Abstract
Objective
Previous studies have shown that intrinsic behavior of subchondral bone marrow stem cells (BMSCs) is influenced by donors and locations. To understand the variability in cartilage repair outcomes following bone marrow stimulation, we tested the hypothesis that in vivo cartilage repair correlates with in vitro biological properties of BMSCs using a rabbit model.
Methods
Full-thickness cartilage defects were created in the trochlea and condyle in one knee of skeletally mature New Zealand White rabbits (n = 8) followed by microdrilling. Three-week repair tissues were analyzed by macroscopic International Cartilage Repair Society (ICRS) scores, O’Driscoll histological scores, and Safranin-O (Saf-O) and type-II collagen (Coll-II) % stain. BMSCs isolated from contralateral knees were assessed for cell yield, surface marker expression, CFU-f, %Saf-O, and %Coll-II in pellet culture followed by correlation analyses with the above cartilage repair responses.
Results
In vivo cartilage repair scores showed strong, positive correlation with cell number, clonogenic, chondrogenic, and matrix production (Coll-II, GAG) potential of in vitro TGF-βIII stimulated BMSC cultures. Trochlear repair showed clear evidence of donor dependency and strong correlation was observed for interdonor variation in repair and the above in vitro properties of trochlear BMSCs. Correlation analyses indicated that donor- and location-dependent variability observed in cartilage repair can be attributed to variation in the properties of BMSCs in underlying subchondral bone.
Conclusion
Variation in cell number, clonogenic, chondrogenic, and matrix production potential of BMSCs correlated with repair response observed in vivo and appear to be responsible for interanimal variability as well as location-dependent repair.
Introduction
Chondral lesions are a commonly occurring knee pathology and are more widespread in medial femoral condyle (MFC) than trochlea. 1 Bone marrow stimulation (BMS) procedures carried out by fracturing or drilling into subchondral bone typically produce a fibrocartilaginous repair. Repair outcomes are affected by defect location suggesting a strong influence of biological characteristics of progenitor cells in the underlying subchondral bone on repair response.2-4 The mechanisms behind these sources of variable cartilage repair still elude us since underlying factors such as bone structure and mechanics, load-bearing condition and role of subchondral bone progenitor cells are largely unknown.
Better repair and increased chondrogenic potential have been observed in rabbit trochlea compared to medial femoral condyle in previously published studies.5,6 BMS cartilage repair in rabbit trochlea was associated with more chondrocytes and larger chondrogenic foci suggesting the greater chondrogenic potential of trochlear progenitor cells compared with condylar progenitors. 5 As evidence of location- and species-dependent cartilage repair, more fibrous repair was observed in ovine trochlea versus condyle with BMS as well as autologous chondrocyte implantation.7,8 In vitro characterization of BMSCs isolated from condyle and trochlea of young and old rabbits showed evidence that trochlear BMSCs were more numerous with higher clonogenic and chondrogenic differentiation potential. 9 Since BMS relies on recruitment of underlying BMSCs for repair of soft and hard tissues, characterization of these progenitors and direct correlation with cartilage repair outcomes may provide important mechanistic insights into location-dependent cartilage repair.
Here, in addition to validating the influence of location on early repair responses in an acute defect rabbit model, we sought to investigate the influence of biological properties of BMSCs isolated from condyles and trochlea on the repair response. The current study was carried out with the hypothesis that the quality of cartilage repair outcome in condyle and trochlea will correlate to the in vitro biological properties of underlying BMSCs at the same 2 sites in the contralateral knee.
Materials and Methods
Study Design and Rabbit Surgical Model
Canadian Council on Animal Care guidelines for the care and use of laboratory animals were observed. The study was approved by the University of Montreal “Comité de déontologie de l’expérimentation sur les animaux” (#13-129). Eight skeletally mature (8-9 months old) female New Zealand White rabbits were used. This number was selected based on several previous studies from our group where a similar number of animals was found to generate statistically significant and relevant differences between groups.5,9 To examine the influence of interanimal variation, a bilateral model was used for the characterization of cartilage repair and in vitro biological properties of BMSCs ( Fig. 1 ). On one knee (n = 8 knees), full-thickness cartilage defects measuring 4 × 4 mm were created on the medial femoral condyle (MFC) and central trochlear groove by complete debridement of noncalcified and calcified cartilage using a flat blade to expose the underlying subchondral bone. Using drilling burrs, 4 subchondral drill holes, 0.9-mm diameter and 6-mm deep, were made in each defect in the trochlea and MFC. 10 Constant cooling irrigation was applied to prevent heat necrosis 10 and remove loose bone debris. After closing in sutured layers, knees were allowed unrestricted motion until sacrifice 3 weeks later. The contralateral knee (n = 8 knees) in each animal was intended for in vitro characterization of BMSCs from condyle and trochlea and received no treatment prior to sacrifice ( Fig. 1 ).
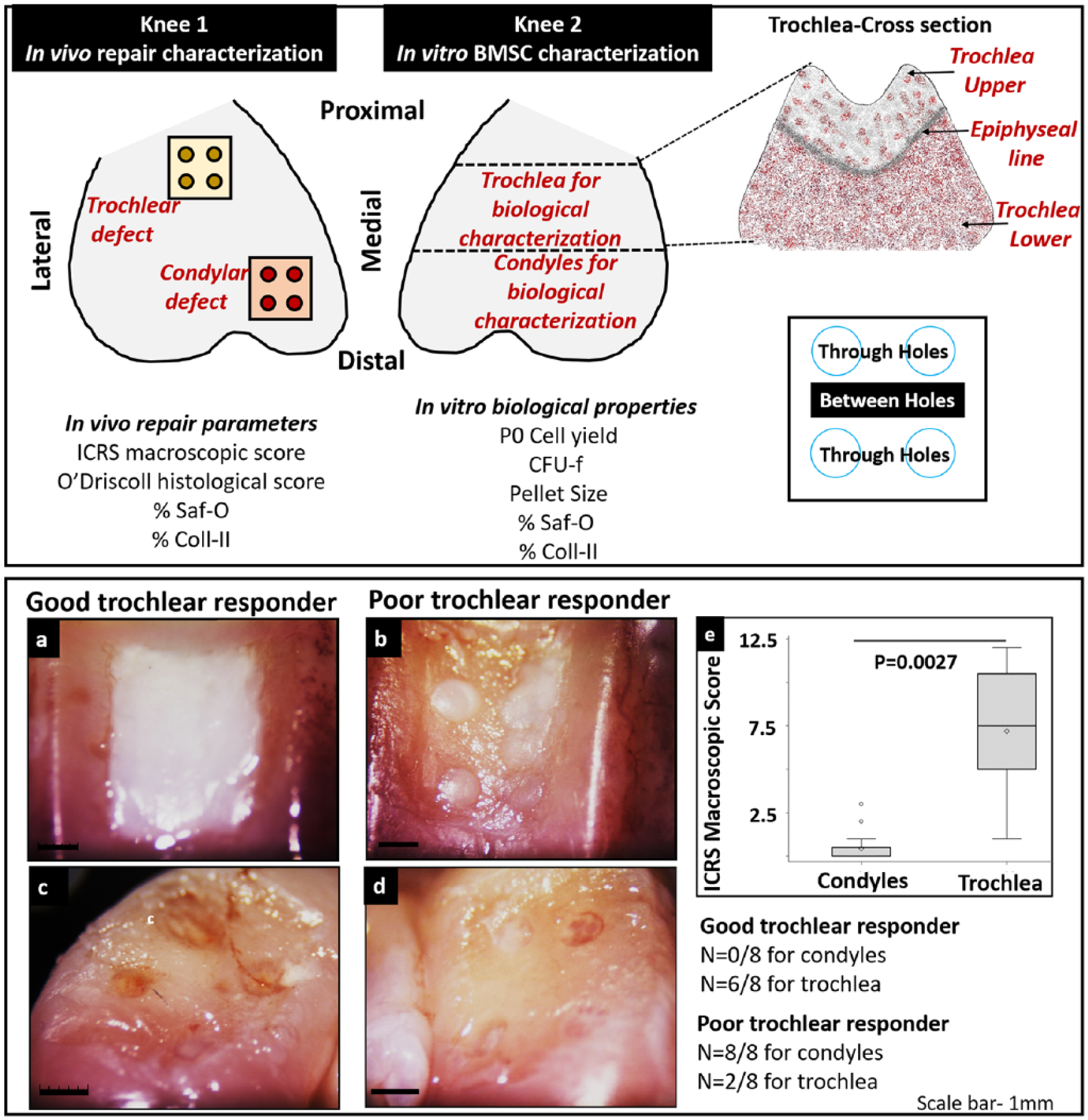
(Top panel) Schematic representing the study design. Condylar and trochlear cartilage repair outcome in one knee from each animal was correlated with in vitro biological properties of bone marrow stem cells (BMSCs) isolated from the condyle, upper trochlea, and lower trochlea from the contralateral knee. Schematic for location for collection of sections for histopathogical analysis shows points for collection of section through (“Through holes”) and in between (“Between holes”) repair holes. (
Characterization of Cartilage Repair
