Abstract
Background:
Bone marrow aspirate (BMA) is a common source for harvesting mesenchymal stem cells (MSCs), other progenitor cells, and associated cytokines and growth factors to be used in the biologic treatment of various orthopaedic pathologies. The aspirate is commonly centrifuged into a concentrated volume that can be immediately administered to a patient using commercially available kits. However, the handling and efficacy of BMA concentrate (BMAC) are still controversial.
Purpose:
To characterize BMA versus BMAC for MSC quantity, potency, and cytokine profile.
Study Design:
Controlled laboratory study.
Methods:
From 8 participants (age, 17-68 years), 30 mL of bone marrow was aspirated by a single surgeon from either the proximal humerus or distal femur and was separated into 2 equal samples. One sample was kept as BMA, and the other half was centrifuged into BMAC. The 2 samples then underwent flow cytometry for detection of MSCs, cell analysis for colony-forming units (CFUs), and cytokine profiling. A 2-tailed t test was used to detect differences between MSCs, CFUs, and cytokine density concentrations between BMA and BMAC.
Results:
The average concentration of MSCs in both BMA and BMAC was 0.001%. Average MSC events detected by flow cytometry were significantly higher in BMA versus BMAC (15.1 and 8.1, respectively; P < .045). Expanded MSCs demonstrated similar phenotypes, but CFUs were significantly increased in BMA compared with BMAC (104 vs 68 CFUs, respectively; P < .001). Total protein concentration and cytokine profiling demonstrated great variability between BMA and BMAC and between patients. Most importantly, BMAC failed to concentrate MSCs in 6 of 8 samples.
Conclusion:
There is great variability in MSC concentration, total protein concentration, and cytokine profile between BMA and BMAC.
Clinical Relevance:
When studying the clinical efficacy of BMAC, one must also evaluate the sample itself to determine the presence, concentration, and potency of MSCs if this is to be considered a cell-based therapy. Further standard operating procedures need to be investigated to ensure reproducible results and appropriate treatments.
Keywords
Recently, mesenchymal stem cells (MSCs) have been investigated as a primary treatment for, as well as augmentation to, numerous orthopaedic procedures. MSCs have the ability to differentiate into multiple musculoskeletal cell types, release cytokines and growth factors, and mitigate the immune response. 5 As such, stem cell therapy is gaining popularity for difficult to treat orthopaedic pathologies such as osteoarthritis, muscle and tendon injuries, and cartilage restoration. 34
One commonly described method of obtaining MSCs is from bone marrow aspiration. 3 Bone marrow aspirate (BMA) not only contains MSCs but also includes platelets, red and white blood cells, and hematopoietic and nonhematopoietic precursors. 5 It is believed that by centrifuging the aspirate, differences in density gradients allow for a concentration of MSCs in an appropriate injectable volume that can be immediately administered to a patient. 5 Other bioactive cells including lymphocytes, neutrophils, monocytes, and platelets, along with growth factors including platelet-derived growth factor (PDGF), transforming growth factor–beta (TGF-β), and bone morphogenic proteins, are also concentrated during this process. 9 This process has been termed “bone marrow aspirate concentrate” (BMAC), and many companies have developed BMAC kits that meet U.S. Food and Drug Administration regulations. 3,17,22
Despite the interest in use of orthopaedic procedures, the use of either BMA or BMAC remains poorly understood, as do the harvesting and processing techniques used to obtain them. Previous studies have suggested that MSCs only represent about 0.001% to 0.01% of the cells in bone marrow concentrate, 27 making it unknown if processes such as BMAC provide enough viable MSCs to have clinical effectiveness. Moreover, often the use of BMA or BMAC in orthopaedic procedures has been improperly defined or advertised as stem cell therapy to render it more appealing. 12,13,18,19,25,28,31
The purpose of this study was to compare the concentration of MSCs, their potency, and the cytokine profile of BMA versus BMAC in a group of patients undergoing surgery. We hypothesized that MSCs obtained from BMA will have superior or equivalent quality and quantity compared with BMAC.
