Abstract
Objective
Bone marrow aspiration and concentration (BMAC) is becoming a more common regenerative therapy for musculoskeletal pathology. In our current pilot study, we studied patients with mild-to-moderate bilateral knee osteoarthritis, compared pain at 12-month follow-up between BMAC-injected and saline-injected knees, and examined cartilage appearance measured by magnetic resonance imaging (MRI) T2 quantitative mapping.
Design
Twenty-five patients with mild-to-moderate bilateral osteoarthritic knee pain were randomized to receive BMAC into one knee and saline placebo into the other. Bone marrow was aspirated from the iliac crests, concentrated in an automated centrifuge, combined with platelet-poor plasma for knee injection, and compared with saline injection into the contralateral knee. Primary outcome measures were T2 MRI cartilage mapping at 6-month and Visual Analog Scale and Osteoarthritis Research Society International Intermittent and Constant Osteoarthritis Pain scores and radiographs at 12-month follow-up.
Results
Constant, intermittent, and overall knee pain remained significantly decreased from baseline at 12-month follow-up (all P ⩽ 0.01), with no apparent difference between BMAC- and saline-treated knees (all P ⩾ 0.54). A similar significant increase from baseline to 12-month follow-up regarding quality of life was observed for both BMAC- and saline-treated knees (all P ⩽ 0.04). T2 quantitative MRI mapping showed no significant changes as a result of treatment.
Conclusions
BMAC is safe to perform and relieves pain from knee arthritis but showed no superiority to saline injection at 12-month follow-up. MRI cartilage sequences failed to show regenerative benefit with single BMAC injection. The mechanisms of action that led to pain relief remain unclear and warrant further studies.
Introduction
Osteoarthritis of the knee is a common and painful degenerative joint disease. Millions of Americans will suffer from it during their lifetime, with 80% of the population showing some radiographic evidence of osteoarthritis by age 65 years.1,2 Commonly, knee replacement will follow when efforts at symptom management are ineffective. 3 The majority of patients who undergo surgery require an extensive rehabilitation and recovery period, while placing an enormous financial burden on our health care system.4,5 The US Department of Health and Human Services has documented 723,086 knee arthroplasties annually, resulting in national charges of approximately 11 billion dollars. 6
In an attempt to lessen this burden, recent efforts at symptom modification from a biochemical and regenerative therapeutic perspective have focused on use of cell-based therapies to treat osteoarthritis. An example of such a therapy concentrates mesenchymal stem cells (MSCs) harvested from bone marrow with the promise of tissue regeneration owing to their chondrogenic potential.7-10 More recent studies have clarified possible alternative therapeutic mechanisms of cellular action, including paracrine, trophic, anti-inflammatory, and immunomodulatory effects of MSCs.11-15 Despite lack of therapeutic clarity, usage of products containing MSCs continues to increase.16-18 One of the earliest sources of concentrating MSCs for therapeutic use is bone marrow aspiration and concentration (BMAC).19-22 The US Food and Drug Administration (FDA) has published guidance pertaining to the use of BMAC as minimally manipulated under CFR 1271 and approved multiple 510K devices that can concentrate bone marrow.23-25 As a result, the number of clinics and practitioners offering this treatment are on the rise as noted by a recent survey of the health care landscape. 16 Nevertheless, despite several publications documenting use of MSCs or BMAC in arthritic disease,26-37 limited evidence exists to confirm efficacy. Some large registry reviews of concentrated marrow products have been performed, 38 but as yet, no therapy or intervention curing degenerative joint disease or halting the progression has been formally approved by the FDA for marketing.23-25
We have previously reported successful safety and feasibility data for a randomized, placebo-controlled pilot trial of the BMAC procedure. 39 We also found marked improvement in pain relief and activity out to 6 months, but knees injected with saline experienced similar improvement as those injected with BMAC. Herein, we expand on this previous pilot study by reporting additional outcomes and 12-month follow-up data. We additionally attempt to clarify BMAC’s regenerative potential by reporting pre- and postinjection magnetic resonance imaging (MRI) T2 quantitative mapping data. We hypothesized that aforementioned pain relief and activity improvement in both BMAC-treated and saline-treated knees would continue out to 12 months, though unlikely contribute to MRI detectable cartilage regeneration.
Methods
Study Population and Data Collection
The institutional review board approved our study, and the FDA granted Investigational New Drug (IND) approval. Twenty-five patients with painful bilateral knee osteoarthritis treated at a single, tertiary care center between November 1, 2013, and February 28, 2015, were included. As appropriate for a pilot study, the sample size of 25 patients was chosen primarily based on feasibility rather than on formal statistical power calculations. Patients had long-standing, mild-to-moderate knee pain (Kellgren-Lawrence grades 1-3) due to bilateral osteoarthritis, despite conventional medical therapy or arthroscopic intervention for a clinically appropriate period of time. Every patient received an intraarticular injection of BMAC into one knee while a saline placebo was injected into the contralateral knee, thereby utilizing each patient as their own control and eliminating the need for sham bone marrow aspirations. For each patient, BMAC and saline were randomly assigned to knees via dynamic allocation. 40 Patients were blinded as to which knee received the cellular intervention.
Outcomes assessed at baseline (i.e., prior to BMAC and saline injection) and 12-month follow-up incorporated several different measures of patient-reported pain, including the Visual Analog Scale (VAS) pain score, the Osteoarthritis Research Society International Intermittent and Constant Osteoarthritis Pain (ICOAP) questionnaire (the 5 outcomes of interest were constant pain score, intermittent pain score, total pain score, how much constant knee pain has affected quality of life, and how much intermittent knee pain has affected quality of life), and algometry measures. Algometry measurements were performed using a handheld applicator that allows the operator to apply pressure increasingly at 3 specific points on the knee: the medial joint line, 1 cm above the medial joint line, and 1 cm below medial joint line. On application, pressure is applied until the patient reaches a pain/pressure level prompting them to press a handheld button, which records a numeric value and also beeps to inform the researcher to stop; a higher value indicates more pain. Pressure values are analyzed by JTECH Medical tracker system, version 5, software (JTECH Medical). All outcomes were measured by an orthopedic research nurse (SEK) independent of the operating physician (SAS). Although only of secondary interest in this study, each of the aforementioned pain- and activity-related outcomes was also compared to the 6-month follow-up time point. Joint surveillance included bilateral radiographs graded by the principal investigator prior to treatment randomization and confirmed by a musculoskeletal radiologist in questionable cases. Quantitative T2 cartilage mapping MRI was performed at baseline, with follow-up MRI at 6 months and radiographs at 12 months.