Animals were sacrificed by an overdose of pentobarbital and the treated knee was characterized for cartilage repair. Photographs of harvested joints were scored using the macroscopic International Cartilage Repair Society (ICRS) scoring system for cartilage repair. 11 Scoring was performed by 2 blinded readers (intraclass correlation coefficient [ICC] = 0.95 for overall repair assessment). Decalcified samples were embedded in OCT and sections were obtained from 3 levels—through proximal and distal holes in addition to between holes ( Fig. 1 ). Sections were stained with Safranin O (Saf-O)/Fast Green and scanned using a Nanozoomer RS system (Hamamatsu, Japan). Two blinded observers scored the digital Saf-O-stained sections using a previously published modified O’Driscoll histological scoring method (ICC 0.86 for total O’Driscoll score). 12 The average score from 2 observers was used to determine final macroscopic ICRS and O’Driscoll score. All 3 sections collected from each defect were also used to determine %Saf-O and %Coll-II in the chondral repair tissue using a previously described method of hue-saturation-value thresholding with ImageJ and in-house Matlab routine software. 12
Isolation and In Vitro Characterization of Condylar and Trochlear BMSCs
The contralateral knee was used for isolation of BMSCs as previously described. 9 Briefly, distal femur ends were separated into condyles, trochlea upper, and trochlea lower compartments ( Fig. 1 ) followed by fragmentation into bone chips (approximately 5 mm), which were then digested with collagenase type XI (Sigma) (3 mg/mL, 2 hours) to obtain the population of collagenase-derived cell BMSCs. Digested bone chips were then seeded as explants to generate a second population of explant-derived cell BMSCs. BMSCs were cultured for a maximum of 2 passages in DMEM/F-12 media supplemented with 10% fetal bovine serum and 1X penicillin-streptomycin. The following biological properties were then characterized using previously reported methods. 9 P0 cell yield was used to estimate the population density of BMSCs originally present in condyles and trochlea. Stemness of trochlear and condylar BMSCs was characterized by CFU-f (colony-forming unit fibroblast) assay. Cell surface marker characterization was done for positive-CD44 (Cell Marque, 144M-96) and CD29 (Millipore, MAB1951Z) and negative-CD34 (Cell Marque, 134M-16) and CD45 (Cell Marque, 145M-97) stem cell markers [9]. BMSC pellets (n = 3 for each group) were used for chondrogenic differentiation for 3 weeks in a 15-mL conical tube in a chondrogenic medium composed of high glucose DMEM (Dulbecco’s modified Eagle medium) containing penicillin-streptomycin, ascorbate, dexamethasone, sodium pyruvate, and bovine serum albumin with or without (control pellets) transforming growth factor–βIII (TGF-βIII). 9 Size of pellets was measured to determine the amount of matrix synthesized as an indicator of chondrogenic differentiation. Saf-O/Fast Green staining and collagen-type II immunostaining were done as described previously. 9 Stained sections of pellets were used to determine the percentage area stained positively for Saf-O (%Saf-O_pellets) and collagen-II (%Coll-II_pellets) using ImageJ and a Matlab routine as described above.
Correlation of Cartilage Repair to BMSC Properties
Correlations between 5 in vitro–assessed biological properties of BMSCs (P0 cell yield, CFU-f, Pellet Size (mm), %Saf-O+ and %Coll-II+ area in pellets) and 4 in vivo–assessed repair parameters (ICRS Score, O’Driscoll Score, %Saf-O+, and %Coll-II+ repair tissue) was carried out for collagenase- and explant-derived BMSCs.
Statistical Analyses
Statistical analysis was performed using SAS Enterprise Guide 5.1 and SAS 9.3. Since several segments were collected from both legs of each rabbit, a mixed model was used to account for the influence of donor, assuming a normal distribution of data with regard to the donor. Fixed effects were location (condyles, upper trochlea or lower trochlea for in vitro characterization, n = 8 knees and condyles and trochlea for cartilage repair characterization, n = 8 knees) while donor was a random effect. Data in figures are presented as mean (diamond); median (line); box: 25th and 75th percentile; whiskers: box to the most extreme point within 1.5 interquartile range. Correlations between in vitro BMSC properties and in vivo repair outcome were analyzed by calculating the Pearson correlation coefficients (P < 0.05 statistically significant). To determine the in vitro variables that best account for the variability in repair outcome, a general linear model (GLM) was applied. For each in vivo repair parameter, variables with the least influence (highest value of P) were sequentially removed in a step wise manner to yield variables with the most significant influence (P < 0.05) on repair outcome.
Results
Repair of Trochlear Defects Was Superior to Condylar Defects and Correlated with In Vitro Properties of BMSCs
The main finding from the current study on the early events in cartilage repair was that the quality and quantity of condylar repair tissue was markedly inferior in contrast to mostly excellent trochlear healing. At 3 weeks postsurgery, 6 out of 8 trochlear defects were filled with glossy, white, and smooth repair tissue flush with adjacent cartilage ( Fig. 1a ). These donors were termed good trochlear responders. The majority of these repair tissues were homogenous with a smooth surface and appeared to bond well with adjacent tissue with occasional cases of heterogeneous tissue with slightly irregular surface and identifiable defect margins. Defects were significantly depressed and incompletely filled in 2 donors identified as poor trochlear responders ( Fig. 1b ). In contrast, all 8 condylar defects were severely depressed with very little fill in addition to distinct residual drill holes and defect margins ( Fig. 1c and d ). Mean ICRS macroscopic scores were significantly higher for trochlea versus condyles (P = 0.0027) ( Fig. 1e ). Moreover, 5 out of 8 trochlear repair tissues were assigned ICRS grade II (nearly normal) while all 8 condylar repair tissues were scored ICRS grade IV (severely abnormal).
Saf-O-stained sections of condyles showed thin, fibrous repair tissue with poor structural integrity in contrast to voluminous tissue with regular surface observed in trochlear defects ( Fig. 2a-h ). Average O’Driscoll scores were significantly reduced for condylar repair tissues versus trochlea (P < 0.0001) ( Fig. 2i ) and associated with lower scores for surface regularity, thickness of repair tissue, cell morphology, health of adjacent cartilage, and negligible GAG (glycosaminoglycan) accumulation. On quantitative histomorphometric analysis, significantly reduced %Saf-O (P = 0.002) ( Fig. 2j ) and %Coll-II (P = 0.0048) ( Fig. 3i ) were observed for condyles.
