lesions between partial- and full-thickness tears, we decided that it would more distinguishable to make a partial tear by scraping and preserving continuity. Thus, the tendon was not completely detached; the articular side remained attached to the greater tuberosity. On the left shoulder, the tendon was sharply dissected from the greater tuberosity for the establishment of a full-thickness tear (Figure 2).

Rotator cuff injury on a 12-week-old Sprague Dawley rat. (A) Partial-thickness rotator cuff tear on the right shoulder, without exposure of the humeral head. Continuity of the damaged tendon remains. (B) Full-thickness rotator cuff tear on the left shoulder, with exposure of the humeral head.
Scaffolds made of resin were used to prevent spontaneous healing of the tendon stump. The split deltoid was repaired. The rats in group 3 received an additional intraperitoneal infusion of GSK-3β inhibitor (2 mg/kg, SB216763; InvivoGen) immediately after wound closure. 13,14,34 The infusion was performed daily for 3 weeks postoperatively. Rats, housed in wire-topped cages, were allowed unrestricted cage activity without any immobilization. Subcutaneous injection of ketoprofen (5 mg/kg) was made for 5 postoperative days for pain control. The cages were exposed to a 12-hour light-dark cycle.
The tendons were harvested at 6 weeks after surgery, as it was considered enough time for healing of a manipulated rotator cuff injury in a rat model. 19 Through the same incision, the remaining supraspinatus was exposed and sharply detached at the humeral insertion with a scalpel at both shoulders. To harvest the full-thickness tear, the tissues were obtained from the unhealed end of the torn rotator cuff. All surgical procedures and tissue harvesting were executed by a single surgeon (H.J.L.).
RNA Extraction and Gene Expression Analysis
The obtained tissues were evaluated using real-time reverse transcriptase polymerase chain reaction (PCR). The tissues were stored in RNAlater solution (Applied Biosystems/Ambion) at –20°C. Total RNA was extracted using an RNeasy Fibrous Tissue Mini Kit (Qiagen), and RNA purification was performed with the 260/280-nm light absorption ratio set at >1.7.
The messenger RNA (mRNA) expression of the following 8 markers was analyzed: caspases 3, 8, and 9 as well as B-cell lymphoma 2 (Bcl-2); Bcl-2–associated X protein (BAX); beclin 1; p53; and GSK-3β. Each marker was chosen to represent the activity of apoptosis, autophagy, or the tendon’s reaction to injury. Caspases are a family of protease enzymes playing essential roles in apoptosis. 12,18,37 BAX and Bcl-2 are regulatory proteins that regulate cell death by inhibiting or inducing apoptosis 7,29,30,33,43 ; beclin 1 is a protein known to have a central role in the regulation of autophagy by interacting with Bcl-2 38,42 ; and p53 is a well-known tumor-suppressing agent, with one of its most important functions being to activate apoptosis and induce autophagy. 9,35,41
Quantitative real-time reverse transcriptase PCR was performed using the iTaq Universal SYBRGreen Supermix (Bio-Rad Laboratories) and an iScript cDNA Synthesis Kit (Bio-Rad Laboratories) on a CFX96 Real-Time Detection System (Bio-Rad Laboratories). The final solution was used directly for PCR amplification. Each cDNA reaction was diluted 5 times, with a calibrator sample containing the transcript of interest, and run in 25 µL of Bio-Rad SYBR Green using 10 pmol of primers designed with Primer-BLAST software (http://www.ncbi.nlm.nih.gov/tools/primer-blast). The PCR program consisted of 1 initial denaturation step (3 minutes at 94°C) and 40 cycles of denaturation (10 seconds at 95°C), annealing (20 seconds at a temperature optimal for each gene), and amplification (30 seconds at 72°C), followed by melt curve determination consisting of 1 denaturation step (10 seconds at 95°C) and 1 annealing step (0.5 seconds at 65°C). Calculation of relative mRNA levels was performed with CFXmanager software according to the 2–▵▵CT method. 31 The values obtained for each gene were normalized to the β2 microglobulin housekeeping gene and the uninjured control. Gene-specific oligonucleotide sequences are listed in Table 1.
Primer Sequences a

a Bcl-2, B-cell lymphoma 2; BAX, Bcl-2–associated X protein; GSK-3β, glycogen synthase kinase 3β.