Methods
This study took place from June 2017 through January 2018. A total of 8 consecutive patients between 17 and 68 years of age consented to participate in the study. All participants were under the care of a single surgeon (P.M.). Patients were included if they underwent surgery for anterior cruciate ligament (ACL) tear or rotator cuff surgery during the study period. Exclusion criteria were history of diabetes, blood disorders, hematological malignancy, use of immunosuppressive medications, or medications with bone marrow–suppressive effects. The study protocol received institutional review board approval, and all participants provided informed consent or informed assent for minors.
Bone Marrow Aspiration and Concentration
Using sterile techniques during the anticipated procedure, 30 mL of bone marrow was aspirated from either the proximal humerus (n = 6) or distal femur (n = 2) using the manufacturer’s described technique (BioCUE MINI Platelet Concentration Kit; Biomet Biologics). The aspiration trocar utilized was fenestrated, seated to the first laser line. The location used was based on convenience of the planned procedure. The bone marrow was aspirated from the greater tuberosity for cuff surgery, specifically at the site of anticipated anchor placement. 24 For ACL surgery, the BMA was aspirated from roof of the notch. 8,20 The aspirate was collected into a 30-mL syringe containing 4 mL of anticoagulant citrate dextrose–solution A (ACD-A) to prevent coagulation. The needle was advanced 1 cm and rotated 90° after each 7 mL was aspirated. Half of the volume was retained as the BMA sample for the study to be used as an internal control. The remainder was centrifuged at 3200 rpm for 15 minutes using the BioCUE MINI Platelet Concentration Kit, to be used as BMAC. A total of 3.5 mL of each BMA and BMAC was washed with phosphate-buffered saline and resuspended in alpha modified Eagle’s medium with 16% fetal bovine serum and seeded in a T25 flask. The media was changed every other day until passage 10. Eight patient samples were cultured; each sample had a BMA and BMAC sample.
Flow Cytometry
We analyzed 100 µL of BMA and BMAC with flow cytometry for MSCs markers, as defined by the International Society for Cellular Therapy. 37 This included positive (cluster differentiation [CD] 90, CD73, and CD105) and negative (human leukocyte antigen–DR isotype [HLA-DR], CD45, CD11b, CD19, and CD34) cocktail markers. Conjugated primary monoclonal antibodies and isotype controls were used as recommended by the manufacturer (BD Biosciences). Cells were analyzed on a FACS Fortessa flow cytometer (BD Biosciences) and analyzed using FCS Express (Denovo Software).
Cell Analysis
An equal number of cells from BMA and BMAC were used for colony-forming unit (CFU) assays. Four different seeding densities were chosen: 50, 100, 200, and 500 cells/cm2. Various seeding densities were used to determine if CFUs were affected by initial plating density. 35 Samples of BMA and BMAC were cultured in triplicate using sterile media and standard laboratory techniques. After 2 weeks, CFUs were circled with a 1.8 cm–diameter self-inking marker. A colony was counted if it was ≥1 mm in diameter. 23,26 Doubling time was calculated for cells derived from BMA and BMAC until passage 10.
Protein Profiling
To test any paracrine or downstream growth factor upregulation, protein quantification of the BMA and BMAC was investigated on all the samples using Bio-Rad protein assay (Bio-Rad Laboratories). After quantification, equal amounts of BMA and BMAC were loaded onto the Proteome Profiler Human XL Cytokine Array (R&D Systems) to analyze the presence of 102 different cytokines.
Statistical Analyses
Statistical analysis was performed utilizing a 2-tailed t test to detect differences between MSCs, CFUs, and cytokine density concentrations between BMA and BMAC. An alpha of 0.05 was set as statistically significant. SPSS Statistics (version 21; IBM Corp) was used for statistical processing.
Results
The average age of patients was 51.25 ± 16.6 years (range, 17-68 years), with an average BMI of 26.96 ± 5.1 kg/m2 (range, 20.7-33.3 kg/m2) (Table 1). There were 6 male and 2 female patients. Per the exclusion criteria, none of the included patients had a history of diabetes, blood disorders, or hematological malignancy, and none used immunosuppressive drugs or drugs with bone marrow–suppressive effects.