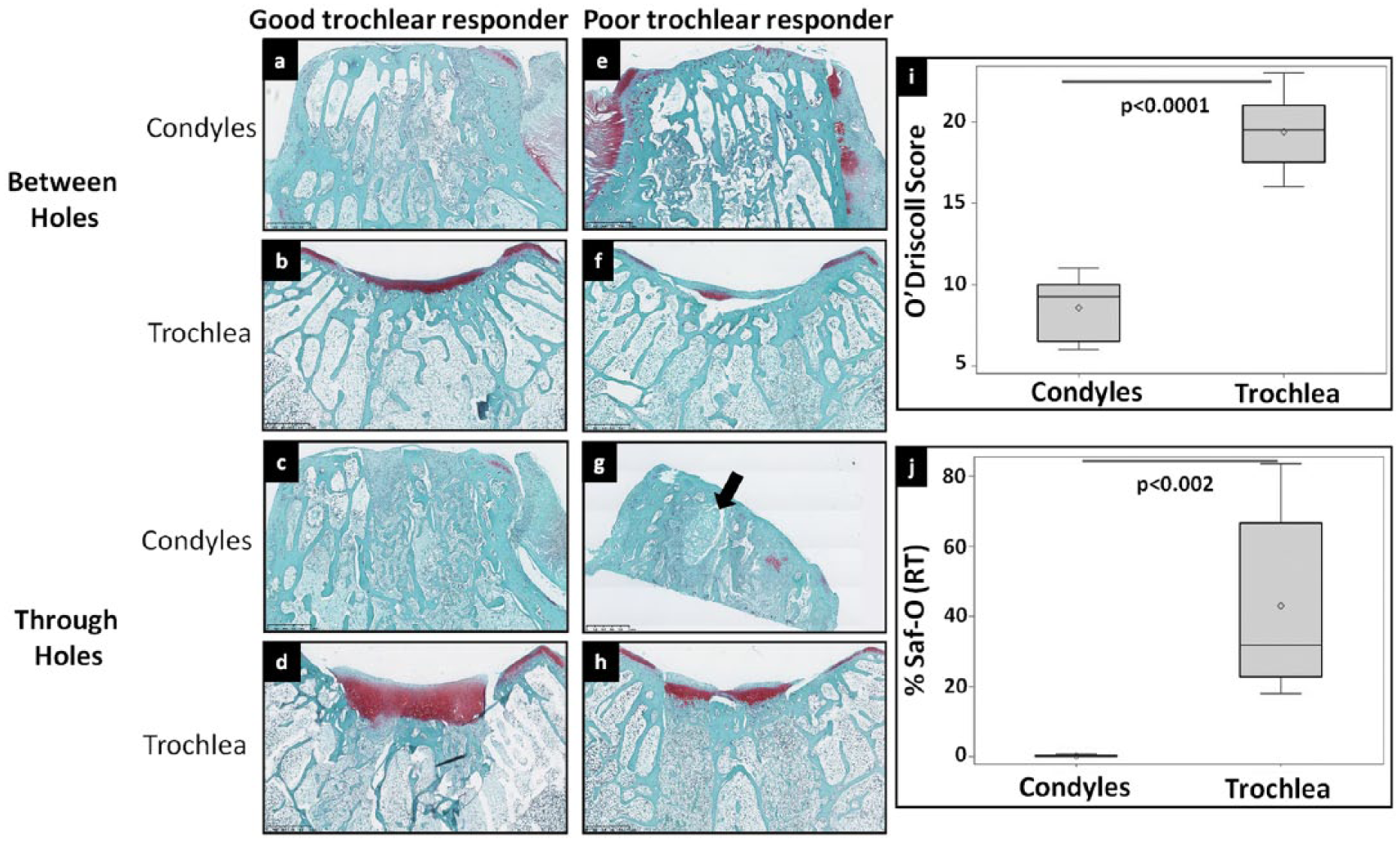
Representative Saf-O/fast green (
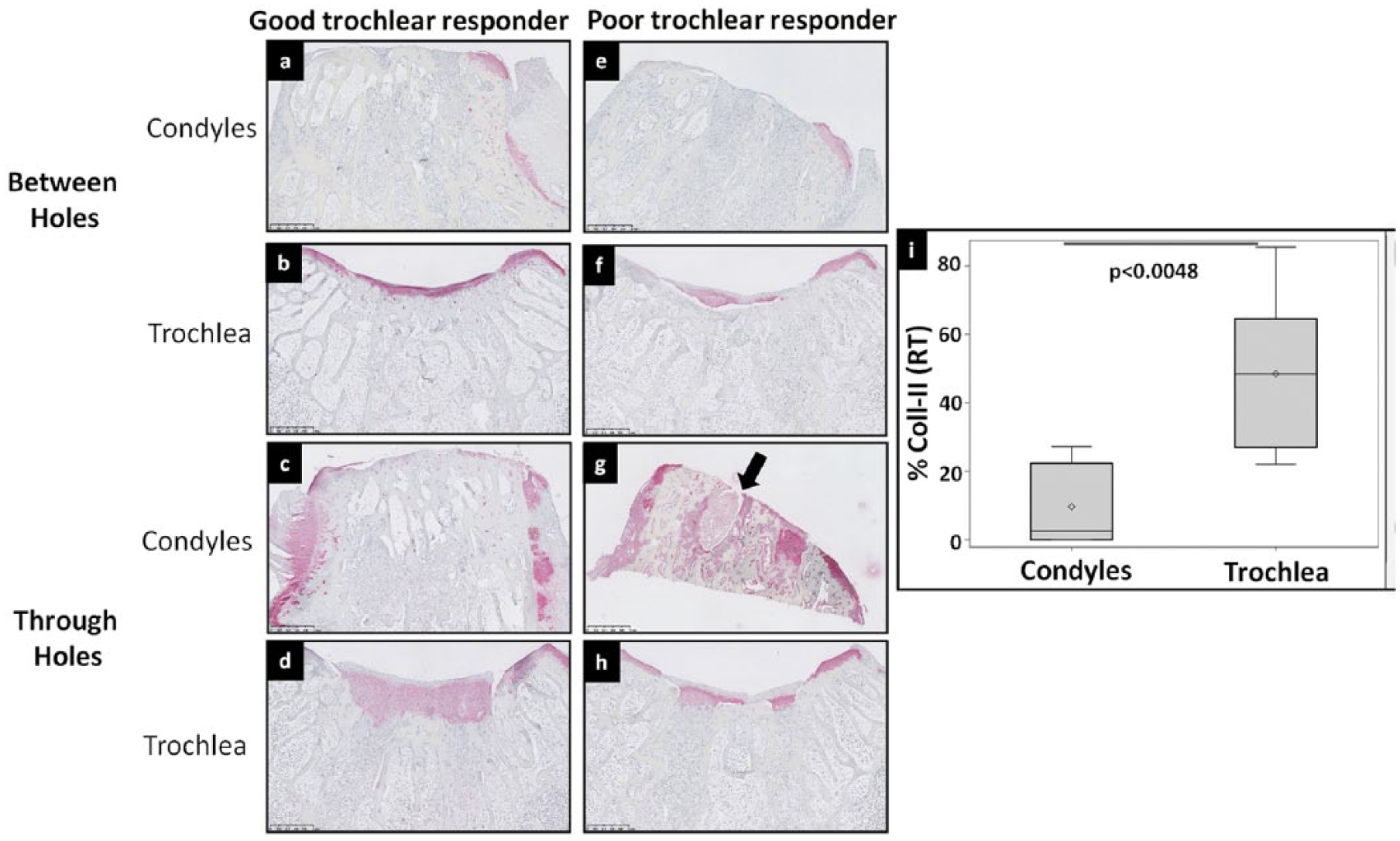
Representative Coll-II (
Clear histological differences were observed between good and poor trochlear responders in vivo. In the sections taken from between holes, thicker repair tissue with abundant GAG was observed in good trochlear responders in contrast to thin, fibrous tissues in poor trochlear responders with negligible GAG expression. In trochlear repair tissue through the microdrill holes, Saf-O staining was observed in mid-deep regions of good trochlear responders with varied levels of hyaline and fibrocartilaginous tissue. In contrast, repair tissue was thinner and more fibrous in poor responders indicated by diminished Saf-O (average %Saf-O −49.46 ± 13.72 in good responders vs 26.62 ± 8.1 in poor responders) ( Fig. 2b vs f and 2d vs h ). The only instance of an osteochondral cyst was observed in the condyle of one of the poor responders ( Fig. 2g ). Trochlear repair tissues also expressed greater levels of collagen type II compared with condyles ( Fig. 3a-h ). Among trochlea, Coll-II was more widespread in good responders while poor responders showed evidence of depleted Coll-II staining (average %Coll-II −56.67 ± 23.64 in good responders vs 20.6 ± 2.29 in poor responders) ( Fig. 3 ).