TUNEL Staining
To detect apoptotic activity at the single-cell level, an apoptotic assay was performed using the deoxynucleotidyl transferase method of end labeling (Promega). A 5-µm–thick paraffin section of tendon tissue was deparaffinized and rehydrated with a graded series of ethanol in water. The tendon tissues were treated with 20 μg/mL of proteinase K (Sigma-Aldrich) for 15 minutes at room temperature. A 0.1% triton X-100 solution was then applied to the cells at 4°C for 5 minutes. The sections were next incubated in a TUNEL solution at 37°C for 1 hour. The reaction was quenched with a stop/wash buffer. After being washed, the sections were mounted using Vectashield Mounting Medium with DAPI (4′-6-diamidino-2-phenylindole, 1.5 μg/mL; Vector Laboratories) and washed 3 times with phosphate-buffered saline (PBS). Each section was observed under a fluorescence microscope in a wavelength range of 520 ± 20 nm (green) and 460 nm (DAPI). Apoptotic cells were identified as those with nuclei stained in green.
Immunohistochemistry
Immunohistochemical staining was performed for the histological assessment of the tendon tissue. After fixing overnight with 10% buffered formalin, each sample was washed and dehydrated through a graded series of alcohol. Each specimen was embedded in paraffin and cut into 4-µm thickness for attachment to the slide. It was treated with Dako REAL Peroxidase-Blocking Solution (Agilent Tech) for 15 minutes, 0.1% Triton X-100 solution for 10 minutes, and 10% normal goat serum (Vector Laboratories) for 20 minutes. Overnight incubation with the primary antibodies (mouse monoclonal antihuman caspase 3, caspase 8, caspase 9, BAX, Bcl-2, beclin 1, p53, and GSK-3β) was performed at 4°C. After washing with PBS, biotinylated secondary antibody (Vectastain Elite ABC Kit; Vector Laboratories) was applied for 20 minutes. To catalyze chromogen development in 3,3′-diaminobenzidine tetrachloride, streptavidin-conjugated peroxidase was applied. After counterstaining with hematoxylin, the stained sections were examined under optical microscopy to determine the distribution of caspase 3, caspase 8, caspase 9, BAX, Bcl-2, beclin 1, p53, and GSK-3β in the control samples and torn cuff tissue with and without GSK-3β inhibitor treatment. The slides were scanned with a Panoramic MIDI Scanner (3DHISTECH), and the amount of stained marker was expressed quantitatively using an automated method. Positively stained pixels were digitally selected on all slides, and the total positivity ratio of immunoreaction for the entire slide was calculated, which is the total number of positively stained pixels divided by the total number of pixels.
Statistical Analysis
Statistical analysis was performed using 1-way analysis of variance. The Dunnett T3 post hoc test was performed for the significant values. All statistical measurements were carried out using SPSS PC Version 15.0 (IBM). P <.05 were considered significant.