Patient Characteristics a

a BMI, body mass index.
Flow cytometry demonstrated the heterogeneity of the analyzed samples. In Figure 1, there is representative scatterplot that highlights the difference in cell populations between BMA and BMAC samples. The quantification of MSCs as detected by flow cytometry for the first 1 million events in each 100-µL sample was significantly higher in BMA versus BMAC samples (Figure 2). The overall concentration of MSCs in BMAC was 0.001%. There was a statistically higher number of stem cells isolated in the aspiration compared with the concentrate (15.1 vs 8.1, respectively; P = .045). We were unable to concentrate MSCs utilizing the BMAC methodology in 6 of 8 samples, and in samples 4 and 5 we were unable to detect any MSC events in the concentrate.

Representative side scatterplot of bone marrow aspirate (BMA) versus BMA concentrate (BMAC) samples. Red, highest cell density; blue, smallest cell density. Flow cytometry was used to identify the different cell populations among BMA versus BMAC samples. Patient 5 is displayed in the top row, and patient 6 is displayed in the bottom row. For patient 5, there are 2 distinct populations in the BMA sample, while 3 populations of cells are apparent in the BMAC. Conversely, for patient 6, there are 3 distinct populations in the BMA sample and a more heterogenous distribution in the BMAC sample. FSC-A, forward scatter; SSC-A, side scatter.

(A) Table and (B) bar graph depicting the number of mesenchymal stem cells (MSCs) detected by flow cytometry. The overall concentration of MSCs in bone marrow aspirate concentrate (BMAC) was 0.001%. There was a statistically higher number of stem cells isolated in the aspiration compared with the concentrate (15.1 vs 8.1, respectively; P = .045). (B) Quantification of MSCs as detected by flow cytometry for the first 1 million events was significantly higher in bone marrow aspirate (BMA) versus BMAC samples.
Despite the difference in concentration, when MSCs were grown and expanded, the phenotypes were similar between BMA and BMAC with the typical spindle-like shape (Figure 3). Doubling times were similar until passage 4, after which they diverged (Figure 4B). However, there was no statistical significance between doubling time in BMA and BMAC when the graphs did diverge (P = .1274). MSCs cultured with initial seeding densities of 100 and 500 cells/cm2 demonstrated significantly more colony-forming units (CFUs) after 2 weeks in BMA as compared with BMAC (P < .05) (Figure 4A).