Pellets cultured without TGF-βIII were distinctly smaller ( Fig. 4a , e , a′ , e′ ) than TGF-βIII-stimulated cultures for both collagenase- and explant-derived BMSCs. Among the pellets stimulated with TGF-βIII, trochlea upper pellets had the largest diameter while condyles generated the smallest pellets for both collagenase-derived (P < 0.0001) and explant-derived BMSCs (P = 0.0013) ( Fig. 4 , Suppl. Figs. 1Sc and h and 2Sb). In general, trochlear pellets had increased GAG and Coll-II in collagenase- and explant-derived BMSCs ( Fig. 4 ). Interestingly, trochlea lower pellets were smaller in poor trochlear responders for both collagenase- and explant-derived BMSCs ( Fig. 4 and Suppl. Fig. 2S). Trochlea upper pellets were associated with abundant GAG and Coll-II in good as well as poor trochlear responders while trochlea lower pellets from poor trochlear responders had reduced Saf-O staining indicating low GAG deposition ( Fig. 4 ). As a further evidence of poor chondrogenic differentiation potential in cells derived from condylar subchondral bone, the matrix of condylar pellets was depleted in GAG and Coll-II as indicated by lower %Saf-O (Suppl. Fig. 1Sd and i) and %Coll-II (Suppl. Fig. 1Se and j). Trochlear pellet matrix had higher %Saf-O (collagenase-derived BMSCs, P < 0.0001 and explant-derived BMSCs, P = 0.0009) and %Coll-II versus condylar pellets (collagenase-derived BMSCs, P < 0.0001 and explant-derived BMSCs, P = 0.0002).
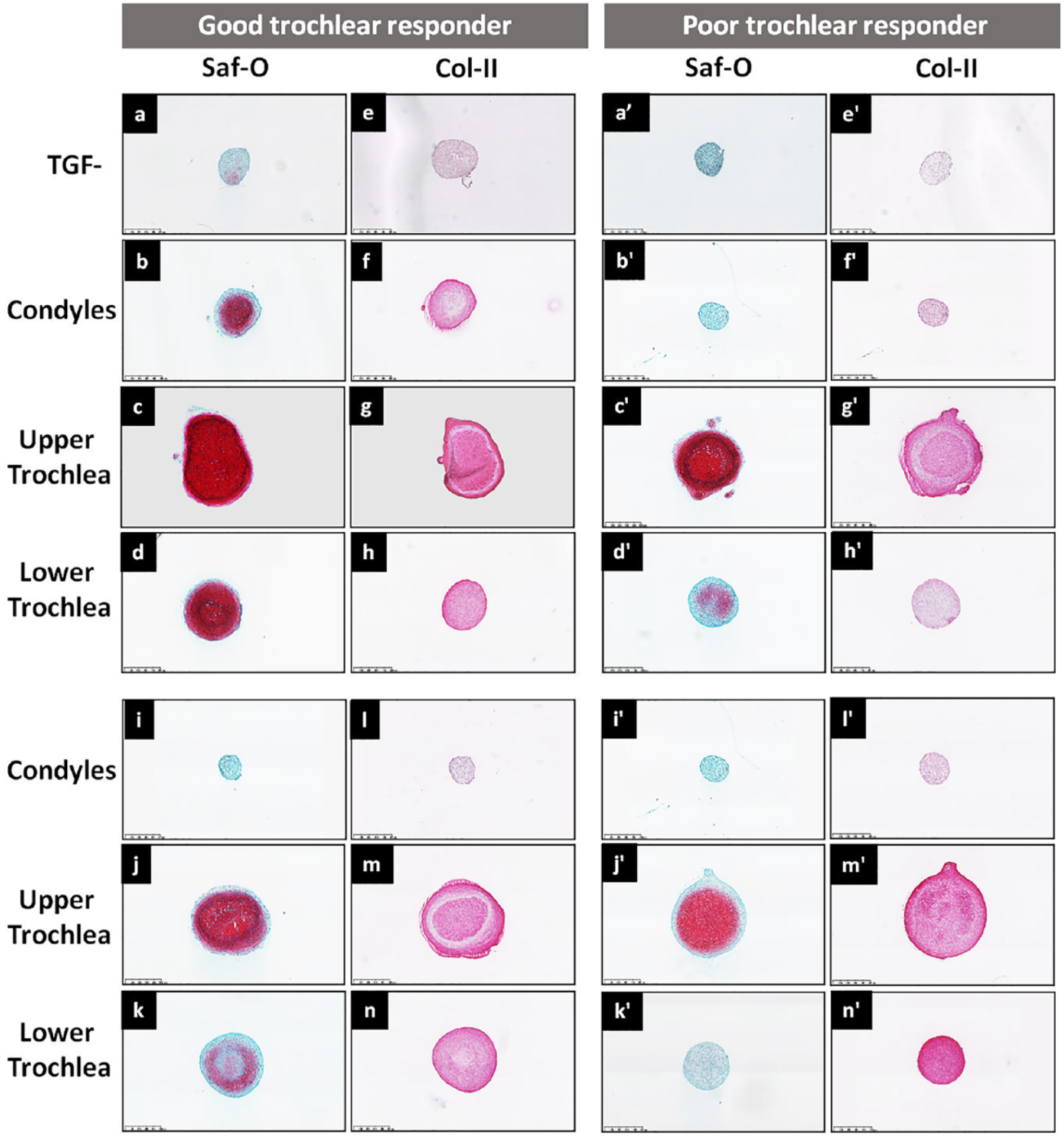
Safranin-O/fast green and collagen type II staining of pellets from good (
Biological Properties of Trochlear Mesenchymal Stem Cells Were Superior versus Condylar BMSCs and Decreased in Poor Trochlear Responders
In vitro biological properties were found to be influenced by location as well as the donor. In comparison with condyles, higher cell yield was observed in trochlea for both collagenase-derived (P = 0.007) and explant-derived BMSCs (P = 0.0008) (Suppl. Fig. 1Sa and f). BMSCs from trochlear segments also had higher clonogenic potential indicated by higher CFU-f for both collagenase (P < 0.0001) and explant-derived BMSCs (P = 0.008) (Suppl. Fig. 1Sb and g). In contrast, the 2 poor trochlea responders showed a noticeable reduction in cell yield and CFU-f in both trochlea upper and trochlea lower (Supplementary Table 1S).