Adverse events, including effusions, warmth, erythema, joint line tenderness, and range of motion, were recorded at 12 months. All patients were discouraged from taking any prescription or over-the-counter pain medication for as long as they were pain-free, and asked to report any rescue medication they needed to use, as well as the duration and frequency they used them. BMAC and saline injections were the only treatment interventions made for the duration of the study.
MRI Information
All patients in the study had bilateral knee MRI examinations performed on the same 3.0-T MR unit utilizing a 15-channel transmit/receive phased-array coil. Imaging of all knees was performed in the early morning in an attempt to minimize any effect of prolonged weight-bearing on T2 relaxation times. Imaging protocol consisted of a sagittal, multiecho, spin-echo, T2-weighted sequence performed with the following parameters: repetition time ms/echo time ms; 2800/13.8, 27.6, 41.4, 55.2, and 69.0; field of view 140 × 140 mm; pixel matrix 320 × 320 mm; flip-angle 180°; slice thickness 4 mm; and bandwidth 160 Hz/pixel. T2 relaxation times were obtained from online reconstructed T2 maps by using pixelwise, monoexponential, nonnegative least squares method analysis (
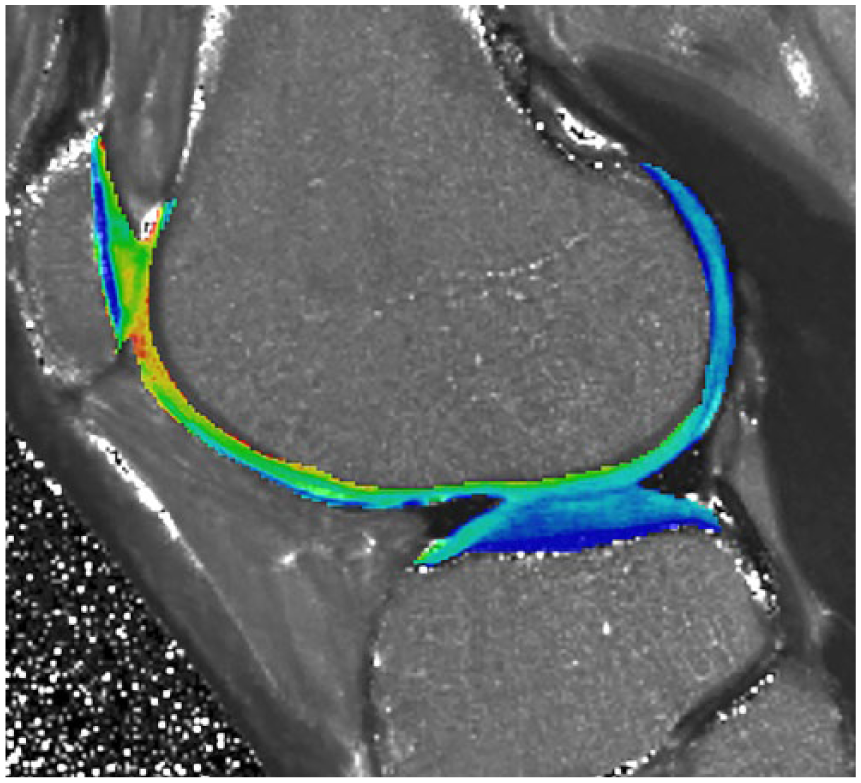
Representative sagittal color-scale T2 map fused with anatomic T2 weighted image (TR 2800/TE 13.8) obtained through the central portion of left lateral femoral condyle and lateral tibial plateau in a study participant at the time of 6-month follow-up.
Surgical Technique
Bone marrow was harvested from each superior iliac crest in the following manner: in a procedure room, the posterior superior iliac spine was manually palpated using a standard technique. 41 After a 1-cm stab wound with an 11-blade scalpel, 26 mL of bone marrow from 3 sites on each iliac crest was harvested using a 11-guage Jamshidi bone marrow aspiration needle for a total of 52 mL of bone marrow aspirate. Aspirated marrow and anticoagulant citrate dextrose solution A were then passed through a sterile 170-µm filter into a separate 60-mL syringe to remove particulate matter. The resulting filtered marrow was transferred to a Magellan Autologous Platelet Separator System (Arteriocyte) for automated centrifugation in a dual spin technique (2800 rpm initial soft spin followed by 3800 rpm hard spin) for resulting marrow cell concentration. The concentration process yielded 6 mL of cellular product; 1 mL of concentrated cells was reserved for analysis that characterized a BMAC product containing a median of 56% mononuclear cells containing 150 × 106 white blood cells, 80 × 106 mononuclear cells, 4.4 × 106 hematopoietic stem cells (HSCs), and 34,000 MSCs in accordance with the minimal criteria for defining MSC set forth by the International Society for Cellular Therapy. These numbers are consistent with other published reports of BMAC (known to have only small numbers of MSCs). A median cell viability of 97% was determined for all samples by flow cytometry using 7-AAD staining.39,42 No activating agent was added to the BMAC.
Patients were blinded to the contents of each injection by a curtain hung just proximal to the patients’ thighs. Both knees were aspirated prior to injection. Treatment cells were combined with 10 mL of previously separated platelet-poor bone marrow plasma to increase the volume of injectate to 15 mL within the knee. This was done to distribute the volume of injectate as best as possible throughout the suprapatellar pouch and knee compartments. Intraarticular injection was performed into the randomly assigned knee through a superolateral approach under continuous ultrasound guidance. The contralateral knee was injected with 15 mL of sterile saline.