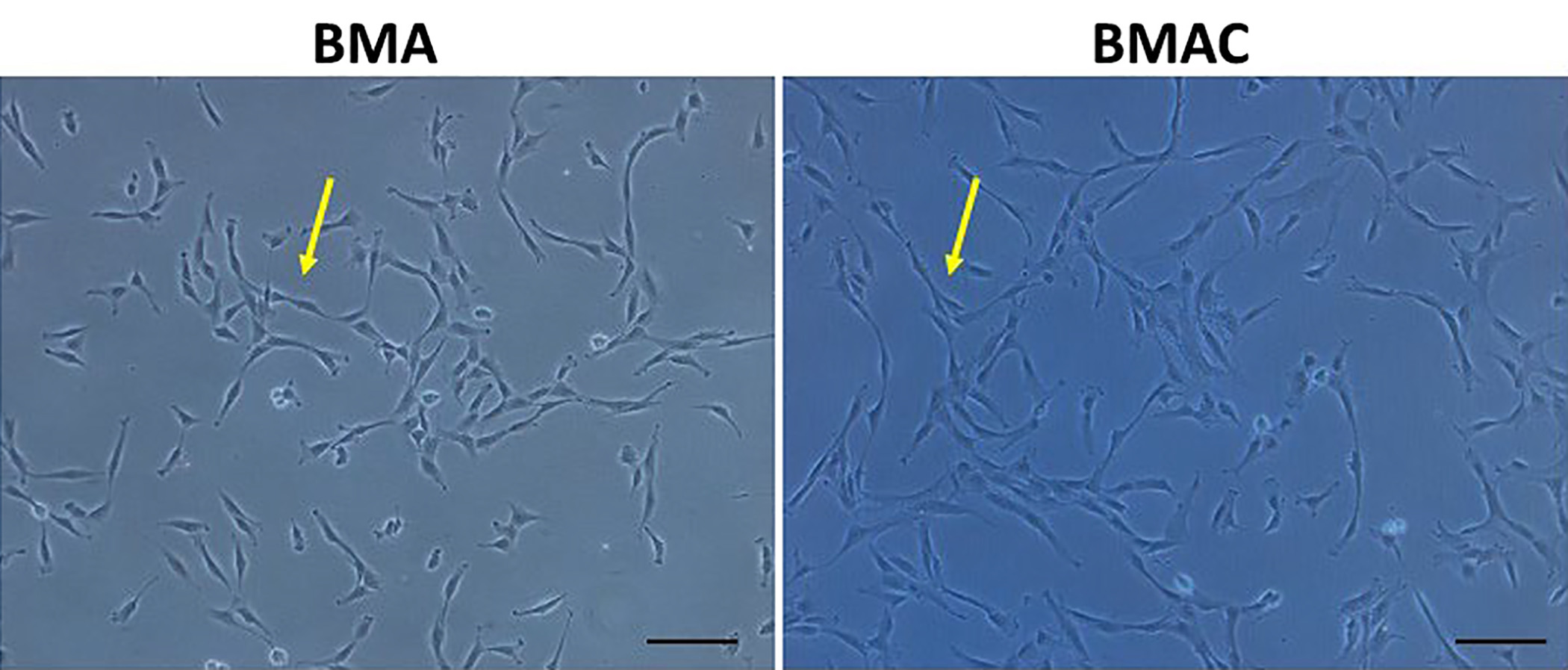
Morphology of mesenchymal stem cells (MSCs) using an inverted microscope. Phenotypes of expanded MSCs are similar between bone marrow aspirate (BMA) and BMA concentrate (BMAC). Both BMA and BMAC yielded MSCs in the typical spindle-like shape (arrows). Scale bar = 10 µM.

(A) Phenotype of bone marrow aspirate (BMA) versus BMA concentrate (BMAC) mesenchymal stem cells (MSCs) and number of colonies with different initial plating number, MSCs cultured with initial seeding densities of 100 and 500 cells/cm2 demonstrated significantly more colony-forming units after 2 weeks in BMA as compared with BMAC (**P < .05, ****P < .001). Scale bar = 10 µM. (B) Mean doubling time of BMAC versus BMA from passages 1 to 10. Doubling times were similar until passage 4, after which they diverged (P = .1274).
The total protein concentration for each sample is quantified in Figure 5A. There is no correlation or clear pattern of protein content between BMA and BMAC or between patients. Half of the patients demonstrated a higher protein concentration in BMAC compared with BMA (patients 2, 4, 6, and 8). We used the Proteome Profiler Human XL Cytokine Array to characterize the protein profile of patients 7 and 8 as a proof of concept (Figure 5B). For patient 7, we found that the protein content in BMAC was 206.05 mg/mL versus 118.0 mg/mL in BMA. Despite this difference, and the considerable amount of protein present, only 5 of 102 screened cytokines were identified using the Proteome Profiler Human XL Cytokine Array: adiponectin, aggrecan, angiogenin, vitamin D binding protein, and tumor necrosis factor–alpha (TNF-α) (Figure 6A). Each cytokine demonstrated a significantly different concentration between BMA and BMAC. Furthermore, the presence of TNF-α, an inflammatory cytokine, was 50 times higher in BMAC than BMA.

(A) Total protein concentration of bone marrow aspirate (BMA) versus BMA concentrate (BMAC) by patient. To test any paracrine or downstream growth factor upregulation, protein quantification of the BMA and BMAC was investigated in all the samples using the Bio-Rad protein assay. Patients 2, 4, 6, and 8 demonstrated higher protein concentration in BMAC compared with BMA. No correlation or clear pattern of protein content between BMA and BMAC or between patients was found. (B) Proteome Profiler Human XL Cytokine Array for patients 7 (left) and 8 (right). Equal amounts of BMA and BMAC were loaded onto the Proteome Profiler Human XL Cytokine Array to analyze the presence of 102 different cytokines.