Expression of cell surface markers was similar between condyles and trochlea. However, expression varied as a function of donor and was found to be weaker for collagenase-derived (Suppl. Fig. 3Sa and b) as well as explant-derived BMSCs (Suppl. Fig. 3Se and f) in both poor trochlear responders. This reduced expression was only observed for CD44 and not CD29 (data not shown)—the other stem cell marker analyzed. Expression of CD34 and CD45 was absent for all donors for collagenase- and explant-derived BMSCs indicating absence of any contamination by cells of hematopoietic origin (Suppl. Fig. 3S).
In Vitro Properties of Collagenase-Derived BMSCs Showed Moderate to Strong Positive Correlations with Early In Vitro Repair Responses
Significant correlations were found between in vitro BMSC properties and early repair response in condyle and trochlea. Robust positive correlations between cell yield, CFU-f, and %Coll-II (pellets) of collagenase-derived BMSCs and all in vivo repair properties studied ( Figs. 5 and 6 ) indicated a strong influence of these BMSCs properties on cartilage repair outcome (r2 ranged from 0.48 to 0.73). Pellet size and %Saf-O (pellets) showed significant, positive but modest correlation with most of the in vivo repair parameters studied (r2 ranged from 0.26 to 0.44) ( Figs. 5 and 6 ).
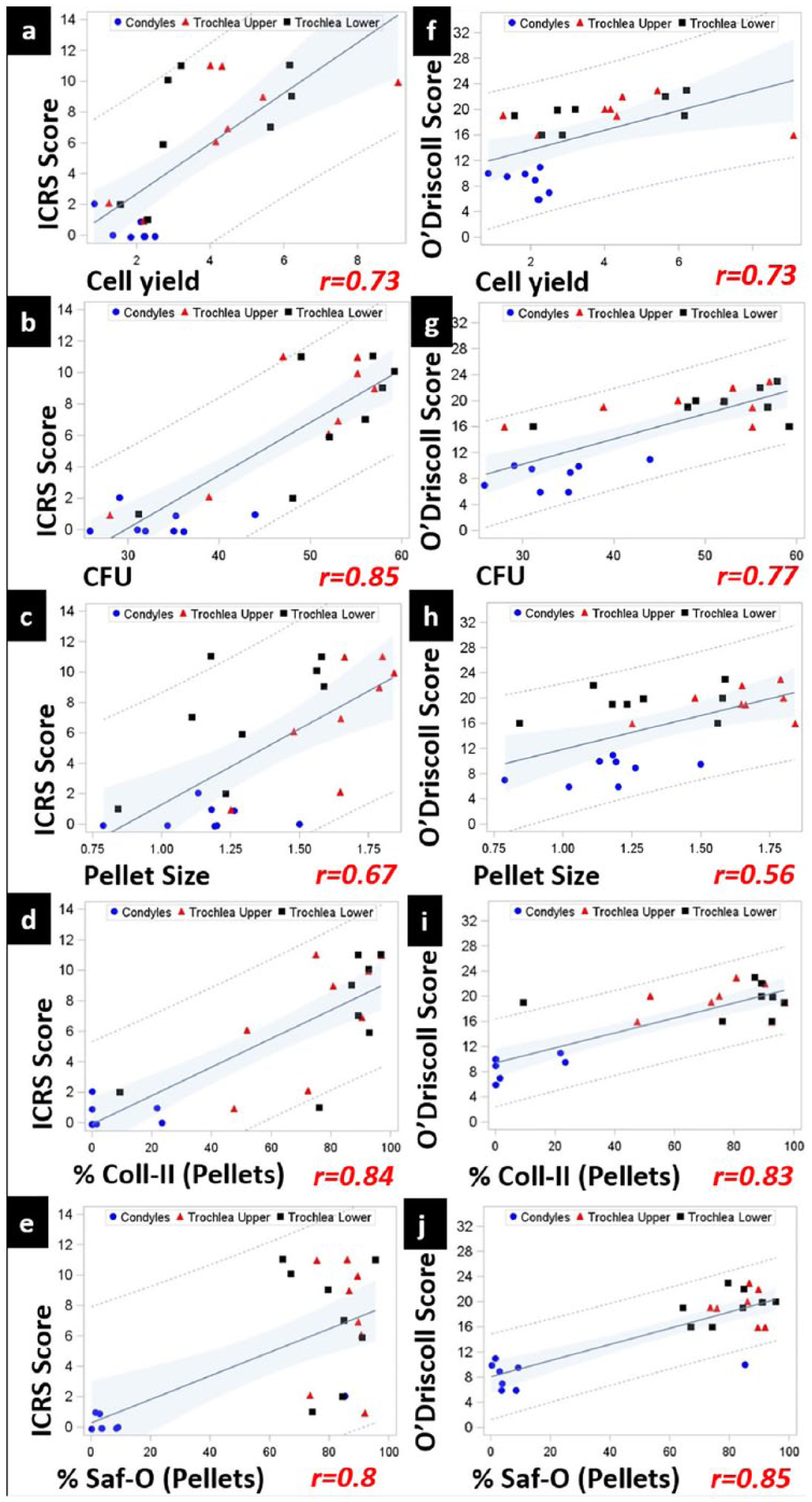
Strong, positive correlation between in vitro biological properties of bone marrow stem cells (BMSCs)—cell yield, CFU, pellet size, %Saf-O (pellets), %Coll-II (pellets), and repair response measured by International Cartilage Repair Society (ICRS) macroscopic scoring and O’Driscoll Score in trochlea versus condyles. N = 8. CI 95%; P < 0.0001-0.0068. RT, repair tissues. Blue circles, condyles; red triangles, trochlea upper; black squares, trochlea lower.