Statistical Analysis
All analyses were performed according to the intention-to-treat principle. Comparisons of VAS, ICOAP, and algometer-assessed pain scores; activity level; Kellgren-Lawrence grade; and MRI T2 values between baseline and follow-up time points were made using a paired Wilcoxon signed rank test for the separate BMAC and placebo knee groups. Changes in each of the aforementioned outcome measures from baseline follow-up time points were also compared between the BMAC and placebo knee groups using a paired Wilcoxon signed rank test. P values of 0.05 or lower were considered statistically significant, and all statistical tests were 2-sided. Statistical analyses were performed using SAS (version 9.4; SAS Institute, Inc.) and R Statistical Software (version 3.2.3; R Foundation for Statistical Computing).
Results
After screening a total of 279 patients, 25 patients were included in the study (Supplemental Fig. 1).
Baseline Demographic Information a .
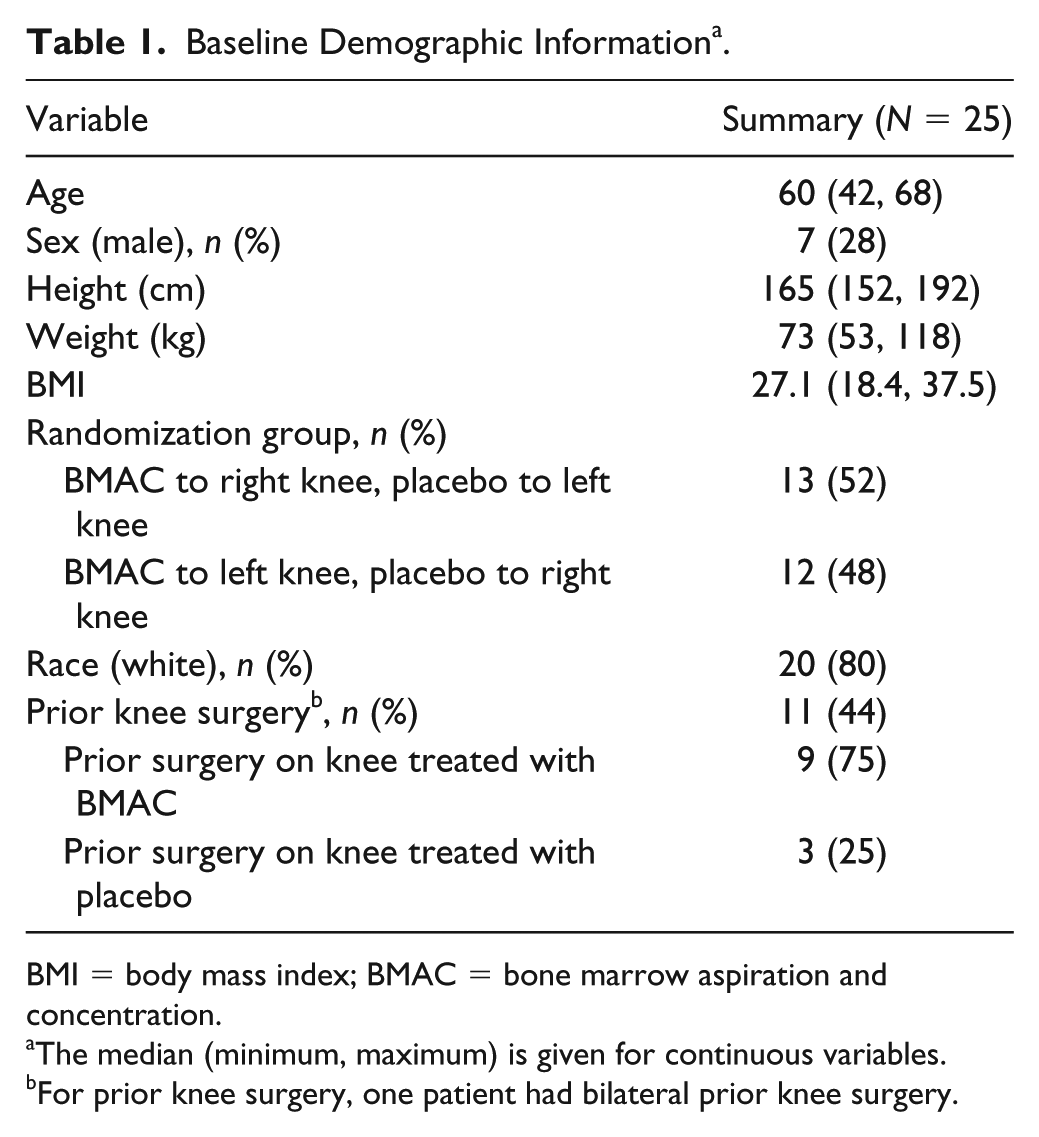
BMI = body mass index; BMAC = bone marrow aspiration and concentration.
The median (minimum, maximum) is given for continuous variables.
For prior knee surgery, one patient had bilateral prior knee surgery.
As shown in
Pain at Baseline and 12-Month Follow-up for Knees Treated with BMAC and Placebo as Measured by the ICOAP Questionnaire, VAS Pain Score, and Algometer-Assessed Pain a .