Results
Gene Expression of Apoptosis- and Autophagy-Related Markers
All evaluations were performed in the same experiment, but the data are expressed in separate graphs for easier interpretation. The levels of caspases 3, 8, and 9 as well as BAX; Bcl-2; beclin 1; p53; and GSK-3β, showed significant increases with partial-thickness tears, and all increases in the partial-thickness tears were significantly restricted by the GSK-3β inhibitor (Figure 3).
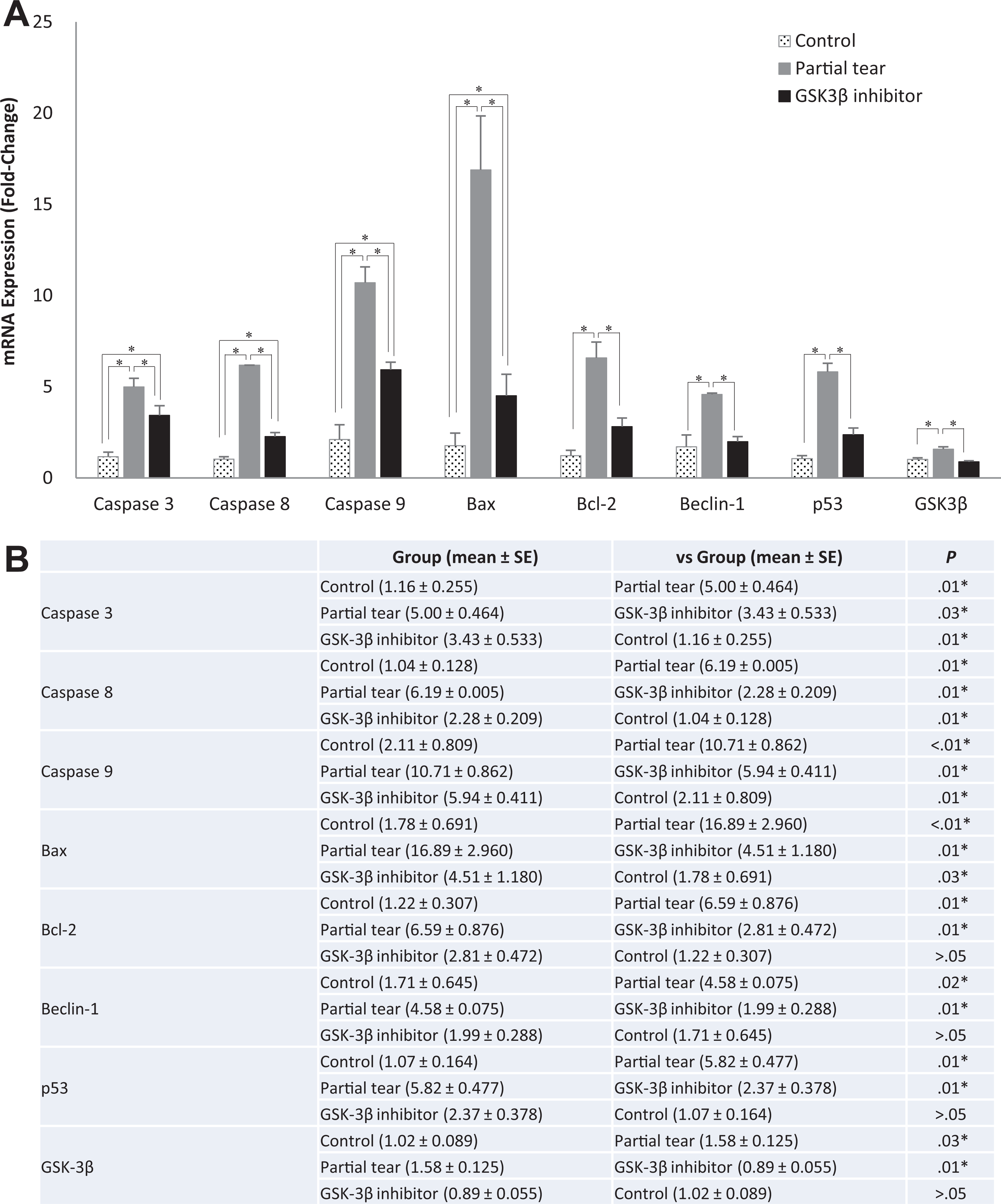
(A) Bar graph and (B) table show the mRNA levels of caspases 3, 8, and 9 as well as BAX; Bcl-2; beclin 1; p53; and GSK-3β after partial-thickness rotator cuff tears with and without GSK-3β inhibitor treatment and in the cuff tissue of the control rats. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). BAX, Bcl-2–associated X protein; Bcl-2, B-cell lymphoma 2; GSK-3β, glycogen synthase kinase 3β; mRNA, messenger RNA.
All significant increases of the evaluated markers were shown in full-thickness tears as in partial-thickness tears; however, the response to the GSK-3β inhibitor was not as prominent as in partial-thickness tears. The increased levels of caspases 3, 8, and 9 with full-thickness tears were not significantly affected by the GSK-3β inhibitor. The BAX, Bcl-2, and beclin 1 levels also increased after full-thickness tears, but there was no significant downregulation by the GSK-3β inhibitor (Figure 4).

(A) Bar graph and (B) table show the mRNA levels of caspases 3, 8, and 9, as well as BAX, Bcl-2, beclin 1, p53, and GSK-3β, after full-thickness rotator cuff tears with and without GSK-3β inhibitor treatment and in the cuff tissue of the control rats. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). BAX, Bcl-2–associated X protein; Bcl-2, B-cell lymphoma 2; GSK-3β, glycogen synthase kinase 3β; mRNA, messenger RNA.
The results for p53 were similar to those of the caspases, increasing with tears and affected by the GSK-3β inhibitor treatment after partial-thickness tears but showing no significant influence of GSK-3β inhibitor on full-thickness tears. The level of GSK-3β increased after partial- and full-thickness rotator cuff tears, and the increase was significantly restricted by GSK-3β inhibitor, showing that the function of GSK-3β inhibitor is well implemented.
TUNEL Staining for Apoptotic Cells
The number of TUNEL-positive cells in each group was compared. The in situ TUNEL assay revealed a significantly large number of positively stained cells in cuff samples from partial- and full-thickness tears, whereas no stained cells were present in the control samples. Fewer apoptotic cells were observed in the samples treated with the GSK-3β inhibitor as compared with the untreated tear samples, with statistical significance with partial-thickness tears (Figure 5).