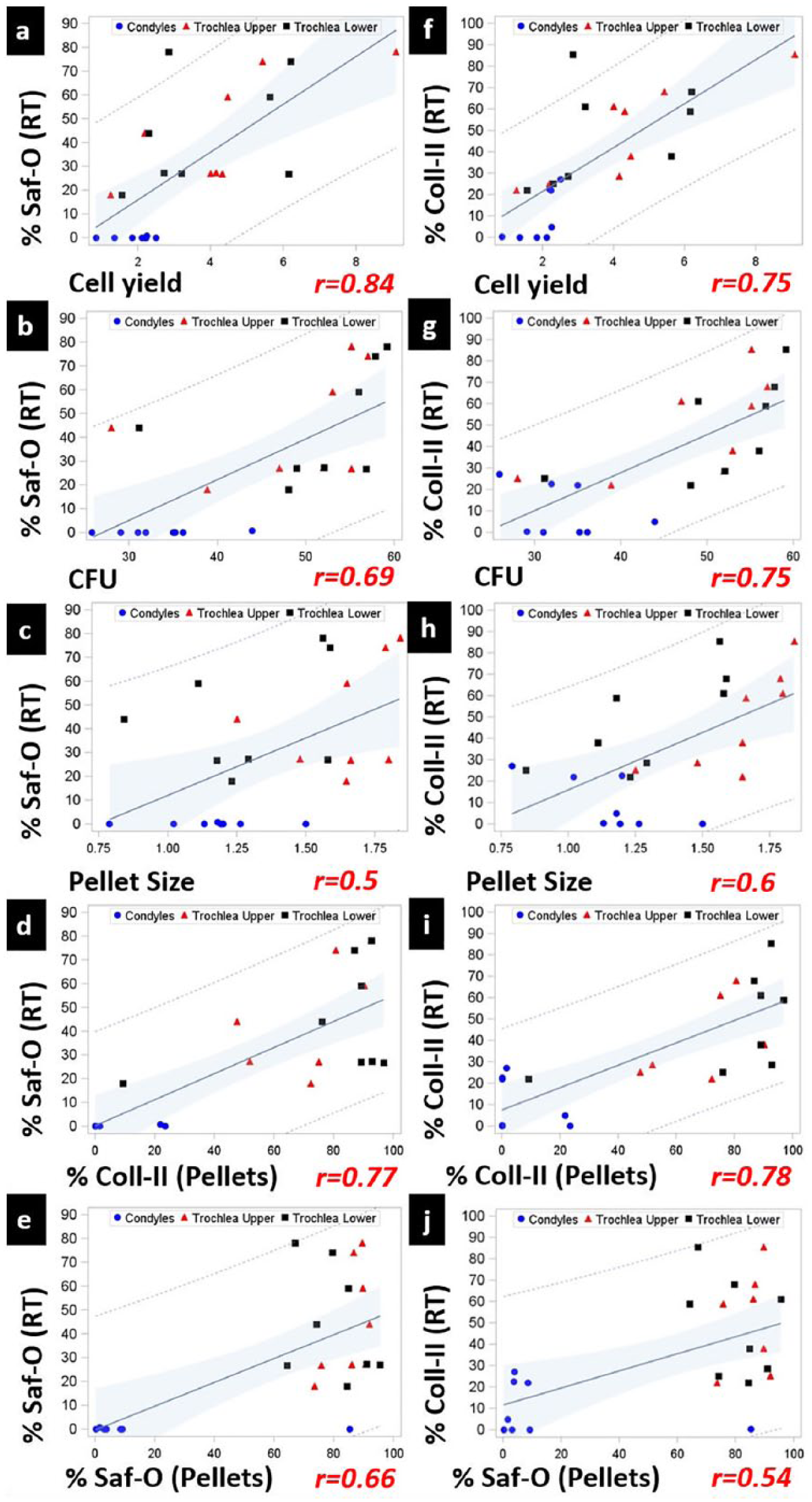
Strong, positive correlation between in vitro biological properties of bone marrow stem cells (BMSCs)—cell yield, CFU, pellet size, %Saf-O (pellets), %Coll-II (pellets), and repair response measured by %Saf-O (RT) and %Coll-II (RT) in trochlea versus condyles. N = 8. CI 95%; P < 0.0001-0.0068. RT, repair tissues. Blue circles, condyles; red triangles, trochlea upper; black squares, trochlea lower.
In contrast, the in vitro biological properties of explant-derived BMSCs showed positive, significant (P < 0.5) but weak correlations with in vivo characterization parameters (r2 ranged from 0.1 to 0.3). As an exception, %Coll-II (pellets) of explant-derived BMSCs demonstrated strong, positive correlations with all the in vivo parameters (r2 ranged from 0.38 to 0.56) (Suppl. Figs. 4S and 5S).
GLM analysis confirmed that cell number, clonogenic, and chondrogenic potential can serve as predictors of repair outcome. However, not all BMSC properties accounted for the variability in repair outcome to an equal extent ( Table 1 ). Macroscopic and O’Driscoll scores were better predicted by clonogenic and chondrogenic potential of pellets (%Saf-O and %Coll-II). On the other hand, matrix content of repair tissues—%Saf-O and %Coll-II—was more predicted by the initial cell population density (P0 cell yield) and chondrogenic potential of pellets (also defined by %Saf-O and %Coll-II). Taken together, cell yield, CFU and %Coll-II provided the maximum explanation for variability in different in vivo repair parameters. Consistent with these GLM analyses, these variables also presented the highest r2 values in the independent pairwise Pearson correlation analysis ( Table 1 and Figs. 5 and 6 ).
General Linear Model (GLM) Analysis of Dependent Variables to Determine the Variables with Maximum Influence on Repair Outcome. a
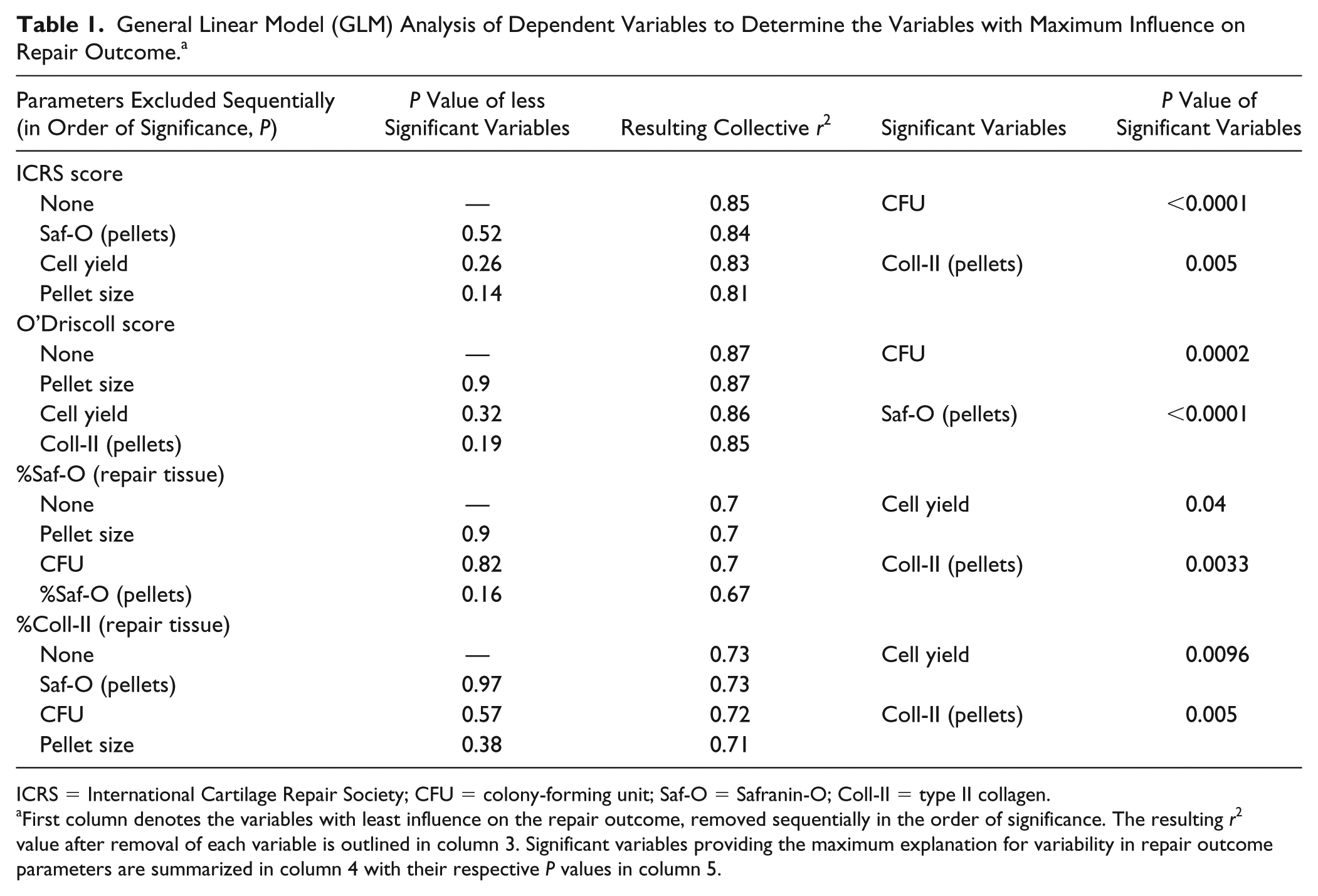
ICRS = International Cartilage Repair Society; CFU = colony-forming unit; Saf-O = Safranin-O; Coll-II = type II collagen.