Patient 8 demonstrated a very different profile. Twenty-three cytokines were identified in both BMA and BMAC (Figure 6B). While adiponectin was significantly higher in concentration in the BMAC of patient 7, it was significantly higher in the BMA of patient 8. Similarly, angiogenin was also significantly higher in the BMAC of patient 7, but higher in the BMA of patient 8. Other regenerative molecules, such as PF4, PFGF-AA, and osteopontin, were significantly higher in concentration in BMA in comparison with BMAC of patient 8.

Proteome Profiler Human XL Cytokine Array for bone marrow aspirate (BMA) versus BMA concentrate (BMAC) in 2 patients. (A) Patient 7 had higher concentrations of protein in BMAC versus BMA, but only 5 of 102 screened cytokines were identified using the Proteome Profiler Human XL Cytokine Array: adiponectin, aggrecan, angiogenin, vitamin D BP, and tumor necrosis factor–alpha (TNF-α). In addition, each cytokine demonstrated a significantly different concentration between BMA and BMAC (*P < .05). (B) In patient 8, a very different cytokine profile was identified compared with patient 7. In this patient, significantly higher concentrations of regenerative molecules such as platelet factor 4 (PF4), platelet-derived growth factor–AA (PFGF-AA), and osteopontin were found in BMA in comparison with cytokines identified in the BMAC sample. a.u., arbitrary unit; BP, binding protein; MIF, macrophage migration inhibitory factor; MMP-9, matrix metallopeptidase–9; RBP4, retinol binding protein 4; SHBG, sex hormone binding globulin; VCAM-1, vascular cell adhesion molecule–1.
Discussion
In this study, we found that there is great variability in MSC concentration, total protein concentration, and cytokine profile between BMA and BMAC techniques. Average MSC events detected by flow cytometry were significantly higher in BMA versus BMAC (15.1 vs 8.1, P < .045), and CFUs were significantly increased in BMA compared with BMAC (104 vs 68, respectively; P < .001). Additionally, total protein concentration and cytokine profiling demonstrated great variability not only between BMA and BMAC but also between patients. We found there was no correlation or clear pattern of protein content between BMA and BMAC or between patients.
It is well known that human BMA contains MSCs. However, the number of MSCs is very low per unit volume; MSCs make up only around 0.01% to 0.02% of the total cell volume. 1,14 Because of the small number of MSCs per unit volume of aspiration, BMAC has been considered as a method to increase the concentration of MSCs in a short time, directly in the operating room. 28 –32 BMAC involves using a density gradient centrifuge in order to concentrate MSCs, and many kits are available. 18,28 However, there are conflicting findings regarding the superiority of BMAC over traditional BMA. Some studies 1,14,30 have found that BMAC is an effective way to concentrate MSCS. Other studies, similar to ours, have found that BMAC does not result in a higher number of MSCs. 29,30,31 In addition, our study, although limited with its small sample number, found that the number of colonies was significantly higher in BMA samples when 100 or 500 cells were plated, suggesting that BMA MSCs have higher viability and are healthier to divide in cell culture. This may be due to the centrifugation process in BMAC having detrimental effects on MSCs in the aspirate. Although the doubling time for BMAC is initially higher, there is not a significant difference between BMA and BMAC MSCs.
In addition, the site of extraction of MSCs may influence the quality of MSCs. We extracted 75% of the MSCs from the proximal humerus, while 25% were extracted from the distal femur. It is possible that had the aspiration occurred at the iliac crest, the differences found between BMA and BMAC would have been less evident. Moreover, the site of extraction may influence the relative quantity of hematopoietic stem cells, which could have an impact on the quantity of MSCs extracted.
