Abstract
Background:
Focal cartilage defects are often debilitating, possess limited potential for regeneration, are associated with increased risk of osteoarthritis, and are predictive for total knee arthroplasty. Cartilage repair studies typically focus on the outcome in younger patients, but a high proportion of treated patients are 40 to 60 years of age (ie, middle-aged). The reality of current clinical practice is that the ideal patient for cartilage repair is not the typical patient. Specific attention to cartilage repair outcomes in middle-aged patients is warranted.
Purpose:
To systematically review available literature on knee cartilage repair in middle-aged patients and include studies comparing results across different age groups.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
A systematic search was performed in EMBASE, MEDLINE, and the Cochrane Library database. Articles were screened for relevance and appraised for quality.
Results:
A total of 21 articles (mean Coleman Methodology Score, 64 points) were included. Two out of 3 bone marrow stimulation (BMS) studies, including 1 using the microfracture technique, revealed inferior clinical outcomes in middle-aged patients in comparison with younger patients. Nine cell-based studies were included showing inconsistent comparisons of results across age groups for autologous chondrocyte implantation (ACI). Bone marrow aspirate concentrate showed age-independent results at up to 8 years of follow-up. A negative effect of middle age was reported in 1 study for both ACI and BMS. Four out of 5 studies on bone-based resurfacing therapies (allografting and focal knee resurfacing implants [FKRIs]) showed age-independent results up to 5 years. One study in only middle-aged patients reported better clinical outcomes for FKRIs when compared with biological repairs.
Conclusion:
Included studies were heterogeneous and had low methodological quality. BMS in middle-aged patients seems to only result in short-term improvements. More research is warranted to elucidate the ameliorating effects of cell-based therapies on the aging joint homeostasis. Bone-based therapies seem to be relatively insensitive to aging and may potentially result in effective joint preservation. Age subanalyses in cohort studies, randomized clinical trials, and international registries should generate more evidence for the large but underrepresented (in terms of cartilage repair) middle-aged population in the literature.
Focal cartilage defects in the knee can cause considerable pain and disability 53 and can impair quality of life to the same extent as osteoarthritis (OA) in patients scheduled for total knee arthroplasty (TKA). 37 Articular cartilage possesses limited repair potential, and therefore cartilage defects frequently progress toward OA. 53,82 Cartilage defects have thus been recognized as a major predictive factor for TKA in patients >45 years old. 22
Currently applied cartilage repair therapies include palliative techniques (ie, chondroplasty), a wide variety of regenerative techniques with differing complexities, and various bone-based cartilage resurfacing techniques. Regenerative treatments include bone marrow stimulation (BMS) techniques such as abrasion arthroplasty (AA), microfracture (MF), 43,58,62,74 and autologous matrix-induced chondrogenesis (AMIC). 53 Cell-based techniques include autologous chondrocyte implantation (ACI); ACI with periosteal flap (ACI-p), collagen flap (ACI-c), or matrix assistance (ACI-m) 8,23,24,44,67 ; and bone marrow aspirate concentrate (BMAC). 29,30,66 Biological cartilage resurfacing via osteochondral autograft transplantation 19 and osteochondral allograft transplantation (OCA) 4,8 as well as metallic and biosynthetic cartilage resurfacing via focal knee resurfacing implants (FKRIs) 38,55,64,69,79 are considered bone-based techniques, as they rely on osseointegration rather than chondrogenesis. 57 Together, these interventions constitute the spectrum of surgical options currently available for cartilage repair.
Large databases have shown that 52% to 60% of cartilage surgeries are performed in patients between 40 and 60 years old. 15,34,58,62 Although there is no official definition, 40 to 60 years is generally referred to as middle age. 10,86 The indication for each cartilage treatment in published guidelines is typically dictated by the size or location of the defect. Patient age is not typically included in these treatment algorithms. 2,7,11,32,53,83 Available randomized controlled studies evaluating the efficacy of cartilage repair typically include an upper age limit as an inclusion criterion. 20 The upper age limit is often set at or around 40 years (ie, the ideal patient). Therefore, the most commonly treated patient is the least represented in the literature and subsequent guidelines.
The ultimate goal of cartilage repair in middle age is joint preservation by postponing or eliminating the need for unicompartmental knee arthroplasty (UKA) or TKA. In our opinion, this could be attained by improving pain and functional performance sufficiently while preserving the native anatomy, at least the bone stock, for potential joint replacement later in life.
