Abstract
Background:
The identification of factors associated with clinical recovery in youth after sports-related concussion could improve prognostication regarding return to play (RTP).
Purpose:
To assess factors associated with clinical recovery after concussion in youth ice hockey players.
Study Design:
Cohort study; Level of evidence, 2.
Methods:
Participants were part of a larger longitudinal cohort study (the Safe to Play study; N = 3353). Included were 376 ice hockey players (age range, 11-17 years) from teams in Calgary and Edmonton, Canada, with 425 physician-diagnosed ice hockey–related concussions over 5 seasons (2013-2018). Any player with a suspected concussion was referred to a sports medicine physician for diagnosis, and a Sport Concussion Assessment Tool (SCAT) form was completed. Time to clinical recovery was based on time between concussion and physician clearance to RTP. Two accelerated failure time models were used to estimate days to RTP clearance: model 1 considered symptom severity according to the SCAT3/SCAT5 symptom evaluation score (range, 0-132 points), and model 2 considered responses to individual symptom evaluation items (eg, headache, neck pain, dizziness) of none/mild (0-2 points) versus moderate/severe (3-6 points). Other covariates were time to physician first visit (≤7 and >7 days), age group (11-12, 13-14, and 15-17 years), sex, league type (body checking and no body checking), tandem stance (modified Balance Error Scoring System result ≥4 errors out of 10), and number of previous concussions (0, 1, 2, and ≥3).
Results:
The complete case analysis (including players without missing covariates) included 329 players (366 diagnosed concussions). The median time to clinical recovery was 18 days. In model 1, longer time to first physician visit (>7 days) (time ratio [TR], 1.637 [95% confidence interval (CI), 1.331-1.996]) and greater symptom severity (TR, 1.016 [95% CI, 1.012-1.020]) were significant predictors of longer clinical recovery. In model 2, longer time to first physician visit (TR, 1.698 [95% CI, 1.399-2.062]), headache (moderate/severe) (TR, 1.319 [95% CI, 1.110-1.568]), and poorer tandem stance (TR, 1.249 [95% CI, 1.052-1.484]) were significant predictors of longer clinical recovery.
Conclusion:
Medical clearance to RTP was longer for players with >7 days to physician assessment, poorer tandem stance, greater symptom severity, and moderate/severe headache at first visit.
Keywords
Our current understanding of factors influencing clinical recovery after sports-related concussion in youth is limited. 12 A better understanding of factors influencing time to clinical recovery will inform concussion management and treatment. Sports-related concussion is a traumatic brain injury induced by biomechanical forces. 18 In Canada, each year approximately 1 in 10 adolescents and young adults (age range, 13-19 years) sustain a sports-related concussion. 10 In youth, 60% of concussions are sustained in sports, and >30% of concussions are expected to be recurrent. 2,20,22 In youth ice hockey (age range, 11-17 years), concussion rates are high, ranging from 2.8 to 4.2 concussions per 1000 game-hours in leagues allowing body checking and 1.1 to 2.31 concussions per 1000 game-hours in leagues not allowing body checking. 4,8,9,11 Studies evaluating time to clinical recovery in children and adolescents have reported that 15% to 30% remain symptomatic >1 month and 11% remain symptomatic for >3 months. 1 –3,22 –24
The long-term effects of concussion in youth are largely unknown. The 5th International Conference on Concussion in Sport reported a standard definition for persistent postconcussive symptoms to ensure consistency in clinical management and research outcomes. 18 Persistent symptoms after sports-related concussion were determined to reflect failure of normal clinical recovery including symptoms that persist beyond expected time frames (ie, >10-14 days in adults and >28 days in children). 5,18 Studies investigating factors influencing clinical recovery have consistently reported prolonged clinical recovery in adolescents compared with children, those evaluated with a headache acutely after concussion, and those with more severe injury (eg, admission to hospital, severity of injury score). 1,23,24 Furthermore, Zemek et al 23 reported that being female, previous concussion history, migraine history, error scores ≥4 on the Balance Error Scoring System (tandem stance), and symptoms of sensitivity to noise and fatigue (in addition to headache) were associated with persistent postconcussive symptoms >28 days. Some contradictory evidence has suggested that higher scores on the postconcussion symptom scale were acutely associated with a reduced likelihood of prolonged clinical recovery in children evaluated at a children’s hospital. 3