In order to evaluate the efficacy of BMAC as a viable option to concentration MSCs, it must be determined if they have any efficacy clinically. There have been a few studies 5,11,12 that have suggested improved outcomes with BMAC injections for various orthopaedic pathologies. In 2014, Gobbi et al 11 demonstrated improvement in radiographic and clinical outcome scores at 3 years of follow-up in 25 patients with grade 4 chondral lesions treated with single-stage BMAC covered by a collagen-based scaffold. The best results were seen in patients younger than 45 years and those with smaller lesions, and thus it was concluded that BMAC is a safe and effective treatment option with no significant complications. 12 Gobbi et al also later compared matrix-induced autologous chondrocyte implantation (MACI) to BMAC in patellofemoral chondral lesions with a minimum 3-year follow-up. 12 They found equivocal improvements in both groups in radiographic and clinical outcome scores at the 2-year follow-up mark. They noted more than 80% complete defect filling on magnetic resonance imaging in those treated with BMAC, and those who underwent second-look arthroscopy with concomitant biopsy revealed hyaline-like cartilage on histology. 12 However, these results are not easily extrapolated to other patient populations with larger lesions and have not been compared with the clinical use of BMA aspirations. Moreover, the combined use of a collagen-based scaffold also introduced another variable, which can affect the final efficacy of BMAC itself.
Other studies 2,13,19,31 were unable to demonstrate improved results with the use of BMAC. A systematic review by Filardo et al 9 identified a number of preclinical and clinical trials looking at MSCs for the treatment of cartilage lesions. However, they concluded that further studies including randomized controlled trials were needed to support the potential of MSCs isolated from BMA or BMAC in cartilage repair. Shapiro et al 31 looked at 25 patients with bilateral knee osteoarthritis. Using the contralateral knee as a control, patients were randomized to receive BMAC injection in one knee and saline placebo in the other. Although both knees saw improvements at 1 week, 3 months, and 6 months, there was no significant difference in those treated with BMAC compared with placebo (P > .09). 31
In this study, we compared the concentration of MSCs, doubling time in vitro, and cytokine analysis between equal samples of BMA versus BMAC collected from the distal humerus or proximal humerus of 8 patients in order to understand which could be of beneficial BMAC use.
Our overall hypothesis was that the concentration procedure might affect the efficacy of the MSCs as well as help concentrate regenerative growth factor, such as that present in platelet rich plasma (PRP), which can lead to better healing. We found that the overall concentration of MSCs in BMAC was consistent with previous literature at 0.001%. 1,14,27 However, we found an inconsistent number of MSCs obtained from BMA between patients and were unable to identify any MSCs in 2 samples of BMAC. We failed to concentrate MSCs via BMAC in 6 of 8 patients. These findings emphasize the lack of consensus in terms of the number of MSCs injected, frequency of injections, volume of BMAC injected, and timing of injections when stem cells from BMAC are used in orthopaedic procedures. In addition, due to the low number of MSCs, it seems improper for us to name BMAC therapy as stem cell therapy.
Recently, Cotter and colleagues 5 reviewed the current literature using BMAC for cartilage defects of the knee. They concluded that the low starting concentration of MSCs in the bone marrow necessitates further investigations as to whether the quantity of bone marrow–derived MSCs results in significant differences in clinical effect. Further higherquality studies are needed to determine the optimal concentration of cells injected and the timing of injections. 5 In order to effectively utilize BMA or BMAC in the clinic, standardization regarding isolation, amount of MSCs injected, frequency of injections, and volume injected is needed. Given the large variation surrounding the number of MSCs that are injected, standardization for clinical practice will most likely be difficult.