When cartilage repair fails, arthroplasty is considered as the last resort. Orthopaedic surgeons perceive a treatment gap for the management of middle-aged patients who have cartilage defects owing to the lack of conclusive evidence identifying a superior cartilage treatment. 6,49,61,64,69 The application of treatments in practice, particularly in the middle-aged patient population, consequently varies greatly among orthopaedic surgeons. 18,73 At the same time, the number of arthroplasty procedures in middle-aged patients has shown an undesirably large increase in the past decade. 50 There is a high risk of revision surgery later in life when TKA is chosen at a relatively young age. 5 Men receiving a knee replacement in their early 50s have a 35% lifetime risk of revision. 5 It is well known that TKA revisions result in inferior outcomes and are associated with high costs. 40
The objective of this study was to systematically review available literature on cartilage repair treatment outcomes in middle-aged and middle-aged versus younger patients. We hypothesized that all cartilage repair techniques performed in middle-aged patients would lead to inferior clinical and imaging outcomes and higher failure rates when compared with those in younger patients.
Methods
Search Strategy and Study Selection
A systematic literature search was performed using MEDLINE, EMBASE, and Cochrane Library databases according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. Included were studies with evidence levels 1 to 4 (based on the Oxford Centre for Evidence-Based Medicine) on cartilage repair in middle-aged patients. The search was conducted with support of a university librarian in December 2020 using the following terms (including its free terms using title and abstract, Medical Subject Headings terms, abbreviations, and single and plural forms): ((“chondral” or “osteochondral” or “cartilage”) and (“wounds and injuries” or “damage” or “defect” or “lesion” or “injury” or “trauma” or “wound”) and (“knee”)) and (“cartilage repair” or “regeneration” or “healing” or “microfracture” or “autologous chondrocyte implantation” or “autologous chondrocyte transplantation” or “matrix assisted chondrocyte implantation” or “autologous matrix-induced chondrogenesis” or “chondrocyte/surgery” or “tissue engineering” or “tissue scaffolds” or “mosaicplasty” or “osteochondral autograft” or “osteochondral allograft” or “resurfacing” or “resurface” or “bone-implant interface” or “implant” or “bone-anchored prosthesis” or “prosthesis and implants” or “prosthesis” or “mosaicplasty” or ((“focal” or “local”) and “arthroplasty”)). This review was registered in the international database PROSPERO (registry No. CRD42020179932).
Results from the databases were uploaded to Endnote X7 (Clarivate Analytics). The combined database was scanned for duplicates and then uploaded to the online systematic review software Rayyan (Qatar Computing Research Institute). 68 This allowed 2 observers (R.M.J. and P.P.W.v.H.) to independently screen titles and abstracts. Article screening and selection was performed using the following inclusion criteria: studies evaluating cartilage repair treatment in patients aged between 35 and 65 years; adding 5 years above and below typical middle age 10,86 extended the search results. Studies with an age comparison between younger- and middle-aged patients with a clear age cutoff as well as with a minimum follow-up of 2 years, with a minimum of 25 patients, involving humans, and written in the English language were also included. Exclusion criteria included comparative studies not including middle-aged patients in the “older” group (upper age limit ≤50 years), mean patient age <40 years for noncomparative studies, animal studies, studies that stated osteoarthritic change defined as grade ≥1 on the Kellgren-Lawrence scale, joints other than the knee, and patellofemoral or tibial defects only. Upon reading the full-text articles assessed for eligibility, we performed snowball sampling to allow the inclusion of relevant referred studies. A consensus meeting was held to sort out potential disagreements.
Data Extraction
Two observers (R.M.J. and P.P.W.v.H.) systematically extracted study data, which included patient characteristics, study design, inclusion and exclusion criteria, study groups, previous and concomitant surgeries, defect characteristics, follow-up time, and study outcomes.
Assessment of Quality and Bias
The Coleman Methodology Score (CMS) was used to assess the quality and bias of each study. 12 This score is used to evaluate, among others, the criteria for participant selection, the study design, surgical and rehabilitation protocols, and assessments of outcomes, providing each study with a score from 0 to 100. Scores are interpreted as excellent (>85), good (70 to 84), fair (55 to 69), or poor (<55).
Results
Study Characteristics
The search strategy yielded 21 studies for inclusion as shown in the PRISMA flow diagram in Figure 1. There were 3 studies on BMS, 28,43,74 9 studies on cell-based techniques, 8,23,24,29 –31,44,65,67 5 studies on bone-based resurfacing techniques, 4,25,38,48,55 and 4 studies comparing different treatments. 16,64,66,69 Table 1 lists the characteristics of all included studies. The level of evidence was level 4 in 18 studies, § level 3 in 2 studies, 16,69 and level 2 in 1 study. 67 Various age cutoff values were used to make a comparison between the middle-aged and the younger patient population, but 40 years was the most commonly used cutoff. The average CMS was 64, with 3 studies 16,28,29 scored as poor, 13 ∥ scored as fair, and 5 4,31,64 -66 scored as good. Patient characteristics, study setup and outcomes, and a summary of results are provided in the evidence table for each study (Supplemental Table S1).