First column denotes the variables with least influence on the repair outcome, removed sequentially in the order of significance. The resulting r2 value after removal of each variable is outlined in column 3. Significant variables providing the maximum explanation for variability in repair outcome parameters are summarized in column 4 with their respective P values in column 5.
Discussion
This study provides tangible evidence of a direct correlation between marrow stimulated repair outcome in condyle and trochlea and inherent biological properties of BMSCs present in these 2 locations. Variability in cell number, clonogenic, and chondrogenic potential of trochlear BMSCs showed strong correlations with quality and quantity of repair tissue in a location- and donor-dependent manner. Cell number, clonogenic, and matrix production potential of BMSCs were strong predictors of macroscopic and histological repair outcomes. Sufficient evidence was found to substantiate the hypothesis that trochlear cartilage repair outcome is superior to condyles and correlates with in vitro biological properties of the BMSCs at the 2 sites in the contralateral knee.
Although repair responses induced by BMS along with characterization of BMSCs has been investigated in animal models before,13-16 this is the first study designed to correlate the early repair response with biological properties of BMSCs from the same location to elucidate the mechanism of location-dependent and interanimal variation observed in cartilage repair. We chose a rabbit model since comparison of the structure of subchondral bone—the primary source of repair in marrow stimulation—has revealed striking similarities between human condyles and rabbit trochlea, making this a relevant preclinical model. 17 Unlike skeletally immature rabbits, >9-month-old skeletally mature rabbits do not display spontaneous repair potential and thus are more suitable choice for this model. Additionally, the proportion of the defect area covered by the perforations fit within the clinical range of hole perforation density. 5 A period of 2 months will most likely provide preliminary evidence of differences in the early events in cartilage repair to provide mechanistic insights into location-dependent variability in cartilage repair.
In human, cartilage lesions mostly affect the medial femoral condyle18,19 and although less frequently observed, trochlear lesions have been reported harder to treat. 20 According to one study, microfracture (MFX) resulted in superior outcome in MFC when compared with other locations. 21 In another evidence of the effect of location, a recent study reporting mid-term outcome after MFX found significantly better improvement in the patellofemoral or lateral lesions compared to medial lesions. 22 However, in a level II, prospective study with a cohort of 49 patients followed for 5 years for repair outcome with matrix assisted autologous chondrocyte transplantation, Filardo et al. 23 observed poorer repair specifically in the patellar lesions compared with trochlear defects emphasizing the need for more accurate and deeper exploration of location-dependent variability in different compartments of knee joint. An influence of location was also observed in a sheep study where MFX generated superior fill and integration in trochlear defects. 7 A recent study explored the correlation between repair of the defect and its location on the femoral head in a rabbit model. Quality and quantity of repair were found to be strongly influenced by the load-bearing role of the location assessed. 24 These species and location dependent differences might be affected by a combination of several factors such as convex versus concave nature of condyle, which may be inept at protecting the newly formed repair tissue. Multiplicity of lesions and symptom duration are other compounding factors. 25 Being the primary weightbearing location in the knee joint adversely affects the ongoing repair in condyles. The location-dependent variability may also result from a difference in the thickness and density of subchondral bone which shows distinct differences between condyle and trochlea. A similar effect of location was also observed when tissue engineered cartilage survived better in trochlea compared with condyles. 26 The anatomic location of the defect is, therefore, an important guiding factor influencing cartilage repair in a patient-specific manner.
Bone marrow stimulated repair is mediated by recruitment of mesenchymal stem cells (MSCs) into access channels and cartilage defect followed by a complex multistep cascade of events, including proliferation, cell-cell interaction and differentiation finally leading to the formation of a repair tissue with varying amounts of fibrous and hyaline cartilage.27,28 It is logical to speculate that the properties of MSCs will also have a profound impact on the repair outcome. A difference in the initial progenitor cell population will likely have a strong bearing on the variability in the repair response. As shown by Mizuta et al. 29 in a rabbit full-thickness defect model, explicit chondrogenesis was preceded by an active phase of expansion in chondroprogenitor cell population. Moreover, a high number of cells in the initial stages of chondrogenesis is a prerequisite since close interaction of closely packed cells during the condensation phase is critical for initiation of chondrogenic differentiation of MSCs. 30 Fennema et al. 31 had reported that the differences in the original number of proliferative cells isolated were mirrored in the growth rate of corresponding BMSC cultures. Clearly, inherent progenitor cell population is a vital consideration in cartilage repair. In the current study, lower cell yield indicated a depleted progenitor cell population in condyles having an impact on the onset and progress of downstream repair process. Low number accompanied with reduced clonogenic potential as indicated by CFU-f assay might attribute to the diminished chondrogenic potential of condyles. It has also been shown that increasing population doublings escalate heterogeneity, reduces multipotency and repair potential. 32
In vitro cultures of BMSCs provide limited insight into the complex and dynamic joint microenvironment during repair; however, studies have shown that cellular behavior of MSC cultures provide a close estimation of their in vivo potency and allude to heterogeneity in the repair outcome. 33 Post et al. 34 characterized 2 murine MSC populations in vitro and found a preexisting propensity toward adipogenic or osteogenic differentiation, which was consistent with in vivo behavior. Moreover, MSC cultures have been shown to be heterogeneous with regard to morphology, 35 which was later correlated to functional heterogeneity and differentiation potency in vitro 36 and repair in vivo. 37 Furthermore, on comparison of stem cells from a variety of niches, Woo et al. 38 revealed marked differences in their proliferation and differentiation propensity despite similar cell surface marker profiles likely due to cellular environment and genetic factors, including epigenetic factors. Stem cells isolated from different locations have also shown differences in their propensities toward a particular lineage—such as a preference for adipogenic and osteogenic differentiation in the case of adipose-derived stem cells and BMSC, respectively. 39 Furthermore, studies have shown that the proliferative and functional profile of MSCs is altered in osteoarthritis.40,41 Taken together, observations from the previous and the current study provide strong evidence of a correlation between location-dependent variation in cartilage repair response and inherent differences in the biological properties of subchondral BMSCs including their number and differentiation potential.
In this study, BMSCs were isolated from both stromal spaces and bone lining regions by means of collagenase digestion and cell outgrowth from explants since cells participating in marrow-stimulated cartilage repair originate from both these regions. While a fraction of cells residing in the bone marrow would be recruited to the defect site, they would undergo rapid proliferation leading to increased localized cell number. Although further studies are needed to locate the origin of the in situ cells migrating into the defect, cells from the subchondral bone matrix are less likely to contribute to the repair owing to the lower cell number, advanced differentiated stage, and constraints of the surrounding matrix. In vitro studies revealed weak correlations between cartilage repair and explant-derived BMSCs, which might suggest a limited contribution of these cells in cartilage repair, likely restricted by their ability to migrate out of bone. Because of low initial yield, these cells might have undergone more population doublings in culture leading to loss of stemness and subsequent chondrogenic potential.