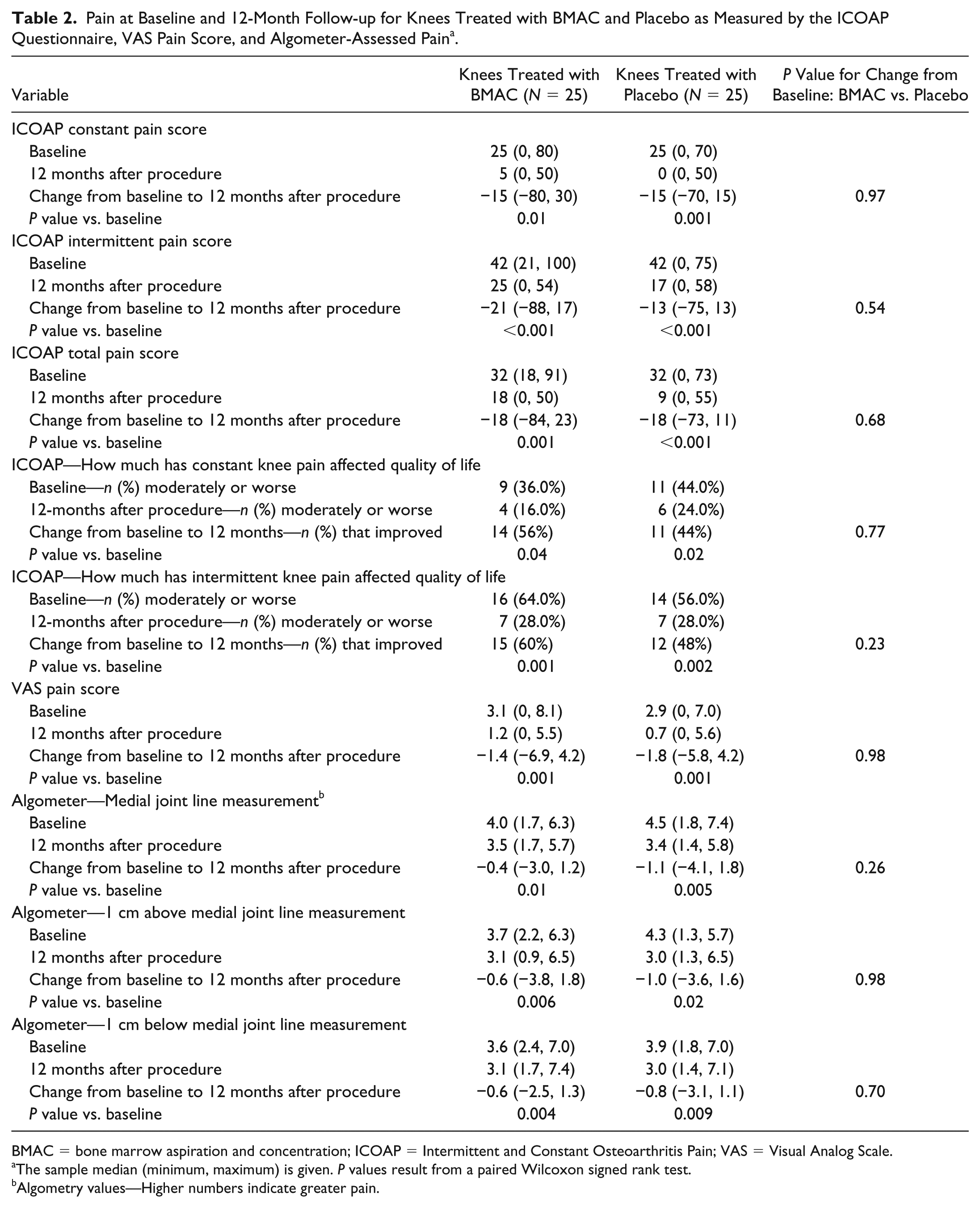
BMAC = bone marrow aspiration and concentration; ICOAP = Intermittent and Constant Osteoarthritis Pain; VAS = Visual Analog Scale.
The sample median (minimum, maximum) is given. P values result from a paired Wilcoxon signed rank test.
Algometry values—Higher numbers indicate greater pain.
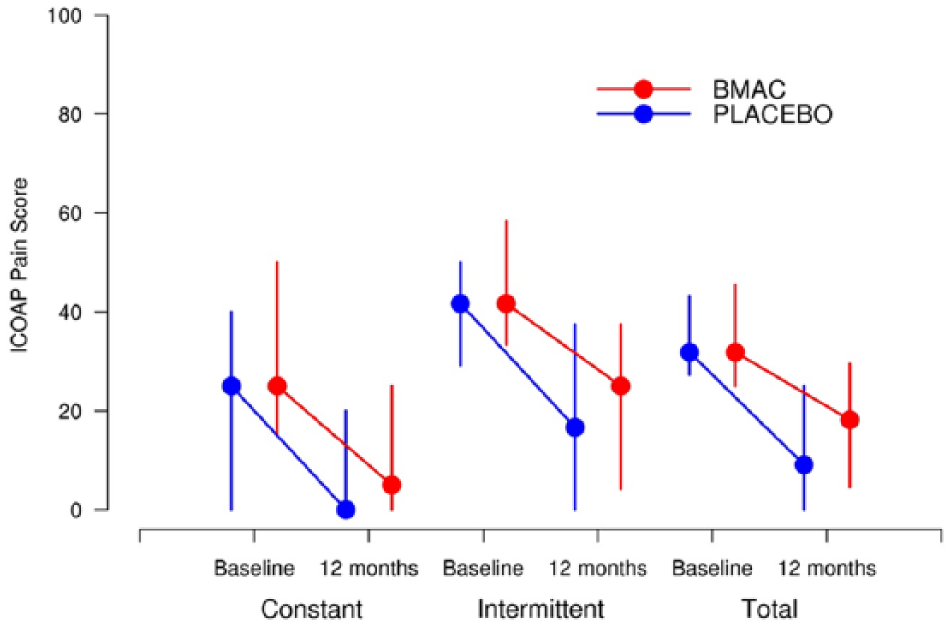
ICOAP constant, intermediate, and total pain score for BMAC and placebo at baseline and 12-month follow-up. Vertical lines extend from the 25th percentile to the 75th percentile. BMAC = indicates bone marrow aspiration and concentration; ICOAP = Intermittent and Constant Osteoarthritis Pain.