In evaluating factors influencing prolonged clinical recovery in adult collegiate athletes, Zuckerman et al 25 reported previous concussion history and acute symptoms, including retrograde amnesia, difficulty concentrating, sensitivity to light, and insomnia, are associated with postconcussion symptoms >1 month. McCrea et al 16 also demonstrated loss of consciousness at time of injury, posttraumatic amnesia, and more severe acute symptoms are associated with prolonged clinical recovery after concussion in a large sample of high school and adult collegiate athletes. A study of the effect of timing of initiation of clinical care on clinical recovery in youth and young adults (age range, 12-22 years) evaluated at a sports medicine concussion clinic reported that patients evaluated >7 days after injury had 5.8 times greater odds of a clinical recovery >30 days. 14 In a systematic review, Iverson et al 12 evaluated factors associated with clinical recovery after concussion and reported that the most consistent factor influencing slower clinical recovery from concussion is the severity of acute and subacute symptoms. The development of subacute problems with headaches or depression was consistently a risk factor for persistent symptoms beyond 1 month. 25 Individuals with a preinjury history of mental health problems, adolescents (compared with children), and girls (vs boys) were at greater risk for persistent symptoms. 12
Given the potential of prolonged clinical recovery in youth and the paucity of prospective studies evaluating sports-related concussion prognosis in youth, this study aimed to investigate factors associated with clinical recovery after sports-related concussion specifically in child and adolescent ice hockey players (age range, 11-17 years). Ice hockey may be unique given the high rates of concussion in youth players and the mechanisms of concussion in youth ice hockey associated with body checking and higher player speeds compared with other high concussion risk youth collision sports such as football and rugby. 4,8,9,11,22
Methods
Study Design
This was a subcohort study including youth ice hockey players participating in the Safe to Play cohort study conducted across 5 ice hockey seasons (2013-2018) in Calgary, Edmonton, and surrounding areas in Alberta, Canada. 4,8,11 This subcohort included players with a suspected concussion that was subsequently diagnosed by a sports medicine physician involved in this study (C.L. and other physicians). Ethics approval was received for this study.
Participants
The sample size for the larger cohort study was based on an a priori evaluation of body-checking policy changes in reducing rates of concussion and all injury across 3 age groups (11-12, 13-14, and 15-17 years) and different levels of play. Details of these studies have been previously reported. 4,8,11 The inclusion criteria were (1) age between 11 and 17 years, (2) participation in male or female leagues, (3) written informed player and parent consent, (4) registration in Hockey Calgary or Edmonton, (5) agreement of the head coach, and (6) agreement of a team designee (eg, manager, safety coach) to collect player participation and injury information. Players were excluded if they had a previous injury or illness preventing full participation in hockey at season commencement.
Procedures
The injury surveillance methodology used has previously been validated in youth ice hockey. 9 This included a preseason baseline questionnaire, Sport Concussion Assessment Tool (SCAT3 in years 1-4 and SCAT5 in year 5), weekly exposure sheet (WES), and injury report form (IRF). 6,7,9,17 Each team designee collected WES data and identified players with a suspected concussion or injury. Team designees were all managers or safety coaches who had received training from a therapist involved in this study (A.M.B., S.S., K.J.S.) on identification of a potential concussion using a concussion recognition tool based on the 2013 and 2017 consensus statements on concussion in sport (4th and 5th International Conferences on Concussion in Sport). 17,18 For all injuries reported on an IRF or if a player’s WES indicated the player was absent owing to injury, a study therapist followed up by telephone or at physician visit. Additional details of injury surveillance procedures have been reported in previous studies. 4,8,9,11
All ice hockey injuries resulting in medical attention, the inability to complete a session, or time loss from hockey were identified by the team designee and recorded on an IRF. All players with a suspected concussion were given the opportunity to follow up with a study sports medicine physician within 72 hours. Standardized diagnostic procedures and follow-up and return-to-play (RTP) protocols were followed by all study physicians based on the 4th (years 1-4) and 5th (year 5) International Consensus Statements on Concussion in Sport. 17,18 Concussions were included if they met the definition for concussion based on the consensus statements. 17,18 Given the change in the standard of care after concussion between years 4 and 5, a change from the RTP protocol to the return-to-sports strategy was implemented after publication of the 5th International Consensus Statement on Concussion in Sport. 18 The main difference was that only 24 to 48 hours of initial rest followed by a gradual return to subsymptom threshold activities was implemented in 2017. 18,21