In another observation, repair outcome in poor trochlear responders was associated with an inferior number, clonogenic, and differentiation potential of BMSCs isolated from trochlea of these donors indicating a strong donor influence. In a recent study, the functional behavior of human MSCs differentiated in 3-dimensional hyaluronic acid gels was found to be strongly influenced by the donor effect. 42 Histological analysis of the repair tissues in the poor trochlear responders showed evidence of poor cartilage repair indicated by thin and poorly attached tissues. The tissues did not stain positive with Saf-O and were mostly fibrous in nature, which is known to be of inferior stability. Poor quality of tissue has been one of the most important limitations of MFX procedure in the past. 43 Earlier studies have reported cultures obtained from different donors to be heterogenous with respect to number and differentiation. 44 Interestingly, our data suggest that inferior repair in poor trochlear responders may be a result of a depleted progenitor cell population further aggravated by a reduced clonogenic potential of BMSCs isolated from the metaphyseal region of trochlea in these donors. These probably had a direct bearing on the chondrogenic potential indicated by depleted GAG expression in trochlea lower pellets. Smaller pellet size further suggests that inferior chondrogenic differentiation potential of metaphyseal BMSCs might be responsible for poor chondrogenic differentiation in poor trochlea responders. The poor potential of these donors seems to be at least partially intrinsic since the cells also had reduced expression of cell surface markers. These observations provide further evidence of correlation of cartilage repair response and inherent properties of local population of BMSCs in the region.
Location-dependent variability in cartilage repair might also arise due to impact of location on cellular interactions, growth and differentiation factors in addition to other mechanical and chemical stimuli. These factors impact the signaling pathways which control the transcription of specific genes contributing to the onset and progress of the repair. For instance, Wnt proteins, TGFβ superfamily, bone morphogenetic proteins (BMPs), fibroblast growth factor (FGF), and insulin-like growth factor (IGF) are some of the several morphogenetic factors, 45 which influence BMSCs possibly controlling the eventual repair outcome. Moreover, in vitro expansion and differentiation of BMSCs has been shown to be influenced by hydrostatic, compression, and shear forces46-48 in addition to mechanical loading and oxygen tension. 49 The downstream differentiation commitment of BMSCs may also be influenced by spatial dimensionality modifying the interaction of BMSCs in trochlea and condyle and their corresponding microenvironment. 50 In addition, the mechanical environment plays a role in the regulation of tissue-specific inhibitors of metalloproteases (TIMPs) and matrix metalloproteases (MMPs), which have been implicated in the differentiation of BMSCs. 51
Despite moderate to strong positive and significant Pearson correlation coefficients observed between all the biological properties analyzed and corresponding repair outcome parameters, GLM analysis revealed that certain correlations were more strongly defined by trochlea data. Taken together, trochlea upper provided maximum explanation for the variability observed in vivo (Suppl. Table 2S). One drawback from our study is that several hypotheses were tested that did not explicitly account for multiplicity in our statistical analyses and correlations and therefore this study should be regarded as a first hypothesis forming study.
Recognizing the underlying cause is the first step toward addressing differences in cartilage repair outcome. As evident from the present study, the differences in the number and stemness of the underlying progenitor cells might be one of the most important factors affecting the marrow stimulated repair outcome. This finding can be helpful in determining the treatment route in the clinical setting in the future. For example, it is clear that a primary cause for poor condylar repair originates from poor progenitor cell population in this region. Clinicians could consider using an approach such as augmented MFX in such cases, which in addition to increasing the recruitment of progenitor cells, also stabilizes them into the defect thereby increasing the density of these cells likely leading to superior repair. Augmentation strategies may also better protect the premature repair tissue during early repair thus improving the overall repair quality and quantity. In a recent study, a similar finding was applied to improve the repair in poorly healing cartilage by administration of intra-articular hyaluronic acid. 42 An alginate gel used to augment MFX also showed superior repair in a canine model. 52 Supporting MSC differentiation with matrices could also reduce hypertrophy and endochondral ossification thereby improving the overall repair outcome. 53 Additionally, a combination of scaffolds and bioactive agents such as platelet-rich plasma or growth factors may also be considered.54-56 Similar considerations will also be relevant in an older patient population.
Conclusion
This study provided substantial evidence to reveal clear differences in biological properties of BMSCs present in condyles and trochlea which are most likely the cause of variation in the repair outcome observed in these 2 locations. It is imperative to carry out functional phenotype analysis both in vitro and in vivo in order to draw parallels between in vitro behavior of BMSCs and corresponding repair outcome in an animal model especially since precise methods for in vivo identification of BMSC and their behavioral patterns still elude us. In the future, detailed molecular profiling of BMSCs needs to be carried out in order to identify the genetic mechanism of the location- and donor-dependent differences. The results from this study will advance the understanding of donor- and site-specific variations in BMS repair response eventually helping in maximizing the efficiency of procedure in a more patient-relevant basis.
Supplemental Material
Dwivedi_2_supplementary_Table_1 – Supplemental material for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model
Supplemental material, Dwivedi_2_supplementary_Table_1 for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model by Garima Dwivedi, Anik Chevrier, Mohamad-Gabriel Alameh, Caroline D. Hoemann and Michael D. Buschmann in CARTILAGE
Supplemental Material
Dwivedi_2_supplementary_Table__2 – Supplemental material for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model
Supplemental material, Dwivedi_2_supplementary_Table__2 for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model by Garima Dwivedi, Anik Chevrier, Mohamad-Gabriel Alameh, Caroline D. Hoemann and Michael D. Buschmann in CARTILAGE
Supplemental Material
Dwivedi_Figure_Supplementary_1 – Supplemental material for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model
Supplemental material, Dwivedi_Figure_Supplementary_1 for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model by Garima Dwivedi, Anik Chevrier, Mohamad-Gabriel Alameh, Caroline D. Hoemann and Michael D. Buschmann in CARTILAGE
Supplemental Material
Dwivedi_Figure_Supplementary_2 – Supplemental material for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model
Supplemental material, Dwivedi_Figure_Supplementary_2 for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model by Garima Dwivedi, Anik Chevrier, Mohamad-Gabriel Alameh, Caroline D. Hoemann and Michael D. Buschmann in CARTILAGE
Supplemental Material
Dwivedi_Figure_Supplementary_3 – Supplemental material for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model
Supplemental material, Dwivedi_Figure_Supplementary_3 for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model by Garima Dwivedi, Anik Chevrier, Mohamad-Gabriel Alameh, Caroline D. Hoemann and Michael D. Buschmann in CARTILAGE
Supplemental Material
Dwivedi_Supplementary_figure_4 – Supplemental material for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model
Supplemental material, Dwivedi_Supplementary_figure_4 for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model by Garima Dwivedi, Anik Chevrier, Mohamad-Gabriel Alameh, Caroline D. Hoemann and Michael D. Buschmann in CARTILAGE
Supplemental Material
Dwivedi_Supplementary_figure_5 – Supplemental material for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model
Supplemental material, Dwivedi_Supplementary_figure_5 for Quality of Cartilage Repair from Marrow Stimulation Correlates with Cell Number, Clonogenic, Chondrogenic, and Matrix Production Potential of Underlying Bone Marrow Stromal Cells in a Rabbit Model by Garima Dwivedi, Anik Chevrier, Mohamad-Gabriel Alameh, Caroline D. Hoemann and Michael D. Buschmann in CARTILAGE
Footnotes
Author Contributions
The authors made substantial contributions in designing the study (GD, AC, CDH, MDB), gathering and analyzing the data (GD, MGA and AC) and drafting the article (GD, AC, CDH, MDB). All the authors approved the submitted manuscript.
Acknowledgments and Funding
We gratefully acknowledge the excellent technical contributions of Jun Sun and Geneviève Picard. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CIHR Operating Grant (MOP 115186).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MDB and CDH are on the Board of Directors and hold shares with Ortho RTi. AC holds shares with Ortho RTi. None of the other authors has any competing interests to declare.
Ethical Approval
The study was approved by the University of Montreal “Comité de déontologie de l’expérimentation sur les animaux” (#13-129).
Animal Welfare
Canadian Council on Animal Care guidelines for the care and use of laboratory animals were observed.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