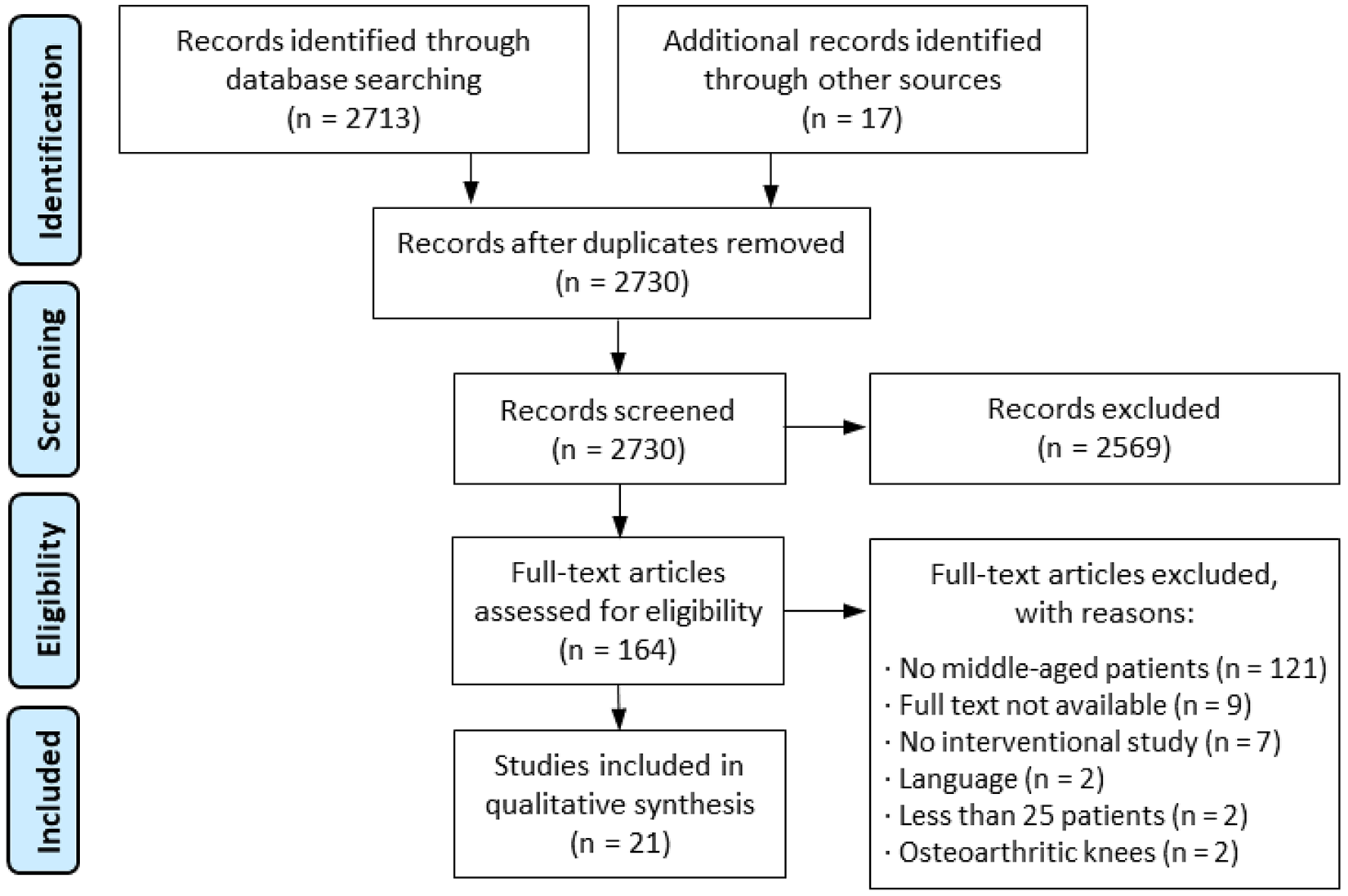
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flowchart showing the yield of the initial search and exclusion of studies leading to the 21 included studies.