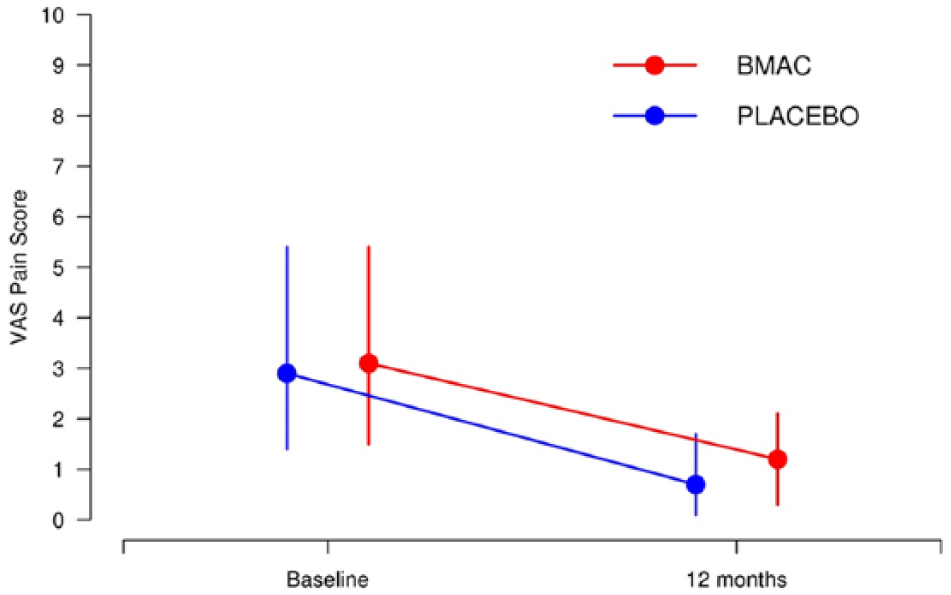
VAS pain score for BMAC and placebo at baseline and 12-month follow-up. Vertical lines extend from the 25th percentile to the 75th percentile. BMAC = indicates bone marrow aspiration and concentration; VAS = visual analog scale.
Activity Level and Kellgren-Lawrence Grade at Baseline and 12-Month Follow-up for Knees Treated with BMAC and Placebo a .
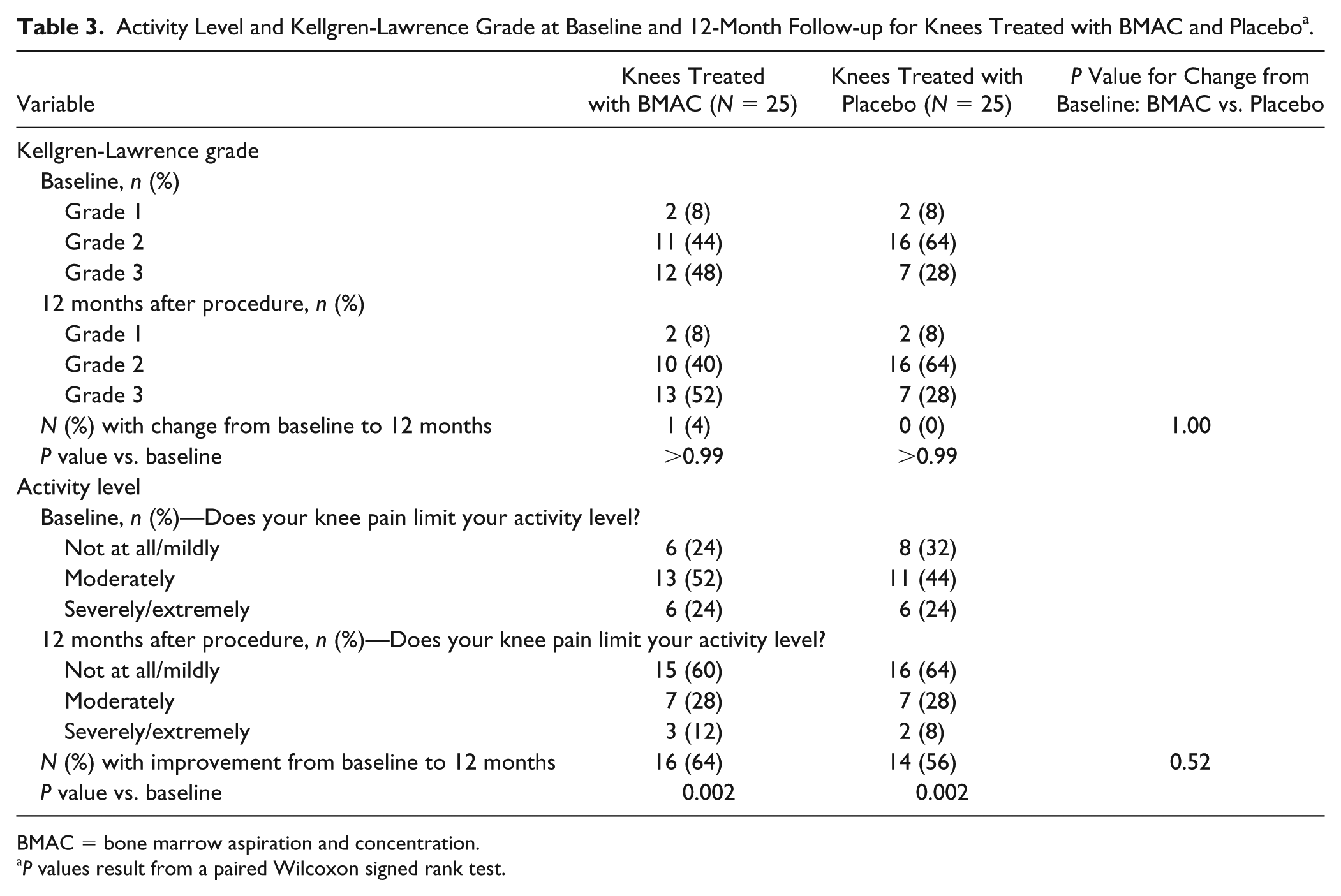
BMAC = bone marrow aspiration and concentration.
P values result from a paired Wilcoxon signed rank test.
Regarding activity level, both BMAC and placebo groups demonstrated a significant improvement in activity level at 12-month follow-up compared with baseline (both P = 0.002;
MRI T2 Values at Baseline and 6 Months for Knees Treated with BMAC and Placebo a .