A SCAT3 or SCAT5 form, including the symptom evaluation and modified Balance Error Scoring System (mBESS) components, was completed during the first physician visit after concussion. 7,17 These 2 components remained unchanged in the 2 versions of the SCAT used. Days to clinical recovery (ie, time to RTP clearance) was defined as the number of days from injury to physician clearance to RTP based on clinical assessment. This clearance was based on resolution of concussion-related symptoms at rest and completion of the RTP protocol/strategy as outlined in the 4th and 5th International Consensus Statements on Concussion in Sport. 17,18
Statistical Analysis
Players with at least 1 concussion diagnosed by a study sports medicine physician were considered in the analyses. Baseline characteristics were reported each year for the participants. Every time a player had a concussion, other characteristics and assessments were also recorded (eg, number of previous concussions, mBESS tandem stance). These characteristics were described with frequencies and proportions. An accelerated failure time (AFT) log-logistic model, 13,15 clustered by participant, was used to find the best combinations of covariates available that influence greater number of days to clinical recovery (ie, number of days to physician clearance to RTP). If the number of days to physician clearance to RTP was not available (ie, if the player did not return to the physician for clearance to RTP), then the date of clearance was based on actual date of RTP based on IRF or WES. This was part of the surveillance methodology in which a study therapist followed up to complete the IRF in case a player did not attend the final physician visit. If RTP did not occur by the end of the study or the participants were lost to follow-up, the participant was right censored (ie, we considered it an incomplete observed data point).
Covariates were identified based on previous research that indicated they were significantly related to days to clinical recovery to RTP in pediatric populations (non–sports- and sports-related concussion) and adult populations. 1,3,23,24 The covariates included were collected at the first physician visit and included the components of the SCAT3 or SCAT5 (ie, symptom evaluation score [range, 0-132 points; a higher score indicates more severe symptoms]; responses to specific symptom evaluation items of none/mild [0-2 points] and moderate/severe [3-6 points]; and mBESS tandem stance [≤3 and ≥4 errors out of 10]) 14,24 as well as the number of previous concussions (0, 1, 2, and ≥3). Other covariates collected at baseline (ie, preinjury at annual time of recruitment in each study year) were age group (11-12, 13-14, and 15-17 years), sex, and league type (body checking, no body checking). The time to first physician visit (≤7 and >7 days) 14,24 was also recorded after first physician visit on the IRF.
Because the symptom evaluation component was derived from the specific symptom items (eg, trouble sleeping, headache, fatigue), we expected collinearity between the overall score and specific symptom scores. Therefore, 2 separate AFT models were used for analyses: model 1 included all covariates identified a priori based on the literature and symptom severity according to overall symptom evaluation score, and model 2 considered all covariates and symptom severity according to specific symptom responses (none/mild vs moderate/severe).
We performed imputation for specific symptoms and the corresponding symptom evaluation scores where data were available for at least 3 specific symptoms. The imputation of specific symptoms for a player was done by substituting the missing value with the median of all other symptom scores for that player; the overall symptom evaluation score was also calculated for that player. A complete case analysis was used in the AFT models. The data characteristics were similar in the excluded data to those in the analyses data. A significance level of .05 was used, and covariates were evaluated as confounders if a difference of >10% in the coefficients was detected. The statistical software R (Version 4.0.0) was used for all analyses. 19
Results
A total of 3353 hockey players (age range, 11-17 years) were recruited from teams in Calgary, Edmonton, and surrounding areas during the 2013-2018 ice hockey seasons. Among these, 376 players were diagnosed by a physician with at least 1 concussion, resulting in 425 concussions in total (Figure 1).

Participant flowchart.