Overview of Included Studies a
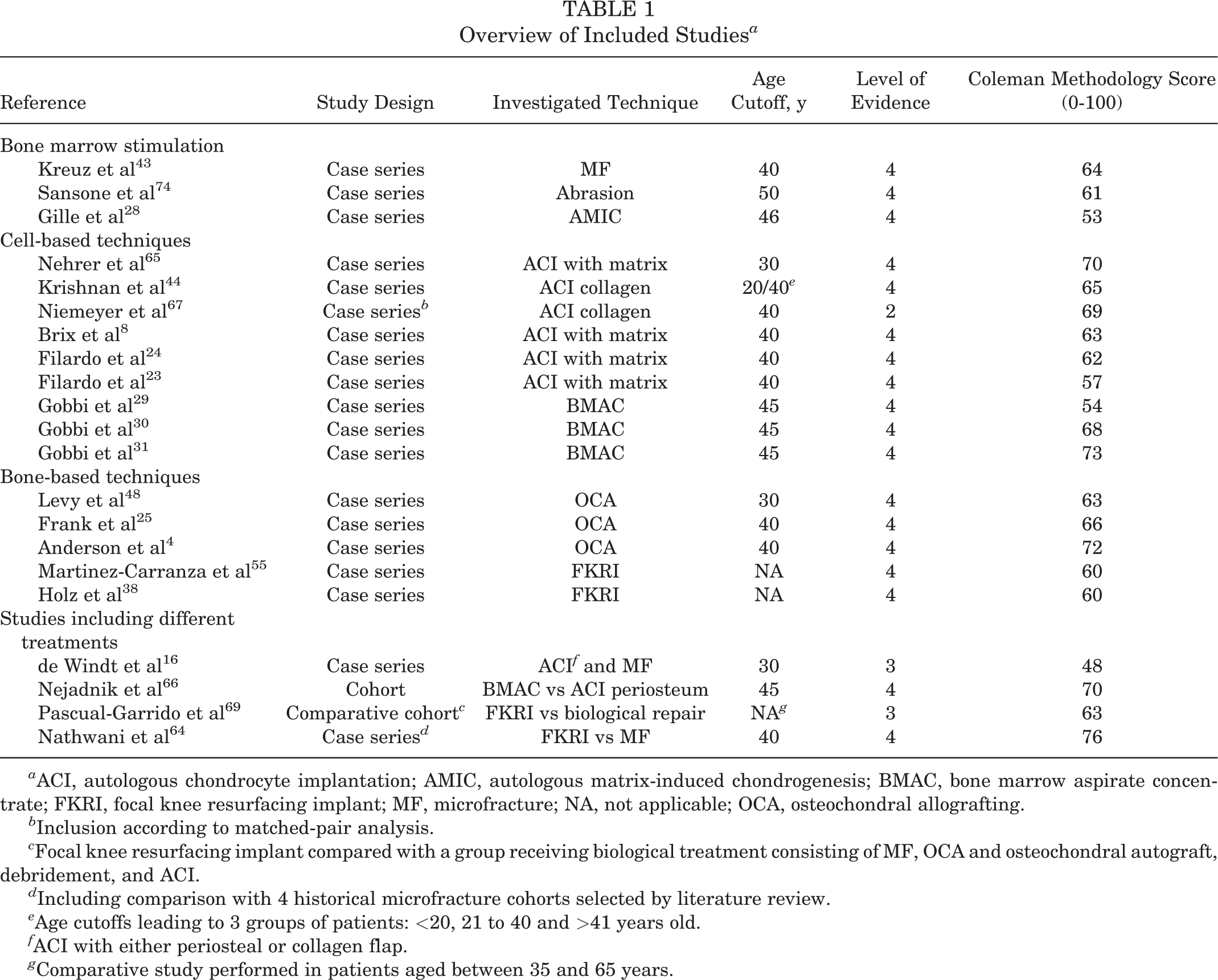
a ACI, autologous chondrocyte implantation; AMIC, autologous matrix-induced chondrogenesis; BMAC, bone marrow aspirate concentrate; FKRI, focal knee resurfacing implant; MF, microfracture; NA, not applicable; OCA, osteochondral allografting.
b Inclusion according to matched-pair analysis.
c Focal knee resurfacing implant compared with a group receiving biological treatment consisting of MF, OCA and osteochondral autograft, debridement, and ACI.
d Including comparison with 4 historical microfracture cohorts selected by literature review.
e Age cutoffs leading to 3 groups of patients: <20, 21 to 40 and >41 years old.
f ACI with either periosteal or collagen flap.
g Comparative study performed in patients aged between 35 and 65 years.
Bone Marrow Stimulation
Kreuz et al 43 showed that MF-treated patients >40 years old experienced significant deterioration in International Cartilage Regeneration & Joint Preservation Society (ICRS) scores between 18 and 36 months postoperatively, whereas patients <40 years did not. After 36 months, patients ≤40 years had significantly better defect filling on magnetic resonance imaging (MRI) scans and significantly better outcomes on the Modified Cincinnati Score (MCS) in comparison with patients >40 years. 43 Sansone et al 74 reported an 84% “high” (70-100 points) Knee Society Score in AA-treated patients <50 years compared with 53.6% in patients aged ≥50 years. Patients >50 years had significantly more defects >4 cm2. There were 12 conversions to arthroplasty in patients aged ≥50 years versus 1 patient in the <50 years subgroup. Finally, Gille et al 28 compared 3 age groups (17-32, 33-46, and 47-65 years) after AMIC cartilage repair and found no significant differences in outcome across all age groups.