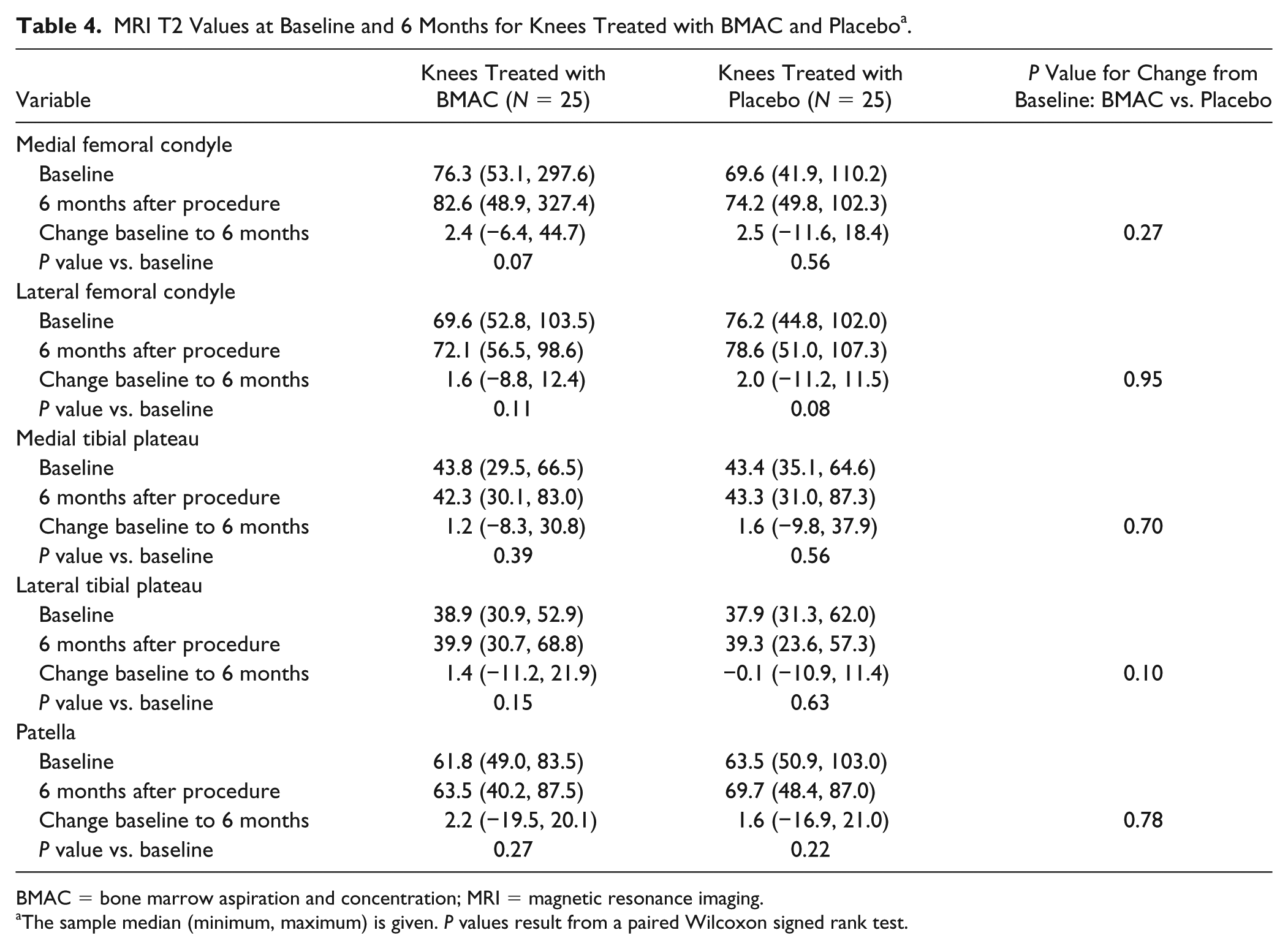
BMAC = bone marrow aspiration and concentration; MRI = magnetic resonance imaging.
The sample median (minimum, maximum) is given. P values result from a paired Wilcoxon signed rank test.
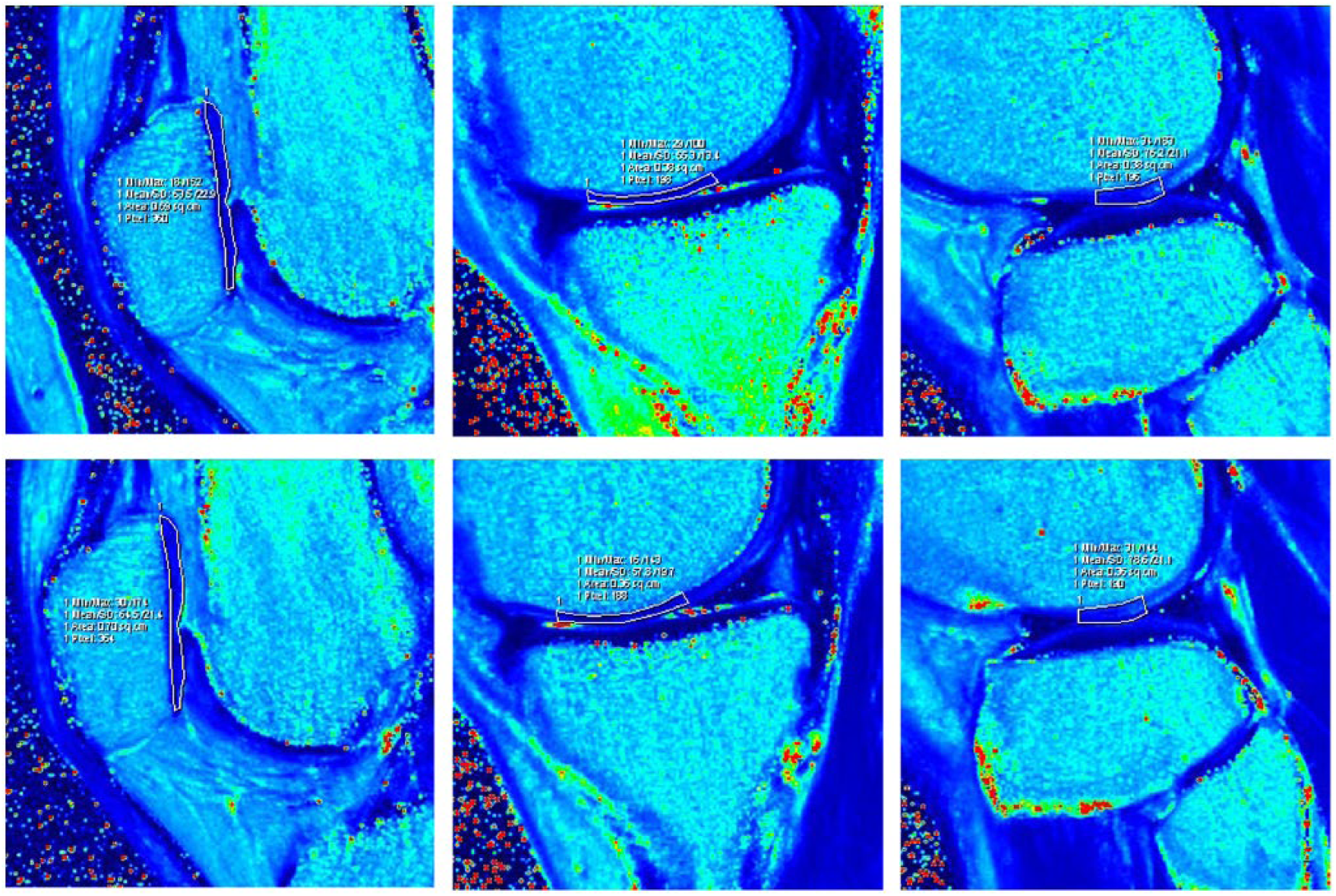
Representative sagittal color-scale T2 maps obtained through the left knee at baseline (top row) and at 6-month follow-up (bottom row) demonstrate how regions of interest were generated by manual segmentation of cartilage in each knee. Selected images above depict (from left to right) measurements obtained at the level of the central patella, central medial femoral condyle, and central lateral femoral condyle (central medial tibial plateau and central lateral tibial plateau were evaluated in the same manner).