Eliminating players with missing covariates in the AFT models, we considered 366 diagnosed concussions from 329 players (female, 50 players; male, 279 players) in our complete case analyses. This included imputation of the symptom evaluation scores for 19 players with a diagnosed concussion and imputation of specific symptom scores for each of the selected specific symptom covariates for 5 players. The median number of days to clearance to RTP from the model was 18. The total numbers of concussions with complete and censored information on RTP were 324 (88.525%) and 42 (11.475%), respectively. Of the 324 concussions included and not right censored, the clinical recovery date was based on clinical clearance for RTP (based on physician assessment) in 258 of 324 (79.6%) cases. In total, of these 324 players, 30 (9.26%) of players with diagnosed concussions had recovered on or before 7 days, 243 (75%) players had recovered on or before 28 days, and 81 (25%) demonstrated prolonged clinical recovery >28 days. The concussion characteristics for the 376 players are presented in Tables 1 and 2.
Diagnosed Concussions at Time of Assessment According to Player Characteristics (N = 425 Concussions) a

a Data are presented as n (%).
Responses to SCAT3/SCAT5 Components for Each Concussion (N = 425 Concussions) a

a Data are presented as mean ± SD or n (%). mBESS, modified Balance Error Scoring System; SCAT, Sport Concussion Assessment Tool.
Model 1
Analysis of model 1 (Table 3) demonstrated that longer time to first physician visit (>7 days) (time ratio [TR], 1.637 [95% confidence interval (CI), 1.342-1.998]) and a higher symptom evaluation score (TR, 1.016 [95% CI, 1.012-1.020]) were associated with longer clinical recovery. The time to RTP among youth ice hockey players who saw a physician >7 days after a concussion was 1.637 times that of youth hockey players who saw a physician ≤7 days. Probability curves for RTP comparing the 2 groups of time to first physician visit are presented in Figure 2. The probabilities in each group corresponded to 13- to 14-year-old male hockey players, playing in a body-checking league, no previous history of concussion, a symptom evaluation score of 12, and tandem stance score ≤3 at the time of first physician visit.
Model 1 Analysis Results (Outcome: Time to RTP Clearance) a

a Bolded values indicate statistical significance based on a 5% significance level. mBESS, modified Balance Error Scoring System; RTP, return to play.
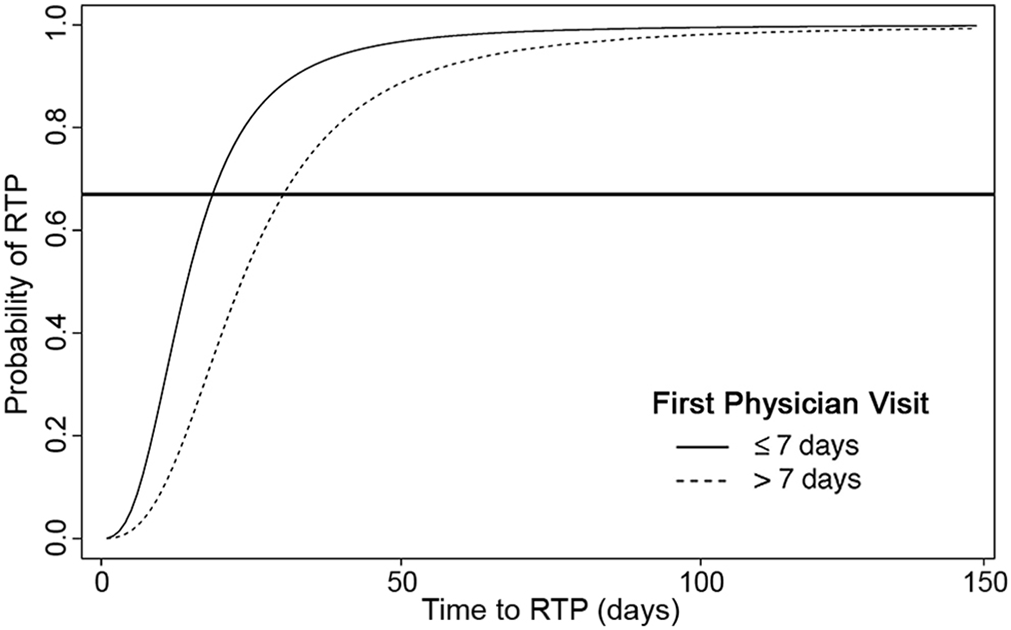
Probability curve for return to play (RTP) by time to first physician visit. Example based on a player in the 13- to 14-year age group playing in a body-checking league, with no previous history of concussion, a symptom evaluation score of 12, and a tandem stance score ≤3 at time of first physician visit. The horizontal line represents the point at which 67% of players (level of covariables as stated) returned to play by 11 days if they saw a physician ≤7 days after injury versus 18 days if they saw a physician at >7 days.