Cell-Based Techniques
Nehrer et al 65 reported significant improvements on the Lysholm and International Knee Documentation Committee (IKDC) scores after ACI-m only when patients were aged <30 years; patients ≥30 years showed no significant improvement. Krishnan et al 44 reported significantly higher MCS scores in younger patients when comparing patients aged <20 years, 21 to 40 years, and ≥41 years. Patients aged <20 years had good/excellent scores in 85.7% of cases, and only 64.2% and 55.9% of patients attained good/excellent scores in the 21 to 40 years and ≥41 years age categories, respectively.
Niemeyer et al 67 reported no significant differences after ACI-c in patients aged ≥40 years compared with patients aged <40 years using the IKDC, Lysholm, MCS, and Tegner activity score (TAS). Brix et al 8 reported significantly better Lysholm score improvement after ACI-m in patients aged <40 years versus ≥40 years (25.8 ± 20.3 vs 5.6 ± 13.2, respectively). Filardo et al 24 reported that all of the patients ≤40 years reached the intended 80% improvement on the IKDC score while none of those aged >40 years reached this level. In a later ACI-m study, Filardo et al 23 showed that IKDC scores were significantly lower for patients >40 years compared with patients <40 years. When scores were standardized to age-normative healthy IKDC values, however, there were no significant differences.
Gobbi et al 29 performed a multivariate analysis comparing outcomes from patients <45 years with those ≥45 years and reported no significant differences. Gobbi et al 30 performed a study using a hyaluronan-based scaffold soaked in BMAC. There were no significant differences in visual analog scale (VAS), IKDC, Knee injury and Osteoarthritis Outcome Score (KOOS), TAS, and MRI between patients aged 45 to 60 years and patients aged 20 to 44 years at a mean final follow-up of 41.3 months. The VAS improvement, TAS, and KOOS Sport and Recreation score at 2 years were significantly higher in the older group compared with the younger group. The authors hypothesized that the lower physical demand in older patients was responsible for this finding. Defect filling on MRI scans was superior in the older group compared with the younger group; however, statistics were not provided, and not all patients were evaluated via MRI. Then, Gobbi et al 31 found no significant differences after BMAC in IKDC, KOOS, TAS, and VAS pain score between patients ≤45 years and >45 years after BMAC surgery.
Bone-Based Techniques
Levy et al 48 performed a logistic regression on OCA-treated patients using failure as the outcome. Failure was defined as graft revision or conversion to an arthroplasty. After a median of 13.5 years, the odds ratio for failure was 3.5 times higher in patients ≥30 years compared with patients <30 years. Frank et al 25 reported no significant differences in IKDC, KOOS, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and 12-Item Short Form Health Survey scores after OCA treatment between patients aged <40 years and ≥40 years. There were no significant differences in time to reoperation, reoperation rate, and failure rate at final follow-up. The older group had significantly better KOOS Symptoms scores compared with the younger group, hypothesized by the authors to be the result of higher expectations in the younger patients.
Anderson et al 4 reported no significant differences in KOOS, IKDC, and 12-Item Veterans RAND Health Survey scores after OCA treatment between patients aged ≤39 years and ≥40 years. Martinez-Carranza et al 55 reported significant improvements in EuroQol Five Dimensions Health Questionnaire, VAS, and KOOS scores in FKRI-treated middle-aged patients 2 years postoperatively. After a mean follow-up of 55 months, 1 patient required conversion to a hemiarthroplasty. Finally, Holz et al 38 performed a multicenter multinational study and reported significant improvements in KOOS and VAS scores in FKRI-treated middle-aged patients. Two out of 75 patients had failed treatment and required implant removal within 2 years. A subanalysis found that previous cartilage repair had no negative effect on the outcomes.
Studies Including Different Treatments
de Windt et al 16 performed a single-variant regression analysis on ACI-p/ACI-c or MF-treated patients and showed that those <30 years had significantly higher KOOS improvements compared with those ≥30 years in both groups. Nejadnik et al 66 compared ACI-p with BMAC. A subanalysis with 45 years as a cutoff was performed in both groups using the IKDC and TAS. There was no significant difference in IKDC score between patients aged <45 and ≥45 years in the BMAC group. The TAS was significantly higher in patients <45 years versus ≥45 years in the ACI group, but there was no significant difference between age groups in the BMAC group.