Flow cytometry using accepted markers to quantify total number of HSCs and MSCs injected were examined separately for BMAC and placebo-treated knees for correlation with change in pain from baseline to 12 months as measured by the ICOAP questionnaire, VAS pain score, and algometer measures. No significant correlations with total HSC or MSC numbers injected and change in pain scores from baseline to 12 months were observed (all P ⩾ 0.15).
Additionally and importantly, no new serious adverse events were encountered at 12-month follow-up (Supplemental Table 2).
Discussion
Cell-based therapies for osteoarthritis are on the rise. True efficacy remains unclear, and potential therapeutic mechanisms of action are even less understood. Our pilot trial, the first to be randomized, blinded, and placebo-controlled, previously demonstrated the bone marrow aspiration and concentration technique and product safety. In the current report, we expand on the previous findings by reporting 12-month follow-up data and attempt to clarify the regenerative potential of BMAC by reporting MRI T2 quantitative mapping data. We hypothesized that the pain relief and improved activity in both BMAC-treated and saline-treated knees that was previously observed at 6 months would continue out to 12 months. We also hypothesized the BMAC treatment would be unlikely to contribute to MRI detectable cartilage regeneration given such known low MSC quantities. The results presented herein confirm our hypothesis and demonstrate that the interventions performed produced significant pain relief, with overall improvement of activity and quality of life, which remained constant at 12-month follow-up but without significant change in quantitative MRI analysis. Similar responses in BMAC- and placebo-treated knees for all measured outcomes continues to cloud clear mechanism of action. 18
Several observations regarding the outcomes of this intervention therefore merit consideration. At 6-month follow-up, we considered possible therapeutic mechanisms of action, the first of which was a very strong placebo effect.39,43 While the power of the placebo effect should not be underestimated, in this study design, where patients would theoretically not expect both knees to improve (due to their knowledge that one knee would receive a placebo), such explanation for our findings remains uncertain. Possibly, pain relief in the saline-treated knee might not be a true placebo effect as strong treatment effects have previously been demonstrated for intraarticular injection of saline.44-47 This is better considered a meaningful treatment effect using a nonharmful injected substance, in this case, a 15-mL saline injection of the knee joint similar to that presented in recent systematic review and meta-analyses.46,47 However, in each of those reviews, only 2 included studies utilizing saline controls followed subjects to a 12-month period. Much like the previously mentioned true placebo effect, such saline-induced, meaningful therapeutic responses have rarely been documented to have such a dramatic, sustained relief as the 12 months of continuous relief we demonstrate here.
In spite of this, it remains difficult to associate the therapeutic elements of BMAC with the improvements seen in both the BMAC- and saline-treated knees given the data presented here. To do so, the treatment effect would likely need to include a systemic mechanism. In this scenario, though the major impact would be expected at the site of delivery, the therapeutic active elements in BMAC (MSCs and other biologically active factors) known to demonstrate paracrine, anti-inflammatory, and immunomodulatory mediators of pain relief in an arthritic joint might produce a similar systemic therapeutic response outside the joint.12-14,48 The stochastic “homing” capability of MSCs to areas of injury has only been described in models where MSCs were injected intravascularly and, thus, the mechanism cannot be extrapolated.43,49-51 However, there is growing evidence to support MSC and growth factors leaving the injection site and entering the circulation. Systemic spikes in the circulation after musculoskeletal injections have been demonstrated for both platelet rich plasma–related growth factors in human trials and also immunofluorescent radiolabeled MSCs in animal models,51-53 leaving open the prospect that circulating systemic elements from the BMAC could exert a therapeutic benefit on either knee. Nonetheless, significant more work remains before definitively associating the BMAC treatment with such mechanisms of pain relief, and for now, no such conclusions can be drawn.
That we saw no evidence of cartilage regrowth at 6-month follow-up MRI or Kellgren-Lawrence classification change on 12-month follow-up radiography suggests it remains unsuitable to call the BMAC procedure regenerative in nature. It should be pointed out that care must be taken when interpreting T2 values in evaluation of articular cartilage damage as its role continues to be elucidated. Quantitative T2 mapping is subject to a number of variables that may affect its ability to accurately detect early cartilage degeneration. T2 values of articular cartilage are dependent on intrinsic changes in water content and collagen structure, orientation, and organization.54-56 T2 values have been shown to vary with patient age, alterations in articular cartilage loading, and depth of cartilage from the articular surface.54,55,57,58 Furthermore, T2 values are influenced by inherent technical limitations of MRI, such as partial volume averaging and chemical shift artifact.55,59-61 A limitation of this study is T2 values were not directly correlated with physical changes in articular cartilage as arthroscopic confirmation was not performed. Reported cases of surgically confirmed cartilage lesions that do not show increased T2 relaxation time indicate T2 mapping sequences should not be used in isolation to evaluate articular cartilage in clinical practice or the research setting. 62 For these reasons, it is possible early increased changes of either cartilage degeneration or repair in this study may have escaped detection with quantitative T2 mapping at 6 months.