(A) TUNEL staining (terminal deoxynucleotidyl transferase–mediated dUTP nick end labeling). Apoptosis determined by TUNEL (green), counterstained with DAPI (blue), and visualized under a fluorescence microscope. (B) Bar graphs and (C) table show the percentage of TUNEL-positive cells in partial- and full-thickness tears. Error bars indicate standard error. Asterisks indicate statistically significant differences between groups (P < .05). GSK-3β, glycogen synthase kinase 3β.
Immunohistochemistry Analysis
Immunohistochemistry revealed increased levels of the 3 caspases, BAX, Bcl-2, beclin 1, p53, and GSK-3β in the samples from partial- and full-thickness tears as compared with the control samples. Significant responses were shown with the treatment of the GSK-3β inhibitor on caspase 3 (P = .01), caspase 8 (P = .01), and caspase 9 (P = .01), as well as BAX (P < .01), Bcl-2 (P = .01), p53 (P < .01), and GSK-3β (P < .01) with partial-thickness tear. In full-thickness tears, significant responses were shown on caspase 8 (P < .01), Bcl-2 (P = .02), and GSK-3β (P = .02) (Figures 6 and 7).

Immunohistochemistry for caspases 3, 8, and 9. *P < .05. F, full-thickness tear; GSK-3β, glycogen synthase kinase 3β; P, partial-thickness tear.

Immunohistochemistry for BAX, Bcl-2, beclin 1, p53, and GSK-3β. *P < .05. BAX, Bcl-2–associated X protein; Bcl-2, B-cell lymphoma 2; F, full-thickness tear; GSK-3β, glycogen synthase kinase 3β; P, partial-thickness tear.
Discussion
The major finding of our study was that with a partial-thickness tear, the levels of all 8 markers tested (caspases 3, 8, and 9; BAX; Bcl-2; beclin 1; p53; and GSK-3β) showed significant increases with the injury and significant downregulation with the GSK-3β inhibitor. The number of TUNEL-positive cells with partial- and full-thickness tears was higher than in the control and GSK-3β inhibitor conditions. Immunohistochemistry revealed increased expression of all the evaluated markers in the torn cuff samples as compared with the control. The expression of the markers in the torn cuff was decreased by treatment with the GSK-3β inhibitor.
This study showed that apoptosis and autophagy, which are known to be related to the pathology of chronic rotator cuff tears, can also be induced in an animal model of acute rotator cuff tears. These induced reactions of apoptosis and autophagy were clearly affected by the GSK-3β inhibitor.
Apoptosis, the physiological process of programmed cellular suicide, is controlled by various proteins, including a family of cysteine proteases known as caspases. 6,16 Different cascades of caspase activity are involved in the 2 pathways of apoptosis: intrinsic and extrinsic. Whereas apoptosis is caspase-dependent and morphologically characterized by chromatin condensation, cell shrinkage, DNA fragmentation, and the formation of apoptotic bodies, autophagy is a caspase-independent process that proceeds through a specific sequence of cellular events causing the bulk degradation of cellular proteins and organelles through the formation of autophagosomes and lysis by lysosomal hydrolases. 27,38 The interplay between apoptosis and autophagy is complicated, and they cross-regulate each other in excitatory and inhibitory manners. Apoptotic and autophagic reactions are positively correlated with the damage caused by rotator cuff tears. 11,17,20,39
We found a significant increase in the level of caspase 8 in partial-thickness tears, which we expected because caspase 8 is involved in the extrinsic apoptotic pathway that responds to the outer stimuli. In the extrinsic pathway, the autoactivation of caspase 8 at the cell surface of the death receptor–inducing signaling complex is followed by the activation of effector caspases, which function as downstream effectors of the cell death program. In an earlier study that evaluated apoptotic activity after a human rotator cuff tear, caspase 9, which is one of the initiators of the intracellular mitochondrial-mediated intrinsic apoptotic pathway, increased regardless of its location on the torn tendon. 20 We also found a significant increase in the expression of caspase 9 after an acute injury to a rat rotator cuff, despite our relatively short follow-up after the imposed injury and without prolonged exposure to risk factors that could affect the condition of the cuff. Intrinsic and extrinsic factors responded to the partial tear of the rotator cuff; caspase 3, which belongs to the common pathway following caspases 8 and 9, additionally increased. In the rats given an infusion of a GSK-3β inhibitor, all of the caspase increases seen with the partial-thickness tears were downregulated. However, neither the increase in the caspases after injury nor the changes induced by the GSK-3β inhibitor were significant after a full-thickness rotator cuff tear.