Pascual-Garrido et al 69 investigated the use of the first-generation metal FKRI and compared it with the use of biological treatments. Only middle-aged patients were included in both groups. The biologically treated group consisted of 15 MF, 10 OCA, 2 osteochondral autograft transplantation, 2 debridement, and 1 ACI-treated patient. Clinical success was defined as ≥20% improvement on the WOMAC score and was significantly higher in the FKRI-treated patients compared with the biologically treated patients: 75% versus 53%. The WOMAC pain and satisfaction domains were significantly higher in the FKRI group compared with the biologically treated group.
Nathwani et al 64 compared a bilayered polymer and metal FKRI cohort with 4 historical MF cohorts in middle-aged patients. FKRI-treated patients aged ≤40 years were also compared with those aged >40 years. The KOOS Quality of Life subscore was significantly higher for the FKRI group compared with all 4 referenced MF cohorts. The KOOS Sport and Recreation and Activities of Daily Living subscale scores were significantly higher for the FKRI group compared with 2 MF cohorts. No significant differences in KOOS, VAS, and the 36-Item Short Form Health Survey were observed when comparing patients aged ≤40 years versus >40 years.
Discussion
The objective of this study was to systematically review literature on cartilage repair treatment outcomes in middle-aged versus younger patients. Included studies had low quality and heterogeneous methodology. The null hypothesis that all cartilage repair techniques in middle-aged patients would lead to inferior clinical outcomes and higher failure rates when compared with those performed in younger patients is rejected. Some cell-based, and most bone-based treatments, were equally effective in middle-aged patients compared with younger patients. The null hypothesis was confirmed for the conventional BMS techniques MF and AA, which indicates that the effectiveness of these techniques is age dependent.
The BMS studies have shown that applying MF or AA in middle-aged patients results in inferior defect filling, fast deterioration of repair tissue, and higher conversion to arthroplasty rates in comparison with application in younger patients. 43,74 Even in younger patients, BMS often leads to biomechanically inferior repair tissue. 51 Advancing age deteriorates the joint homeostasis and cellular potency, resulting in impaired chondrogenesis and hence lower quality of the repair tissue. 75,81,82 It has been hypothesized that the treated defect is unable to withstand the detrimental pothole effect. 45 In addition, the subchondral bone (SB) starts thickening and forms cysts due to aging, OA, or chronic cartilage defects. 26,72 The standard MF awl causes widespread microarchitecture disturbances in the SB, leading to further sclerosis 27 and cyst formation after MF, 76 with good correlation between the severity of SB changes and poor clinical outcomes. 78 SB alterations may also explain how BMS jeopardizes consecutive regenerative treatments. 48,60,71,84 Failure rates of subsequent cartilage repair treatments after failed BMS are up to 3 times higher. 60
Despite these known disadvantages, the advantages of BMS are its technical ease, low costs, high availability, and being a single-stage procedure. 1 BMS is the most commonly applied regenerative technique. 15 The widespread use of MF in middle-aged patients has been reported. 34,58,62 Salzmann et al 73 surveyed German-speaking orthopaedic surgeons and found that >30% of the respondents did not see age as a limiting factor for MF and only 11.6% upheld 40 years as an upper age limit for MF. Similar numbers were seen in a more recent Turkish survey. 18 The AMIC study of Gille et al 28 showed that supplementing BMS with an overlying matrix can provide good clinical outcomes in middle-aged patients. This is consistent with most preclinical BMS studies, which show the mitigating effects of biological stimuli on the detrimental SB disturbances after MF. 76
Cell-based techniques provide biological stimuli without violating the SB. The ACI-c study by Niemeyer et al 67 and all 3 BMAC studies showed age-independent results. 29 –31 However, 5 other ACI studies with longer follow-up periods showed a negative effect of advancing age on clinical outcomes. 8,23,24,44,65 ACI relies on the injection of laboratory-cultured chondrocytes. Chondrocyte yield and potency in middle-aged patients is often diminished compared with that in younger individuals. 82 This may lead to rapid deterioration of the repair tissue. Good clinical outcomes in middle-aged patients were reported for BMAC, 29 –31,66 with a follow-up of up to 8 years. 31 Results were superior to ACI-p at 2 years postoperatively. 66 BMAC contains high concentrations of platelets, which include a significant number of cytokines, chemokines, and growth factors that elicit a trophic effect. 13 Several studies have shown that the iliac crest yields the highest concentrations of bone marrow–derived mesenchymal stem cells. 13 BMAC may thus possess a more potent composition than that derived during ACI or MF, as shown in a review of multiple animal studies. 13 Substantiated by satisfactory results up to 8 years, postponing arthroplasty with BMAC seems to be possible. In addition to these advantages, BMAC requires only 1 surgery instead of 2 when compared with ACI.