A different potential benefit surrounding BMAC treatment is that MSCs become “activated” during the centrifuge processing, allowing them to quickly differentiate into the desired cell lineage. This can also be induced by adding a collagen matrix. 12,27 To test this theory, we cultured MSCs using 4 different initial seeding densities. However, after 2 weeks, we discovered a significantly higher proliferation of cells in the aspiration as compared with the concentrate for half of the cultures. This suggests that the cells may actually become less potent, or more stressed as they are processed and concentrated. Although the MSCs may be more concentrated, their ability to proliferate and differentiate may be affected, suggesting that their use in clinical practice would have little utility. The MSCs may in some way become damaged during the ultracentrifugation process, ultimately affecting their healing potential and clinical use. 29
Another theory is that the beneficial effect of BMAC treatment could be due to augmented protein content instead of the actual presence of MSCs. 6,21 For this reason, we identified and compared the cytokine profile of BMA versus BMAC in order to determine if BMAC had a superior treatment effect not because of the quantity of MSCs obtained, but because of the superior cytokine profile. We anticipated finding higher concentrations of regenerative growth factors such as aggrecan, vitamin D, or platelet derived growth factor (PDGF). It has been suggested the BMAC promotes regeneration by delivering MSCs in addition to platelets and noncellular growth factors such as PDGF and vascular endothelial growth factor (VEGF). 30 PDGF recruits cells involved in the musculoskeletal repair process (ie, MSCs, osteoblasts, osteocytes, and tenocytes) and upregulates angiogenesis. 7,10,15,25,32,33,38 VEGF helps to initiate angiogenesis after tissue injury. 16 However, we were unable to find any clear pattern or correlation because of the great variability both between BMAC and BMA and between patients. Although we would expect to see a higher concentration of these factors in BMAC because of the increased concentration of MSCs, we found no difference in the sample we analyzed. Moreover, in one instance, we unexpectedly found a higher concentration of TNF-α in BMAC, a proinflammatory cytokine. The increase in TNF-α may have kept the MSCs in the concentration from becoming stressed. However, this finding is particularly concerning because MSCs should have regenerative and anti-inflammatory properties, associated with a decrease in TNF-α. The large variability suggests that further investigated is warranted to determine the appropriate method to isolate MSCs. 36 In addition, the effect of the BMAC method on MSCs needs to be further investigated.
Moreover, the clinical efficacy of using a low versus high quantity of MSCs for treatment must be determined. Studies have not yet determined the minimal amount of MSCs necessary to result in a clinically significant difference in outcome. Although the quantity of MSCs needed for injection is still under investigation, our study suggests that the quality of the MSCs extracted also has importance. The variability and difference in doubling time and cytokine expression suggest that the quality of MSCs is just as important a factor to consider as the quantity of MSCs injected.
This study does come with limitations related to the small number of patients included in it. The average age was 51.25 years, and a previous study has shown a decrease in MSC quantity and quality with increasing age. 4 There is also the potential for confounding in harvesting and processing techniques, which was minimized by having all procedures performed by a single surgeon. We were also unable to control for other patient factors, such as sex or harvest location, which has previously been shown to affect MSC concentration. 3 Finally, this study does not evaluate the clinical efficacy of BMAC versus BMA MSCs, as it focused on the basic science of MSCs rather than clinical outcomes.
Conclusion
There is great variability in MSC concentration, protein concentration, and cytokine profile between BMA and BMAC and between patients. Because of the low inherent concentration of MSCs in bone marrow, it is difficult to reliably isolate and concentrate these cells. Furthermore, the significantly lower CFUs in BMAC suggest a lower potency of MSCs compared with BMA. Further studies are needed to determine proper harvesting and processing techniques to reliably standardize the concentration of MSCs injected before they are introduced as “stem cell treatment” in orthopaedic procedures. However, this study strongly suggests that claims of BMAC’s superiority over BMA need to be questioned, and its use limited until additional research is performed to further delineate MSC viability, differentiation, and efficacy between the 2 different techniques. Finally, clinical trials need to be conducted to evaluate specific MSC populations and growth factors to clinical therapeutic effects among BMAC and BMA.
Footnotes
Final revision submitted July 22, 2021; accepted August 24, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: B.J.S. has received education payments from Medinc of Texas and hospitality payments from Wright Medical and Smith & Nephew. D.L. has received education payments from Arthrex/Medinc of Texas and consulting fees from Zimmer Biomet. P.C.M. has received research support from Arthrex and DePuy, education payments from Medinc of Texas, consulting fees from Smith & Nephew, speaking fees from Aastrom Biosciences and Vericel, and honoraria from Vericel. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Houston Methodist Research Institute (Pro00015718).