The median value of the symptom evaluation score and the mode value of each of the other covariables were chosen to calculate probabilities to demonstrate that the probability of RTP for players who see a physician at ≤7 days is higher than that of players who see a physician >7 days after concussion. Furthermore, with every 1-unit increase in symptom evaluation score, the time to RTP for youth ice hockey players increased by 1.016-fold. Poorer tandem stance was not statistically significant in model 1 (95% CI, 0.997-1.417), but it was a significant covariable in model 2 (see below). Age group, sex, league type, and concussion history at first visit were not associated with time to clinical recovery but were included in the model as confounders.
Model 2
Analysis of model 2 (Table 4) demonstrated that longer time to first physician visit (>7 days) (TR, 1.698 [95% CI, 1.399-2.062]), headache (moderate/severe) (TR, 1.319 [95% CI, 1.110-1.568]) at first physician visit, and poorer tandem stance score (≥4 errors) (TR, 1.249 [95% CI, 1.052-1.484]) at first physician visit were associated with time to clinical recovery. Thus, time to RTP among youth ice hockey players who saw a physician >7 days after a concussion was 1.698 times that of youth hockey players who saw a physician at ≤7 days. Furthermore, time to RTP among youth ice hockey players with ≥4 errors in tandem stance after a concussion was 1.249 times that of youth hockey players with ≤3 errors in tandem stance. Finally, time to RTP among youth ice hockey players who had moderate or severe headache after a concussion was 1.319 times that of youth hockey players who had mild or no headache symptoms. Age group, sex, league type, concussion history, and other specific symptoms at first visit were not found to influence time to clinical recovery but were included in the model as confounders.
Model 2 Analysis Results (Outcome: Time to RTP Clearance) a

a Bolded values indicate statistical significance based on a 5% significance level. mBESS, modified Balance Error Scoring System; RTP, return to play.
Discussion
This prospective cohort study investigated factors associated with clinical recovery after sports-related concussion specifically in youth ice hockey players. The proportion of youth ice hockey players with prolonged clinical recovery (not cleared to RTP by physician by ≤28 days) was 25% (for those not censored). Consistent with this, 20% of elite youth ice hockey players were not cleared at 30 days after injury. 22 Zemek et al 23 reported that 30% of children and adolescents (age range, 5-17 years) reporting to the emergency department had persistent symptoms for >28 days. Kontos et al 14 reported that 61% of patients (age range, 12-22 years) assessed at a sports medicine clinic had not yet recovered at 30 days. This larger proportion with prolonged clinical recovery may have been related to loss to follow-up in 61.1% of this sample. 14 In the current study, 9.26% players with diagnosed concussions (of those who were not censored) were clinically recovered on the basis of clearance to RTP by 7 days, similar to the rates reported across multiple sports in youth and adult collegiate athletes (10%). 16
In both models analyzed in this study, longer time to first physician visit (>7 days) was consistently associated with longer clinical recovery. This is consistent with the results of the study by Kontos et al, 14 in which patients evaluated >7 days after concussion had a 5.8 times greater odds of prolonged clinical recovery (>30 days) than did patients evaluated at ≤7 days. Furthermore, 81.3% of patients evaluated >7 days after concussion had prolonged clinical recovery compared with 48% of patients evaluated within 7 days. 14 This may indicate that earlier clinical management and initiation of RTP protocol (eg, symptom limiting activities, light aerobic activity) may lead to earlier resolution of symptoms and RTP. An alternative possibility is that players seeking physician care >7 days after concussion may be seeking care because they have more persistent or intense symptoms. However, Kontos et al reported no differences between those evaluated after or before 7 days on symptom severity. In that study, the median symptom evaluation score was also similar in players evaluated within 7 days (12/132) and >7 days (13/132) after concussion.