Bone-based techniques replace the complete osteochondral unit, including the affected SB in a single surgery, and provide a mechanically resilient articulating layer. Good clinical outcomes in middle-aged patients with consistent treatment effects across age groups have been shown. 4,25,38,55,64,69 Only Levy et al 48 showed that OCA-treated patients were subject to age-related differences. In contrast to the more recent 2 OCA studies that both employed the dowel technique, 4,25 82% of the participants in the study by Levy et al 48 were treated via the shell technique. The shell technique requires large SB surface contact and additional internal fixation materials. More surgical trauma is consequently induced, resulting in higher failure rates. 51 Bone-based techniques do not require chondrogenesis; an animal study showed that osteochondral allografts are capable of adequate osseointegration, but a persistent gap between host and donor cartilage typically remains. 57 Osseointegration relies on the same physiological process as found in fracture healing, 54,77 which has been shown to be sufficient into old age. 39 Replacement of the SB may also be an important contributor to pain relief. It is becoming increasingly accepted that the innervated SB is responsible for pain perception in cartilage damage. 47 Previous cartilage repair has no effect on FKRI outcomes. 38 Rehabilitation after bone-based techniques is generally short. 39 A recent OCA cost-effective analysis concluded that OCA is highly cost-effective over a 30-year period and able to eliminate or postpone the need for the first TKA. 61 The varying availability of allografts remains an ongoing drawback, particularly limiting its use in Europe. 56 FKRIs are readily available and do not have availability issues. 64,69 FKRIs in 2 included studies were compared with biological-based treatments in middle-aged patients: 1 study compared FKRI outcomes with a cohort of different biological treatments consisting mainly of MF and OCA, 69 whereas the other study compared the FKRI outcomes with 4 historical MF cohorts. 64 Both studies showed superiority of the FKRIs in comparison with their control group. 64,69 No differences in outcome between patients ≤40 and >40 years for the FKRIs were observed. 64 Long-term follow-up of FKRIs is scarce, and available long-term evidence from the first-generation metal FKRI raised concerns because of high failure rates based on OA progression. 46 Novel metal FKRIs with patient-specific surface geometries have resulted in very low failure rates up to 55 months. 38,55 Cartilage-mimicking polymers, such as in the study by Nathwani et al, 64 pose great potential for prevention of OA progression when using FKRIs. 59,64 Failed FKRIs that are explanted however result in SB voids, and there are concerns that this may necessitate more extensive revision arthroplasty implants. 33 More FKRI research is warranted.
Limitations
Cartilage repair literature is known for its low quality. 36 The average CMS of articles included in this study was 64 points, and 86% of the studies were of evidence level 4. The methodologic heterogeneity hampered data assimilation for a meta-analysis.
Concomitant or previous injuries and concomitant procedures were not always or only poorly described but could have important implications. For instance, unloading the repaired cartilage compartment via a high tibial osteotomy could have potential synergistic effects on cartilage defect repair. These underreported but potentially influencing variables make comparison of cartilage defect interventions difficult.
Defect size varied among the included studies as a consequence of the size-based treatment algorithms. Sansone et al 74 showed significantly larger defects in patients >40 years, 3 studies did not show any significant difference, 4,23,67 and the remaining studies did not provide statistics on defect size differences. It remains unclear whether older patients have larger defects, but if so, this should further discourage MF usage in older patients given the consensus that MF leads to inferior outcomes in larger defects. 21
Proven OA was out of the scope of this review to prevent clouding of results. However, long-present defects in middle-aged patients are perhaps already an expression of early OA (EOA). 52 OA leads to an impaired joint homeostasis that jeopardizes repair. 52 Kim et al 41 confirmed that MF could not prevent progression of radiological EOA after MF cartilage repair in middle-aged patients. Deterioration of clinical results occurred after 1 year. 41 de Windt et al 17 conducted a review on cartilage defect repair in patients with EOA and concluded that ACI was able to postpone the need for arthroplasty in the short and midterm. Wang et al 85 showed OCA graft survival rates of 75% after 5 years in middle-aged patients with EOA.
FKRIs have previously been denoted as partial arthroplasties, 63 indicating that their classification as a cartilage repair technique is still under debate. The indications for FKRIs are limited to the treatment of focal cartilage lesions, with radiographic OA or severe lesions (ICRS grade 3 to 4) on the opposing cartilage surface as absolute contraindication. 38,64 UKAs are primarily indicated for diffuse medial or lateral compartment OA, thus both femoral and tibial OA. Owing to this important difference, we consider unipolar FKRIs to be a metallic or biosynthetic cartilage resurfacing technique similar to auto- and allografts, which we consider to be biological cartilage resurfacing.