BAX is a regulatory protein known for its proapoptotic activity. Similar to the results for the caspases, BAX increased significantly with a partial injury to the cuff and was affected by the treatment of GSK-3β inhibitor. Meanwhile, Bcl-2, which is known to have antiapoptotic activity by binding to BAX, showed a pattern similar to that of BAX. According to the study by Fabis et al, 7 BAX and Bcl-2 expression showed opposing results depending on the site of the torn cuff. However, we obtained these markers from the same site in this experiment, so we assume that the expression of pro- and antiapoptotic regulators was upregulated to maintain homeostasis within the acutely damaged rotator cuff. Bcl-2 also plays an important role in suppressing autophagy by binding to beclin 1. 29,30,33 Beclin 1, which is a substrate of caspases 3, 7, and 8 in the intrinsic and extrinsic apoptotic pathways, is involved in the initiation of autophagosome formation. 38
The relationship between the factors related to apoptosis and autophagy is highly complicated and varies by circumstance and timing. Research about these activities is common in chronic diseases such as cancer, in which subjects are exposed to the risk factors for enough time to respond to them. However, despite the uncertainty of these reactions in acute settings, apoptotic and autophagic activity was clearly shown in response to the acute injury of the rotator cuff. Furthermore, these activities were downregulated by the GSK-3β inhibitor, especially in the partial-thickness tears. The results of this study are cellular data, not functional data, and cannot be interpreted as induction of actual healing of the injured tendon. However, regarding the finding of this study showing the response of apoptotic and autophagic factors to the GSK-3β inhibitor, we believe that it is valuable to be able to modulate the deterioration that can actually affect the condition of the torn cuff.
This study has limitations that require consideration. First, the experimental period was not long enough to guarantee the complete observation of the full cascade of reactions. Also, to clearly reveal the mechanism and influence of one factor on another, a periodic analysis is needed to compare differences at different time points. We analyzed the responses after the cuff injury at a single time point and thus cannot explain the cascade of reactions in the torn cuff. However, this one-time analysis was objective enough to verify the existence of certain reactions to the injury and treatment. Second, we used a rat model of acute rotator cuff injury in this study. Although it shares many anatomic features with human cuff tendons, it is not possible to reproduce the same chronic process that leads to rotator cuff tears in humans. These histological alterations and manipulations made on rats cannot be precisely adapted to the changes on movement or ambulation. In addition, we were not able to figure out the effect of resin scaffold that was inserted to prevent spontaneous healing of the cuff on the interaction of GSK-3β inhibitor. Third, although the expression of several mRNA transcripts changed as a result of the tear and the treatment with the GSK-3β inhibitor, we examined the expression of transcripts from the whole tendon and did not localize changes to specific cell types or locations within the tendon. Fourth, we used a bilateral model and did not include sham surgery. This study was designed to conserve rats and decrease surgical time, but patients do not usually have surgery on both shoulders at the same time unless it is a trauma setting and completely necessary. Given these reasons, we were not able to evaluate the effects of the surgical approach to the shoulder, and surgery on one side would affect the use of the tendon on the other. An additional group with no tear should be treated with inhibitor as another control for the effects of the inhibitor alone.
Despite these limitations, this work provides the novel insight that apoptosis and autophagy are involved in acute rotator cuff tears, and using a GSK-3β inhibitor can coherently affect those processes after partial-thickness rotator cuff tears. Future work will elucidate the mechanism of apoptosis and autophagy in chronic rotator cuff tears, potentially leading to a practical pharmacological application of a GSK-3β inhibitor to regulate and minimize the deterioration of torn rotator cuffs and ultimately lead to actual healing. Also, the data reported in this study would benefit from more experiments, particularly the impact of GSK-3β inhibition after a rotator cuff tear in a surgical repair group. Such an experiment would provide legitimacy to the therapeutic potential of GSK-3β inhibitors.
Conclusion
Apoptotic and autophagic activity, which is known to be observed in the biopsies of chronic human rotator cuff tears, can be induced by an acute rotator cuff tear in a rat model. The reactions were prominent in partial rotator cuff tears and were downregulated by treatment with a GSK-3β inhibitor.
Footnotes
Final revision submitted July 12, 2021; accepted August 24, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding was received from the National Research Foundation of Korea (research grant to H.J.L.; 2017R1D1A1B0303672). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Catholic University of Korea (CUMC-2017-0212-03).