An additional area of consideration regarding T2 mapping and the therapeutic potential of BMAC is the expected and previously demonstrated normal progression of knee arthritis, shown to be anywhere from 4% to 14% per year. 63 Despite the challenges mentioned above, there is some evidence in several prospective trials that utilizing T2 mapping in assessment of articular cartilage can predict progression of knee osteoarthritis, especially early on in the disease process.38,61,64,65 Utilizing similar cartilage mapping techniques, we saw no evidence of joint deterioration in our patients, albeit at an earlier time point, 6 months.
Several limitations of our study are important to acknowledge. First, it is important to point out that due to the relatively small sample size of our pilot study, power to detect differences is limited, and therefore the possibility of a type II error (i.e., a false-negative finding) is important to consider. One cannot conclude that no true difference exists simply based on the lack of a statistically significant P value in our study, and our findings should be interpreted with this in mind. Second, due to the small sample size we were unable to perform any subgroup analysis, such as comparisons between BMAC-treated and placebo-treated knees separately according to Kellgren-Lawrence grade at baseline. Finally, we did not collect data regarding validated activity scales or other functional scores; patients were only asked to what degree their knee pain limits their activity level. However, this limitation is tempered to a degree by the fact that ICOAP scores (which we did collect data on) have been shown to be correlated with KOOS (Knee Injury and Osteoarthritis Outcome Score) and WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) patient-reported outcomes measures, and therefore it is relatively likely that trends for these other scores would mirror those that we observed for ICOAP.
Future studies must include larger populations and compare BMAC treatment in unilateral knee joint disease to placebo control in order to eliminate the possibility of a systemic effect. Investigations like those using protocols for platelet-rich plasma, which has emerged as a viable treatment option for knee arthritis pain, have clarified optimal dosing of cellular types and counts, frequency of treatment, and delivery mechanisms. 66 The hope is that human cell therapy will ultimately lead to regenerative mechanisms for osteoarthritis and other orthopedic conditions. 67 Continued investigation into next-generation therapies like BMAC are necessary, given the still unmet needs of patients seeking conservative, nonsurgical treatment options for osteoarthritis.
Conclusion
We have previously shown BMAC has the ability to safely and efficiently concentrate mononuclear progenitor cells containing small numbers of MSCs for intraarticular use. Based on the 12-month outcomes of our study, the first to be randomized, blinded, and saline-controlled, the BMAC technique is, at present and based on findings thus far, only a pain-relieving modality, not necessarily superior to other injection techniques for osteoarthritis. Additionally, in the absence of clear evidence for cartilage regeneration based on early T2 mapping, mechanisms of action to explain this pain relief remain unclear. For now, available autologous, same-day, minimally manipulated human cell–based products such as BMAC remain restorative at best, without true regenerative evidence.
Supplemental Material
CONSORT_Flow_Diagram_BMAC_MRI_Cartilage – Supplemental material for Quantitative T2 MRI Mapping and 12-Month Follow-up in a Randomized, Blinded, Placebo Controlled Trial of Bone Marrow Aspiration and Concentration for Osteoarthritis of the Knees
Supplemental material, CONSORT_Flow_Diagram_BMAC_MRI_Cartilage for Quantitative T2 MRI Mapping and 12-Month Follow-up in a Randomized, Blinded, Placebo Controlled Trial of Bone Marrow Aspiration and Concentration for Osteoarthritis of the Knees by Shane A. Shapiro, Jennifer R. Arthurs, Michael G. Heckman, Joseph M. Bestic, Shari E. Kazmerchak, Nancy N. Diehl, Abba C. Zubair and Mary I. O’Connor in CARTILAGE
Supplemental Material
MGH_supplemental_tables_Cartilage – Supplemental material for Quantitative T2 MRI Mapping and 12-Month Follow-up in a Randomized, Blinded, Placebo Controlled Trial of Bone Marrow Aspiration and Concentration for Osteoarthritis of the Knees
Supplemental material, MGH_supplemental_tables_Cartilage for Quantitative T2 MRI Mapping and 12-Month Follow-up in a Randomized, Blinded, Placebo Controlled Trial of Bone Marrow Aspiration and Concentration for Osteoarthritis of the Knees by Shane A. Shapiro, Jennifer R. Arthurs, Michael G. Heckman, Joseph M. Bestic, Shari E. Kazmerchak, Nancy N. Diehl, Abba C. Zubair and Mary I. O’Connor in CARTILAGE
Footnotes
Author Contributions
Shapiro: Concept and design, experiment, collection of data, analysis and interpretation of data, drafting of the article, generation of figures, collection of images, critical revision of the article for importation intellectual content, and final approval of the article.
Arthurs: Experiment, drafting of article, critical revision of the article for important intellectual content, and final approval of the article.
Heckman: Collection of data, analysis and interpretation of data, drafting the article, generation of figures, collection of images, critical revision of the article for important intellectual content, and final approval of the article.
Bestic: Analysis and interpretation of data, drafting of the article, critical revision of the article for important intellectual content, and final approval of the article.
Kazmerchak: Concept and design, experiment, collection of data, drafting of the article, and final approval of the article.
Diehl: Collection of data, analysis and interpretation of data, drafting the article, generation of figures, collection of images, critical revision of the article for important intellectual content, and final approval of the article.
Zubair: Concept and design, analysis and interpretation of data, drafting of the article, and final approval of the article.
O’Connor: Concept and design, drafting of the article, critical revision of the article for important intellectual content, and final approval of the article.
Acknowledgments and Funding
Mark Wentworth for regulatory support and compliance, Alison Dowdell for manuscript assistance, and Cheryl Desmond for laboratory support. Funding provided by Mayo Clinic Center for Regenerative Medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from Mayo Clinic Institutional Review Board.
Informed Consent
Written informed consent was obtained from all subjects before the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