We must note that UKAs may also be a viable treatment option for younger and active patients with cartilage damage with excellent functional outcomes up to 11 years. 9,35 However, subsequent UKA revision procedures have drawbacks that warrant reservations in their use for the indication of focal cartilage defects. Upon OA disease progression, both biological and metallic or biosynthetic (eg, FKRIs) resurfacing implants can be converted to UKA/TKA with relative ease, 38,80 whereas one-third of UKA revisions require additional surgical measures to compensate for loss of bone stock. 14 UKA revisions are associated with high failure rates 14,70 and exhibit poorer clinical outcomes than those of primary procedures, both similar to TKA revisions. 70 To the best of the authors’ knowledge, such typical arthroplasty-related drawbacks have not yet been reported for revision of cartilage resurfacing (eg, FKRI conversions to arthroplasty). For these reasons, we believe that, in line with allografts, FKRIs should be considered as a joint-preserving option ahead of UKA for each individual middle-aged patient with a focal cartilage defect. UKAs are considered a joint replacement technique disrupting the native knee anatomy and thus are outside of the scope of this review.
The natural decline in knee performance with advancing age may be a confounding factor. The largest natural decrease in normative IKDC data has been reported to occur between the ages of 51 and 65 years, followed by the decline between the ages of 35 and 50 years. 3 Hence, long-term follow-up studies in patients >40 years overlap with the period of the largest natural decline in knee performance, as was confirmed by Filardo et al. 23 Without age-normative standardization, no significant differences between middle-aged and younger patients were observed for ACI, 67 OCA, 25 BMAC, 30,31,66 and FKRI, 38,55,64,69 which highlights the age independency of these treatments.
Interpreting the 3 studies of Gobbi et al 29 –31 as separate studies might be biasing the results, as it is conceivable that the patients included in these studies overlap.
The commonly used threshold age of 40 years to characterize middle age is arbitrary in the context of cartilage repair. Chronological age and biological age are not the same, particularly for a knee with a history of previous injury and intervention. Biomarkers that differentiate in joint homeostasis are critically needed. 42 Until then, orthopaedic surgeons should be aware of the findings in this study when treating middle-aged patients.
With these limitations in mind, it becomes apparent that the present study is significantly hampered. More frequent age subanalysis in cohort studies and comparative randomized controlled trials in middle-aged patients are warranted. Correction to age-normative data seems imperative when analyzing patients on the verge of natural decline. Furthermore, international registries collecting all patient, defect, and surgical characteristics combined with biomarkers could aid in bias-free identification of successful treatments in middle-aged patients and the prognostic variables to aid in treatment selection.
Conclusion
There is a paucity of available evidence for cartilage repair in middle-aged patients, and drawing definite conclusions is severely limited by inadequate methodology and reporting in the included studies. Importantly, no prospective randomized study comparing different therapies such as BMS versus cell- or bone-based therapies specifically in middle-aged patients has been performed to date. MF is still the most frequently performed cartilage repair technique in middle-aged patients. The included studies suggest that conventional BMS leads to inferior outcomes in middle-aged patients compared with younger patients. Literature shows various effects of age categorization on clinical outcomes for cell-based techniques, particularly in the long term. Bone-based techniques can be considered to be relatively insensitive to aging. This review has indicated that not all cartilage treatments are affected equally by age and that joint preservation in middle-aged patients is possible. However, further methodically sound research is warranted for all cartilage repair techniques to elucidate their joint preservation potential.
Supplemental Material for this article is available at http://journals.sagepub.com/doi/suppl/10.1177/23259671211031244.
Supplemental Material
Supplemental Material, sj-pdf-1-ojs-10.1177_23259671211031244 - A Systematic Review of Focal Cartilage Defect Treatments in Middle-Aged Versus Younger Patients
Supplemental Material, sj-pdf-1-ojs-10.1177_23259671211031244 for A Systematic Review of Focal Cartilage Defect Treatments in Middle-Aged Versus Younger Patients by Ralph M. Jeuken, Pieter P.W. van Hugten, Alex K. Roth, Ufuk Tan Timur, Tim A.E.J. Boymans, Lodewijk W. van Rhijn, William D. Bugbee and Pieter J. Emans in Orthopaedic Journal of Sports Medicine
Footnotes
Notes
Acknowledgment
This work was performed under the framework of Chemelot InSciTe, a private-public institute for biomaterial research and upscaling.
Final revision submitted March 3, 2021; accepted March 22, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: W.D.B. has received consulting fees from JRF Ortho and Arthrex. P.J.E. has received consulting fees from Episurf and KioMed. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