In our study, poorer tandem stance (≥4 errors) at the time of first physician visit was associated with a longer clinical recovery in model 2, similar to the findings of Zemek et al 23 in children reporting to the emergency department. Greater overall symptom severity (model 1) and moderate/severe headache symptom severity (model 2) were also associated with longer clinical recovery, consistent with the findings of McCrea et al. 16 In this cohort, age group, sex, body checking status, number of previous concussions, and other specific symptoms (eg, neck pain, sensitivity to noise, fatigue, dizziness) were not associated with time to clinical recovery. The age range (11-17 years) in this study was narrow compared with that of other studies reporting prolonged clinical recovery in adolescents compared with younger children. 12,23 Sex did not influence clinical recovery in this study, possibly related to the small number of girls (n = 50). Body-checking policy was considered a covariable because physician decisions to approve RTP may be more conservative for players in leagues that allow body checking; however, results did not support this. The number of previous concussions was not associated with time to clinical recovery, despite findings reported in other studies. 23,25 Zemek et al 23 considered patients with previous concussions with >7 days to clinical recovery and demonstrated a greater odds of persistent symptoms (odds ratio, 2.2 [95% CI, 1.7-3.0]). Time to clinical recovery from previous concussion was not evaluated in this study.
Limitations
While a standard concussion definition and harmonized protocol leading to physician clearance to RTP was established across study physicians, differences in clinician decisions to clear a player to RTP after concussion may still have occurred. Furthermore, the determination of clinical recovery was based on the physician’s clinical decision to RTP (79.6% of cases) or athletic therapist follow-up assessment, and neurophysiological outcomes that were not clinically assessed may not have fully returned to a preconcussion state. Additionally, players may have been exposed to various management and treatment strategies that were not accounted for in this study. The main difference in concussion management based on the 4th (years 1-4) and 5th (year 5) International Consensus Statements on Concussion in Sport was that only 24 to 48 hours of initial rest followed by a gradual return to subsymptom threshold activities was implemented in 2017. 17,18 However, the distribution of time to clinical recovery did not change between years 1 to 4 and year 5, so the effect of this change was not apparent and therefore would not have influenced our results.
In the absence of standard objective outcomes for concussion diagnosis or persistent postconcussion symptoms (eg, imaging, fluid biomarkers, clinical measures), models examining factors influencing concussion outcomes lack the precision available for other diseases. Migraine history and attention deficit hyperactive disorder were not considered in these analyses based on only 4% of players reporting a migraine history (23.5% missing data) at baseline and 4% of players reporting attention deficit hyperactive disorder (no missing data).
The homogeneity of youth ice hockey players limits the generalizability of these findings.
Conclusion
This study highlights the importance of early assessment and appropriate management by a clinician after sports-related concussion in youth. In addition, poorer scores on tandem gait, higher symptom evaluation score, and greater headache severity were associated with longer time to clinical recovery in child and adolescent ice hockey players after concussion. These findings support inclusion of symptom severity and postural stability measures acutely after concussion. Further study is required to consider other potential objective measures (eg, oculomotor, neurocognitive, fluid biomarkers, imaging) influencing time to clinical recovery after sports-related concussion in youth.
Footnotes
Clinical Implications
This study supports timely access to physician assessment, which is associated with reduced clinical recovery time and may be related to early appropriate management. Further, early symptom severity can inform management strategies and clinical decision making regarding early treatment after sports-related concussion in youth.
Acknowledgment
The authors acknowledge Hockey Canada; Hockey Calgary; Hockey Edmonton; and all research staff, coaches, players, and parents involved for their time and support in completing this research project.
Final revision submitted February 4, 2021; accepted March 24, 2021
One or more of the authors has declared the following potential conflict of interest or source of funding: The Sport Injury Prevention Research Centre is one of the International Research Centres for Prevention of Injury and Protection of Athlete Health supported by the International Olympic Committee. We acknowledge funding from Canadian Institutes of Health Research, Alberta Innovates Health Solutions, Hotchkiss Brain Institute, and Alberta Children’s Hospital Foundation. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the research ethics boards at the University of Calgary (ethics ID: REB14-0348 and REB14-2209) and University of Alberta (ethics ID: REB Pro00024093).
